
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmacy, Institute of Technology and Management, GIDA, Gorakhpur, Uttar Pradesh, India, 273209.
Dipeptidyl peptidase 4 (DPP-4) inhibitors, or gliptins, are oral antidiabetic medications that enhance the production of incretin by inhibiting the degradation of endogenous “insulinotropic” GIP and GLP-1 in vivo (glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide). Incretin hormones play a key role in the homeostasis of blood glucose and initiate the secretion of insulin in an incretin-dependent manner, while also inhibiting glucagon function. With DPP-4 inhibition, postprandial GIP and GLP-1 plasma levels are increased both in fasting and after meals, lowering the minimally hypoglycemic level of postprandial glycemic hunger. Their gastric emptying is also delayed, and they elicit satiety, which can negate their weight-neutral effects. DPP-4 inhibitors (pleiotropic) in addition to glycemic treatment exhibit pleiotropic characteristics (despite potential cardio- and/or normotensive effects). They have been associated with improved endothelial dysfunction, oxidative stress, and inflammation—all of which are essential in the prevention of diabetic complications. As for DPP-4 inhibitors (sitagliptin, a commonly used agent, saxagliptin, linagliptin, and alogliptin), they are commonly associated with mild to moderate side effects, with nasopharyngitis and gastrointestinal symptoms being the most prevalent among them. DPP-4 inhibitors play a prominent role due to their once-daily dosing, safety profile, and glucose-dependent mechanism of action, representing a standard in the management of type 2 diabetes. Their ability to tightly control blood glucose with minimal side effects, both in monotherapy and combination therapy, is why they are commonly employed. Determining whether they provide durable benefits in diabetic patients may require more research to answer questions regarding long-term cardiorenal outcomes.
Diabetes mellitus, especially type 2 diabetes (T2D), is a rapidly growing global health problem. As of 2021, the International Diabetes Federation (IDF) estimates that approximately 537 million adults have diabetes globally, and this number is forecast to rise to 783 million by 2045. This dramatic increase highlights the immediate public health requirements and emerging therapeutic opportunities urgently needed. Diabetes was the eighth leading cause of death in 2021 and drastically affects the quality of life for those who have this disease. Moreover, diabetes is very costly from an economic perspective: by 2021, global diabetes healthcare costs were over $966 billion, and this is expected to surpass $1 trillion according to estimates from the Global Burden of Disease study by 2045 [1], [2]. Different physiological mechanisms that govern the homeostasis of glucose, including incretin hormones, are involved in many aspects of glucose physiology. The two types of incretin hormones that have critical roles in incretin signaling, glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP), bind to specific members of the G-protein-coupled receptor (GPCR) family: the GLP-1 receptor (GLP-1R) and the GIP receptor (GIPR), respectively. Upon receptor binding, both of these hormones stimulate adenylate cyclase, resulting in the enhancement of cyclic adenosine monophosphate (cAMP) in β-cells. This sequence leads to glucose-dependent insulin secretion through sensitization, thus ensuring optimal maintenance of glucose levels. Still, this function is tightly regulated by dipeptidyl peptidase-4 (DPP-4), an enzyme that contributes to the rapid hydrolysis and inactivation of incretin hormones, resulting in decreased insulin secretion [3]. Dipeptidyl peptidase-4, or DPP-4, is the enzyme that hydrolyzes incretin hormones such as glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1). Dipeptidyl peptidase-4 (DPP-4) cleaves these hormones, acting in an inhibitory manner to regulate the physiological action of endogenous incretin. By cleaving these hormones, DPP-4 inhibits their functions, impacting physiological processes [4], [5]. The DPP-4 inhibitors, which are oral antidiabetic drugs of a specific class, have now become one of the main therapeutic categories for T2D. They work by blocking the action of the DPP-4 enzyme and increasing the half-life of endogenous GLP-1 and GIP. Hence, this leads these hormones to work for a longer time, and as a consequence, pancreatic β-cells become more sensitive to increases in blood glucose, causing the release of insulin and a decrease in glucagon. DPP-4 inhibitors have the advantage of decreasing glucagon by augmenting levels of incretin hormones, particularly driving insulin secretion in hyperglycemia to avoid glucagon overproduction. It has been observed that this group of drugs can block DPP-4 activity by over 80–90%, resulting in pronounced reductions in postprandial glucose levels and improved glycemic control [6], [7]. This article reviews the mode of action for DPP-4 inhibitors and their application in the treatment of T2D. In this review, we highlight the drugs in this class, describe their role in glucose homeostasis preservation and the specificities/differences among them, as well as clinical results, safety profiles, and off-label indications within different metabolic diseases. We also delve into the ongoing research along with future prospects of DPP-4 inhibitors, suggesting an increasing role for these agents in applications beyond the management of diabetes. With the increasing global burden of T2D, understanding and optimizing the use of DPP-4 inhibitors is important to address the progressive rise in the prevalence of this disease.
2. Structure and Function of DPP-4:
2.1. Molecular structure and enzymatic activity of DPP-4:
2.1.1. Molecular Structure: DPP-4 (766 amino acid residues and binds with proline and alanine at the N-terminal of the peptide) [8]. The protein DPP-4 is localized to a small pocket known as the catalytic pocket, which has serine at position 630. This protein harbors a stalk extending from the cells, with a stalk anchored to the cell membrane via a tethered rigid linker coupled to a transmembrane region. It contains a short arm at the other end as well, which is inside the cytoplasm. Two DPP-4 proteins come together to form one pair on the cell membrane. These pairs can then be cleaved at a particular bond to release a full-length DPP-4 that goes free into the circulation [5].
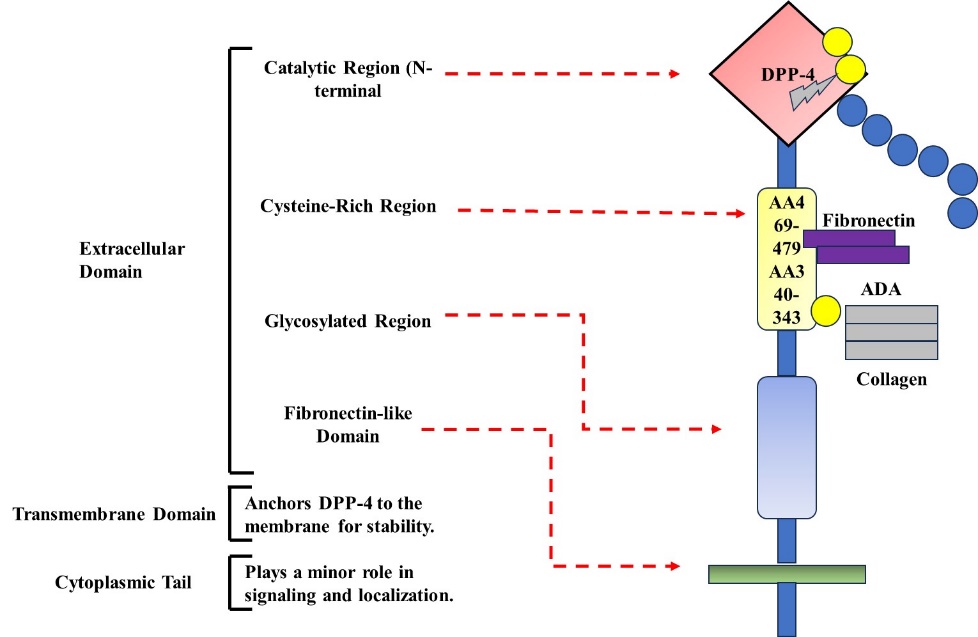
Figure 1. Tridimensional structural organization of Dipeptidyl Peptidase-4 (DPP-4) with its locations for extracellular, transmembrane, and intracytoplasmic regions, as well as the main functional domains.
2.1.2. Enzymatic Activity: DPP-4 is an enzyme that degrades proteins by first removing dipeptides. It works best on proteins where the second amino acid is proline or alanine. It can also cleave proteins where the second position is glycine, serine, valine, or leucine, but more slowly. It cannot cut proteins if proline is in the third position [5]. DPP-4 inhibitors degrade certain entero-hormones (e.g., glucose-dependent insulinotropic peptide (GIP) and glucagon-like peptide-1 (GLP-1)), or incretins, which are primarily produced within minutes [9].
2.2. Tissue distribution and biological functions beyond glucose metabolism:
2.2.1. Tissue Distribution: DPP-4 is present in many organs and tissues of the human body (the intestinal and renal epithelium, blood vessel walls, pancreas, liver, gland cells, gut, as well as lymphocytes) [5]. Plasma contains the soluble form of DPP-4. This protein mainly comes from seminal plasma, kidneys, and endothelial cells—the cells lining blood vessels—as well as a couple of populations of white blood cells (lymphocytes) [9].
2.2.2. Biological Functions Beyond Glucose Metabolism: DPP-4 is a protein that not only digests other proteins, but also plays a crucial role in important body processes, such as how cells grow, move, and connect with their environment, known as the extracellular matrix [5]. DPP-4 also fulfills its non-proteolytic functions as a receptor or co-stimulatory protein in the field of immunology [10]. DPP-4 also cleaves cytokines and chemokines, contributing to inflammation [11].
2.3. DPP-4 involvement in immune regulation, cardiovascular function, and inflammation:
2.3.1. Immune Regulation: DPP-4, also known as CD26, is an immune protein that functions as a co-stimulatory molecule to amplify the functions of other immune system components [5]. B cells: DPP-4 is located in B cells and may help their development and function when the bones self-renew [10]. DPP-4 is indispensable for immune responses, T-cell activation, and maturation, and it often functions as a sole receptor of DPP-4 in isolation from its catalytic action [12].
2.3.2. Cardiovascular Function: DPP-4 is present on the endothelial cells [5], [9].
2.3.3. Inflammation: Inflammation is regulated by DPP-4 clearance of proteins that modulate immune responses. Additionally, its soluble form, sDPP-4, directly interacts with immune cells [12]. Experimental inhibition of DPP-4 could block inflammation. In human beings, it showed that soluble DPP-4 correlated with MCP-1 and up-regulation of interleukin-8 (IL-8) and interleukin-6 (IL-6), both mediators that initiate inflammation. These mediators are called pro-inflammatory cytokines and are the main culprits for launching and sustaining inflammation. Therefore, inhibiting the cytokines that induce inflammation could decrease inflammation and its associated effects in our bodies by targeting DPP-4. Better knowledge of how DPP-4 interacts with these cytokines may provide novel therapeutic strategies for inflammatory diseases [11].
2.4. Pathophysiological role of DPP-4 in diabetes and metabolic disorders:
2.4.1. Diabetes and Metabolic Disorders: Changes in DPP-4 expression and/or activity are linked to metabolic disorders such as obesity and diabetes [5]. DPP-4 blood activity is several times higher in type 1 and type 2 diabetes than it is in healthy people. This increased activity is strongly associated with HbA1c, the level reflecting average blood sugar over weeks. Moreover, rapid degradation of GLP-1 occurs since DPP-4 inactivity is upregulated. The reason GLP-1 is important is that it stimulates the body to secrete a little bit of insulin. Faster degradation of GLP-1 means the body might not release enough insulin, especially for people who have type 2 diabetes [9].
2.4.2. DPP-4 inhibitors: DPP-4 inhibitors are drugs that prevent the degradation of incretin hormones, such as GLP-1 (an incretin hormone that actively contributes to blood sugar control) in the DPP-4 rich incretin pathway. These drugs help reduce blood sugar by protecting GLP-1 from degradation. They trigger a rise in insulin when blood sugar levels are high, lower glucagon (the hormone that can cause blood sugar levels to rise), and slow gastric emptying. Consequently, they help to control blood sugar levels better at the same time [9].
3. Mechanism of Action of DPP-4 Inhibitors:
Dipeptidyl peptidase-4 (DPP-4) is a medicine used to reduce blood sugar. DPP-4 is one of the most important classes of drugs in diabetes treatment. They work by inhibiting an enzyme (DPP-4) that increases the quantities of various hormones. The hormones tell the pancreas to secrete more and higher amounts of insulin (the hormone that reduces blood sugar). DPP-4 inhibitors decrease the production of liver glucose, which prevents blood sugar from going too high. Together, these functions enable DPP-4 inhibitors to help individuals with diabetes maintain better regulation of their blood sugar [13]. DPP-4 inhibitors increase glycemic control in patients with type 2 diabetes through multiple mechanisms.
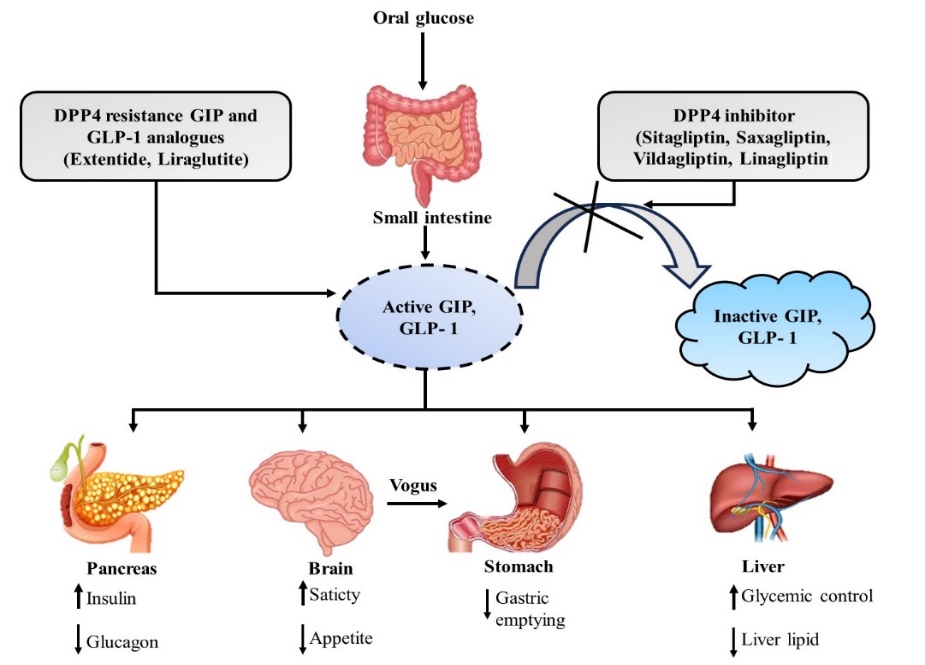
Figure 2. DPP-4 inhibitors prevent enhancing insulin secretion, incretin degradation, and reducing glucagon release for better glucose control
3.1. Inhibition of incretin degradation and prolonged action of GLP-1 and GIP: As DPP-4 inhibitors, they work by elevating the amounts of incretin hormones in the body. DPP-4 inhibitors raise levels of these hormones, which include glucose-dependent insulinotropic polypeptides (GIP) and glucagon-like peptide-1 (GLP-1). These hormones regulate blood sugar levels. DPP-4 inhibitors help the body manage blood sugar levels better by elevating these hormones, especially in diabetic patients who have to deal with their blood sugar. A high level of GIP and GLP-1 binding causes increased insulin secretion, leading to stronger control over glucose in the body [6]. GLP-1 is rapidly degraded by DPP-4 under normal circumstances. Inhibiting DPP-4 enables the body to retain the active form of GLP-1 for a longer time [14].
3.2. Enhanced insulin secretion and reduced glucagon levels: DPP-4 inhibitors prevent the degradation of GLP-1 and GIP, which in turn enhances the response of the pancreas's β-cells to blood sugar. They reduce the hormone glucagon, lowering blood sugar levels and making it easier to manage blood sugar levels [6]. Raising unmodified GLP-1 levels improves pancreas functioning. It causes more insulin release and less glucagon, which helps keep blood sugar down [15].
3.3. β-cell preservation and insulin sensitivity improvement: The DPP-4 inhibitors help the pancreas by supporting insulin-producing β-cells in the body. Experimental studies confirm that these inhibitors improve the function of β-cells in general and help to prevent poor pancreatic health [16].
3.4. Impact on gastric emptying and appetite regulation: The action of DPP-4 inhibitors does not affect the rate of gastric emptying. By contrast, GLP-1 receptor agonists delay gastric emptying, but DPP-4 inhibitors do not [6].
4. Classification and Pharmacokinetics of DPP-4 Inhibitors:
4.1. Classification of DPP-4 Inhibitors
As an oral antidiabetic drug, the Dipeptidyl peptidase-4 inhibitors (DPP-4) class plays an important role in treating type 2 diabetes mellitus (T2DM) [1]. They work by inhibiting the DPP-4 enzyme, which is responsible for degrading incretin hormones. This inhibition leads to increased insulin secretion and reduced glucagon levels by preventing the degradation of these hormones. DPP-4 inhibitors can be classified by chemical type, binding mode, and regulatory approvals.
4.1.1. Chemical Nature:
4.1.2. Binding mechanisms:
DPP-4 inhibitors' binding modes can be grouped into three main classes that interact in different manners with specific sites on the DPP-4 enzyme:
4.1.3. FDA-Approved DPP-4 Inhibitors: DPP-4 inhibitors are used to treat blood sugar levels in people with diabetes. Modulation of DPP-4 enzymes includes teneligliptin, vildagliptin, saxagliptin, linagliptin, sitagliptin, anagliptin, alogliptin, gemigliptin, etc. They block the DPP-4 enzyme, which increases postprandial insulin secretion from these medications. This process assists in the reduction and maintenance of blood sugar levels. If you are diabetic, your doctor may recommend one of the following inhibitors to manage your blood sugar [7], [20]. Sitagliptin (the first of the DPP-4 inhibitors) was launched on the market in 2006 [7]. Since then, newer agents have been developed with enhanced pharmacokinetic properties while considering safety issues. A very good example is linagliptin, which is predominantly excreted through the bile rather than through the kidneys; hence, its dose should not be changed in renal impaired patients [24]. Some recent clinical trials have also examined combination therapies with other antidiabetic agents, including dipeptidyl peptidase-4 (DPP-4) inhibitors, sodium-glucose cotransporter-2 (SGLT2) inhibitors, or metformin for potential additive outcomes [25].
4.1.4. Recent Advances in DPP-4 Inhibitor Research
Finally, there has been a shift in the therapeutic landscape of DPP-4 inhibitors toward glycemic control in recent years in research. So far, they have also been studied for their anti-inflammatory and cardioprotective properties in subjects with T2DM who are at high risk for cardiovascular diseases [26], [27]. Recent improvements in molecular modeling have enabled the design of new inhibitors with higher selectivity for DPP-4 than for other dipeptidyl peptidases, thus minimizing side effects [28]. Investigations on fixed-dose combinations (FDCs) containing DPP-4 inhibitors as monotherapy are also underway to improve treatment simplification and medication adherence. Patients with cardiovascular (CV) risk, for instance, FDCs planned with sitagliptin and metformin or empagliflozin have shown superiority versus monotherapy on the grounds of treatment efficacy in clinical trials [29].
4.1.5. Comparison of Binding Affinities, Potency, and Selectivity: Saxagliptin displays efficacy at around a 50 nM concentration. Vildagliptin and sitagliptin have IC50 values of 62 nM and 19 nM, respectively. Linagliptin is the most powerful DPP-4 inhibitor with an IC50 of 1 nM, making it the most efficacious of these drugs [30]. Linagliptin is a drug that specifically acts upon the DPP-4 enzyme. It is approximately 44,000 times more selective for DPP-8 dpp-4 than DPP-8 and about 10,000 times more selective than DPP-9, which results in Linagliptin primarily targeting DPP-4 with very little effect on DPP-8 or DPP-9 [30].
4.2. Pharmacokinetics
4.3. CYP450 Metabolism vs. Renal Elimination: Sitagliptin is not extensively metabolized by the liver enzymes cytochrome P450 3A4 and 2C8. Therefore, it should ideally have minimal interactions with other drugs that are metabolized through these pathways or the p-glycoprotein transport system. If you are on other medications that are metabolized by these pathways, major drug interactions with sitagliptin are thought to be unlikely. Thus, in patients on many drug regimens, sitagliptin is the best choice to minimize unexpected adverse effects or problems. You may feel more confident that sitagliptin is safe to take with other drugs and doesn't interfere with its hepatic metabolism. This is particularly important for patients regularly dosed on medications who would like their treatment to be free from drug interactions [31]. Unlike other DPP-4 inhibitors, linagliptin is renally excreted very little and is not significantly eliminated by the kidneys. Hence, patients with kidney problems can take it without having to change the dose [32].
4.4. Drug-Drug Interactions and Contraindications: Clinical trials failed to identify any significant interactions, other than those affecting Sitagliptin pharmacokinetics, which could potentially impact the dose [31]. If you are going to start any GLP-1 receptor agonists, please stop the DPP-4 inhibitors [7].
5. Therapeutic Applications of DPP-4 Inhibitors:
5.1 Treatment of Type 2 Diabetes Mellitus (T2DM)
5.1.1. Effects on Glycemic Control: DPP-4 inhibitors prevent the inactivation of incretin hormones (glucagon-like peptide-1 (GLP-1) and gastric inhibitory polypeptide (GIP)). These hormones:
In addition, DPP-4 inhibitors help to reduce long-term HbA1c levels (approximately 0.5–1%) through significant reductions in fasting and postprandial glucose levels, which allows for better long-term glycemic control. Unlike sulfonylureas, they do not cause hypoglycemia, making them the preferred option for many patients [33].
5.1.2. Monotherapy vs. Combination Therapy: DPP-4 inhibitors may be used as monotherapy in individuals with mild to moderate diabetes who are non-compliant or intolerant of metformin and other first-line agents. However, because of their lesser magnitude of glucose lowering, they are prescribed more commonly as an addition to another type of medicine rather than as monotherapy:
In patients with progressive β-cell dysfunction, combination therapy is particularly advantageous as it focuses on multiple pathways in glucose regulation and overall increases diabetes control [33].
5.2. Benefits in Specific Patient Groups:
5.2.1. Elderly Patients: Hypoglycemia and weight gain are common problems in elderly diabetics receiving insulin or sulfonylureas, especially when the class of medications powerfully affects glucose. The popular regimen of DPP-4 inhibitors is as follows for this population:
5.2.2. Renal-Impaired Individuals: Chronic kidney disease is a major risk factor for diabetes, and many antidiabetic drugs are either contraindicated or need dose modifications in patients with renal impairment. DPP-4 inhibitors:
Furthermore, some DPP-4 inhibitors have specific features that make them safe to use in patients with CKD without the need for dose adjustment and are therefore necessary in this subset [34].
5.2.3. Cardiovascular Risk Groups:T2DM patients commonly have a high burden of cardiovascular diseases (CVD), such as heart attacks and stroke. Studies on the anti-CVD cardiovascular effects of DPP-4 inhibitors are extensive. That is:
With this in mind, DPP-4 inhibitors represent a good choice for patients with high cardiovascular risk, but they are not a first-line consideration for cardiovascular protection compared to SGLT2 inhibitors or glucagon-like peptide-1 receptor agonists, which showed greater CV benefits [35], [27].
5.3. Cardiovascular and Renal Benefits
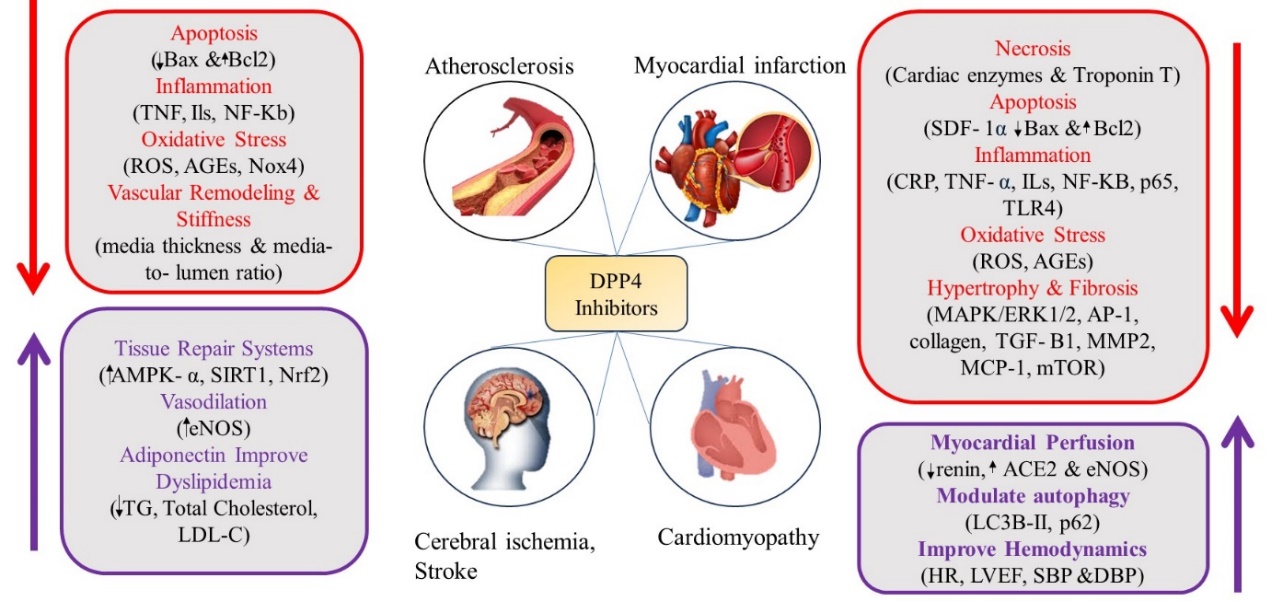
Figure 3. Cardiovascular Protective Properties of DPP-4 inhibitors: protection of endothelial function, reduction of inflammation, and improvement in glucose metabolism.
Cardiovascular Outcome Trials:
Impact on Cardiovascular Health: DPP-4 Inhibitors for a Healthy Heart: Controlling Cholesterol and Blood Pressure Issues. They fight inflammation, protect cells from damage, provide benefits to blood vessels, and reduce protein excretion in urine [27].
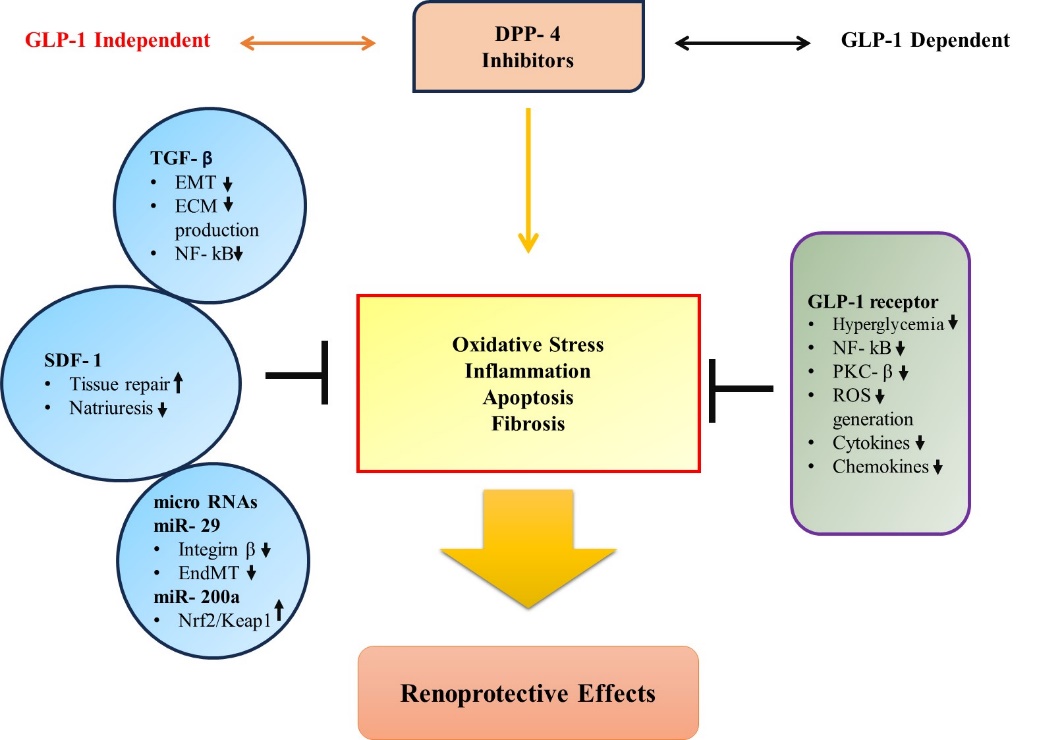
Figure 4. Schematic representation of the kidney-protective actions of DPP-4 inhibitors, including both GLP-1-dependent and independent effects on renoprotection
Renoprotective Effects: DPP-4 inhibitors may protect the kidney by reducing albuminuria. This suggests that DPP-4 inhibitors might be beneficial for kidney health [27].
5.4. Emerging Non-Diabetic Applications
Anti-inflammatory Effects: DPP-4 inhibitors can modify the immune response to start in a manner with an additional line [36].
Neuroprotective Potential: Gliptins are being investigated for their effects on brain and nerve protection. However, so far most of the research work done has been on animals [36].
Anti-cancer Properties: Gliptins are being evaluated for their ability to prevent and treat cancer in part by focusing on disparate elements of the tumor microenvironment. They inhibit an enzyme called DPP-IV, which can cause the body to function differently, potentially affecting how tumors develop and spread. Investigators are exploring whether gliptins possess these properties that might have utility in the treatment of cancer [36].
Effects on Obesity and Metabolic Syndrome: DPP-4 inhibitors are medications that are not associated with weight gain. They may be helpful in nonalcoholic fatty liver disease (NAFLD), which is also a risk factor for heart disease [27], [33].
6. Clinical Trials and Safety Profile:
In 2006, dipeptidyl-peptidase-4 (DPP-4) inhibitors were launched for the treatment of type 2 diabetes. These drugs stimulate insulin secretion and reduce glucagon, which can be beneficial for blood sugar control. They often bring HbA1c levels down by between 0.5 to 1%, although the actual decrease varies from person to person depending largely on their blood sugar at diagnosis and other medications they may be taking. DPP-4 inhibitors are similarly effective as sulfonylureas in improving glucose control and are equivalent to metformin or even pioglitazone, especially in those who have not been taking diabetes medications previously [7].
Adverse effects
Most people are able to tolerate DPP-4 inhibitors fairly well. They are very unlikely to cause serious side effects and have a very small risk of hypoglycemia, which means that blood sugar can get too low. As a general rule, people do not tend to experience side effects from using DPP-4 inhibitors or have dangerously low blood sugar [14]. Sore throats and skin issues may occur in a lot of people, but unless these problems are severe, they do not indicate the discontinuation of the drug. Over the years, researchers have followed this drug to see who it harms in certain organ systems and haven't seen any significant red flags in the heart or blood vessels [7].
Pancreatitis
The US Food and Drug Administration (FDA) and the European Medicines Agency highlighted concerns over whether incretin-based therapies may be damaging to the pancreas by conducting extensive studies. Following their extensive reviews, they concluded that there is no direct relationship between these treatments and pancreatitis [7], [14]. DPP-4 inhibitor label warnings about the danger of acute pancreatitis (sudden and severe inflammation of the pancreas) [14].
Cardiovascular risk
Research trials (such as EXAMINE, SAVOR-TIMI-53, TECOS, and CARMELINA) have shown that certain DPP-4 inhibitors do not adversely impact the heart and are cardiovascularly safe [7], [14]. Though all of the studies focused on the same primary endpoint, hospitalizations due to heart failure, the results differed across studies. Research one found that saxagliptin was associated with more heart failure admissions [7].
Risk vs. benefit analysis in long-term therapy
DPP-4 inhibitors are a safe and well-tolerated drug class, ideal for older adults, type 2 diabetes patients, and those with kidney manifestations. These medications are favored by patients because they have fewer side effects, which is particularly important for individuals with other kidney issues or the elderly who may be taking other nephrotoxic drugs. They effectively control blood sugar without straining the kidneys, which is extremely important for diabetic patients who already have kidney problems. Therefore, DPP-4 inhibitors are a safe choice for the elderly and those with kidney disease, as they help control diabetes without the risk of causing further harm [7]. These drugs are co-prescribed with metformin to lower blood sugar without causing hypoglycemia and reducing the risk of obesity. They can be used in conjunction with other anti-diabetic drugs, such as SGLT-2 inhibitors [7].
Post-marketing surveillance and safety updates
It is essential to monitor the drugs when they are ready to be picked up for any additional safety issues. DPP-4 inhibitors were among the first to cause side effects since they target many proteins in the body — so, in some sense, at onset. In general, the majority of studies suggest that the risk of side effects with DPP-4 inhibitors is equivalent to that of a placebo (inactive pill) [14].
7. Recent Advances and Future Perspectives:
7.1. Development of Dual and Multi-Target Inhibitors
Researchers have been able to develop new medicines for treating type 2 diabetes and metabolic syndrome across several areas simultaneously. Mechanism: There is one type of such medicine that stimulates two receptors in the body, the PPAR alpha and PPAR gamma. The reason is to increase the body's usage of insulin for blood sugar regulation and decrease blood triglycerides. Thus, these drugs help improve the control and health status of type 2 diabetic patients as well as other metabolic disorders [37]. Researchers are evaluating strategies to increase GLP-1 release with other drugs (e.g., DPP-4 inhibitors + GPR119 agonists, etc.) [38]. Fixed Dose Combination (FDC) of anti-diabetic drugs that lowers blood glucose is a priority. These drug combinations have the same efficacy for people with diabetes to effectively manage their blood sugar levels [39].
7.2. Personalized Medicine and Pharmacogenomics
New Era of Diabetes Care: Pharmacogenomics for Personalized Medicine — The dawn of personalized diabetes treatment combines studies of drugs with genomics to develop individualized medical regimens for patients. It aims to tailor drug therapy for every patient based on their unique genetics and how they will respond to certain medications. This approach helps in choosing an ideal treatment with the least possible side effects or harm. It is quite different from how diabetes treatment used to be, with limited choices for doctors and the necessity to experiment with a few medications and doses in order to find one that works for the patient. Now, under personalized care, physicians can prescribe the best drug and dosage to a patient from the outset. The individualized approach means better health outcomes and mitigates adverse drug reactions. This is a significant advancement in healthcare, moving towards personalized diabetes care based on each patient's unique genetic profile [40].
7.3. Combination Therapies and Fixed-Dose Formulations
In most cases for the treatment of type II diabetes (T2DM), several classes of drugs are taken together to control blood sugar levels. The drugs in question are classified as antihyperglycemic agents; each has a different mechanism of action to achieve the same result. They are equally effective when taken together in one pill (fixed-dose combination, FDC) as when they are administered separately in two doses. They are also similar in effect and well tolerated by the body. FDCs improve treatment adherence, as past research examining historical data finds. This, in turn, leads to effective control of blood sugar and less expensive diabetes management. Being on any combination therapy early in the disease is associated with enduring advantages, sometimes termed "legacy effects," in which benefits lag behind for a while [39], [41].
7.4. Future challenges:
Advancements have been made, but some key issues still remain. First, there is clinical inertia, the fact that sometimes individuals do not move forward or change treatments. Another problem is that patients do not always follow their treatment regimens. Additionally, it is important to determine how well the new drugs perform in the long run and whether they are safe. It is equally important to assess the value of these new drugs. Once the drugs are approved for use, they must be monitored carefully for any new adverse events and to ensure they continue to be effective at controlling diabetes over time [42].
CONCLUSION:
Dipeptidyl peptidase 4 (DPP4) inhibitors have emerged as a crucial class of anti-diabetic medications offering glycemic control with a low risk of hypoglycemia. The incretin-based mechanism of action for these agents causes them to suppress glucagon in a glucose-dependent fashion while simultaneously inducing insulin secretion. Therefore, they offer an ideal treatment strategy against type 2 diabetes mellitus (T2DM), in particular. In contrast to the majority of anti-diabetic compounds, DPP-4 inhibitors do not produce weight gain as a side effect, which is crucial for limiting body weight gain during long-term therapy for diabetes. While generally safe and effective, long-term cardiovascular and immune implications remain understudied and thus represent major unanswered concerns. Some studies propose potentially cardioprotective and renoprotective actions, while others highlight an enhanced vulnerability of some DPP-4 inhibitors to cause heart failure. Additionally, life-threatening yet very rare adverse effects, such as pancreatitis and joint pain, should be monitored closely. Further studies propose that these agents might be involved in something beyond diabetes for the treatment of neurodegenerative diseases. They could provide a privileged entry to augment their impact on other physiological pathways that are outside of what their current limited classification implies. Continued research will be required to examine the long-term safety implications of DPP-4 inhibitors, as well as to discover off-target and congeneric clinical applications in order to fully realize these advantages. This would render them safe and effective for life, meaning that they would not just be part of the toolbox in diabetes, but also for other chronic disease care, leading to better outcomes.
REFERENCES


Tejas Pandey*, Priyanka Sonker, A Comprehensive Review on Dipeptidyl Peptidase-4 (DPP-4) Inhibitors: Mechanism, Therapeutic Applications, and Future Perspectives, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 5, 5108-5124. https://doi.org/10.5281/zenodo.15560637
 10.5281/zenodo.15560637
10.5281/zenodo.15560637