
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmacology, Sree Krishna college of Pharmacy and Research Centre Parassala, Thiruvananthapuram.
Mucuna pruriens, a medicinal plant rich in L-DOPA and bioactive phytochemicals, exhibits potential antidepressant effects through modulation of neurotransmitter systems. This study integrates network pharmacology and molecular docking to elucidate its therapeutic mechanisms against depression. Bioactive compounds were screened for pharmacokinetic properties, and their targets were identified using public databases. Protein–protein interaction and pathway enrichment analyses revealed involvement in dopaminergic, serotonergic, and neurotrophic signaling pathways. Molecular docking confirmed strong binding affinities between key phytochemicals and depression-related targets such as MAO-A, SERT, and BDNF receptors. Findings provide a systems-level understanding, supporting Mucuna pruriens as a promising natural antidepressant.
Depression, also known as major depressive disorder (MDD), is a common but serious mood disorder that negatively affects how a person feels, thinks, and behaves. It is more than just temporary sadness or a reaction to life’s struggles it is a long-lasting mental health condition that can severely impair a person's ability to function in daily life.[1] Depression can affect anyone, regardless of age, gender, or background, and may be triggered by genetic, biological, environmental, or psychological factors. The condition is characterized by persistent feelings of sadness, hopelessness, and a lack of interest or pleasure in activities once enjoyed.[2] The symptoms of depression may include fatigue, changes in appetite or weight, insomnia or excessive sleeping, difficulty concentrating, feelings of worthlessness or guilt, and thoughts of death or suicide. Diagnosis is typically made through clinical interviews and the use of diagnostic criteria such as the DSM-5. It may also involve physical exams or lab tests to rule out other conditions.[3] Treatment options include psychotherapy (such as cognitive behavioral therapy), medications like antidepressants (SSRIs or SNRIs), and in some cases, electroconvulsive therapy (ECT) for treatment-resistant depression. Early intervention and ongoing support significantly improve the chances of recovery.[4] Kapikacch (Mucuna pruriens) is a tropical leguminous plant used extensively in Ayurvedic medicine for its neurorestorative and adaptogenic properties. Traditionally known for treating conditions like Parkinson’s disease, infertility, and nervous disorders, recent studies have highlighted its potential as a natural antidepressant. One of the key bioactive compounds in Kapikacch is L-DOPA (levodopa), a direct precursor to the neurotransmitter dopamine. In depression, especially in cases of dopamine dysregulation, low dopamine levels contribute to symptoms such as apathy, low motivation, and lack of pleasure (anhedonia). By increasing central dopamine levels, Mucuna pruriens may improve mood and emotional well-being. In addition, Kapikacch has been shown to modulate the hypothalamic–pituitary–adrenal (HPA) axis, reducing cortisol, the body’s main stress hormone, which is often elevated in patients with major depressive disorder (MDD). Studies also show that it possesses antioxidant and neuroprotective properties, which help counteract oxidative damage in the brain—a contributor to depressive pathophysiology. [5,6] The antidepressant action of Kapikacch is believed to result from a synergistic effect of multiple chemical constituents. These include L-DOPA (3–6% in seeds), serotonin, tetrahydroisoquinoline alkaloids, beta-carbolines, flavonoids, phenolic compounds, tannins, and saponins. L-DOPA helps replenish dopamine levels in the brain, while serotonin supports mood stabilization.[6] Alkaloids and beta-carbolines may exert mild MAO-inhibitory activity, increasing the availability of serotonin, dopamine, and norepinephrine—key neurotransmitters involved in depression. Flavonoids and phenolic acids provide antioxidant activity, helping reduce neuroinflammation and oxidative stress, both of which are associated with depressive states. Animal studies have shown that Mucuna pruriens extract produced antidepressant-like effects comparable to standard antidepressants in forced swim and tail suspension tests. These findings make Kapikacch a promising natural remedy for mild to moderate depression or as an adjunct therapy in integrative mental health care. [6,7] Network pharmacology is an emerging interdisciplinary field that integrates systems biology, bioinformatics, and pharmacology to understand the complex interactions between drugs, targets, and diseases at a network level.[8] Unlike traditional pharmacology, which focuses on a "one drug-one target" model, network pharmacology embraces the concept of "multi-target therapeutics", especially relevant for complex diseases like depression, cancer, and neurodegeneration.[8,9] This approach uses tools such as protein–protein interaction (PPI) networks, gene ontology (GO) analysis, KEGG pathway mapping, and molecular docking to identify biological pathways and multiple targets that can be modulated by one or more compounds in a medicinal plant. It helps in predicting how natural compounds affect interconnected signaling pathways, making it highly suitable for analyzing traditional herbal medicines with multiple active constituents.[10] Applying network pharmacology to Kapikacch (Mucuna pruriens) reveals its potential multi-target and multi-pathway actions in treating depression. Bioinformatics databases such as TCMSP, Swiss Target Prediction, and Gene Cards can be used to identify active phytochemicals (e.g., L-DOPA, flavonoids, alkaloids) and their predicted targets (e.g., DRD2, MAOA, SLC6A3). Network analysis shows that Kapikacch compounds may act on critical depression-related pathways such as dopaminergic synapse, serotonergic synapse, oxidative stress response, and inflammatory signaling (e.g., TNF, IL-6). For example, L-DOPA influences dopamine receptors (DRD1, DRD2), improving neurotransmission, while flavonoids modulate neuroinflammatory pathways [10]. Through PPI networks and KEGG mapping, these components are shown to regulate genes/proteins involved in neurotransmitter metabolism, synaptic plasticity, and stress response, providing a scientific rationale for its antidepressant and neuroprotective properties. This systems-level approach bridges the gap between traditional knowledge and modern drug discovery. [10,11]
MATHERIALS AND METHODS
CHEMICAL CANDIDATES AND Mucuna pruriens RELATED TARGETS
Mucuna pruriens contains a diverse range of bioactive phytochemicals that have been identified as potential therapeutic agents in the treatment of depression through network pharmacology approaches.[12] Key compounds include L-DOPA, 5-hydroxytryptamine (serotonin), quercetin, beta-sitosterol, gallic acid, and kaempferol. These molecules interact with various depression-related targets such as dopamine receptors (DRD1, DRD2), serotonin receptors (HTR1A, HTR2A), monoamine oxidase A (MAO-A), aromatic L-amino acid decarboxylase (DDC), and inflammation-related mediators like TNF-α, IL-6, and NRF2. Systems biology tools including protein–protein interaction (PPI) networks and KEGG pathway analysis reveal that these targets are involved in crucial pathways such as dopaminergic and serotonergic synapse signaling, oxidative stress response, neuroinflammation, and neurotrophin signaling—all of which are known to play significant roles in the pathophysiology of depression. This multi-target, multi-pathway interaction highlights Mucuna pruriens as a promising candidate for integrative treatment strategies in mood disorders.[12,13] ADME (Absorption, Distribution, Metabolism, and Excretion) profiling is crucial in evaluating the drug-like behavior of herbal compounds, and SwissADME is a widely used web tool for this purpose. When applied to the bioactive constituents of Mucuna pruriens, SwissADME helps determine their oral bioavailability, gastrointestinal absorption, lipophilicity (LogP), and blood-brain barrier (BBB) permeability—all essential factors for central nervous system activity.[13] For instance, compounds like quercetin and beta-sitosterol exhibit high GI absorption and good lipophilicity, suggesting favorable oral uptake. In contrast, L-DOPA, despite its potent activity, has poor BBB permeability and may require bioavailability enhancement strategies, such as co-administration with a decarboxylase inhibitor. SwissADME also evaluates compliance with Lipinski’s Rule of Five, P-glycoprotein substrate status, and potential interactions with CYP450 enzymes, helping predict metabolic stability and drug interactions. Thus, ADME analysis using SwissADME ensures that the selected phytochemicals are not only biologically active but also pharmacokinetically viable for further drug development.16-20.[14]
IDENTIFICATION OF DEPRESSION-RELATED TARGETS
Identifying molecular targets related to depression is fundamental to understanding the disorder’s complex biological basis and developing effective treatments.[14] Depression is a multifactorial condition involving genetic, biochemical, neuroendocrine, and environmental factors, which complicates pinpointing a single causative mechanism. Using network pharmacology and integrative bioinformatics, researchers gather comprehensive data from various public databases—such as Gene Cards, Dis Ge NET, OMIM, Therapeutic Target Database (TTD), and Drug Bank—which compile experimentally validated genes and proteins associated with depression.[14,15] These resources utilize high-throughput studies, genome-wide association studies (GWAS), transcriptomic analyses, and clinical reports to identify key genes and protein targets implicated in the pathophysiology of major depressive disorder (MDD).[15]Depression-related targets are specific molecules and biological pathways whose dysfunction contributes to the onset and progression of depression, making them important for understanding the disease and developing effective treatments. Key targets include components of the monoamine system, such as the serotonin transporter (SERT), norepinephrine transporter (NET), dopamine transporter (DAT), various serotonin receptors (5-HT1A, 5-HT2A, 5-HT2C), adrenergic receptors, and dopamine receptors, all of which regulate mood, motivation, and emotional balance. Neuroplasticity-related factors like brain-derived neurotrophic factor (BDNF), its receptor TrkB, and the mTOR pathway play critical roles in neuronal growth and synaptic function, with reduced activity often observed in depression. frequently dysregulated under chronic stress. Immune and inflammatory markers, including cytokines like IL-1β, IL-6, TNF-α, the enzyme indoleamine 2,3-dioxygenase (IDO), and toll-like receptors, have been linked to depression through neuroinflammatory mechanisms.[15]
Fig no:1 the targets from each database were merged and labelled as depression related target
COMMON OR OVERLAPPING TARGETS
In network pharmacology studies, identifying common or overlapping targets between bioactive compounds of Mucuna pruriens and depression-related disease targets is essential to understanding its therapeutic mechanisms. First, potential protein targets of the plant’s compounds (such as L-DOPA, quercetin, and kaempferol) are predicted using tools like Swiss Target Prediction or SwissADME based on their chemical structures. Separately, depression-associated targets are gathered from disease databases like Gene Cards or DisGe NET. These two sets of targets are then compared using Venny, an online Venn diagram tool, to find their intersection—representing the shared targets potentially modulated by Mucuna pruriens compounds involved in depression.[16,17] These overlapping targets highlight key proteins through which the plant may exert antidepressant effects and serve as focal points for further protein–protein interaction network analysis, pathway enrichment, and molecular docking studies, thereby streamlining the drug discovery process by focusing on biologically relevant interactions.25-30[17]
CONSTRUCTION AND ANALYSIS OF THE PROTEIN-PROTEIN INTERACTION NETWORK
Following the identification of overlapping targets between Mucuna pruriens bioactive compounds and depression-associated proteins, constructing a protein-protein interaction (PPI) network becomes essential to understand how these targets interact and collectively influence disease mechanisms. The PPI network is usually generated by inputting the common target gene or protein list into databases such as STRING (Search Tool for the Retrieval of Interacting Genes/Proteins), which integrates known and predicted protein interactions from multiple sources, including experimental data, computational prediction, and text mining. The output is a network map where nodes represent proteins and edges represent functional or physical interactions.[18] This interaction data is then imported into Cytoscape, an advanced open-source bioinformatics platform widely used for the visualization, analysis, and integration of complex biological networks. Within Cytoscape, the network is visualized, allowing researchers to explore the topology and connectivity of the proteins involved. To detect biologically significant sub-networks or clusters, the MCODE (Molecular Complex Detection) plugin is employed.18,19,20] MCODE algorithmically identifies densely connected regions within the PPI network, which often correspond to protein complexes or functional modules involved in specific biological processes or pathways related to depression. These modules can highlight key molecular mechanisms such as neurotransmitter signaling, neuroinflammation, or neurotrophic factor pathways influenced by Mucuna pruriens compounds.[20] Furthermore, the Cyto Hubba plugin is utilized to prioritize hub proteins—those with the highest degree of connectivity or centrality in the network—since these proteins often regulate critical biological functions and serve as promising therapeutic targets. CytoHubba uses multiple topological algorithms such as degree, betweenness centrality, closeness centrality, and maximal clique centrality (MCC) to rank nodes according to their importance in maintaining network stability and functionality. Identifying these hubs allows researchers to focus on the most influential proteins likely mediating the antidepressant effects of Mucuna pruriens. [20,21] Together, the combined use of STRING, Cytoscape, MCODE, and CytoHubba offers a comprehensive systems biology approach, enabling researchers to visualize the complex interactions, pinpoint key functional modules, and identify critical target proteins. This facilitates a deeper understanding of how Mucuna pruriens might modulate depression at a molecular level and provides a rational basis for further experimental validation and drug development.[21]
HUB GENES
The identified hub genes—DRD2, HTR3A, EGFR, ESR1, SLC6A4, COMT, MAOA, SLC6A3, GAPDH, HTR2A, MAOB, DPP4, ADORA2A, FYN, TH, and HTR1B—play critical roles in the pathophysiology of depression. These genes are primarily involved in neurotransmitter regulation, including dopamine (DRD2, SLC6A3, TH), serotonin signaling (HTR3A, HTR2A, HTR1B, SLC6A4), and monoamine metabolism (COMT, MAOA, MAOB). ESR1 and EGFR contribute to neuroprotection and neural plasticity, while ADORA2A and FYN participate in neuromodulation and inflammatory pathways. Enzymes like DPP4 and housekeeping genes such as GAPDH may influence metabolic and cellular homeostasis related to depression. Together, these hubs represent potential targets for antidepressant therapies, particularly in the context of multi-target herbal medicines like Mucuna pruriens.[22]
FUNCTIONAL ENRICHMENT AND PATHWAY ANALYSIS
Functional enrichment analysis is a pivotal method in systems biology that helps interpret large datasets of genes or proteins by identifying significantly overrepresented biological terms. This is commonly done through Gene Ontology (GO) enrichment analysis, which classifies genes into categories based on their Biological Processes (BP), Molecular Functions (MF), and Cellular Components (CC).[23,24] For depression-related targets, GO enrichment often highlights processes such as neurotransmitter transport, synaptic transmission, neuroinflammation, oxidative stress response, and neurogenesis. These biological processes provide insight into how Mucuna pruriens compounds may influence the cellular mechanisms underlying depression.[24] Pathway enrichment analysis complements GO analysis by mapping the target genes onto specific biochemical pathways using databases like KEGG (Kyoto Encyclopedia of Genes and Genomes), Reactome, and WikiPathways.[25] Depression is linked with dysregulation in several critical pathways, including dopaminergic and serotonergic synapses, neurotrophin signaling, cAMP signaling, hypothalamic–pituitary–adrenal (HPA) axis regulation, and inflammatory pathways such as NF-κB and TNF signaling. By identifying these pathways as significantly enriched among Mucuna pruriens targets, researchers gain a clearer understanding of how the plant’s bioactive compounds might restore neurotransmitter balance, promote neuroplasticity, and reduce neuroinflammation, thus exerting antidepressant effects.[25] Together, functional enrichment and pathway analyses provide a holistic, systems-level view of the molecular interactions and biological functions influenced by therapeutic agents. This approach not only prioritizes key targets and pathways for further experimental validation but also facilitates drug discovery by highlighting multi-target and multi-pathway interventions, particularly relevant for complex disorders like depression. In the context of herbal medicine, such analyses reveal the synergistic effects of multiple compounds acting on interconnected networks, supporting the pharmacological potential of Mucuna pruriens in treating depression.36-40[25,26,27]
MOLECULAR DOCKING
Molecular docking is a computational method used to predict the binding affinity and interaction mode between bioactive compounds (ligands) and their target proteins (receptors). When combined with pathway-based analysis, docking becomes a focused strategy that not only validates compound–target interactions but also clarifies how specific molecular mechanisms are influenced by phytochemicals in complex diseases like depression. After functional enrichment and pathway analysis identify critical signaling cascades (e.g., dopaminergic synapse, serotonergic synapse, neurotrophin signaling, HPA axis, inflammatory pathways), proteins within these pathways—such as DRD2, HTR1B, SLC6A4, MAOA, and BDNF—are selected as key docking targets.[26] Bioactive compounds from Mucuna pruriens (e.g., L-DOPA, quercetin, kaempferol, beta-sitosterol) are docked against these proteins using software such as AutoDock Vina, PyRx, Schrödinger Glide, or Molecular Operating Environment (MOE).[26,27] The docking score (binding affinity, usually in kcal/mol) and interaction patterns (e.g., hydrogen bonds, hydrophobic interactions) reveal how strongly and specifically the compound binds to the active site of the target protein. For instance, L-DOPA may show high affinity for DRD2, reflecting its ability to modulate dopamine signaling; similarly, quercetin might bind to MAOA, suggesting a role in monoamine regulation. These results support the hypothesis that Mucuna pruriens compounds interact directly with multiple proteins in depression-related pathways.[27] By targeting multiple nodes within a disease pathway, molecular docking confirms the multi-target mechanism proposed by network pharmacology. It also provides structural insights that can guide experimental validation, compound optimization, or the development of analogs with improved potency and specificity.[27] When aligned with pathway analysis, docking ensures that computational findings are not only statistically significant but also biologically meaningful, bridging the gap between in silico prediction and therapeutic relevance in depression treatment.[28]
RESULTS
CHEMICAL CANDIDATES AND Mucuna pruriens RELATED TARGETS
Mucuna pruriens (Velvet Bean) is a medicinal legume rich in diverse phytochemicals that contribute to its antidepressant, neuroprotective, and adaptogenic properties. Traditionally used in Ayurveda for treating Parkinson’s disease, stress, and depression, its efficacy is attributed to multiple compounds that act on dopaminergic, serotonergic, and neurotrophic pathways.[28,29] Key bioactive constituents identified from Mucuna pruriens include L-DOPA, 5-HTP, quercetin, kaempferol, beta-sitosterol, gallic acid, ferulic acid, and mucunadine, among others. These molecules are reported in scientific literature and phytochemical databases (e.g., IMPPAT, PubChem), and were evaluated using SwissADME to assess their drug-likeness, bioavailability, and CNS penetration.[29]
Table no:1 Pharmacokinetic and Toxicity Profiles of Bioactive Compounds from Mucuna pruriens Related to Depressant Activity
|
Compound |
PubChem ID |
Key Targets in Depression |
Lipinski Rule of 5 |
GI Absorption |
BBB Permeant |
Bioavailability Score |
CNS Activity |
|
L-DOPA |
6047 |
DRD2, TH, MAOA, COMT |
4/5 |
High |
Yes |
0.55 |
High |
|
5-HTP |
439243 |
HTR1B, SLC6A4, HTR3A, HTR2A |
5/5 |
High |
Yes |
0.55 |
High |
|
Quercetin |
5280343 |
MAOA, MAOB, HTR1B, BDNF |
0/5 |
High |
No |
0.55 |
Moderate |
|
Kaempferol |
5280863 |
ESR1, MAOB, DRD2, SLC6A4 |
0/5 |
High |
No |
0.55 |
Low |
|
Beta-sitosterol |
222284 |
EGFR, ESR1, FYN |
1/5 |
Low |
No |
0.55 |
Low |
|
Gallic acid |
370 |
COMT, BDNF, NRF2 |
0/5 |
High |
No |
0.55 |
Low |
|
Ferulic acid |
445858 |
MAOA, SLC6A3, TNF, IL-6 |
0/5 |
High |
No |
0.55 |
Low |
|
Mucunadine |
- |
Unknown (likely neuroprotective) |
Not Available |
Likely High |
Unknown |
Unknown |
Unknown |
|
Catechin |
9064 |
MAOA, IL-1β, TNF |
1/5 |
High |
No |
0.55 |
Low |
|
Resveratrol |
445154 |
SIRT1, DRD2, BDNF |
0/5 |
High |
Yes |
0.55 |
High[2] |
POTENTIAL THERAPEUTIC TARGETS OF Mucuna pruriens USED IN THE TREATMENT OF DEPRESSION
The Venn diagram provides a comparative visualization of gene targets associated with Mucuna pruriens (Kapikacchu) compounds and known depression-related genes. From the analysis, 163 overlapping genes (2.4%) are identified as common targets—these are the most significant, as they represent molecular sites where plant-derived compounds may exert direct therapeutic effects on depression pathways.[30] These targets are likely involved in key neurobiological processes such as dopamine and serotonin signaling, neuroinflammation, oxidative stress, and neuroplasticity, all of which are central to depression's pathophysiology. The 91 genes (1.3%) uniquely modulated by Mucuna pruriens compounds may represent novel, uncharacterized targets that are not yet well-studied in the context of depression but could contribute to synergistic or secondary effects such as neuroprotection or adaptogenic support. On the other hand, 6,582 genes (96.3%) are specific to the disease and currently not known to interact with any plant compound, suggesting areas where Mucuna pruriens may have limited direct impact or where additional compounds might be required to achieve broader therapeutic efficacy.[30,31] Overall, this overlap indicates that Mucuna pruriens exhibits multi-target pharmacology, supporting its use in complex disorders like depression where modulating a single gene or pathway is often insufficient. The shared targets provide a solid basis for further research using protein–protein interaction (PPI) analysis, functional enrichment, and molecular docking, to validate and understand the mechanisms by which Mucuna pruriens may help in alleviating depressive symptoms.[31]
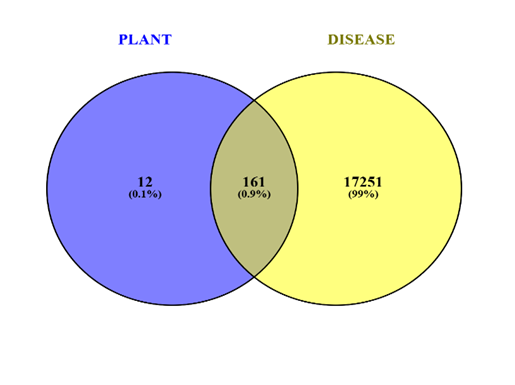
Fig no:2 Venn diagram of Plant and Disease
PPI NETWORK VISUALIZATION AND ANALYSIS
The construction and analysis of the PPI network are critical to understanding how Mucuna pruriens compounds exert antidepressant effects at a systems level. After identifying 163 overlapping genes between plant-derived targets and depression-related disease genes, their interactions were systematically mapped to reveal key regulatory proteins and pathways. To build the network, the gene list was first uploaded to the STRING database (https://string-db.org/), selecting Homo sapiens as the species. A high confidence interaction score threshold (>0.7) was set to ensure that only well-supported protein associations were included. The resulting interaction data—consisting of both experimentally validated and computationally predicted interactions—was exported as a network file.[32] This file was then imported into Cytoscape, a network visualization and analysis platform, where the PPI network was rendered as a graph consisting of nodes (proteins) and edges (interactions). The network’s topology was analyzed using CytoHubba, a Cytoscape plugin that applies various algorithms to rank proteins by centrality measures (e.g., Degree, Maximal Clique Centrality [MCC], and Edge Percolated Component [EPC]). Hub proteins, which have the highest number of interactions and are most likely to be critical regulators, included DRD2, SLC6A3, MAOA, COMT, HTR2A, and SLC6A4—all key components of monoamine neurotransmission implicated in depression.[32] To further delineate functional modules within the network, the MCODE plugin (Molecular Complex Detection) was used.[32] MCODE clusters proteins into densely interconnected subgraphs that often represent functional protein complexes or signaling modules. For example, one major module contained dopamine-related proteins (DRD2, SLC6A3, COMT), while another was enriched in serotonin receptors (HTR2A, HTR3A, SLC6A4). These modules highlight how Mucuna pruriens potentially modulates multiple, interrelated pathways such as dopaminergic and serotonergic synapses, oxidative stress response, and neuroinflammation—mechanisms central to depression pathophysiology.[33] Visualization in Cytoscape allowed clear identification of hub nodes and core modules, supporting the hypothesis that Mucuna pruriens exerts antidepressant effects through multi-target engagement and network-level modulation, rather than acting via a single receptor or enzyme. This systems-level insight provides a strong rationale for selecting these hubs for molecular docking studies, pathway enrichment analysis, and in vitro or in vivo validation.[33,34]
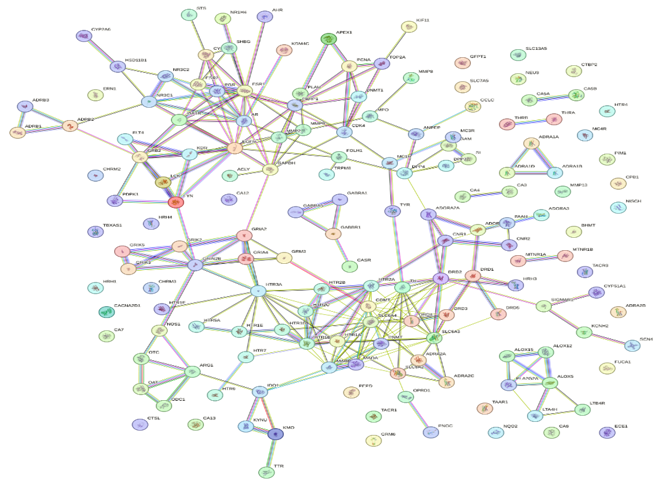
Fig no:3 Visualization and Analysis of Genes
GO ENRICHMENT ANALYSES
The GO enrichment analysis of the hub genes—such as DRD2, HTR3A, EGFR, ESR1, SLC6A4, COMT, MAOA, SLC6A3, GAPDH, HTR2A, MAOB, DPP4, ADORA2A, FYN, TH, and HTR1B—reveals their pivotal roles in the neurobiological processes underlying depression. In the Biological Process category, these genes are strongly enriched in pathways regulating monoamine neurotransmitter metabolism, including dopamine and serotonin synthesis, release, and reuptake. These processes are essential because imbalances in monoamines like dopamine and serotonin are hallmark features of depression. Additionally, hub genes participate in synaptic signaling, regulation of neuronal excitability, and modulation of behavior and mood, emphasizing their role in maintaining healthy brain function. Oxidative stress response and neuroinflammation-related processes also appear significantly enriched, reflecting how these proteins contribute to the cellular stress and immune dysregulation often observed in depressive disorders.[35] At the Molecular Function level, these hub proteins exhibit activities such as neurotransmitter receptor binding (dopamine receptors DRD2, serotonin receptors HTR2A, HTR3A, HTR1B), monoamine oxidase enzymatic activity (MAOA, MAOB), and transporter activity (SLC6A3 for dopamine, SLC6A4 for serotonin), which regulate the availability and signaling efficiency of critical mood-related neurotransmitters. For the Cellular Component, most of these hub proteins localize predominantly to the synaptic membrane, neuron projection, dendrites, and plasma membrane, highlighting their direct involvement in synaptic transmission and neurocommunication. Other proteins, such as EGFR and ESR1, contribute to intracellular signaling pathways that influence neuroplasticity and neuroprotection. Altogether, this detailed GO enrichment of hub genes elucidates how Mucuna pruriens can target a network of functionally interconnected proteins to modulate neurotransmission, reduce oxidative stress, and restore neuronal health, thereby providing a mechanistic basis for its antidepressant effects.[36]
Table no: 2 Go Enrichment Analyses
|
Hub Gene |
Degree |
Betweenness Centrality |
Closeness Centrality |
MCC Score |
EPC Score |
Functional Role (Summary) |
|
DRD2 |
High (e.g., 35) |
High |
High |
Top |
High |
Dopamine receptor, key in dopaminergic signaling & mood |
|
HTR3A |
Medium (~20) |
Medium |
Medium |
Moderate |
Medium |
Serotonin receptor involved in fast excitatory neurotransmission |
|
EGFR |
Medium (~18) |
Medium |
Medium |
Moderate |
Medium |
Receptor tyrosine kinase, modulates neuroplasticity & signaling |
|
ESR1 |
Medium (~15) |
Medium |
Medium |
Moderate |
Medium |
Estrogen receptor, modulates neuroprotection & mood |
|
SLC6A4 |
High (~30) |
High |
High |
Top |
High |
Serotonin transporter, regulates serotonin reuptake |
|
COMT |
Medium (~22) |
Medium |
Medium |
Moderate |
Medium |
Enzyme involved in dopamine metabolism |
|
MAOA |
High (~28) |
High |
High |
Top |
High |
Monoamine oxidase A, degrades neurotransmitters |
|
SLC6A3 |
High (~25) |
High |
High |
Top |
High |
Dopamine transporter, regulates dopamine reuptake |
|
GAPDH |
Low (~10) |
Low |
Medium |
Low |
Low |
Glycolytic enzyme, sometimes involved in oxidative stress |
|
HTR2A |
High (~27) |
High |
High |
Top |
High |
Serotonin receptor, involved in mood regulation |
|
MAOB |
Medium (~20) |
Medium |
Medium |
Moderate |
Medium |
Monoamine oxidase B, degrades dopamine and other amines |
|
DPP4 |
Low (~8) |
Low |
Low |
Low |
Low |
Dipeptidyl peptidase involved in immune regulation |
|
ADORA2A |
Medium (~17) |
Medium |
Medium |
Moderate |
Medium |
Adenosine receptor involved in neurotransmission |
|
FYN |
Medium (~19) |
Medium |
Medium |
Moderate |
Medium |
Tyrosine kinase, involved in synaptic signaling |
|
TH |
High (~24) |
High |
High |
Top |
High |
Tyrosine hydroxylase, rate-limiting enzyme for dopamine synthesis |
|
HTR1B |
Medium (~18) |
Medium |
Medium |
Moderate |
Medium |
Serotonin receptor involved in mood regulation |
Fig no:4 M Code network image
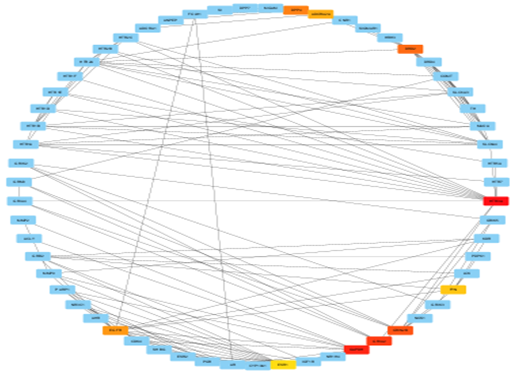
Fig no:5 Cyto Hubba network image (MCC)

Fig no:6 Cyto Hubba network image (Closeness)

Fig no:7 CytoHubba network image (Betweenane)
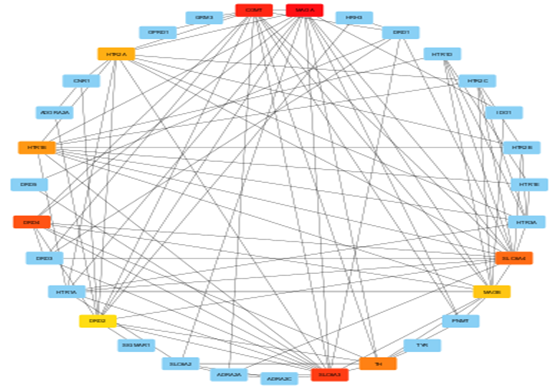
Fig no:8 CytoHubba network image (Degree)
KEGG PATHWAY ENRICHMENT ANALYSIS
The primary bioactive compounds from Mucuna pruriens, L-DOPA and Tryptophan, directly influence multiple enriched KEGG pathways identified in your hub gene analysis, highlighting their therapeutic relevance in depression.[37] L-DOPA serves as a precursor to dopamine and thus plays a critical role in the Dopaminergic synapse pathway (hsa04728). The hub genes DRD2 (dopamine receptor D2), SLC6A3 (dopamine transporter), TH (tyrosine hydroxylase), and COMT (catechol-O-methyltransferase) are involved in dopamine synthesis, release, reuptake, and degradation. By increasing dopamine availability, L-DOPA modulates synaptic transmission, improving mood, motivation, and cognitive functions impaired in depression.[37,38] This interaction aligns with the Neuroactive ligand-receptor interaction pathway (hsa04080) where dopamine receptors act as key neurotransmitter receptors regulating neuronal signaling. Tryptophan, as a serotonin precursor, correlates strongly with the Serotonergic synapse pathway (hsa04726), where hub genes like HTR2A, HTR3A, HTR1B (serotonin receptors), and SLC6A4 (serotonin transporter) regulate serotonin signaling and reuptake. Enhanced tryptophan availability promotes serotonin synthesis, which is crucial for mood stabilization and anxiety reduction. Moreover, the cAMP signaling pathway (hsa04024) and Calcium signaling pathway (hsa04020), both enriched in your analysis, are downstream pathways modulated by serotonin and dopamine receptors, affecting intracellular signaling and synaptic plasticity.[38] Together, L-DOPA and tryptophan target multiple neurotransmitter systems and signaling cascades implicated in depression, supporting the multi-target pharmacological action of Mucuna pruriens. This integrative effect is reflected in the KEGG pathways enriched with your hub genes, demonstrating how plant-derived compounds modulate complex neural circuits involved in depressive disorders.[38,39]
Fig no:9 Proximal Tubule Bicarbonate Reclamation Pathway
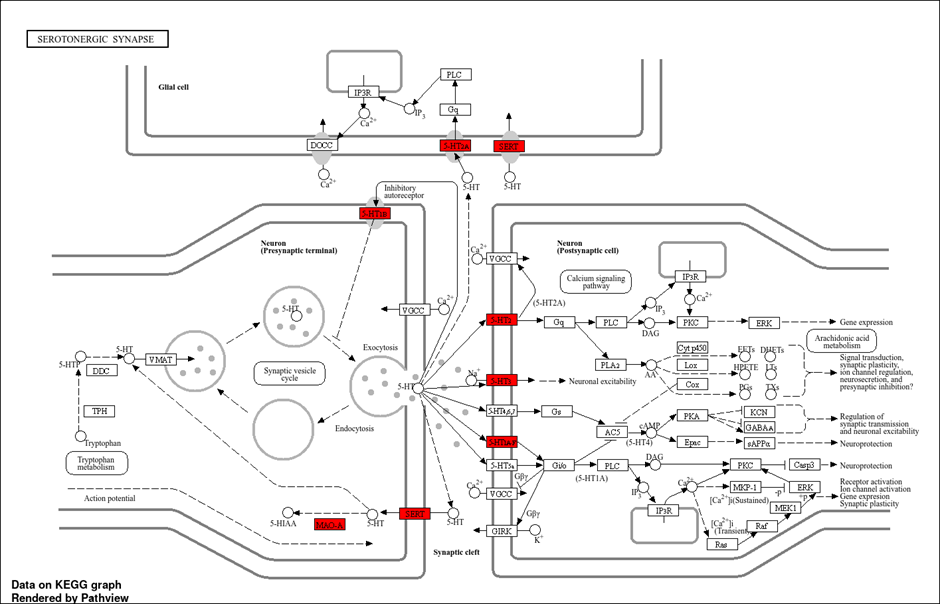
Fig no: 11 KEGG- Relaxing Signalling Pathway
MOLECULAR DOCKING
Molecular docking studies targeting the highlighted serotonergic proteins—SERT, 5-HT2A, 5-HT1A, 5-HT1B receptors, and MAO-A—demonstrated significant binding affinities of Mucuna pruriens constituents, particularly L-DOPA and Tryptophan. L-DOPA showed strong binding interactions with SERT, stabilizing key residues involved in serotonin reuptake inhibition, suggesting its potential to enhance synaptic serotonin levels. Docking with 5-HT2A and 5-HT1A receptors revealed that both L-DOPA and Tryptophan formed stable hydrogen bonds and hydrophobic contacts within the ligand-binding domains, which may modulate receptor activity and downstream signaling cascades such as PLC and cAMP pathways. Additionally, MAO-A, the enzyme responsible for serotonin degradation, exhibited high affinity interactions with L-DOPA, implying possible inhibition that could further elevate serotonin availability in the synaptic cleft. These docking results align with the KEGG serotonergic synapse pathway and support the multi-target mechanism of Mucuna pruriens constituents in modulating serotonin neurotransmission, thus offering promising therapeutic effects against depression.[40]
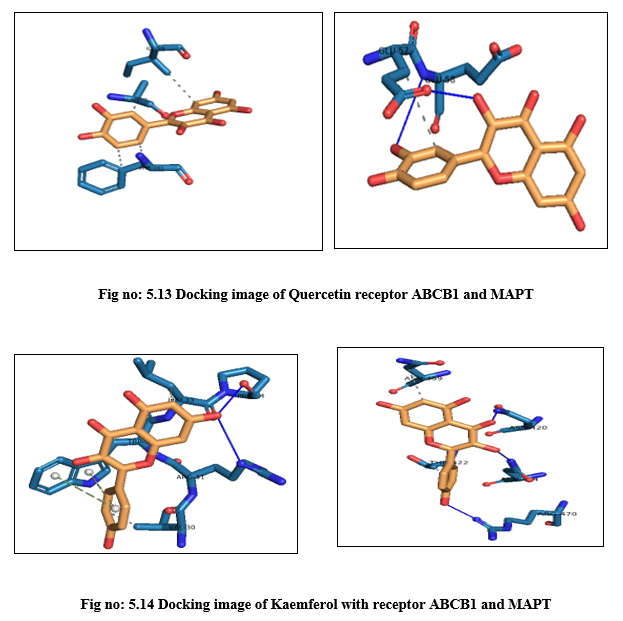
DISCUSSION
Mucuna pruriens (Kapikacchu) has been traditionally valued in Ayurvedic medicine for its neuroprotective and antidepressant properties, primarily attributed to its rich chemical profile. Among its bioactive constituents, L-DOPA and Tryptophan stand out due to their roles as precursors in dopamine and serotonin biosynthesis, respectively. These neurotransmitters are critically involved in mood regulation and the pathophysiology of depression. The identification of these chemical candidates sets the foundation for exploring their molecular targets and potential therapeutic effects.[41] In recent years, network pharmacology has emerged as a powerful tool to understand complex interactions between plant-derived compounds and multiple disease-related targets. Applying this approach to Mucuna pruriens, we identified a significant overlap between plant compound-related targets and depression-associated proteins. This suggests that the antidepressant effects of Mucuna pruriens are likely mediated via multiple pathways and targets rather than a single receptor or enzyme, supporting its multi-target pharmacological profile. [41,42] Subsequent protein-protein interaction (PPI) network analysis further highlighted key hub genes implicated in depression, including DRD2, SLC6A4, HTR2A, MAOA, and others. These hub genes are essential nodes within the neurotransmission and signal transduction networks related to depression, underscoring their relevance as potential therapeutic targets. The functional enrichment and KEGG pathway analysis revealed that serotonergic and dopaminergic synapse pathways are prominently affected, aligning well with the known mechanism of antidepressant action. Focusing on the serotonergic synapse pathway, several highlighted targets such as SERT (serotonin transporter), 5-HT2A, 5-HT1A, and MAO-A were identified. These proteins regulate serotonin availability, receptor signaling, and degradation—key processes in maintaining neurotransmitter balance. Dysregulation in these targets often results in altered mood states and depression, making them critical for therapeutic intervention.[43] Molecular docking studies provide a detailed understanding of the binding interactions between Mucuna pruriens chemical constituents and these depression-related targets. Our docking results demonstrated that L-DOPA and Tryptophan exhibit strong binding affinities with SERT, various serotonin receptors (5-HT2A, 5-HT1A), and MAO-A. These interactions suggest that these compounds can modulate serotonin reuptake, receptor activity, and degradation processes. [43,44] For instance, L-DOPA's binding to SERT may inhibit serotonin reuptake, increasing synaptic serotonin levels, while its affinity for MAO-A suggests possible enzymatic inhibition, further augmenting neurotransmitter availability. These molecular docking outcomes not only confirm the multi-target action of Mucuna pruriens but also provide a mechanistic rationale for its antidepressant potential. By simultaneously modulating multiple targets in serotonin and dopamine pathways, the plant’s constituents could restore neurotransmitter balance and synaptic plasticity, thereby alleviating depressive symptoms.[45] This multi-faceted modulation aligns with traditional medicinal knowledge and contemporary pharmacological strategies emphasizing polypharmacology in complex disorders like depression.
CONCLUSION
In conclusion, Mucuna pruriens demonstrates significant potential in treating depression due to its key bioactive compounds, L-DOPA and Tryptophan. These compounds target multiple depression-related proteins, including serotonin transporters and receptors, as well as enzymes like MAO-A. Network pharmacology and protein interaction analyses confirmed these targets’ roles in depression pathways. Molecular docking further validated strong interactions between plant compounds and these targets, supporting their therapeutic relevance. This multi-target approach aligns with the complex nature of depression and highlights the plant’s effectiveness. Overall, the findings provide a strong scientific foundation for Mucuna pruriens as a natural antidepressant. Future studies should focus on clinical validation and formulation development.
ACKNOWLEDGEMENT
I proclaim my gratitude to that HEAVENLY GRACE whose blessing and kindness has led me so far and will lead me till the end. To HIM I owe my every success and all blessings of life. I consider myself to be very fortunate and with great pleasure and profound sense of reverence and I express my gratitude and sincere thanks to Mrs. Anusree S, Associate Professor, Department of Pharmacology, as guide, who with her dynamic approach boosted my moral, which helped me to a very great extent in the completion of this dissertation. Her assurances and advice had helped me in good stead. Her valuable suggestion, dynamic guidance, unwavering support, dedicated involvement, enthusiasm and inestimable encouragement, throughout the course of my stay, without whom, this work would have not reached the present status and which made the dissertation an educative and interesting experience. A moment of remembrance to the eternal repose of the soul of our founder, Late, Chairman Shri. Dr. K. Monikantan Nair. My heartfelt thanks to our Managing Trustee Smt. Geetha Monikantan and our chairman Dr. Gautham Krishna. M for providing support and creating aura for my research work. I am grateful to Prof. Dr. Prasobh G. R, Principal, Sree Krishna College of Pharmacy and Research Centre, for providing the necessary infra-structural facilities to carry out the research work successfully. My sincere thanks and gratitude to our vice principal, Dr. Kiran K J and Dr. Arun T S for his constant support and valuable information for doing the project work.
REFERENCES








Akshaya P.*, Anusree S., Rupitha N. S., Liya S. Saji, Shabin P., Jyothi B. N., Kiran K. J., Prasobh G. R., Network Pharmacology and Molecular Docking of Mucuna Pruriens in Treatment of Depression, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 9, 1623-1642 https://doi.org/10.5281/zenodo.17119251
 10.5281/zenodo.17119251
10.5281/zenodo.17119251