
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
1 Chatrapati Shivaji Maharaj University, Panvel,
2 Associate professor, Victoria College of pharmacy
3 Associate professor, Narayana pharmacy college, Nellore 524003
4 New College of Pharmacy, Kolhapur
5 Assistant Professor, Rupesh Badhan Institute of Pharmacy, Pimpalner, Sakri, Dhule
6 Samarth Institute of Pharmacy, Belhe, Pune
Cancer immunotherapy has revolutionized modern cancer treatment by utilizing the host immune system to target and destroy cancer cells. Of the many cancer immunotherapy strategies, immune checkpoint inhibitors, including monoclonal antibodies against programmed cell death protein-1 (PD-1), programmed death ligand-1 (PD-L1), and cytotoxic T-lymphocyte-associated antigen-4 (CTLA-4), have shown tremendous clinical results in the treatment of cancer. Despite the impressive results, biologics have shown limitations, including their high production costs, poor oral bioavailability, inability to penetrate cancer tissues, and immune-related adverse effects.[1] Recently, small molecule inhibitors have emerged as promising agents in cancer immunotherapy, considering their favorable pharmacokinetic properties, oral bioavailability, and ability to penetrate cancer tissues, in addition to their cost-effectiveness. These agents have shown tremendous potential in modulating the immune response by inhibiting intracellular targets and interfering with protein-protein interactions, which play a role in cancer immune evasion.[2] This review article attempts to provide an in-depth overview of the design, development, and mechanism of action of small molecule inhibitors targeting cancer immunotherapy pathways, including the PD-1/PD-L1, CTLA-4, indoleamine 2,3-dioxygenase 1 (IDO1)-mediated tryptophan metabolism, and stimulator of interferon genes (STING) pathways. Special emphasis has been given to structure-based drug design, molecular docking, and recent advancements in medicinal chemistry, which have greatly contributed to the discovery of these promising agents. Additionally, the review attempts to highlight the challenges in cancer immunotherapy, including the achievement of specificity in targeting protein-protein interactions, overcoming off-target effects, and drug resistance, in addition to emerging strategies, including multi-target directed ligands and artificial intelligence in drug discovery.[3]
1. Mechanisms of Action of Small Molecule Inhibitors in Cancer Immunotherapy
Small-molecule inhibitors exert therapeutic effects in cancer immunotherapy by modulating key immune regulatory pathways and overcoming tumor-induced immune suppression. Unlike monoclonal antibodies, these molecules can penetrate intracellular targets and disrupt critical signaling cascades involved in immune evasion. The major mechanisms are described below[5]
2. Inhibition of Immune Checkpoint Pathways
Small molecule inhibitors can block immune checkpoint interactions such as PD-1/PD-L1 and CTLA-4, which are commonly exploited by tumor cells to evade immune surveillance.[6] These inhibitors disrupt protein–protein interactions (PPIs) between receptors and ligands.
Blocking PD-1/PD-L1 restores T-cell activation and enhances cytotoxic immune response.
CTLA-4 inhibition promotes T-cell priming and proliferation.
Result: Reactivation of anti-tumor immune response[7]
3. Modulation of Enzymatic Immunosuppressive Pathways
Certain tumors overexpress enzymes that suppress immune function.
Example: Indoleamine 2,3-dioxygenase 1 (IDO1)
IDO1 degrades tryptophan → leads to T-cell suppression
Small molecule inhibitors block IDO1 activity → restore immune function
Result: Reduction of tumor-induced immune tolerance[8]
4. Activation of Innate Immune Signaling (STING Pathway)
Small molecules can act as agonists of innate immune pathways.
STING activation → production of type I interferons
Enhances dendritic cell activation and T-cell priming
Result: Enhanced immune recognition of tumor cells[9]
5. Targeting Intracellular Signaling Pathways
Small molecules can penetrate cells and modulate intracellular pathways involved in immune regulation.
Inhibit signaling proteins like kinases (e.g., JAK/STAT pathway)
Regulate cytokine production and immune cell activity
Result: Improved immune-mediated tumor killing[10]
6. Disruption of Tumor Microenvironment (TME)
Tumor microenvironment plays a key role in immune suppression.
Small molecules reduce immunosuppressive cells (Tregs, MDSCs)
Improve infiltration of cytotoxic T-cells into tumor
Result: Conversion of “cold tumors” into “hot tumors”
7. Induction of Immunogenic Cell Death (ICD)
Some small molecules induce a type of cell death that stimulates immune response.
Release of tumor antigens
Activation of antigen-presenting cells
Result: Enhanced adaptive immune response
8. Multi-Target Modulation
Many small molecules are designed to act on multiple targets simultaneously.
Simultaneous inhibition of checkpoints and enzymes
Synergistic immune activation
Result: Improved therapeutic efficacy
Cancer remains a leading cause of mortality worldwide. Traditional therapies such as chemotherapy and radiation have limitations including toxicity and resistance. Immunotherapy, particularly immune checkpoint blockade, has transformed cancer treatment. However, monoclonal antibodies face challenges such as high cost and poor tissue penetration. Small molecule inhibitors provide a complementary strategy with advantages in pharmacokinetics and chemical modification flexibility.[11]
Clinical Applications of Small Molecule Inhibitors as Adjuvants in Cancer Immunotherapy
Small molecule inhibitors are widely utilized as adjuvants in cancer immunotherapy to enhance therapeutic efficacy, overcome resistance, and improve immune-mediated tumor destruction. Unlike monoclonal antibodies, these agents can penetrate intracellular targets and modulate key signaling pathways involved in immune regulation and tumor progression. Their integration with immunotherapeutic approaches has shown promising clinical outcomes across multiple cancer types.[12]
Clinical Applications (with Examples)
1. Combination with Immune Checkpoint Inhibitors
Small molecule inhibitors are used alongside checkpoint inhibitors to enhance T-cell activation and overcome immune suppression.
Example: IDO1 inhibitors (Epacadostat) + anti-PD-1 therapy
Application: Melanoma, lung cancer
Outcome: Improves T-cell proliferation and reduces tumor immune evasion
2. Use of Kinase Inhibitors as Immunomodulators
Kinase inhibitors modulate signaling pathways that regulate immune responses and tumor growth.
Example: Imatinib (BCR-ABL inhibitor), Sorafenib (RAF/VEGFR inhibitor)
Application: Leukemia, hepatocellular carcinoma
Outcome: Enhances immune response and reduces immunosuppressive cells
3. Modulation of Tumor Microenvironment (TME)
Small molecules improve immune cell infiltration by altering tumor conditions.
Example: VEGF inhibitors (Sunitinib)
Application: Renal cell carcinoma
Outcome: Converts “cold tumors” into “hot tumors” (more responsive to immunotherapy) [13]
4. Activation of Innate Immunity (STING Agonists)
Small molecules activate innate immune pathways to boost anti-tumor immunity.
Example: STING agonists (e.g., ADU-S100 – under clinical trials)
Application: Solid tumors
Outcome: Increased interferon production and T-cell priming
5. Combination with Adoptive Cell Therapy (CAR-T Cells)
Small molecule inhibitors enhance the efficacy of engineered immune cells.
Example: Ibrutinib (BTK inhibitor) with CAR-T therapy
Application: B-cell malignancies
Outcome: Improves CAR-T cell expansion and persistence
6. Epigenetic Modulation
Small molecules regulate gene expression to enhance immune recognition of tumors.
Example: Vorinostat (HDAC inhibitor), Azacitidine (DNA methyltransferase inhibitor)
Application: Leukemia, lymphoma
Outcome: Increases tumor antigen expression and immune sensitivity
7. Targeting Metabolic Pathways
Tumor metabolism is targeted to restore immune cell function.
Example: IDO inhibitors (Epacadostat), Arginase inhibitors
Application: Advanced cancers
Outcome: Reverses immunosuppressive metabolic environment[14]
T-Cell Differentiation and Its Role in Cancer Immunotherapy (Theory)
T lymphocytes play a central role in cancer immunotherapy by mediating immune responses against tumor cells. The development and functional specialization of T cells occur primarily in the thymus, where thymocytes undergo a series of differentiation stages before giving rise to distinct T-cell subsets with specialized roles in immune surveillance and tumor eradication.
Thymocyte development begins at the double-negative (DN) stages, particularly DN3 and DN4, where T-cell receptor (TCR) gene rearrangement occurs. Depending on the nature and strength of TCR signaling, thymocytes differentiate into two major lineages: conventional αβ T cells and unconventional innate-like T cells. Strong agonist signaling through the γδ TCR promotes the development of innate-like T cells, whereas signaling through the αβ TCR leads to the formation of conventional CD4? and CD8? T cells.[15]
Conventional T cells, especially CD8? cytotoxic T lymphocytes (CTLs), are critical for tumor cell elimination. These cells recognize tumor-associated antigens presented on major histocompatibility complex class I (MHC-I) molecules via their TCR. Upon activation, CTLs release cytotoxic molecules such as perforin and granzymes, which induce apoptosis in tumor cells. CD4? helper T cells further support this response by secreting cytokines that enhance CTL activation and proliferation.[16]
In contrast, unconventional or innate-like T cells—including γδ T cells, invariant natural killer T (iNKT) cells, mucosal-associated invariant T (MAIT) cells, and innate-like T cells (ILTCs)—exhibit rapid, antigen-independent or semi-specific responses. These cells bridge innate and adaptive immunity and contribute to early tumor recognition and immune activation.
A key regulatory factor in the differentiation and expansion of innate-like T cells is interleukin-15 (IL-15). Binding of IL-15 to its receptor (IL-15R) activates intracellular signaling pathways, particularly the STAT5 pathway, promoting survival, proliferation, and cytotoxic differentiation of effector T cells. These effector cells express markers such as NK1.1 and produce cytotoxic mediators, enhancing anti-tumor immunity.[17]
Within the tumor microenvironment, both conventional and innate-like T cells infiltrate tumor tissues and interact with tumor cells. However, tumor cells often evade immune destruction by upregulating immune checkpoint molecules such as programmed death-1 (PD-1) and its ligand PD-L1. Engagement of PD-1 inhibits T-cell activation and leads to functional exhaustion of cytotoxic T cells.
This immunosuppressive mechanism is a major target in cancer immunotherapy. Small molecule inhibitors and immune checkpoint blockers aim to disrupt these inhibitory pathways, restore T-cell function, and enhance tumor cell killing. Additionally, modulation of cytokine signaling and tumor microenvironment by small molecules further improves immune cell infiltration and activity.[18]
In summary, the differentiation of thymocytes into diverse T-cell subsets and their functional activation within the tumor microenvironment are critical determinants of effective anti-tumor immunity. Understanding these mechanisms provides a strong foundation for the development of novel small molecule-based immunotherapeutic strategies.[19]
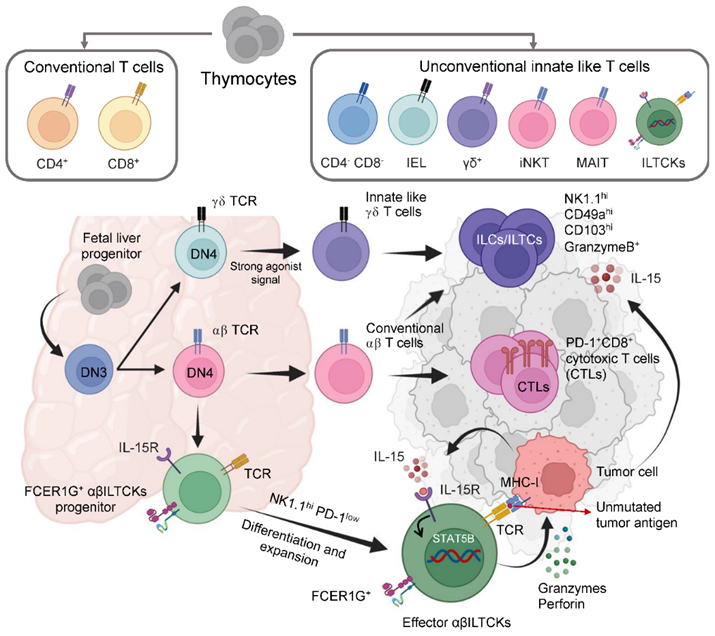
Fig 1. Mechanisms of T-Cell Mediated Anti-Tumor Immune Response
2. CANCER IMMUNOTHERAPY TARGETS
2.1 PD-1/PD-L1 Pathway
The Programmed Cell Death Protein-1 (PD-1) is an immune checkpoint receptor expressed on activated T-cells, while its ligand PD-L1 (Programmed Death-Ligand 1) is expressed on tumor cells and antigen-presenting cells. This pathway plays a crucial role in maintaining immune homeostasis and preventing autoimmunity by downregulating excessive immune responses.
Under normal physiological conditions, the binding of PD-L1 to PD-1 transmits an inhibitory signal to T-cells, leading to reduced proliferation, cytokine production, and cytotoxic activity. However, tumor cells exploit this mechanism by overexpressing PD-L1 on their surface. This interaction suppresses T-cell activity, allowing cancer cells to evade immune detection and destruction.[20]
To counteract this immune evasion, small molecule inhibitors and immune checkpoint inhibitors are developed to block the PD-1/PD-L1 interaction. By disrupting this pathway, these agents restore T-cell function, enhance immune responses, and promote the destruction of tumor cells. This approach has become a cornerstone in cancer immunotherapy, significantly improving outcomes in various malignancies such as melanoma and non-small cell lung cancer.[21]
2.2 CTLA-4 Pathway
Cytotoxic T-Lymphocyte-Associated Protein 4 (CTLA-4) is an important immune checkpoint receptor expressed on activated T-cells and regulatory T-cells (Tregs). It plays a critical role in controlling early stages of T-cell activation, primarily within lymphoid organs.
Under normal conditions, T-cell activation requires two signals: antigen recognition and co-stimulation via the binding of CD28 on T-cells to B7 molecules (CD80/CD86) on antigen-presenting cells. CTLA-4 competes with CD28 for binding to B7 molecules but has a much higher affinity. When CTLA-4 binds to B7, it delivers an inhibitory signal that suppresses T-cell activation, proliferation, and cytokine production, thereby maintaining immune tolerance and preventing autoimmunity.[22]
Tumors exploit this regulatory pathway by enhancing CTLA-4-mediated inhibition, leading to reduced T-cell activation and allowing cancer cells to escape immune surveillance. Blocking CTLA-4 using inhibitors or small molecules prevents this inhibitory signaling, thereby enhancing T-cell activation and proliferation. This results in a stronger immune response against tumor cells.
Thus, targeting the CTLA-4 pathway is a key strategy in cancer immunotherapy, as it boosts the body's immune system to recognize and eliminate cancer cells more effectively.
2.3 IDO1 Enzyme
Indoleamine 2,3-dioxygenase 1 (IDO1) is a key immunoregulatory enzyme involved in the metabolism of the essential amino acid tryptophan through the kynurenine pathway. It is expressed in various cells, including tumor cells and antigen-presenting cells within the tumor microenvironment.
Under physiological conditions, IDO1 plays a role in maintaining immune tolerance by regulating T-cell activity. However, in cancer, tumor cells upregulate IDO1 expression to create an immunosuppressive environment. IDO1 catalyzes the degradation of tryptophan into kynurenine metabolites, leading to local depletion of tryptophan and accumulation of immunosuppressive by-products.
Tryptophan depletion inhibits T-cell proliferation and induces T-cell anergy or apoptosis, while kynurenine metabolites promote the differentiation of regulatory T-cells (Tregs) and suppress effector T-cell function. This dual effect significantly weakens the anti-tumor immune response, allowing tumor cells to evade immune surveillance.[23]
Targeting IDO1 with small molecule inhibitors aims to restore immune function by preventing tryptophan depletion and reducing immunosuppressive metabolite formation. This enhances T-cell activity and improves the efficacy of cancer immunotherapy, especially when used in combination with immune checkpoint[24]
2.4 STING Pathway
The Stimulator of Interferon Genes (STING) pathway is a crucial component of the innate immune system that detects cytosolic DNA and initiates immune responses against infections and cancer. STING is an adaptor protein located on the endoplasmic reticulum of immune cells such as dendritic cells and macrophages.
When abnormal DNA (e.g., from tumor cells or pathogens) is present in the cytoplasm, it is recognized by cyclic GMP-AMP synthase (cGAS), which produces cyclic GMP-AMP (cGAMP). This molecule binds to and activates STING, triggering downstream signaling pathways that lead to the activation of transcription factors such as IRF3 and NF-κB.[25]
Activation of the STING pathway results in the production of type I interferons and other pro-inflammatory cytokines. These mediators enhance antigen presentation, activate dendritic cells, and promote the priming and activation of T-cells, thereby linking innate and adaptive immunity.
In cancer, the STING pathway is often underactive, allowing tumor cells to escape immune detection. Small molecule STING agonists are designed to activate this pathway, thereby boosting innate immune responses and enhancing anti-tumor immunity. These agents are being explored as promising therapeutic strategies, particularly in combination with immune checkpoint inhibitors.[26]
3. SMALL MOLECULE INHIBITORS: MECHANISM OF ACTION
Small molecule inhibitors are low molecular weight compounds designed to selectively interact with specific target proteins or enzymes involved in cancer progression and immune regulation. Due to their small size, they can easily penetrate cell membranes and access intracellular targets, making them highly effective in modulating signaling pathways.
These molecules exert their effects primarily by binding to target proteins or enzymes, often at active or allosteric sites. This binding can inhibit enzymatic activity or alter protein conformation, thereby disrupting downstream signaling processes essential for tumor growth and survival.[27]
Another important mechanism is the blockade of protein–protein interactions (PPIs). Many immune checkpoint pathways, such as PD-1/PD-L1, rely on interactions between proteins. Small molecule inhibitors can interfere with these interactions, preventing inhibitory signaling and restoring immune cell function.[28]
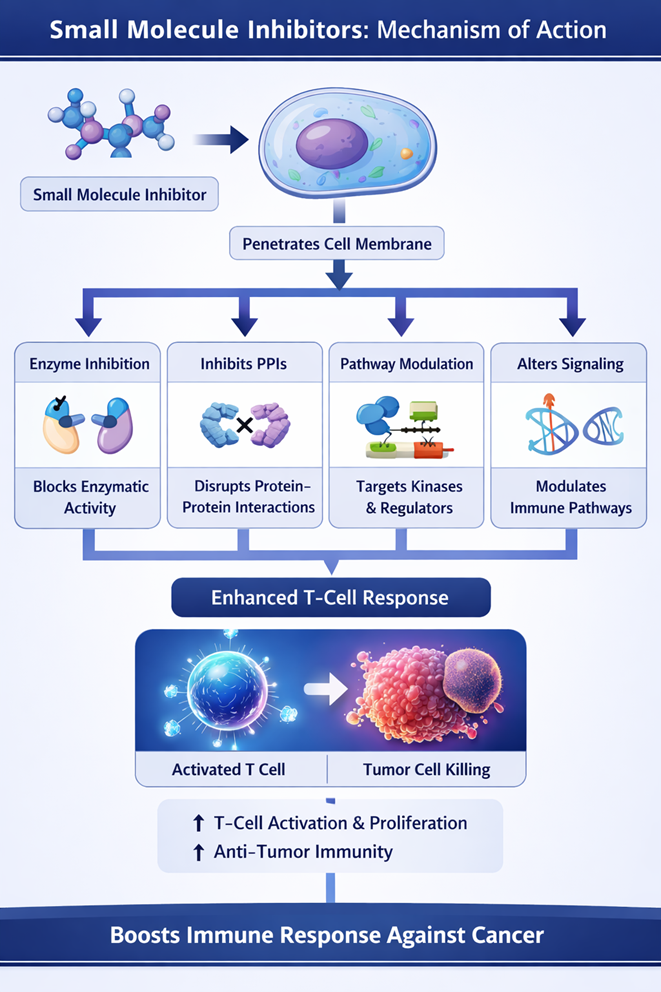
Additionally, small molecules modulate immune signaling pathways by targeting key regulators such as kinases, transcription factors, or metabolic enzymes (e.g., IDO1). This modulation can either suppress tumor-promoting pathways or activate immune-stimulatory pathways.[29]
A critical outcome of these actions is the enhancement of T-cell activation and proliferation. By removing inhibitory signals and promoting stimulatory pathways, small molecule inhibitors boost the cytotoxic activity of T-cells against tumor cells, thereby strengthening anti-tumor immunity.[30]
Overall, small molecule inhibitors provide a versatile and targeted approach in cancer immunotherapy, with the potential for oral bioavailability, better tumor penetration, and combinational use with other therapies.[31]
4. DRUG DESIGN STRATEGIES
The development of small molecule inhibitors for cancer immunotherapy relies on advanced drug design strategies that enable the identification, optimization, and validation of effective therapeutic agents.[32]
Structure-Based Drug Design (SBDD) involves the use of three-dimensional structural information of target proteins, often obtained through techniques like X-ray crystallography or NMR spectroscopy. By understanding the active or binding sites of proteins, researchers can design molecules that fit precisely into these sites, enhancing specificity and efficacy while minimizing off-target effects.
Ligand-Based Drug Design (LBDD) is applied when the structure of the target protein is unknown. This approach uses information from known active compounds (ligands) to identify key chemical features responsible for biological activity. Techniques such as pharmacophore modeling and similarity analysis help in designing new compounds with improved activity.[33]
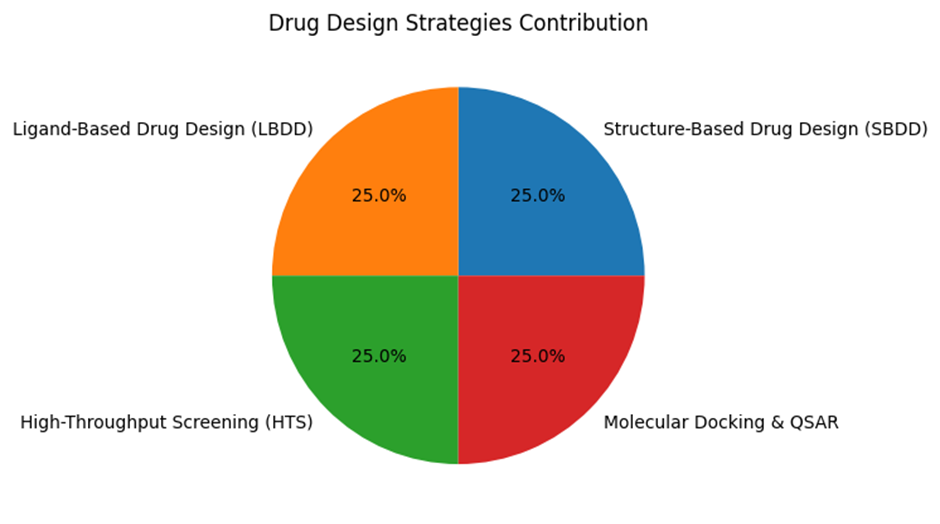
High-Throughput Screening (HTS) is a powerful method that allows the rapid testing of thousands to millions of compounds against a biological target. It helps in identifying potential lead compounds that exhibit desired biological activity, which can then be further optimized through medicinal chemistry approaches.[34]
Molecular Docking and Quantitative Structure–Activity Relationship (QSAR) Studies are computational techniques widely used in modern drug discovery. Molecular docking predicts the binding orientation and affinity of a small molecule with its target protein, while QSAR establishes relationships between chemical structure and biological activity. These methods help in optimizing lead compounds, reducing time and cost in drug development.[35]
Overall, these integrated strategies accelerate the discovery and development of novel small molecule inhibitors with improved selectivity, potency, and safety profiles for cancer immunotherapy.[36]
Table: Drug Design Strategies in Small Molecule Development[37]
|
Strategy |
Principle |
Key Techniques/ Tools |
Advantages |
|
Structure-Based Drug Design (SBDD) |
Utilizes 3D structure of target proteins to design drugs |
X-ray crystallography, NMR spectroscopy, molecular modeling |
High specificity, rational design, reduced off-target effects |
|
Ligand-Based Drug Design (LBDD) |
Uses information from known ligands when target structure is unknown |
Pharmacophore modeling, similarity analysis |
Useful without protein structure, faster lead identification |
|
High-Throughput Screening (HTS) |
Rapid screening of large compound libraries against targets |
Automated assays, robotics, screening platforms |
Identifies lead compounds quickly, large-scale analysis |
|
Molecular Docking & QSAR Studies |
Predicts interaction and correlates structure with activity |
Docking software, QSAR modeling, computational tools |
Cost-effective, reduces experimental workload, lead optimization |
5. ADVANTAGES OVER BIOLOGICS
A. Small molecule inhibitors offer several advantages over biologics (such as monoclonal antibodies) in cancer immunotherapy, making them an attractive therapeutic option.
B. One of the major advantages is their suitability for oral administration. Unlike biologics, which are typically administered via intravenous or subcutaneous routes, small molecules can often be taken orally, improving patient compliance and convenience.[38]
C. Small molecules also exhibit better tumor penetration due to their low molecular weight and physicochemical properties. They can easily diffuse across cell membranes and penetrate deep into solid tumors, reaching intracellular targets that are often inaccessible to large biologic molecules.
D. Another key benefit is lower immunogenicity. Biologics, being protein-based, have a higher potential to trigger immune reactions such as the formation of anti-drug antibodies. In contrast, small molecules are less likely to induce such immune responses, leading to improved safety and tolerability.[39]
E. Additionally, small molecules are associated with cost-effective manufacturing. Their chemical synthesis is generally more straightforward, scalable, and less expensive compared to the complex biotechnological processes required for producing biologics. This can enhance accessibility and affordability of treatment.
F. Overall, these advantages make small molecule inhibitors a promising and practical alternative or complement to biologic therapies in cancer immunotherapy.[40]
6. CHALLENGES
1. Despite their significant potential, small molecule inhibitors in cancer immunotherapy face several important challenges that can limit their clinical success.
2. One major difficulty is targeting protein–protein interactions (PPIs). These interactions often involve large, flat, and dynamic surfaces without well-defined binding pockets, making it challenging for small molecules to effectively disrupt them with high specificity and affinity.[41]
3. Another critical issue is off-target toxicity. Due to their ability to interact with multiple proteins, small molecules may unintentionally affect non-target pathways, leading to adverse effects and reduced safety profiles. Achieving high selectivity remains a key objective in drug design.
4. There is also limited clinical success in certain pathways, particularly with some targets like IDO1 inhibitors. Although preclinical studies showed promising immunomodulatory effects, several clinical trials have failed to demonstrate significant therapeutic benefits, highlighting the complexity of tumor biology and immune regulation.[42]
5. Additionally, drug resistance poses a significant hurdle. Tumor cells can develop resistance through various mechanisms, such as mutations in target proteins, activation of alternative signaling pathways, or adaptation of the tumor microenvironment, ultimately reducing the long-term efficacy of these therapies.[43]
7. RECENT ADVANCES
Recent years have witnessed significant progress in the development of small molecule inhibitors for cancer immunotherapy, driven by advances in molecular biology, computational tools, and translational research.
One major advancement is the development of PD-L1 small molecule inhibitors. Unlike monoclonal antibodies, these small molecules are designed to disrupt PD-1/PD-L1 interactions with improved tumor penetration and oral bioavailability. Some compounds have shown promising preclinical and early clinical results, offering a potential alternative to antibody-based therapies.[44]
Another important trend is the emergence of dual-target inhibitors (multi-target drugs). These agents are designed to simultaneously modulate multiple pathways involved in tumor progression and immune suppression, such as combining checkpoint inhibition with kinase or metabolic pathway targeting. This multi-pronged approach enhances therapeutic efficacy and reduces the likelihood of resistance.
Combination therapies with checkpoint inhibitors have also gained considerable attention. Small molecule inhibitors are increasingly being used alongside established immune checkpoint inhibitors to achieve synergistic effects. For example, combining IDO1 inhibitors or STING agonists with PD-1/PD-L1 blockers can enhance T-cell responses and improve overall treatment outcomes.
Furthermore, AI-based drug discovery approaches are revolutionizing the field. Artificial intelligence and machine learning techniques are being used to predict drug–target interactions, optimize lead compounds, and accelerate the drug development process. These technologies significantly reduce time, cost, and failure rates in identifying effective small molecule therapeutics.
8. FUTURE PERSPECTIVES
The future of small molecule inhibitors in cancer immunotherapy is highly promising, with ongoing research focused on improving precision, efficacy, and safety through innovative approaches.
One key direction is the adoption of personalized medicine approaches, where treatment strategies are tailored based on an individual patient’s genetic, molecular, and immunological profile. This enables the selection of the most effective small molecule inhibitors for specific tumor types, enhancing therapeutic outcomes and minimizing unnecessary toxicity.
Closely related is the advancement of biomarker-guided therapy. The identification of predictive biomarkers, such as PD-L1 expression levels, tumor mutational burden, or immune signatures, helps in selecting patients who are most likely to respond to specific treatments. This approach improves clinical success rates and supports more rational use of immunotherapeutic agents.
The integration of nanotechnology is another emerging frontier. Nanocarrier-based drug delivery systems can enhance the stability, bioavailability, and targeted delivery of small molecule inhibitors. This allows for improved accumulation in tumor tissues, reduced systemic toxicity, and better control over drug release.
Additionally, efforts are being directed toward achieving improved selectivity and safety profiles. Advanced drug design techniques, including structure-based optimization and AI-driven modeling, are helping to develop highly selective compounds that minimize off-target effects while maintaining strong therapeutic efficacy.
9. CONCLUSION
Small molecule inhibitors represent a rapidly evolving and promising frontier in cancer immunotherapy. Their unique advantages, including oral bioavailability, better tumor penetration, and cost-effective production, position them as valuable alternatives or complements to biologic therapies.
Although challenges such as off-target toxicity, difficulty in targeting protein–protein interactions, and variable clinical success remain, continuous advancements in medicinal chemistry, molecular biology, and computational approaches are significantly accelerating their development. Innovations such as AI-driven drug design, biomarker-guided therapy, and combination treatment strategies are further enhancing their therapeutic potential.
Overall, small molecule inhibitors are expected to play a crucial role in the future of cancer treatment, either as standalone therapies or in combination with existing immunotherapies, ultimately contributing to more effective and personalized cancer care.
REFERENCES






Vanita Mehetre, Chavala Vasanthi, C. L. Sindhura, Ankita Shinde, Heena Mahajan, Nilam Ichake, Small Molecule Inhibitors for Cancer Immunotherapy Targets, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 4, 1914-1926. https://doi.org/10.5281/zenodo.19523261
 10.5281/zenodo.19523261
10.5281/zenodo.19523261