
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmaceutics, Shivajirao.S. Jondhale College of Pharmacy, Asangaon, District Thane, 421601, Maharashtra, India
Menopause is a natural physiological transition marked by permanent loss of ovarian function and reduced estrogen levels, leading to vasomotor symptoms, psychological disturbances, urogenital atrophy, metabolic changes, and increased risk of osteoporosis and cardiovascular diseases. Although hormone replacement therapy (HRT) is effective, its long-term use is associated with adverse effects such as hormone-dependent cancers and cardiovascular complications.Growing interest has focused on plant-derived phytoestrogens as safer alternatives. Clitoria ternatea L. (Blue Pea), rich in flavonoids, anthocyanins, and phytosterols, has shown promising phytoestrogenic and therapeutic potential. Preclinical evidence suggests mild estrogen receptor-? interaction along with antioxidant, anti-inflammatory, neuroprotective, and bone-protective effects. Experimental studies indicate its ability to alleviate menopausal symptoms and improve lipid, bone, cognitive, and emotional health with minimal toxicity. However, evidence is largely limited to in-vitro and animal studies, and clinical validation is insufficient. This review summarizes the phytochemistry, phytoestrogenic mechanisms, therapeutic evidence, formulation strategies, and safety profile of C. ternatea, highlighting its potential role in menopausal health management.
Menopause is a natural biological process that marks the permanent cessation of menstruation due to the gradual depletion of ovarian follicular activity. Clinically, menopause is diagnosed after twelve consecutive months of amenorrhea and typically occurs between 45 and 55 years of age, although the onset may vary due to genetic, nutritional, environmental, and lifestyle factors [1]. The menopausal transition is characterized by a progressive decline in ovarian estrogen and progesterone production, resulting in significant endocrine and metabolic alterations [2].The reduction in estrogen levels during menopause leads to a variety of short-term and long-term health consequences. Common symptoms include vasomotor disturbances such as hot flashes and night sweats, sleep disorders, mood swings, anxiety, depression, and impaired cognitive function [3,4]. In addition to these symptoms, chronic estrogen deficiency contributes to long-term conditions such as osteoporosis, cardiovascular diseases, dyslipidemia, insulin resistance, and neurodegenerative disorders, thereby significantly affecting the quality of life of postmenopausal women [5].Hormone replacement therapy (HRT) has traditionally been considered the most effective treatment for managing menopausal symptoms by restoring declining estrogen levels. Clinical evidence suggests that HRT reduces vasomotor symptoms, improves urogenital health, and prevents bone loss [6]. However, findings from large randomized clinical trials, particularly the Women’s Health Initiative, have demonstrated that prolonged use of HRT is associated with increased risks of breast and endometrial cancer, venous thromboembolism, stroke, and cardiovascular events [7,8]. Consequently, current guidelines recommend the use of HRT at the lowest effective dose for the shortest possible duration and only in selected patients [9].
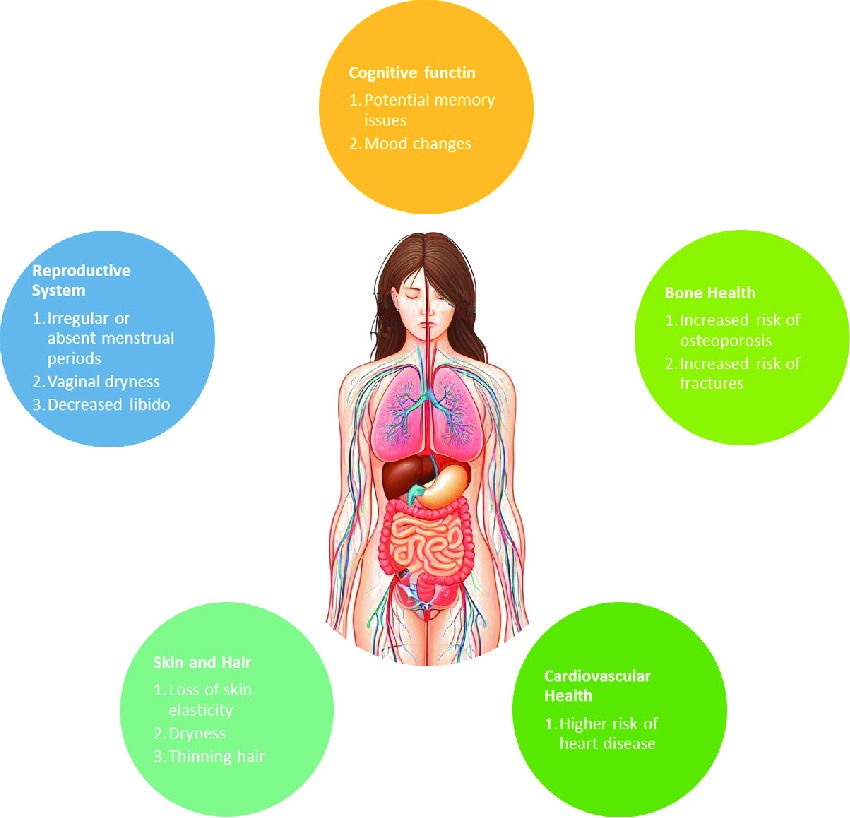
Fig 1: Physical and Cognitive Effects of Menopause
Due to these limitations, there has been increasing interest in alternative therapeutic strategies derived from natural sources. Among these, phytoestrogens have gained considerable attention as safer alternatives to synthetic hormones. Phytoestrogens are plant-derived compounds that share structural similarity with 17β-estradiol and exert estrogen-like effects by binding to estrogen receptors, particularly estrogen receptor-β, thereby acting as selective estrogen receptor modulators [10,11].
Several phytoestrogen-rich plants such as Glycine max (soybean) and Trifolium pratense (red clover) have been studied for menopausal symptom management; however, variability in efficacy and safety concerns have encouraged the exploration of alternative medicinal plants [12]. In this context, Clitoria ternatea L. (family Fabaceae), commonly known as blue pea or butterfly pea, has emerged as a promising medicinal plant. Traditionally used in Ayurvedic and Unani systems of medicine, Clitoria ternatea has been reported to possess memory-enhancing, anxiolytic, antidepressant, and female reproductive health-supporting properties [13]. Phytochemical studies reveal that the plant is rich in flavonoids, anthocyanins (ternatins), triterpenoids, and phytosterols, many of which exhibit antioxidant, anti-inflammatory, neuroprotective, and estrogen-modulating activities [14,15]. Preclinical evidence suggests that these bioactive constituents may help alleviate menopausal symptoms, improve bone and lipid metabolism, and support cognitive and emotional well-being [16]. Therefore, the present review aims to critically evaluate the phytoestrogenic and therapeutic potential of Clitoria ternatea in menopausal management, with emphasis on its phytochemical profile, mechanisms of action, therapeutic evidence, formulation approaches, safety considerations, and future research prospects

Fig 1.1: Hormonal changes across the menopausal transition showing the decline in estrogen and progesterone with a compensatory rise in FSH and LH.
1.1 Pathophysiology of Menopause
Menopause is primarily caused by the progressive depletion of ovarian follicles, which leads to a decline in ovarian steroidogenesis and eventual cessation of ovulation. As follicular reserve diminishes, the ovaries become less responsive to gonadotropins, resulting in a significant reduction in circulating estrogen and inhibin levels. This hormonal decline is a fundamental biological event underlying the menopausal transition [17].
The reduction in inhibin and estrogen disrupts the normal negative feedback regulation of the hypothalamic–pituitary–ovarian axis. As a consequence, circulating levels of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) increase markedly and serve as important biochemical indicators of menopause [18].
Estrogen plays a crucial role in maintaining skeletal integrity by regulating bone remodeling. During menopause, estrogen deficiency accelerates osteoclastic bone resorption and reduces osteoblastic bone formation, leading to decreased bone mineral density and an increased risk of osteoporosis and fragility fractures in postmenopausal women [19].
In addition to skeletal effects, estrogen deficiency contributes to cardiovascular alterations. Reduced estrogen levels adversely affect lipid metabolism, endothelial function, and vascular tone, thereby increasing low-density lipoprotein cholesterol levels and promoting atherosclerotic changes, which elevate cardiovascular risk after menopause [20]. Estrogen also exerts significant effects on the central nervous system by modulating neurotransmitters involved in mood, cognition, and thermoregulation. Declining estrogen levels are associated with alterations in serotonergic and dopaminergic pathways, contributing to mood disturbances, cognitive complaints, and vasomotor instability commonly observed during menopause [21]. Furthermore, estrogen maintains the structural and functional integrity of urogenital tissues. Estrogen deficiency leads to thinning of the vaginal epithelium, reduced collagen content, and decreased blood flow, resulting in urogenital atrophy and associated symptoms such as vaginal dryness, dyspareunia, and urinary discomfort [22].

Figure 1.1.1. Stages of menopause: Perimenopause, Menopause, and Post menopause.
1.2 Symptoms and Clinical Manifestations of Menopause
Menopause is accompanied by a constellation of physiological and psychological changes that arise primarily from declining estrogen and progesterone levels. These hormonal alterations affect multiple organ systems, leading to diverse symptoms that vary in intensity and duration among women [21]. The severity of symptoms depends on age, genetics, nutrition, and the rate of hormonal decline [22].
1. Vasomotor Symptoms
Vasomotor symptoms, including hot flashes and night sweats, are the hallmark manifestations of menopause. They result from hypothalamic thermoregulatory instability due to estrogen withdrawal, leading to episodic vasodilation and perspiration. Fluctuations in neuropeptides, serotonin, and norepinephrine also contribute to these temperature dysregulations [23].
2. Psychological and Cognitive Changes
Many women experience mood swings, anxiety, irritability, depression, and sleep disturbances. These are linked to altered neurotransmitter activity, particularly serotonin and dopamine pathways, which are modulated by estrogen. Chronic sleep loss and stress may further exacerbate cognitive decline, memory impairment, and reduced attention span [24,25].
3. Urogenital and Sexual Symptoms
Declining estrogen levels cause atrophy of the vaginal epithelium and urogenital tissues, resulting in vaginal dryness, burning, itching, and dyspareunia (painful intercourse). Additionally, urinary incontinence and recurrent infections are common due to atrophic urethral mucosa. Collectively, these are termed Genitourinary Syndrome of Menopause (GSM) [26,27].
4. Musculoskeletal and Skeletal Changes
Estrogen plays a key role in maintaining bone density. Its decline leads to an imbalance between bone formation and resorption, increasing the risk of osteopenia and osteoporosis. Women may experience joint pain, stiffness, and muscle weakness due to bone loss and changes in connective tissue [28,29].
5. Metabolic and Cardiovascular Effects Postmenopausal women exhibit increased central adiposity, insulin resistance, and adverse lipid profiles (elevated LDL, reduced HDL), predisposing them to metabolic syndrome and cardiovascular disease. Estrogen deficiency also impairs endothelial function, promoting atherosclerosis [30,31].
6. Dermatological and Hair Changes Reduced estrogen levels affect collagen and elastin synthesis, leading to dry, wrinkled skin, and thinning hair. These cosmetic changes are often early but underappreciated signs of estrogen deficiency [32].
Table 1. Systemic effects of menopause and underlying mechanisms
|
System Affected |
Common Symptoms |
Underlying Mechanism (as per review studies) |
|
Vasomotor |
Hot flashes, night sweats |
Estrogen withdrawal → altered hypothalamic thermoregulation and neuropeptide activity |
|
Psychological / Cognitive |
Mood swings, anxiety, depression, sleep loss |
Decline in estrogen-mediated serotonin & dopamine pathways |
|
Urogenital |
Vaginal dryness, dyspareunia, urinary urgency |
Atrophy of vaginal & urethral epithelium; loss of mucosal elasticity |
|
Skeletal / Musculoskeletal |
Joint pain, stiffness, osteoporosis |
Increased bone resorption due to decreased estrogen and calcium absorption |
|
Metabolic / Cardiovascular |
Central obesity, dyslipidemia, hypertension |
Estrogen deficiency alters lipid metabolism and endothelial function |
|
Dermatological |
Dry skin, hair loss, wrinkles |
Reduced collagen synthesis and dermal elasticity from low estrogen |
Effects of Estrogen Deficiency
Estrogen deficiency during menopause produces widespread systemic effects due to its central role in regulating multiple physiological systems [1,2]. In the nervous system, reduced estrogen disrupts hypothalamic thermoregulation and neurotransmitter balance, leading to hot flashes, mood swings, and sleep disturbances [17,21,24].
In the urogenital system, estrogen loss causes thinning of the vaginal epithelium and reduced lubrication, resulting in vaginal dryness and genitourinary syndrome of menopause (GSM), along with increased susceptibility to vaginal and urinary tract infections [26,27,95].
Estrogen deficiency adversely affects the cardiovascular system by altering lipid metabolism, promoting dyslipidemia and increasing the risk of atherosclerosis and cardiovascular disease [31,38,41].
In the skeletal system, declining estrogen accelerates bone resorption and reduces bone mineral density, leading to osteoporosis and increased fracture risk [18,28,29].
Additionally, estrogen plays an important role in maintaining skin and hair integrity; its deficiency results in skin dryness, wrinkles, loss of elasticity, and thinning of hair [32].
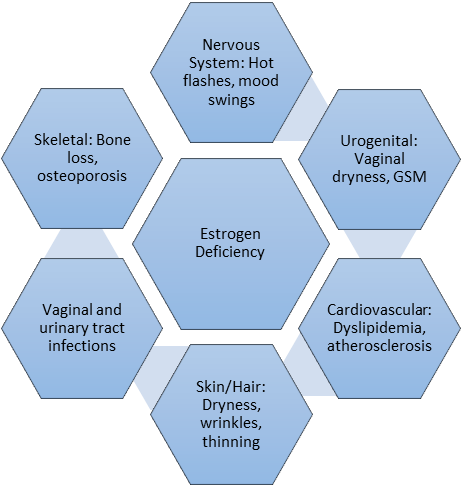
Fig1.2.1: Systemic manifestations of estrogen decline during menopause.
1.3 Global Impact of Menopause
Menopause represents a major physiological milestone in a woman’s life, affecting over 1 billion women globally, with approximately 47 million new entrants into menopause each year [33]. The average age of onset ranges between 45 and 55 years, though this varies with ethnicity, nutrition, and environmental exposure [34]. According to the World Health Organization (WHO), by 2030, over 1.2 billion women worldwide will be postmenopausal, and nearly 75% will experience moderate to severe symptoms [35]. The menopausal transition is associated not only with physiological disturbances but also with significant social and psychological burdens. Studies have reported increased incidences of anxiety, depression, and reduced quality of life scores among postmenopausal women, particularly in developing countries where awareness and access to medical support remain limited [36]. Socioeconomic disparities, lack of healthcare infrastructure, and cultural stigma often prevent timely diagnosis and management of menopausal disorders [37].
From a public health perspective, menopause has become an emerging non-communicable health challenge due to its association with osteoporosis, cardiovascular disease, metabolic syndrome, and cognitive decline. These chronic conditions contribute substantially to morbidity, mortality, and healthcare costs worldwide [38]. Thus, understanding the global epidemiology and societal impact of menopause is essential for developing region-specific management strategies that combine lifestyle interventions, pharmacological treatments, and safe complementary therapies.
1.4 Risk Factors and Conventional Management Approaches
The onset, severity, and duration of menopausal symptoms vary widely among women, influenced by several genetic, environmental, and lifestyle-related factors. Common risk factors include early ovarian failure, smoking, malnutrition, sedentary lifestyle, chronic stress, and surgical menopause following oophorectomy [39]. Additionally, women with a family history of premature menopause, autoimmune disorders, or exposure to endocrinedisrupting chemicals exhibit a higher risk of early menopausaltransition [40].
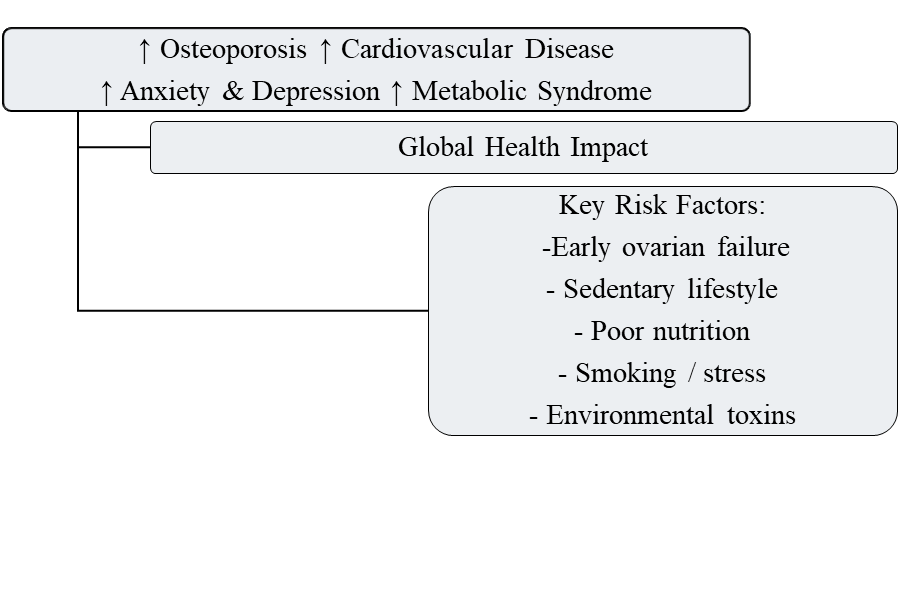
Fig1.3: Global prevalence and risk factors of menopause
1.5 Hormone Replacement Therapy (HRT)
For decades, Hormone Replacement Therapy (HRT) has remained the gold standard for managing menopausal symptoms by replenishing declining estrogen and progesterone levels [41]. It effectively alleviates vasomotor instability, urogenital atrophy, and osteoporosis risk. However, long-term use of HRT is associated with serious adverse effects such as breast and endometrial cancer, venous thromboembolism, and stroke, as evidenced by the Women’s Health Initiative (WHI) trial [42]. Consequently, HRT is now prescribed at the lowest effective dose for the shortest possible duration, often limited to severe cases under clinical supervision.
Non-Hormonal and Lifestyle Interventions
To reduce dependence on HRT, non-hormonal therapies—including selective serotonin reuptake inhibitors (SSRIs), gabapentin, and clonidine—are used for vasomotor symptom relief. Lifestyle modifications such as regular exercise, balanced diet, smoking cessation, and stress management have demonstrated beneficial effects on overall well-being [43]. Nutritional supplements containing calcium, vitamin D, and omega-3 fatty acids also help maintain bone and cardiovascular health during menopause.
Need for Phytoestrogen-Based Alternatives
Given the limitations and risks of long-term HRT, there has been a global shift toward natural, plant-derived alternatives that provide estrogenic benefits without significant side effects. Phytoestrogens, found abundantly in legumes, soy, red clover, and Clitoria ternatea, mimic endogenous estrogen activity by selectively binding to estrogen receptor β (ER-β), which regulates bone density, cognitive function, and cardiovascular protection [44].
Recent research emphasizes that phytoestrogen-rich herbal extracts exhibit multifunctional actions—antioxidant, anti-inflammatory, and hormone-modulating properties—offering safer and more holistic management of menopausal symptoms [45]. Moreover, the growing preference for herbal formulations has encouraged scientific exploration into novel delivery systems such as topical gels, oral capsules, and vaginal suppositories, which enhance bioavailability and localized action while minimizing systemic effects.
This growing evidence base provides the rationale for exploring the phytoestrogenic and therapeutic potential of Clitoria ternatea as a promising botanical intervention for menopause.
1.6 Clitoria ternatea
Botanical and Phytochemical Profile of Clitoria ternatea L. (Blue Pea)Clitoria ternatea L. (Fabaceae), commonly known as Blue Pea, Butterfly Pea, or Aparajita, is a perennial climber native to tropical Asia and widely cultivated across India, Thailand, and Africa [46]. Traditionally, the plant has been used in Ayurvedic, Thai, and Chinese medicine for memory enhancement, stress relief, wound healing, and female reproductive disorders [47].

Fig1.6: Representative image of Clitoria ternatea L. (Blue Pea).
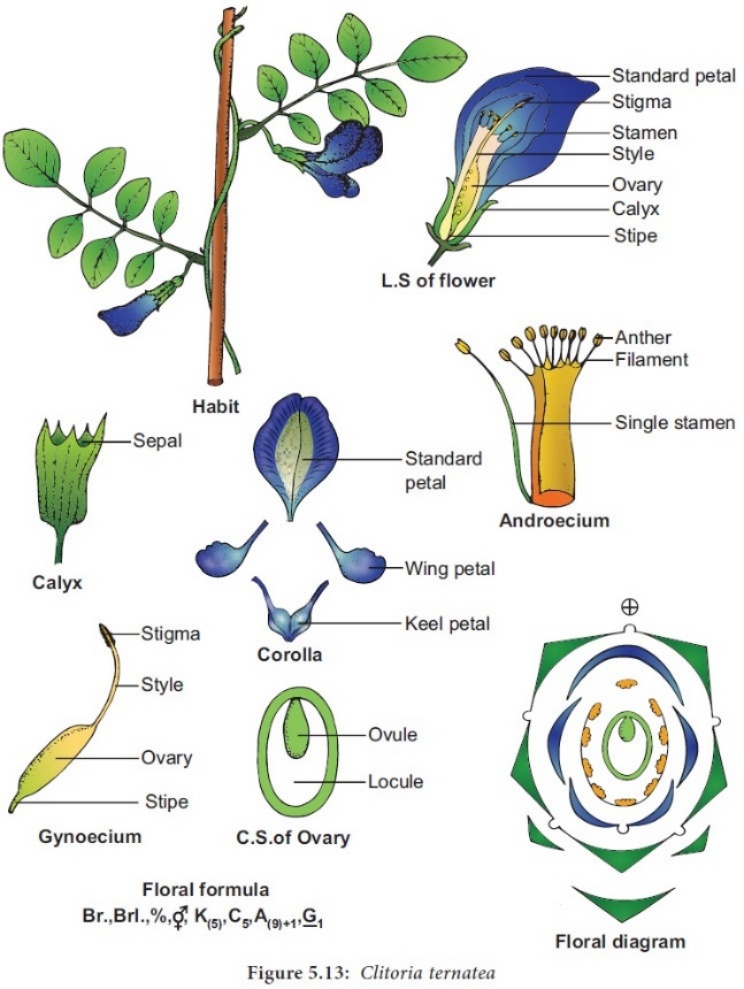
Fig 1.6.1: Botanical illustration of Clitoria ternatea L. showing habit, floral parts (calyx, corolla, androecium, gynoecium), floral diagram, and floral formula.
Table 2. Scientific classification of Clitoria ternatea L. (family Fabaceae), summarizing its botanical hierarchy from kingdom to species.
|
Scientific Classification |
|
Kingdom Plantae Divisions Spermatophyte Subdivision Angiospermae Class Fudicots Subclass Rosids Order Fabales Family Fabaceae/Leguminosae Subfamily Faboideae/Papilionoideae Tribe Phaseoleae Subtribe Clitoriinae Genus Clitoria Species Clitoria ternatea L |
1.Botanical Characteristics plant is a twining herb bearing bright blue or white flowers with a characteristic butterfly shape.
The roots are cylindrical and branched, while the leaves are pinnate with 5–7 leaflets.
Phytochemically, every part of the plant—roots, flowers, leaves, and seeds—contains bioactive constituents that contribute to its diverse pharmacological activities [46, 48].
2. Phytochemical Constituents
Comprehensive phytochemical analyses reveal a wide range of secondary metabolites, particularly flavonoids, anthocyanins, triterpenoids, alkaloids, steroids, and phenolic acids [49].
Dried flower powder
↓
Solvent extraction (Ethanol / Methanol / Hydroalcoholic)
↓
Filtration and concentration under reduced pressure
↓
Crude extract
↓
Phytochemical screening (Flavonoids, Anthocyanins, Sterols)
↓
HPLC / LC–MS profiling for marker compounds
↓
Standardized extract for formulation development
Fig1.6.2: Extraction and Standardization Process
3. Pharmacological Relevance
Multiple review studies have reported that C. ternatea possesses antioxidant, anti-inflammatory, nootropic, anxiolytic, antidiabetic, and lipid-modulating properties, all of which align with postmenopausal therapeutic needs [47, 55].Its flavonoid and anthocyanin fractions can mimic estrogenic signaling through selective binding to estrogen receptor-β, providing mild hormonal support without the carcinogenic risks of synthetic estrogens [56].
1.7 Phytoestrogenic Mechanisms of Clitoria ternatea in Menopause
Menopause is characterized by a natural decline in estrogen production, resulting in widespread physiological alterations across various organ systems. Phytoestrogens—plant-derived compounds structurally similar to 17β-estradiol—can mimic or modulate estrogenic activity by binding to estrogen receptors (ERs), particularly estrogen receptor-β (ERβ), which is associated with neuroprotective and bone-sparing effects [57]. Clitoria ternatea (Blue Pea) contains diverse bioactive flavonoids, anthocyanins, and phytosterols that demonstrate such estrogenic and antioxidant actions without the adverse effects of synthetic hormone therapy [58,59].
1.7.1 Phytoestrogenic Compounds in C. ternatea
Several phytochemicals in C. ternatea, such as quercetin, kaempferol, apigenin, and β-sitosterol, possess partial structural similarity to 17β-estradiol, allowing them to bind weakly to ERβ [60]. These compounds act as selective estrogen receptor modulators (SERMs) — stimulating estrogenic activity in estrogen-deficient states while preventing overstimulation when estrogen is adequate [61]. This property makes them safer than traditional hormone replacement therapies (HRT), which can overstimulate ERα and elevate cancer risks [62].
1.7.2 Interaction with Estrogen Receptors
Upon receptor binding, these phytoestrogens initiate transcriptional pathways that upregulate genes responsible for bone remodeling, lipid regulation, and antioxidant enzyme synthesis [63]. Experimental studies indicate that C. ternatea extracts enhance estrogenic biomarkers such as alkaline phosphatase (ALP) and modulate uterine tissue histology, without causing hyperplasia or carcinogenic effects [64].In menopausal conditions, activation of ERβ in bone and neuronal tissue contributes to bone strength and emotional stability, while ERα suppression in reproductive tissue minimizes unwanted side effects [65].
1.7.3. Antioxidant and Anti-inflammatory Mechanisms
Oxidative stress and inflammation exacerbate menopausal symptoms and metabolic disturbances. The anthocyanins and flavonoids in C. ternatea (notably ternatins and quercetin derivatives) scavenge reactive oxygen species (ROS), inhibit lipid peroxidation, and suppress inflammatory cytokines such as IL-6 and TNF-α [66]. This dual antioxidant–anti-inflammatory effect supports hormonal equilibrium, cardiovascular function, and neuroprotection during menopause [67,68].
1.7.4. Neuroprotective and Anxiolytic Effects
Estrogen is crucial for cognitive and emotional stability. Its withdrawal during menopause leads to mood fluctuations, anxiety, and cognitive decline. Studies summarized in review articles show that C. ternatea extract elevates brain acetylcholine levels and enhances antioxidant enzyme activity (SOD, CAT, GPx), reducing anxiety-like behavior and improving memory in animal models [69]. Such findings highlight its potential as a natural neuroprotective phytoestrogen [70].
1.8 Therapeutic and Experimental Evidence of Clitoria ternatea in Menopausal Management
Clitoria ternatea has gained increasing attention for its diverse pharmacological activities that align with menopausal health needs. Multiple review and experimental studies confirm that its bioactive compounds exert beneficial effects on hormonal balance, bone density, cognitive function, and cardiovascular health through mild estrogenic, antioxidant, and neuroprotective mechanisms [73–75].
1.8.1 Hormonal Modulation
Preclinical and review studies reveal that extracts of C. ternatea (flower and root) mildly elevate circulating estrogen levels and improve uterine tissue histology without inducing hyperplasia or carcinogenic effects [76].The flavonoid fraction—particularly quercetin and kaempferol—acts through selective activation of estrogen receptor-β (ERβ), restoring homeostasis in the hypothalamic–pituitary–ovarian (HPO) axis and modulating luteinizing hormone (LH) and follicle-stimulating hormone (FSH) secretion [77].This balanced hormonal activity mimics natural estrogen signaling and may help alleviate vasomotor symptoms and mood disturbances in postmenopausal women [78].
1.8.2. Bone-Protective Effects
Osteoporosis and bone fragility are major health issues after menopause due to estrogen deficiency. Studies summarized in review papers demonstrate that phytochemicals like β-sitosterol and kaempferol stimulate osteoblastic proliferation and inhibit osteoclastic resorption [79].In ovariectomized animal models, C. ternatea extracts significantly increased bone mineral density (BMD), calcium retention, and alkaline phosphatase (ALP) levels compared to controls [80].Such bone-preserving activity indicates a potential role for C. ternatea as a natural, plant-derived bone-supportive therapy [81].
1.8.3. Neuroprotective and Mood-Enhancing Properties
Estrogen deficiency contributes to depression, anxiety, and cognitive decline during menopause. Studies in animal and clinical models indicate that C. ternatea extract enhances acetylcholine concentration, improves antioxidant enzyme levels, and reduces lipid peroxidation in the hippocampus [82].Its anthocyanin-rich fraction (ternatins A–G) also prevents neuronal apoptosis and improves memory and learning behavior, aligning with its traditional Ayurvedic use as a “medhya rasayana” or brain tonic [83].Therefore, C. ternatea represents a promising neuroprotective phytoestrogenic agent for menopausal mood and cognition support [84].
1.8.4. Cardiometabolic and Antioxidant Activity
Postmenopausal women are at greater risk for metabolic syndrome and cardiovascular diseases due to estrogen decline. Studies report that C. ternatea extract improves lipid profiles by increasing HDL and reducing LDL and triglycerides [85].Its antidiabetic and anti-inflammatory effects—mediated by modulation of TNF-α and IL-6 expression—further contribute to cardiovascular protection [86].Such effects suggest that C.ternatea may provide holistic metabolic support in menopause while reducing oxidative damage [87].
1.8.5. Comparative Insights
Compared with other phytoestrogenic herbs such as Pueraria mirifica and Glycine max (soy), Clitoria ternatea exhibits a balanced estrogenic profile characterized by moderate receptor affinity, potent antioxidant action, and minimal proliferative risk [88].Its dual action—both endocrine and antioxidant—makes it particularly suitable as a complementary or alternative approach to synthetic hormone replacement [89].
1.9 Formulation Aspect – Suppository Development Using Clitoria ternatea
Recent advances in herbal pharmaceutics have demonstrated that vaginal suppositories can serve as an efficient, patient-friendly route for localized phytoestrogen delivery during menopause. This dosage form provides targeted estrogenic activity directly at the urogenital tissues, avoiding first-pass hepatic metabolism and minimizing systemic side effects commonly observed with oral hormone therapy [92,93].
The Clitoria ternatea-based suppository utilizes standardized flower extracts rich in flavonoids, anthocyanins, and β-sitosterol, which exhibit estrogen-modulating, antioxidant, and mucosal-repair properties [94]. These compounds improve epithelial regeneration and restore vaginal elasticity, addressing dryness and discomfort associated with Genitourinary Syndrome of Menopause (GSM) [95].
Choice of Base and Composition Formulation studies suggest that cocoa butter and polyethylene glycol (PEG) are ideal bases for phytoestrogenic suppositories due to their smooth melting behavior and biocompatibility [96].A standardized extract of C. ternatea can be incorporated (5–10% w/w) in a PEG–cocoa butter blend, allowing controlled drug release and enhanced mucosal adherence [97]. The use of hydroalcoholic extracts ensures maximum solubility of flavonoids and anthocyanins, which are responsible for the estrogenic and anti-inflammatory effects [98].
Pharmacotechnical Considerations
For effective clinical performance, the suppository must maintain physical stability, uniform drug distribution, and a melting point of 34–37°C, ensuring it melts easily at body temperature [99].Quality control parameters such as hardness, weight variation, and disintegration time should be optimized as per pharmacopeial guidelines [100].Furthermore, in-vitro dissolution studies can be performed using simulated vaginal fluid to evaluate sustained phytoestrogen release and mucosal penetration efficiency [101].
1.9.1 Mechanistic Overview
When inserted vaginally, the suppository melts and releases the phytoestrogen-rich extract directly into the mucosal tissue, where compounds such as kaempferol, quercetin, and β-sitosterol bind to ERβ receptors in epithelial and stromal cells [102].This localized mechanism enhances lubrication, reduces oxidative damage, and promotes cellular regeneration—providing symptomatic relief without systemic hormone exposure [103].
Dried Flower Powder
↓
Solvent Extraction (Ethanol / Methanol / Hydroalcoholic)
↓
Filtration and Concentration under Reduced Pressure
↓
Crude Extract
↓
Phytochemical Screening (Flavonoids, Anthocyanins, Sterols)
↓
HPLC / LC–MS Profiling for Marker Compounds
↓
Standardized Extract for Formulation Development
↓
Incorporation into PEG / Cocoa Butter Base → Vaginal Suppository
Fig1.9.1: Extraction and formulation steps of Clitoria ternatea for phytoestrogenic suppository.
In traditional and modern therapeutic contexts, Clitoria ternatea is employed in several dosage forms to manage menopausal discomforts. Clinical and preclinical reviews indicate that its flower and root extracts are the main pharmacologically active parts used for therapy [106,107].
Oral Herbal Preparations:
Standardized ethanolic or hydroalcoholic extracts (200–400 mg/day) have been formulated into capsules or teas to provide systemic antioxidant and mild estrogenic support. Regular intake helps regulate mood, hot flashes, and bone metabolism [108].
Topical and Vaginal Formulations:
Incorporation of C. ternatea extract into vaginal gels, creams, and suppositories enables localized estrogen receptor activation in mucosal tissues, improving lubrication, reducing dryness, and relieving genitourinary syndrome of menopause (GSM) [109].
Combination Phytoformulas:
Herbal products combining C. ternatea with other phytoestrogen sources (e.g., Glycine max, Trifolium pratense) show synergistic action—balancing estrogen levels while providing antioxidant and anti-stress benefits [110].
Experimental Use in Dietary Supplements:
Blue-pea flower extract powders are being investigated as nutraceutical additives insmoothies and health drinks for their anthocyanin-rich, hormone-modulating potential [111].
Collectively, these administration routes demonstrate that Clitoria ternatea can be utilized as both a systemic and localized phytoestrogenic therapy to alleviate menopausal symptoms, either alone or as part of combination herbal formulations.
Table 3. Comparative Overview of PCOS, PCOD and Menopause
|
Parameter |
PCOS (Polycystic Ovary Syndrome) |
PCOD (Polycystic Ovarian Disease) |
Menopause |
|
Life stage |
Reproductive age (adolescence–30s) |
Reproductive age |
Post-reproductive age |
|
Nature of condition |
Endocrine and metabolic disorder |
Ovarian functional disorder |
Natural physiological transition |
|
Primary cause |
Hormonal dysregulation with insulin resistance |
Ovarian hormonal imbalance |
Ovarian follicular depletion |
|
Estrogen status |
Fluctuating or excess estrogen |
Irregular estrogen secretion |
Marked estrogen deficiency |
|
Androgen levels |
Significantly elevated |
Moderately elevated |
Usually reduced |
|
Ovulatory status |
Chronic anovulation |
Irregular ovulation |
Ovulation permanently absent |
|
Menstrual pattern |
Oligomenorrhea / amenorrhea |
Irregular cycles |
Permanent cessation of menstruation |
|
Metabolic involvement |
High insulin resistance, obesity |
Mild–moderate metabolic changes |
Dyslipidemia, weight gain |
|
Common symptoms |
Hirsutism, acne, infertility |
Weight gain, cycle irregularity |
Hot flashes, mood changes |
|
Psychological impact |
Anxiety, depression |
Mood instability |
Depression, irritability |
|
Long-term risks |
Type-2 diabetes, infertility |
Infertility |
Osteoporosis, cardiovascular disease |
|
Oxidative stress |
Elevated |
Moderately elevated |
Elevated |
|
Therapeutic focus |
Hormonal regulation & metabolic control |
Cycle regulation & ovarian support |
Symptom relief & bone protection |
PCOS is a common hormonal disorder affecting women of reproductive age, characterized by irregular menstrual cycles, hyperandrogenism, ovarian cyst formation, and possible metabolic and fertility complications.
PCOD is a relatively milder condition mainly involving ovarian cysts and menstrual irregularities, often influenced by lifestyle factors, and usually associated with fewer systemic effects than PCOS.
Menopause is a natural physiological stage marking the permanent cessation of menstruation due to a decline in estrogen and progesterone levels, typically occurring between 45–55 years of age, and is associated with vasomotor, psychological, and skeletal changes.
Table 4. Currently Available Pharmacological Therapies for Menopausal Management
|
Drug Category |
Generic Name |
Brand Name |
Indication |
Limitations / Remarks |
|
Estrogen therapy |
Conjugated estrogens |
Premarin® |
Hot flashes, vaginal atrophy, osteoporosis prevention |
Risk of breast cancer, thromboembolism on long-term use |
|
PPEstrogen therapy |
Estradiol valerate |
Progynova® |
Vasomotor symptoms, bone protection |
Not suitable in estrogen-dependent cancers |
|
Transdermal estrogen |
Estradiol patch |
Climara®, Estraderm® |
Menopausal symptoms, bone loss |
Skin irritation, costly |
|
Combined HRT |
Estradiol + Norethisterone acetate |
Activelle® |
Vasomotor symptoms with uterine protection |
Increased cardiovascular risk |
|
Combined HRT |
Conjugated estrogens + Medroxyprogesterone |
Prempro® |
Severe menopausal symptoms |
Long-term safety concerns |
|
SERMs |
Raloxifene |
Evista® |
Osteoporosis prevention |
Does not relieve hot flashes |
|
SERMs |
Bazedoxifene + Conjugated estrogens |
Duavive® |
Bone protection, menopausal symptoms |
Limited availability |
|
Antidepressants |
Paroxetine |
Brisdelle® |
Hot flashes |
Nausea, sleep disturbance |
|
Antiepileptic |
Gabapentin |
Neurontin® |
Vasomotor symptoms |
Dizziness, fatigue |
|
Antihypertensive |
Clonidine |
Catapres® |
Hot flashes |
Hypotension, dry mouth |
|
Bisphosphonates |
Alendronate |
Fosamax® |
Osteoporosis |
GI irritation |
|
Bisphosphonates |
Risedronate |
Actonel® |
Bone loss |
Long-term jaw osteonecrosis risk |
|
RANKL inhibitor |
Denosumab |
Prolia® |
Severe osteoporosis |
Expensive, injection |
|
Vaginal estrogen |
Estriol cream |
Ovestin® |
Vaginal dryness |
Local irritation |
|
Vaginal estrogen |
Estradiol tablets |
Vagifem® |
Genitourinary syndrome |
Requires long-term use |
|
Herbal therapy |
Soy isoflavones |
Menopace®, Isoflav® |
Mild menopausal symptoms |
Variable efficacy |
|
Herbal therapy |
Black cohosh |
Remifemin® |
Hot flashes |
Limited clinical evidence |
CONCLUSION
Menopause is a critical stage in a woman’s life that significantly affects physical health, emotional well-being, and quality of life. Although hormone replacement therapy remains effective for symptom relief, its long-term risks limit its widespread and prolonged use. Therefore, the exploration of safer, natural alternatives has become increasingly important in modern menopausal management.
Clitoria ternatea emerges as a promising phytotherapeutic agent due to its rich phytochemical composition and broad pharmacological profile. The presence of flavonoids, anthocyanins, and phytosterols enables this plant to exert mild estrogen-like effects along with antioxidant, anti-inflammatory, and neuroprotective actions. These combined properties make it suitable for addressing multiple menopausal complications such as bone loss, mood disturbances, cognitive decline, and urogenital discomfort.
Preclinical evidence indicates that Clitoria ternatea supports hormonal balance and improves menopausal health with minimal toxicity. Furthermore, novel drug delivery systems such as vaginal suppositories enhance its therapeutic applicability by providing localized action and reducing systemic side effects. However, despite promising findings, clinical validation in postmenopausal women is still limited.
In conclusion, Clitoria ternatea holds significant potential as a safe and effective phytoestrogen-based alternative or adjunct to conventional hormone therapy. Future research should focus on clinical trials, standardized formulations, and regulatory evaluation to establish its role in evidence-based menopausal care.
FUTURE PROSPECTS
Menopause-related health challenges and the safety concerns associated with long-term hormone replacement therapy have increased global interest in plant-derived phytoestrogens as alternative therapeutic options. In this context, Clitoria ternatea L. demonstrates strong future potential due to its estrogen-modulating, antioxidant, neuroprotective, and bone-supportive properties. The presence of bioactive flavonoids, anthocyanins, and phytosterols enables this plant to provide mild estrogenic support while minimizing adverse effects commonly associated with synthetic hormones [118,119].
Additionally, Clitoria ternatea holds considerable potential for incorporation into nutraceuticals and functional food products designed for long-term menopausal support. Standardized herbal supplements containing C. ternatea extracts may help improve antioxidant defense, emotional well-being, bone health, and overall quality of life in postmenopausal women. Combination formulations with other phytoestrogen-rich plants may further enhance therapeutic outcomes through synergistic mechanisms [119,120].
One of the most promising future directions involves the development of advanced and targeted delivery systems for Clitoria ternatea extracts. Novel formulation approaches such as vaginal suppositories, topical gels, and localized delivery systems offer site-specific therapeutic action, particularly for managing genitourinary syndrome of menopause. These formulations may enhance bioavailability at the target site while reducing systemic exposure and improving patient compliance [122].
Future research focusing on molecular mechanisms, estrogen receptor selectivity, and formulation optimization will help strengthen its clinical applicability. With proper clinical validation, standardization, and regulatory approval, Clitoria ternatea has the potential to emerge as a safe and effective phytoestrogen-based intervention for menopausal health management.
LIMITATIONS:
Despite its promising therapeutic potential, several limitations currently restrict the widespread clinical use of Clitoria ternatea in menopausal management. The most significant limitation is the lack of well-designed human clinical trials. Most of the existing evidence supporting its phytoestrogenic and therapeutic effects is derived from in-vitro and animal studies, which may not accurately represent clinical efficacy and safety in postmenopausal women [123].
Another major limitation is the absence of standardized extraction and formulation protocols. Variations in plant source, cultivation practices, harvesting conditions, and extraction methods can lead to significant differences in phytochemical composition, affecting reproducibility and therapeutic consistency. This lack of standardization poses challenges for dose optimization, quality control, and regulatory acceptance of Clitoria ternatea-based formulations [121,122].
Furthermore, comprehensive pharmacokinetic data regarding absorption, bioavailability, metabolism, and elimination of Clitoria ternatea bioactive compounds remain limited. Information related to long-term safety, herb–drug interactions, and estrogen receptor modulation in vulnerable populations is also insufficient, particularly in women with pre-existing hormone-sensitive conditions [123].
Therefore, large-scale clinical trials, long-term safety evaluations, and regulatory validation are essential future requirements before Clitoria ternatea can be recommended for routine clinical use in menopausal therapy.
REFERENCES






Shweta Pandey, Nikita Pagare, Manisha Nangude, Akshata Palve, Sujal Nikam, Harshal Nathe, Phytoestrogenic and Therapeutic Potential of Clitoria Ternatea (Blue Pea) In Menopause: A Comprehensive Review, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 2605-2625. https://doi.org/10.5281/zenodo.18668754
 10.5281/zenodo.18668754
10.5281/zenodo.18668754