
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Aryakul group of colleges, Aryakul College of Pharmacy and Research, Lucknow, Uttar Pradesh.
Drug delivery technology has evolved significantly in the medical field since the discovery of liposomes. Liposomes are gaining popularity as adaptable drug delivery systems because of their biocompatibility, effectiveness, targeting capability, and enhanced in vivo performance. Liposomal drug delivery may be used for controlled and prolonged drug release, reduced systemic toxicity, and enhanced pharmacokinetic and pharmacodynamic characteristics of the drug. Liposome-encapsulated medicines have superior viability and less off-target impacts when they are effectively and latently focused on to the tumor-specific area. One of the foremost unmistakable zones in ponder right presently is the creation of multifunctional liposomes that target cell organelles, are long acting (PEGylated), and work with a few drugs. The coordinates employments of liposomal innovation for medications and diagnostics have been the most accentuation in later a long time. The field of liposomal drug delivery is reviewed in the present work, along with current advancements in the delivery of antifungal, antibiotic, anti-inflammatory, and anti-cancer medications, as well as the effectiveness of liposomal technology in moving from idea to clinical acceptance.
Liposomes are for the most part made up of cholesterol and phospholipids. Phospholipids with a hydrophilic head and a hydrophobic tail bunch include the lipid bilayer. Water is drawn to the head and repulsed by the tail, which is made up of a long hydrocarbon chain. Liposomes primary component, phospholipids, promptly combines with skin lipids to progress medicate localization and entrance within the skin's layers. Liposome soundness is expanded and layer penetrability is diminished by the cholesterol retained into the lipid layer. Liposomes in fluid arrangement can acknowledge hydrophobic components within the lipid stage and encapsulate hydrophilic materials within the fluid compartment due to their bilayer structure. They are made up of one or more phospholipid bilayers, which were at first distinguished by British hematologist Alec D. Bangham within the 1960s. The primary depiction of swelling phospholipid frameworks was given by analysts in 1965. The term "bangosomes" and afterward "liposomes" were utilized to assign a run of encased phospholipid bilayer structures composed of single bilayers inside a couple of a long time. It was appeared by early pioneers like Gregoriadis and Perrie that liposomes might entangle solutions and be utilized as sedate conveyance vehicles. Since of this structure, liposomes have created into a really effective medicate conveyance strategy that raises the restorative list of medicines by expanding their bioavailability, expanding their half-life, and bringing down their harmfulness. These vesicles can transport medicines to their target locale through detached or dynamic focusing on forms since they are biocompatible and have a inclination to imitate cell layers. This essentially brings down unfavorable impacts and raises the drugs helpful esteem. Hence, liposomes have been utilized in numerous areas of restorative science, extending from oncology to irresistible infections and quality treatment. The capacity of liposomes to typify drugs either inside their fluid center or inside their lipid bilayer enables them to secure labile drugs from corruption and permits for controlled discharge, consequently making them an alluring vehicle for a number of restorative operators. A reliable and effective method for arranging liposomes is essential to ensure the quality, safety, and effectiveness of liposomal drug delivery systems. Traditional methods, like thin film hydration, face challenges such as poor control over size, low drug loading efficiency, and inconsistency between batches, which can stop them from being used widely or approved by regulators. A good and standardized approach allows for the consistent production of liposomes with uniform size, drug content, and surface features—all of which directly influence how the drug spreads in the body, how it is released, and its ability to target specific areas. Reproducibility is also important for meeting pharmaceutical quality standards and ensuring patient safety. Advanced techniques such as microfluidic mixing provide precise control over liposome formation, improving drug delivery efficiency and versatility while reducing inconsistency. Therefore, using a dependable and efficient method is key to successfully developing and bringing liposome-based drug delivery systems to market.
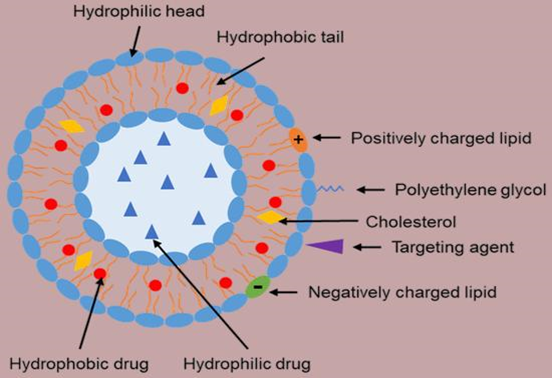
Figure 1: Structure of Liposome
Classification of Liposomes
1. Based on Composition
2. Based on Lamellarity (Number of Bilayers)
3. Based on Size
4. Based on Preparation Method
5. Based on Application
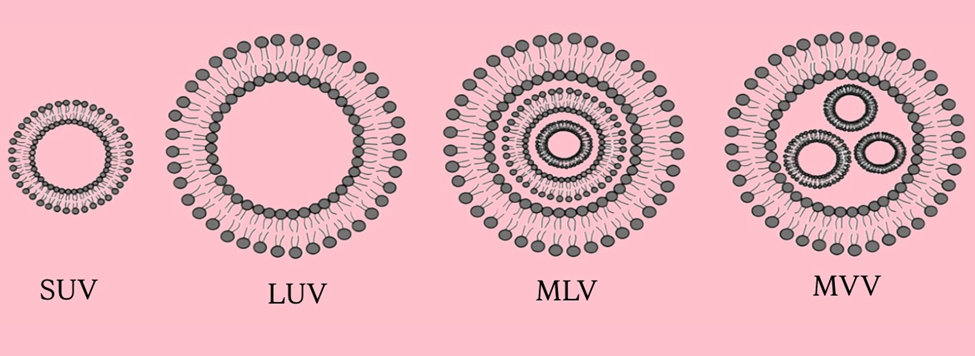
Figure 2: Types of Liposomes
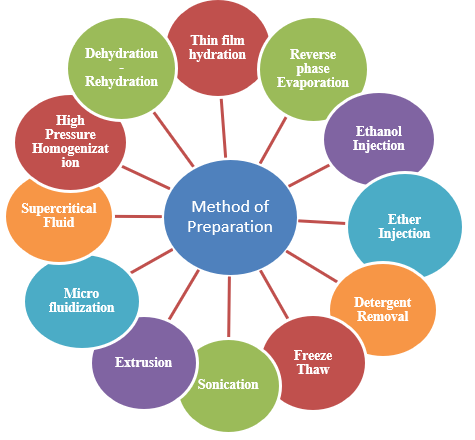
Figure 3: Method of Preparation of Liposomes
Preparation of Liposome Thin-Film Hydration Method
1.Selection of Lipids
Selecting the right lipids for your liposome formulation was the first and most important step in the procedure. In fact, almost every aspect of the final nanoparticle—from size and drug encapsulation effectiveness (and consequently drug content) to cellular absorption and biodistribution—is determined by their selection. Depending on the application, common liposomal formulations include soy phosphatidylcholine (SPC), cholesterol (Chol), polyethylene glycol (PEG), and other particular lipids. As demonstrated by PEG, the percentage of lipids is quite important. Its concentration usually falls between 0.5 and 3%, and this range can nearly triple the size of liposomes or nanoparticles.
2. Dissolution of Lipids in an organic solvent
To create a lipid suspension, the second step entails dissolving lipids in an organic solvent, usually chloroform or a chloroform-methanol mixture. However, less abrasive solvents like methanol or pure ethanol can be employed. For this purpose, a round-bottom flask is usually the best choice because it makes the following procedure easier. At that point, hydrophobic drugs and lipophilic API can also dissolve in the solvent.
3.Formation of Thin Lipid Film - The lipid-solvent mixture should be moved to a rotating evaporator after dissolving. It will be used to completely evaporate the organic solvent at lower pressure, creating a thin lipid layer at the flask's bottom. To get rid of any remaining solvent after evaporation, the thin film needs to be vacuum-dried for a few hours.
4. Hydration of Lipid Film- The another step of the method includes the hydration of the dried lipid film with a reasonable watery stage, such as refined water, buffer arrangement, or saline containing the hydrophilic API. The hydration medium can too be preheated when utilizing lipids with a tall phase-transition temperature (Tc). Putting in contact the lipids with the fluid stage will trigger the self gathering handle of the liposome, driving to the arrangement of multilamellar vesicles (MLVs).
5. Post-processing step: Purification - Following the arrangement prepare, substances like unencapsulated drugs and contaminants regularly stay within the encompassing fluid. Another key component of post-production includes expelling any extra natural solvents utilized within the prepare. Undoubtedly, in the event that these solvents wait within the last item, they can destabilize the liposomes and be hazardous for utilize. These substances must subsequently be expelled in an extra refinement step. Common strategies to evacuate them incorporate tengential stream filtration, ultra-centrifugation, dialysis. In any case, these refinement forms can be time-consuming and may altogether affect the ultimate nanoparticle characteristics whereas lessening the ultimate abdicate of liposomes.
Advantages and Disadvantages of Thin Film Hydration in Liposome Preparation
Pros of Thin Film Hydration Method
Simple and Cost-Effective: Requires basic laboratory equipment like rotary evaporators, vacuum pumps, and glassware. Ideal for academic and foremost articulation studies.
Versatile for Various Lipids: Compatible with a wide range of lipids (e.g., phosphatidylcholine, cholesterol, etc.), making it adaptable for different formulations.
Suitable for Hydrophobic and Hydrophilic Drugs: Hydrophobic drugs can be incorporated into the lipid bilayer. Hydrophilic drugs can be put inside the water-based center.
Scalable at Laboratory Level: Can be used to prepare small to moderate volumes (mg to g scale), which is useful for research and preclinical studies.
Good for Preliminary Studies: Useful for screening different lipid compositions, drug-to-lipid ratios, and encapsulation efficiencies.
Does not need special microfluidic devices: Avoids using complicated and expensive equipment that might not be found in every laboratory.
Cons of Thin Film Hydration Method
Polydispersity (Wide Size Distribution): Liposomes formed are usually heterogeneous in size; further processing like extrusion or sonication is required to reduce size and polydispersity index (PDI).
Low Encapsulation Efficiency for Hydrophilic Drugs: The aqueous core volume is small compared to total volume, leading to poor loading of water-soluble drugs.
Time-Consuming: Requires multiple steps (film formation, drying, hydration, size reduction), which are labor-intensive and slow.
Difficult to Scale-Up for Industrial Use: Challenges in translating this method from lab to large-scale production due to manual operations and poor control over liposome characteristics.
Residual Solvent Problems: Organic solvents like chloroform and methanol, which are used when making lipid films, need to be fully removed to prevent harmful effects.
Stability Problems: The liposomes created might not stay stable for long because the drug inside could get oxidized or leak out from the lipid layer.
Need for Additional Processing: Size reduction techniques (like sonication or extrusion) are often mandatory to obtain nanosized liposomes suitable for drug delivery.
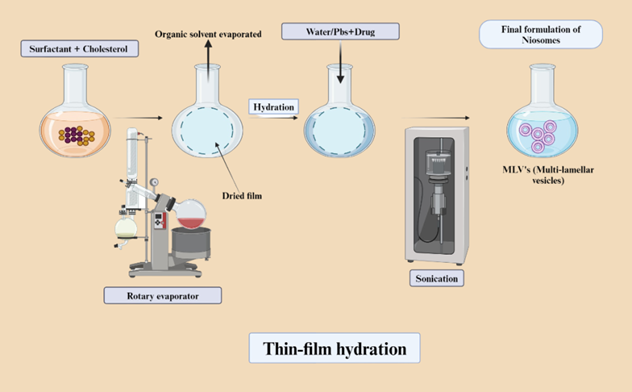
Figure 4: Thin Film Hydration Process
Microfluidics Technique: A Cutting-Edge Approach Surpassing Thin Film Hydration for Liposome Synthesis-
The microfluidic technique for liposome preparation is based on the principle of controlled self-assembly of lipid molecules within micro-scale channels through rapid and precise mixing of immiscible or partially miscible fluids. In this method, lipids are first dissolved in a water-miscible organic solvent, such as ethanol or isopropanol, forming the organic phase. This lipid-containing solution is then introduced into a microfluidic device and brought into contact with an aqueous phase (such as water or a buffer) under laminar flow conditions. Due to the small dimensions of the microfluidic channels, the fluids flow in a highly ordered and parallel fashion (laminar flow), and mixing occurs primarily via diffusion rather than turbulence. As the organic solvent diffuses into the aqueous phase, the local solvent polarity changes, leading to a reduction in lipid solubility. This causes the lipids to self-assemble into bilayered vesicles or liposomes through spontaneous precipitation and thermodynamically driven aggregation. The whole process is controlled by factors like the flow rate ratio between the water and oil parts, the total flow rate, and how much lipid is used. These factors help control the size of the liposomes, how varied their sizes are, and how well the drug is trapped inside. In some setups, like hydrodynamic flow focusing, the oil part is sandwiched between two streams of water, which helps mix better and makes the liposomes very uniform. This method is reliable, can be scaled up, and works well for making liposomes with the same physical and chemical traits every time, which is great for use in medicine and healthcare.
Components of the Microfluidic System for Liposome Preparation
Microfluidic systems have become a popular and controlled way to make liposomes. They allow for precise control of fluid movement at a very small scale. These systems can be split into two main types: (1) microfluidic chip systems, which are often used in small lab settings, and (2) microfluidizer systems, which use high pressure and are good for making large amounts. Each system has important parts that work together to help lipids come together and form liposomes. Below is a detailed look at each of these parts.
1. Microfluidic Chip-Based System Components
These systems use tiny chips made through microfabrication to carefully mix lipid and water-based solutions while keeping the flow steady and controlled. The main parts include:
a) Microfluidic Chip
The microfluidic chip is the main part of the system where liposomes are actually made. It has tiny channels with diameters between 10 and 500 micrometers. These channels are made from materials like PDMS, glass, or silicon. The design of the chip might include shapes like T-junctions, Y-junctions, or flow-focusing channels, which help control how the fluids come together. In the area where mixing happens, the organic and aqueous liquids mix under laminar flow, allowing lipids to come together naturally through diffusion to form liposomes.
b) Syringe Pump
Syringe pumps are used to deliver the lipid solution, which is mixed in a water-mixing organic liquid like ethanol, and the aqueous buffer into the microfluidic chip. They control the flow of these liquids very precisely. The speed at which the liquids flow determines the flow rate ratio and the total flow rate, which are important for making liposomes of the right size, shape, and with good efficiency. The pumps provide a steady and smooth flow without any pulsing, which helps in making liposomes consistently and reliably each time.
c) Inlet Ports
These are the places on the chip where syringes or tubes with the lipid and water solutions are attached. Usually, there are two or more of these openings so that several liquids can be added at the same time. The way these openings are made helps the liquids enter evenly and mix properly in the tiny channels inside.
d) Mixing Junction / Hydrodynamic Focusing Zone
This is the important part of the chip where the liquid made of fats and the water mix closely and blend by moving into each other. In designs that use hydrodynamic flow focusing, the fat liquid is made narrower and kept centered between two water streams. This helps the liquids mix better, making it easier and more even for liposomes to form on their own. How well the mixing happens in this area directly influences the size and uniformity of the liposomes made.
e) Temperature Control System
Some phospholipids need to be heated above their gel-to-liquid crystalline transition temperature (Tm) to dissolve properly and form structures on their own. To help with this, microfluidic systems usually have heating parts or temperature controls. This keeps both the lipid and water parts at the right temperature during the whole process.
f) Outlet Channel / Collection Reservoir
The outlet is where the freshly made liposome mixture leaves the chip. It is linked to a container or tube that carries the mixture to the next steps. These steps may involve removing solvent, filtering the mixture, adding medicine, or testing its properties.
2. Microfluidizer-Based System Components
Microfluidizers are advanced, high-pressure homogenization devices widely used for large-scale and industrial liposome production. They operate based on the principle of forcing fluids through microchannels at very high velocities and pressures, generating intense shear and impact forces that facilitate liposome formation.
a) High-Pressure Pump
This pump is the main part that makes the microfluidization process work. It creates pressures between 10,000 and 30,000 psi, which pushes the fluid mixture through special chambers. The strong shear and cavitation forces from this process break apart large multilamellar vesicles into smaller unilamellar vesicles, which results in nanosized and evenly distributed liposomes.
b) Interaction Chamber
The interaction chamber is where the lipid and water streams are made to meet or go through tight microchannels under high pressure. It can have different shapes, like Y-shaped for two streams to collide or Z-shaped for a flowing, zigzag path. These chambers help mix the streams thoroughly, release energy, and break down large lipid groups into smaller pieces, which results in making liposomes that are all about the same size.
c) Feed Reservoirs / Tanks
Feed reservoirs store the lipid and water-based parts before they go into the high-pressure system. These tanks can be mixed and kept at a steady temperature to ensure the mixture is even and at the right heat. In some setups, the already blended mix is kept in one tank and then sent through a microfluidizer later.
d) Cooling System
High-pressure homogenization generates significant heat due to frictional forces, which may destabilize thermosensitive drugs or lipids. To counteract this, the system is equipped with a cooling jacket, ice bath, or heat exchanger coils that maintain the process temperature within a safe range, typically below 30–40°C.
e) Recirculation Loop
A recirculation loop allows the fluid to pass through the microfluidizer multiple times (cycles), enabling further refinement of liposome size and uniformity. The number of cycles can be adjusted depending on the desired liposome characteristics.
f) Outlet and Collection Vessel
After processing, the liposome dispersion exits through the outlet and is collected in a sterile container. This product may then undergo additional steps such as filtration, solvent removal (via dialysis or rotary evaporation), or drug loading.
Advantages and Disadvantages of Microfluidization in Liposome Preparation
ADVANTAGES (Pros):
DISADVANTAGES (Cons):
.jpg)
Figure 5: Microfluidization Process
Comparison Table 1: Microfluidization vs Thin Film Hydration
The following table provides a detailed comparison between microfluidization and thin film hydration methods for liposome preparation. It shows that microfluidization is better when it comes to controlling size, making larger amounts, getting consistent results, and being more efficient overall.
|
Criteria |
Microfluidization |
Thin Film Hydration |
|
Particle Size Control |
Produces Nano sized, uniform liposomes with low PDI |
Produces larger MLVs; needs post-processing |
|
Reproducibility |
High, due to controlled flow and pressure |
Low to moderate; operator-dependent |
|
Scalability |
Easily scalable from lab to industry |
Difficult to scale; suited for lab scale |
|
Processing Time |
Fast and efficient |
Time-consuming and multi-step |
|
Encapsulation Efficiency |
High, especially with active loading |
Moderate to low without optimization |
|
Post-Processing Needs |
Minimal |
Requires sonication or extrusion |
|
Suitability for Sensitive Drugs |
Good; with integrated cooling system |
May expose drugs to heat during hydration |
|
Batch-to-Batch Consistency |
Excellent |
Variable |
|
Cost of Equipment |
High initial cost |
Low to moderate |
Evaluation Table 2: Microfluidization vs. Thin Film Hydration for Liposome Preparation
|
Parameter |
Thin Film Hydration |
Microfluidization |
|
Principle |
Lipid film is formed, dried, and hydrated with aqueous phase |
Lipid and aqueous phases are mixed at high pressure in microchannels |
|
Size Control |
Poor control; requires post-sizing (e.g., sonication, extrusion) |
Excellent control; produces uniform liposomes directly |
|
Particle Size |
200–1000 nm (initial); size reduced later |
50–150 nm (depending on conditions) |
|
Polydispersity Index (PDI) |
Higher PDI (>0.3), less uniform size |
Low PDI (<0.2), highly uniform |
|
Reproducibility |
Low; manual steps cause batch variation |
High; automated and consistent process |
|
Scalability |
Difficult to scale up; labor-intensive |
Easily scalable for industrial production |
|
Encapsulation Efficiency (EE) |
Low to moderate; especially for hydrophilic drugs |
High (especially with active loading techniques) |
|
Process Time |
Long (film formation, hydration, size reduction) |
Shorter and continuous process |
|
Sterility Control |
Difficult; multiple open steps |
Easier; closed, continuous system |
|
Equipment Cost |
Low to moderate |
High (microfluidizer equipment is expensive) |
|
Temperature Sensitivity |
Mild conditions possible |
High shear may generate heat; temperature control required |
|
Suitability for Doxorubicin |
Not ideal; low EE without active loading |
Ideal; compatible with active (remote) loading using ammonium gradient |
|
Liposome Type |
MLVs or LUVs initially |
SUVs or LUVs, highly uniform |
|
Batch vs Continuous |
Batch process |
Continuous or semi-continuous process |
|
Ease of Operation |
Simple and easy for small scale |
Technically complex, needs trained personnel |
Future Prospects: Microfluidization vs Thin Film Hydration in Liposome Preparation
In the fast-moving world of nanomedicine and delivering drugs more precisely, microfluidization stands out as a better choice than the old thin film hydration method. Thin film hydration has always been popular because it's simple, cheap to start with, and easy to use in a lab. But it has some big problems that make it hard to use in bigger factories or real-world medicine. These problems include not being able to scale up easily, not being able to control the size and consistency of liposomes well, differences between batches, and taking a long time to process. Plus, thin film hydration can expose delicate drugs to changing temperatures and strong mechanical forces, which might make them less stable or less effective. Microfluidization, which is a high-pressure, continuous flow method using microfluidic technology, solves these issues. It gives much better control over how big the particles are, makes the results more consistent, and improves how well drugs are enclosed inside liposomes. It also allows for quick and scalable production that fits with the rules for making medicines safely (GMP) and designing quality into the process (QbD), which are important for getting drugs approved. New microfluidizers also have temperature control, which helps protect sensitive molecules like mRNA, siRNA, and proteins from damage during processing. This makes them perfect for new kinds of treatments, such as vaccines and gene therapy. The fact that microfluidization is already used in approved liposomal drugs and in some COVID-19 mRNA vaccines shows how important it is in industry and how it might become the top choice for making liposomes. So, microfluidization is likely to become the main method for preparing liposomes in the future, pushing forward innovation and making things more efficient in both research labs and drug factories. Many companies already make liposomal drug delivery systems using traditional methods like thin-film hydration. These systems have proven to work well in real-world use and have passed strict medical and legal checks. Some well-known examples include AmBisome® for fungal infections, DaunoXome® for cancer treatment, DepoDurTM for pain management, and Doxil®/Caelyx® for treating breast and ovarian cancers. However, these drugs are made in batches, which can be tough to manage when making large amounts or keeping the drug particles very small. On the other hand, a new technology called microfluidic synthesis is gaining a lot of attention. Big pharmaceutical companies and new startups are using continuous microfluidic systems to develop new drug candidates like mRNA-lipid nanoparticles, targeted nanocarriers, and improved PEGylated liposomes. This technology allows for precise control over the size of the drug particles, consistent results, and easier scaling up of production. Although no liposomal product has been officially approved that is made entirely with microfluidic methods, this technology is seen as very promising for future medicines. Microfluidic-based drug formulations are expected to support personalized medicine and faster clinical development, and may eventually replace traditional thin-film methods like those used for Doxil® and AmBisome® in the coming years.
CONCLUSION
This thesis compared thin film hydration and microfluidization for liposome drug delivery, analyzing their principles, strengths, and limitations. Thin film hydration is simple, cost-effective, and suitable for lab-scale research but struggles with inconsistent particle size, lower encapsulation efficiency, and poor scalability. Microfluidization, however, excels in producing smaller, uniform liposomes with higher encapsulation efficiency and reproducibility, making it ideal for industrial-scale production and targeted therapies. On the other hand, microfluidization is better at making liposomes that are smaller, more uniform, and can hold more drug. It also makes the results more reliable, which is important for making medicine on a large scale and for targeted treatments. Microfluidization gives better control over the size and shape of the liposomes, which helps drugs stay stable and work better in the body. Even though it costs more and needs more technical skill, it’s better for making large quantities and handling complex mixtures. Because of this, microfluidization is the better choice for making high-quality liposomes for real-world use. Thin film hydration is still useful for early research and creating new drug formulas. But microfluidization has clear advantages that make it better for developing new and efficient drug delivery systems. In the end, microfluidization is the future for making liposomes that are high quality, easy to scale, and efficient. Even though thin film hydration will still be used in research, microfluidization has the potential to drive major changes in medicine, leading to better patient care and new, advanced drugs.
REFERENCES


Priyanshi Gupta, Priyanka Kesharwani*, A Comparative Review of Thin Film Hydration and Microfluidic Techniques for Liposome Based Drug Delivery, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 7, 3207-3220. https://doi.org/10.5281/zenodo.16400902
 10.5281/zenodo.16400902
10.5281/zenodo.16400902