
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Bachelor of Pharmacy, Womens College Of Pharmacy, Peth-Vadgoan
Modern synthetic medicines are highly effective in treating diseases but often come with various side effects. In contrast, crude drugs are generally less potent in curing illnesses but tend to have fewer adverse effects. Parasites, particularly helminths, have been a significant concern in the medical field for centuries, affecting both humans and animals. Many medicinal plants are traditionally believed to have anthelmintic properties and are widely used by indigenous communities worldwide. Based on these traditional claims, numerous medicinal plants have been scientifically examined for their potential anthelmintic activity using various in vitro and in vivo methods. This review highlights key pharmacological studies and preliminary research on medicinal plants, their extracts, and bioactive compounds, which could serve as potential leads in developing novel herbal drugs.
In poor countries, intestinal parasites pose a major danger to animal output. Chemotherapy is still often employed to manage helminths, although parasites of greater economic consequence have developed anthelmintic resistance. Worldwide, helminthiasis, which is brought on by helminth infection, is a significant barrier to cattle productivity. As previously said, chemotherapeutics continues to be the mainstay for treating helminthiasis by overcoming many obstacles, including toxicity and chemical residues, high costs, the inability of medications to be modified, and their unavailability in distant locations. Due to many limitations, including the high expense of these medications, the typical emergence of nematode-resistant populations, and the potential for environmental and animal product contamination, control options are being sought. Due to their ecological acceptability and sustainable supply, the use of medicinal herbs for the aforementioned issue has been more widely accepted since ancient times.[1]
1) Snake Plant:

Fig No.1: Draceana trifasciata
The biological source of the snake plant is Dracaena trifasciata a member of the family Asparagaceae. It Is referred to as the Sansevieria trifasciata. The leaves of this plant contains β-sitosterol, ruscogenin, neoruscogenin, and two spirostan sapogenins: 25S-ruscogenin and sansevierigenin.". Initial phytochemical and physicochemical analyses of the leaves revealed the presence of alkaloids, α-amino acids, glycosides, carbohydrates, reducing sugars, phenolic compounds, flavonoids, steroids, and trace amounts of terpenoids. The in vitro anthelmintic potential of D. trifasciata leaf extracts was evaluated against Fasciola hepatica. The study demonstrated that varying concentrations of the extracts resulted in parasite death over different average time periods. In traditional medicine, both the roots and leaves are utilized to treat a range of conditions, including cough, asthma, colic, abdominal discomfort, diarrhea, hemorrhoids, menorrhagia, piles, high blood pressure, sexual debility, foot sores, leprosy, nutritional deficiencies, swollen glands, rheumatism, and snake bites. In Chinese medicine, a decoction of the plant is employed for treating cough, bronchitis, detoxification, inflammation, skin boils, injuries from trauma, and snake bites. It also contains pharmacological activities such as analgesic, antipyretic, anti-ulcerative, anti-diabetic, antibacterial, antioxidant, cytotoxic, anthelmintic, hepatoprotective, anti-alopecia, wound healing, etc.[2]
2) Aloe Vera:

Fig No.2: Aloe berbadensis Miller
The biological source of aloe vera is Aloe berbadensis Millerbelonging to the family Liliaceae. It is also known as Kumari or Musabbar in some demographic areas. It is a shrubby or arborescent, perpetual, xerophytic, juicy, pea-green coloured plant. There are over 300 species of aloe vera in the whole world. It contains chemical constituents such as three isomers of alones, barboloin and isobarbaloin, which constitute the purported crystalline aloin, showing within the pharmaceutical at from 10 to 30% constituents are unclear aloin, sap, emodin and Aloe-emodin[3].The preliminary phytochemical screening of alcoholic extracts of aloe vera contains gums, mucilage, resins, alkaloids, steroids, flavonoids, tannins, glycosides and saponins. From these constituents, the mucilage, glycosides, flavonoids and phenolic compounds are responsible for the anthelmintic activity of aloe vera[4]. The anthelmintic activity of aloe is tested by using the Fasciola hepatica worm. The aqueous aloe vera extracts show low anthelmintic activity compared to the ethanolic extracts. The polyphenol oxidase enzyme present in the aqueous extract decreases the polyphenol level, which is inactive in ethanolic extract. Also the ethanolic extracts contain higher concentrations of resins, gums and mucilage[4]. Hence, it is concluded that Aloe berbadensis Miller shows potential anthelmintic activity. And also the synergistic effect is shown by the alcoholic as well as the aqueous extracts of the Ale berbadensis Miller + Boswellia serrata combination[4]. Aloe vera is cathartic, carminative, deobstuent, diuretic, stomachic, emmenagogue, and anthelmintic. Aloe vera juice is used as skin medicine, for dyspepsia, amenorrhea, smolders, colic, hyperdenosis, hepatopathy, and splenopathy. It is also used to treat ulcerative colitis and weight ulcers.[3]
3) Walnut:

Fig No.3: Juglans regia Linn
The biological source of walnuts is Juglans regia Linn a member of the family Juglandaceae. It is commonly known as Echte walnut or Akhrot[5]. The leaves, bark and kernels of walnut contain alkaloids, saponins, tannins, flavonoids, cardiac glycosides, carbohydrates, steroids and various amino acids[5]. The stem of walnut contains steroids, tannins, flavonoids and cardiac glycosides which are responsible for its anthelmintic activity[6]. The death time and paralysis time tests were performed, and it was concluded that the acetone and aqueous extracts exhibited moderate anthelmintic activity against the Indian earthworm Pheretima posthuma, whereas the benzene and methanolic extracts showed notable effectiveness, surpassing that of the standard drug albendazole[6]. The acetone extracts of the stem of walnut also possess significant anthelmintic activity at all dilutions against Eisenia foetida in relation to the standard drug albendazole[7]. The walnut possesses different therapeutic uses such as analgesic, anti-inflammatory, antipyretic, anticancer, antimicrobial and antihypertensive. In folk medicine, walnuts are employed in the management of various health conditions. [5]
4)Tamarind:

Fig No.4: Tamarindus indica Linn
The biological source of tamarind is Tamarindus indica Linn, a member of the family fabaceae. It is commonly known as the Chincha, Imli and Amlika in Ayurveda [8]. It is rich in nutrients. The aqueous and ethanolic extracts of whole seeds and pulp powder of the plant contain alkaloids, cardiac glycosides, tannins, saponins, flavonoids and reducing sugars. The active chemical constituent of the tamarind is the tartaric acid[8]. The tamarind leaves contain carbohydrates, flavonoids, pectins, tartaric acid, dihydrobutanedioic acid and tannins. From which tannins are responsible for its anthelmintic activity[9]. The death time and paralysis time were studied on the Indian earthworm Pheretima posthuma and it was determined that Tamarindus indica Linn juice demonstrates considerable anthelmintic effects in comparison to the standard drug, piperazine citrate. [9]. The traditional uses of tamarind are antidiabetic, antiulcer, anti-inflammatory, laxative, hepatoprotective, antifertility and alleviate sunstroke. The pulp of the tamarind is also used for beverages and candies as a flavouring agent.[8]
5) Papaya:

Fig No.5: Carica papaya
The biological source of papaya is Carica papaya belonging to the family Caricaceae[10]. Papaya is rapidly growing, short-lived woody plant that can reach heights of 10-12 feet[10]. The papaya possesses potent antimicrobial activity. Papaya fruit is an antioxidant as it contains carotene, vitamin C, vitamin B, flavonoids, folate and minerals such as potassium and magnesium[10]. Papaya leaves contain a variety of compounds, including proteolytic enzymes like papain and chymopapain, alkaloids such as caprin and carpasemine, sulfur-containing substances like benzyl isothiocyanate, as well as flavonoids, triterpenes, carotenoids, tannins, organic acids like vanillic, ferulic, caffeic, and coumaric acids, saponins, and oils.[10]. The papaya seeds are anthelmintic in nature[10]. Also, the papaya’s latex exhibits anthelmintic activity at different concentrations as it contains papain, chymopapain, caricain, glycyl endopeptidase and papaya lipase[11]. Death time and paralysis time of papaya latex at different concentrations were performed by using the Indian earthworm Pheretimaposthuma. It was concluded that the Piperazine citrate works by causing flaccid paralysis in experimental worms, which results in their expulsion through peristalsis. Comparing the latex of papaya showed significant anthelmintic activity, outperforming the control group with Piperazine citrate (10mg/ml).[11]
6) Sicklepod:

Fig No.6: Cassia tora Linn
The biological source of sicklepod is Cassia tora Linn belonging to the family Leguminosae. In Ayurveda, it is referred to as Chakramard, while in Unani, it is called Panwar.. It is an annual foetid plant, 30-90 cm high[12]. The phytochemical screening states that it contains anthraquinone glycosides, flavonoids, phenolic compounds, tannins, saponins, terpenoids, steroids, alkaloids,resins, mucilage and sugars[12]. From all these constituents carbohydrates, tannins, glycosides, terpenes and steroids are responsible for sickle pod's anthelmintic activity [13]. The anthelmintic properties of the alcoholic extracts from the seeds of sicklepod were tested against the two types of worms Pheretimaposthuma and Ascaridia galli. The extracts were combined with distilled water to prepare three concentrations: 25, 50, and 100 mg/ml. At the highest concentration of 100 mg/ml, both extracts exhibited significant activity. For comparison, piperazine citrate was used as the standard drug at the same concentration, while distilled water served as the control. Additionally, the methanolic extract at concentrations of 10, 25, and 50 mg/ml demonstrated notable anthelmintic activity when compared to the standard drug albendazole (20 mg/ml), assessed by both death time and paralysis time. Traditionally, sicklepod is known for its wide range of medicinal properties, including its roles as a laxative, antiseptic, antioxidant, and antiperiodic. It is also utilized in treating conditions such as leprosy, ringworm, bronchitis, heart diseases, liver diseases, hemorrhoids, as well as ophthalmic and skin disorders. [12]
7) Turmeric:

Fig No.7: Curcuma longa
The biological source of turmeric is Curcuma longa belonging to the family Zingiberaceae. Preliminary phytochemical investigations of turmeric rhizome were conducted using various solvents such as aqueous, acetone, ethanol, chloroform, and methanol extracts. The extracts were analyzed using standard precipitation and coloration tests. The findings indicated the presence of bioactive compounds such as carbohydrates, proteins, alkaloids, glycosides, terpenes, steroids, flavonoids, tannins, and saponins. Among these, curcumin was identified as the most active and effective chemical constituent. These extracts were tested using common precipitation and coloration reactions. The results revealed the presence of bioactive compounds including carbohydrates, proteins, alkaloids, glycosides, terpenes, steroids, flavonoids, tannins and saponins. And the most active and most effective chemical constituent is curcumin. These compounds are believed to contribute to the therapeutic properties of turmeric including its potential anthelmintic and antimicrobial activities[14]. The study tested the anthelmintic activity of turmeric rhizome extracts against Haemonchus spp. parasites ingoats. Both the aqueous (AE) and methanolic (ME) extracts showed dose-dependent effects, with the ME proving more effective than the AE. The paralysis and death times of worms exposed to different concentrations of the extracts, with results indicating that higher concentrations led to better anthelmintic activity. At 1 mg/ml, the ME caused paralysis in 139 min and death in 175.3 min, outperforming the AE which took 159.7 min for paralysis and 198 min for death. These results were compared with the standard drug, albendazole, which caused paralysis in 32 min and death in 57.3 min. The extracts contained tannins, saponins, phenols, and alkaloids, which are responsible for antiparasitic activity. The study suggests that turmeric could be explored further as a cost-effective treatment for parasitic infections [15]. Turmeric rhizomes are known to purify the blood, act as a tonic for the brain and heart, and are used in the treatment of leukoderma, piles, bronchitis, asthma, tumors, tuberculosis, spleen enlargement, and to control leucorrheal and gonorrheal discharges. Ancient Hindu texts describe turmeric as an aromatic stimulant and carminative. [14,15]. Recently, turmeric powder has been used in traditional medicine to treat gastrointestinal diseases, particularly biliary and hepatic disorders, diabetic wounds, rheumatism, inflammation, sinusitis, anorexia, coryza, and cough. Turmeric is known for its various health benefits, including anticancer, anti-diabetic, antioxidant, hypolipidemic, anti-inflammatory, antimicrobial, anti-fertility, anti-venom, hepatoprotective, nephroprotective, and anticoagulant properties, and it also exhibits anti-HIV activity, helping to combat AIDS. [14,15]
8) Chicory:

Fig No.8: Cichorium intybus
The biological source of chicory is Cichorium intybus a member of the family Asteraceae. It is commonly known as Coffee weed or Blue daisy [16]. The chicory contains phytochemicals such as polyphenols, terpenoids, saponins, volatile oils, amino acids, carbohydrates, vitamins, fatty acids, alkaloids, and phytosterols[16]. From all these chemical constituents, sesquiterpenes are responsible for its anthelmintic activity[17]. The anthelmintic activity of chicory on Cooperia oncophorawas tested using in vitro testing, The study concluded that chicory extract inhibited egg hatching and induced paralysis in adult worms in a dose-dependent manner. While no in vivo effects were observed in previous cattle studies, the researcher suggests this may be due to host-mediated factors or digestive conditions, rather than a lack of activity in the chicory extract. The results support chicory as a potential natural alternative to reduce reliance on anti-parasitic drugs in livestock[17]. Medicinally chicory is useful as an antioxidant, anti-inflammatory, cardioprotective, nephroprotective, hepatoprotective, gastroprotective, sedative, cardiovascular, immunological, anticancer, antidiabetic and antimicrobial.[18]
9)Betel Leaves:

Fig No.9: Piper betel Linn
The biological source of betel leaves is Piper betel Linn a member of the family Piperaceae. It is also known as Paan in Indian languages, used for improving the digestion process. Initial phytochemical screening revealed the presence of alkaloids including piperine, nicotine, and berberine, phenolic compounds like eugenol, hydroxy-chavicol, and chavibetol, flavonoids such as estragole and anethole, vitamins like β-carotene, α-tocopherol, and vitamin C, as well as acids including hexadeconic and chlorogenic acid, along with eugenol acetate and safrole[19]. All of these tannins, saponins, alkaloids, glycosides,etc. are responsible for their anthelmintic activity[20]. The death time and paralysis time were measured using Pheretima posthuma with test concentrations of 10, 20, 40, 60, and 80 mg/ml, compared to the standard drug albendazole (10 mg/ml). It was concluded that the methanolic extracts of betel leaves possess wormicidal properties. Hence, it may be used as an anthelmintic[20]. The betel leaves are used as digestant, anti-inflammatory, carminative, anti-allergic, antimutagenic, anticancer and anti-tumor. And also used in nutraceutical foodstuffs and products.[19]
10) Milkweed::

Fig No.10: Calotropis procera
The biological source of milkweed is Calotropis procera belonging to the family Apocyanaceae. Plants of this genus are referred to as milkweed because they produce a white, sticky latex from various parts of the plant. This Calotropis procera species is more poisonous than other species like Calotropis gigantica[21]. It is also known as Sodom Apple and Rakta Arka in Ayurveda[21,22]. It is a highly toxic plant, with stems and the roots are more toxic than the leaves. The plant contains harmful glycosides such as calotropin, calotoxin, uscharin, and calactin, which can lead to serious health consequences. Latex is especially dangerous, being caustic and neurotoxic, leading to symptoms such as vomiting, diarrhoea, convulsions, and death. It can cause eye damage and vision issues if it enters the eyes. But the plant also has a defense mechanism where the concentration of calactin increases when attacked by insects, preventing grazing by animals. Additionally, a protein in the latex called osmotin protects the plant against pathogens[21]. The phytochemical studies reveal that Calotropis procera contains cardenolides, flavonoids, sterols, oxypregnanes, triterpenoids, glycosides, etc.[21]. In vivo, studies on sheep infected with mixed species of nematodes revealed that milkweed extracts exhibit strong anthelmintic activity. The crude aqueous extract(CAE), showed the most significant reduction in egg count (88.4%), followed by the crude powder(CP) at 77.8%, and the crude methanolic extract(CME) at 20.9%. These results suggest that the aqueous extract of milkweed has excellent potential as a natural anthelmintic agent for controlling parasitic infections in livestock[21]. In vitro studies on Haemonchus contortus showed that both aqueous and fresh latex of milkweed exhibit dose-dependent wormicidal activity. At higher concentrations, the latex caused paralysis and death of the worms within 60-90 min, similar to the effects of Piperazine citrate. These results suggest that milkweed latex has potential as a natural anthelmintic agent for controlling parasitic infections[21]. The plant has various medicinal properties. When dried, it acts as a tonic, anthelmintic, and expectorant. The roots are used as a laxative and for treating bronchitis, asthma, leprosy, eczema, and paralysis. The latex treats vertigo, hair loss, toothache, fevers, joint swelling, and paralysis. The leaves help with joint pain and swelling.[22]Top of FormBottom of Form
Phytochemicals responsible for the anthelmintic activity:
These phytochemicals present in the above plants are responsible for the anthelmintic activity. [23,24]
|
Sr.No. |
Phytoconstituents |
Structure |
Details |
|
1 |
Quercetin |

|
IUPAC Name: 2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4-chromenone Mol. Formula: C15H10O7 Mol. Wt: 302.23 g/mol Physical Description: Quercetin presents as yellow-colored needles or a yellow powder. It transitions to its anhydrous form at temperatures between 203-207 °F. Solutions in alcohol have a distinctly bitter taste. Melting Point: 601 to 603 °F
|
|
2 |
Kaempferol |

|
IUPAC Name: 3,5,7-trihydroxy-2-(4-hydroxyphenyl)chromen-4-one Mol. Formula: C15H10O6 Mol. Wt: 286.24 g/mol Physical Description: Solid Melting Point: 277 °C |
|
3 |
Aloin |

|
IUPAC Name: (10S)-1,8-dihydroxy-3-(hydroxymethyl)-10-[(2S,3R,4R,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]-10H-anthracen-9-one Mol. Formula: Mol. Wt: 418.4 g/mol Physical Description: A yellow-brown compound with a bitter taste. Melting Point: 148°C |
|
4 |
Aloe-emodin |

|
IUPAC Name: 1,8-dihydroxy-3-(hydroxymethyl)anthracene-9,10-dione Mol. Formula: C15H10O5 Mol. Wt: 270.24 g/mol Physical Description: Solid Melting Point: 221 - 223 °C |
|
5 |
Juglone |
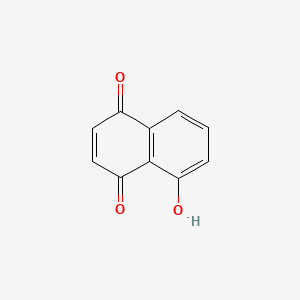
|
IUPAC Name: 5-hydroxynaphthalene-1,4-dione Mol. Formula: C10H6O3 Mol. Wt: 174.15 g/mol Physical Description: Solid Melting Point: 155 °C |
|
6 |
Crysophanic Acid/ Crysophanol |
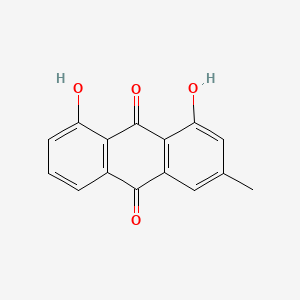
|
IUPAC Name: 1,8-dihydroxy-3-methylanthracene-9,10-dione Mol. Formula: C15H10O4 Mol. Wt: 254.24 g/mol Physical Description: Chrysophanic acid presents as golden-yellow plates or a brown powder, and as yellow hexagonal or monoclinic-shaped needles. Melting Point: 196 °C |
|
7 |
Rhein |
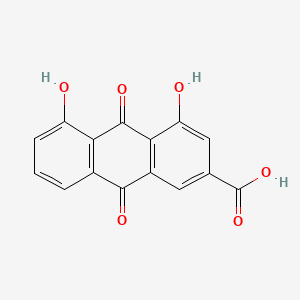
|
IUPAC Name: 4,5-dihydroxy-9,10-dioxoanthracene-2-carboxylic acid Mol. Formula: C15H8O6 Mol. Wt: 284.22 g/mol Physical Description: Rhein is found as yellow needles (when extracted from methanol) or as a yellow-brown powder. Melting Point: 610 °F |
|
8 |
Emodin |
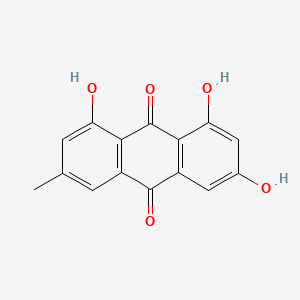
|
IUPAC Name: 1,3,8-trihydroxy-6-methylanthracene-9,10-dione Mol. Formula: C15H10O5 Mol. Wt: 270.24 g/mol Physical Description: Emodin manifests as orange-colored needles or powder. Melting Point: 493 to 495 °F |
|
9 |
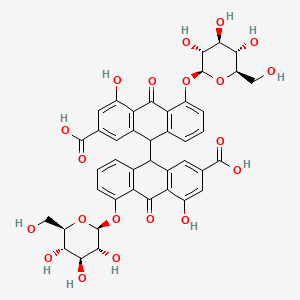
Sennoside |
|
IUPAC Name: 9-[2-carboxy-4-hydroxy-10-oxo-5-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-9H-anthracen-9-yl]-4-hydroxy-10-oxo-5-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-9H-anthracene-2-carboxylic acid Mol. Formula: C42H38O20 Mol. Wt: 862.7 g/mol Physical Description: A yellow-brown powder with a subtle odor and taste, and only slightly soluble in water. Melting Point: 189°C to 225°C
|
|
10 |
Curcumin |

|
IUPAC Name: (1E,6E)-1,7-bis(4-hydroxy-3-methoxyphenyl)hepta-1,6-diene-3,5-dione Mol. Formula: C21H20O6 Mol. Wt: 368.4 g/mol Physical Description: Yellow Melting Point: 356 to 361 °F |
|
11 |
Demethoxycur-cumin |

|
IUPAC Name: (1E,6E)-1-(4-hydroxy-3-methoxyphenyl)-7-(4-hydroxyphenyl)hepta-1,6-diene-3,5-dione Mol. Formula: C20H18O5 Mol. Wt: 338.4 g/mol Physical Description: Solid Melting Point: 168 °C |
|
12 |
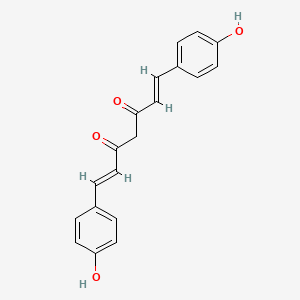
Bis-demethoxyc-rcumin |
|
IUPAC Name: (1E,6E)-1,7-bis(4-hydroxyphenyl)hepta-1,6-diene-3,5-dione Mol. Formula: C19H16O4 Mol.Wt: 308.3 g/mol Physical Description: Solid Melting Point: 179–182 °C |
|
13 |
Papain |

|
IUPAC Name: 2-[[2-[[2-[(2-aminoacetyl)amino]acetyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-5-(diaminomethylideneamino)pentanoic acid Mol. Formula: C19H29N7O6 Mol. Wt: 451.5 g/mol Physical Description: Colorless, odorless Melting Point: Papain doesn’t have melting point. |
|
14 |
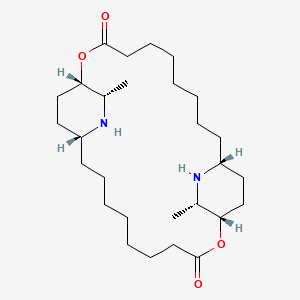
Carpaine |
|
IUPAC Name: (1S,11R,13S,14S,24R,26S)-13,26-dimethyl-2,15-dioxa-12,25-diazatricyclo[22.2.2.211,14]triacontane-3,16-dione Mol. Formula: C28H50N2O4 Mol. Wt: 478.7 g/mol Physical Description: White, crystalline Melting Point: 121°C |
|
15 |
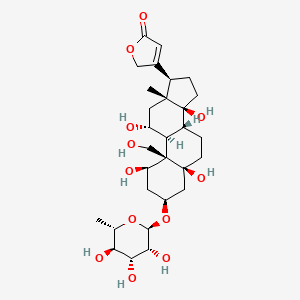
Quabain |
|
IUPAC Name: 3-[(1R,3S,5S,8R,9S,10R,11R,13R,14S,17R)-1,5,11,14-tetrahydroxy-10-(hydroxymethyl)-13-methyl-3-[(2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy-2,3,4,6,7,8,9,11,12,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl]-2H-furan-5-one Mol. Formula: C29H44O12 Mol. Wt: 584.7 g/mol Physical Description: Quabain is seen as odorless, white crystals or a crystalline powder in its octahydrate form. It is used to induce rapid digitalization in cases of acute congestive heart failure and is also recommended for treating atrial or nodal paroxysmal tachycardia and atrial flutter. Melting Point: 392 °F |
|
16 |
Cichorin |
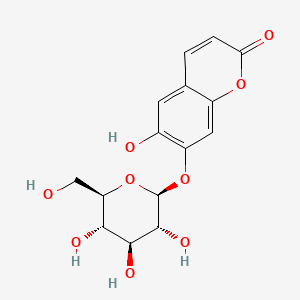
|
IUPAC Name: 6-hydroxy-7-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxychromen-2-one Mol. Formula: C15H16O9 Mol. Wt: 340.28 g/mol Melting Point: 213–215°C |
|
17 |
Eugenol |

|
IUPAC Name: 2-methoxy-4-prop-2-enylphenol Mol. Formula: C10H12O2 Mol. Wt: 164.20 g/mol Physical Description: Eugenol is a clear, colorless to pale yellow or amber liquid with a clove-like aroma and a spicy, pungent taste. Melting Point: 15.4 to 15.6 °F |
|
18 |
Carvacrol |

|
IUPAC Name: 2-methyl-5-propan-2-ylphenol Mol. Formula: C10H14O Mol. Wt: 150.22 g/mol Physical Description: A thick, colorless liquid; according to [Hawley], it is a clear, deep yellow liquid. Melting Point: 1 °C |
CONCLUSION
Many common medicinal plants have been used traditionally for their readily available and potent anthelmintic properties in a range of animals. It is anticipated that thorough in vivo and in vitro research will identify the precise elements that give these plant extracts their anthelmintic action as well as the associated molecular pathways. Ancient texts mention several traditional plants that can be used as anthelmintics. Furthermore, the use of common medications must be required in the future. The development or study of novel medicinal plants is also required to lower the cost of medications. The herbal remedies or medicinal plants that are employed as anthelmintics and have many other uses are presented in this overview. Future people research in India may benefit from this review, which supports the idea that evaluating them may hasten the development of novel, potent medicinal drugs like anthelmintics.
REFERENCES




Gurav Vasudha, Parit Pratiksha, Repe Sakshi, Kamble Ravina, Exploring The Anthelmintic Potential of Commonly Available Medicinal Plants: A Comprehensive Review, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 6, 1358-1372. https://doi.org/10.5281/zenodo.15612840
 10.5281/zenodo.15612840
10.5281/zenodo.15612840