
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Lokmanya Tilak Institute of Pharmacy, Navi Mumbai, Maharashtra 410210
Gelatin is a natural polymer extracted from collagen, which has been widely used as a pharmaceutical excipient due to its properties such as biocompatibility, biodegradability, and gel-forming ability. Its applications include encapsulation of drugs in hard and soft gelatin capsules, stabilizers in vaccines, and coatings for controlled drug release. Gelatin-based formulations improve the stability of drugs, protect APIs from environmental factors, and enhance patient compliance. Gelatin, obtained from the processing of chicken feet, fish skins, and bovine bones, can be synthesized via acid or alkali treatments and by hydrolysis and purification processes. The application of this material ranges from improving the flavor and texture of food products to applications in pharmaceutical capsules, adhesives, and drug delivery systems. Gelatin also finds use in advanced biomedical applications, including tissue engineering, wound dressings, and drug encapsulation. Recent developments involve the manufacture of recombinant gelatin, nanoparticle-based formulations, and plant-derived alternatives. These developments have helped address issues including allergic reactions and unavailability leading to a strengthened importance of gelatin in drug development. This review focuses on the synthesis, functional properties, and evolving applications of gelatin as a versatile excipient in pharmaceutical formulations.
Gelatin is a natural polymer obtained from the hydrolytic degradation of collagen, a structural protein in animal connective tissues. [1] In general, gelatin contains amino acids such as glutamic acid, aspartic acid, arginine, proline, hydroxyproline, lysine, isoleucine, methionine, leucine, and valine. [2] It is usually supplied as translucent sheets, granules, or powder (Fig 1.1). It is light-amber to faintly yellow, tasteless, odorless, brittle when dry, and rubbery when moist.[3] Primarily obtained from animal body parts, gelatin is a by-product of the meat industry and is extensively used in food, pharmaceuticals, cosmetics, and other industries. [4] In food production, gelatin enhances flavor, texture, and nutritional value for items such as mousses, stews, and gelled desserts. Its high protein content makes it a valuable ingredient in these applications. Used for producing various drug and vitamin capsules as protection against the attack of light, oxygen, microbes, or other contaminants within a pharmaceutical process, gelatin has a larger use as an application in photo-graphic film emulsions and paper while utilized for cosmetics, valued for being of excellent functional properties, i.e., strong rheology with efficient emulsion or gelation abilities [5]. Possesses biocompatibility and a very good and successful biodegradation profile- appropriate for leading edge applications including applications in biomedical imaging and engineering. This versatility and eco-friendliness ensure that gelatin remains an indispensable material in many industries for functional and sustainable innovation. [6]

Figure 1.1. Image of gelatin powder, gelatin granules and sheet
DISCOVERY AND HISTORY
A versatile and widely used substance, gelatin has an extended history throughout the centuries. It is mainly extracted from animal collagen and is used extensively in food, medicine, photography, and industry. Its development can be attributed to human ingenuity in extracting and refining natural resources to meet diverse needs. [7-8] (fig 1.2)
Gelatin development Timeline
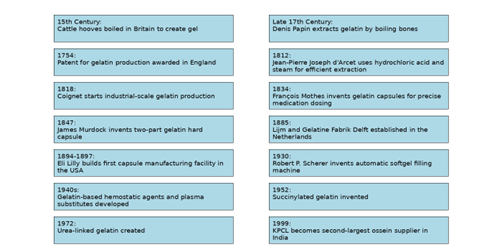
Figure1.2.History and Timeline of Gelatin
SYNONYMS
Byco, Cryogel , gelatine, Instagel, Solugel, Hydrolyzed collagen, gelfoam, gelofusin, emagel, spongiofort, gelatina.[8]
TYPE
Gelatins are mainly of two types on basis of their synthesis:
Gelatins are mainly of two types on basis of their capsule formation:
PHYSICOCHEMICAL PROPERTIES
The chemical and physical properties of gelatin are as follows: [11-14] (Table 1.1)
Table 1.1 physicochemical properties of gelatin
|
Properties |
Description |
|
Structure |
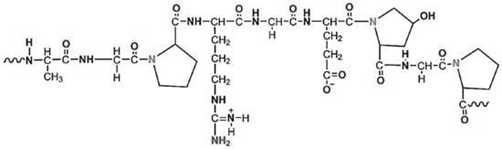
|
|
Chemical name |
Gelatin |
|
CAS register number |
9000-70-80 |
|
Solubility |
Practically insoluble in acetone, chloroform, ethanol (95%), ether, and methanol. Soluble in glycerin, acids, and alkalis, although strong acids or alkalis cause precipitation. In water, gelatin swells and softens, gradually absorbing between five and 10 times its own weight of water. Gelatin is soluble in hot water, forming a jelly, or gel, on cooling to 35–40OC |
|
Melting point |
28-31OC for mammalian derived gelatin 11-28o C for fish derived gelatin |
|
Molecular weight |
varying from 15 000–250 000 |
|
Molecular formula |
Not specific as it contains large group of complex amino acids |
|
Taste |
Tasteless |
|
Odor |
Odorless |
|
Density |
1.325 g/cm3 for type A 1.283 g/cm3 for type B. |
|
Acidity/ alkanity |
pH = 3.8–6.0 (type A) pH = 5.0–7.4 (type B). |
|
Isoelectric point |
7–9 for type A 4.7–5.3 for type B |
|
Moisture content |
9–11% |
|
Gel strength |
Low Bloom 30–150 Medium Bloom 150–225 High Bloom 225–325 |
|
Viscosity |
4.3–4.7 mPa s for a 6.67% w/v aqueous solution at 60oC; 18.5–20.5 mPa s for a 12.5% w/v aqueous solution at 60oC. |
METHOD OF SYNTHESIS
The common steps involves in synthesis of Gelatin are Pre- Treatment, Extraction, Purification, Thickening and Drying.[14]
There are various method by which gelatin is synthesized by using animal skin and bones.
Chicken boiler feet with skin were supplied by a slaughterhouse and kept at −21 ?C for two weeks until subsequent analysis. The chicken feet (2 kg/batch) were thawed at 4?C for 24 hours and afterwards rinsed with tap water. The methods of extraction included rinsing with hot water at a temperature of 65-95°C. The sanitized chicken feet were immersed in a 10% (v/w) solution of 0.1 N HCl at 18°C for 24 hours. Following the acid treatment, the feet's pH was neutralized for 48 hours using running tap water.[15] For extraction at varying temperatures (65, 75, 85, and 95 ?C), the feet were put in polyethylene bags, vacuum sealed, and then heated at these different temperatures for 2 hours. The obtained gelatin was frozen at -70 °C and dehydrated at -40 °C with the help of a freeze-dryer. The gelatin was dried until it attained a consistent weight for 48 hours in the freeze-dryer.[16] (fig 1.3)
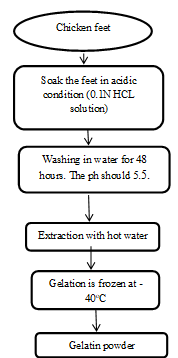
Figure 1.3 Flow chart of synthesis of Gelatin from chicken feet [17]
Gelatin was obtained from the skins of silver carp. The skins of silver carp were taken off, they were descaled, and leftover meat was manually eliminated. The treated skins were rinsed well with distilled water and cut into small pieces. Skin samples were immersed in 0.01 M NaOH with 1% H2O2 at 4 °C while being gently stirred. The solution was replaced every 8 hours for a total of 3 times to eliminate non-collagenous proteins and pigments. Skins treated with alkaline were rinsed using distilled water to achieve neutral pH. The sample was treated with 10% isopropanol for 4 hours at 4°C with gentle stirring to remove fat, then washed extensively with distilled water. The defatted skins were immersed in 0.05 M acetic acid along with a skin for 4 hours at 4oC while being gently stirred to swell the collagen content in the fish skin matrix. The skins treated with acid were rinsed with distilled water to achieve a neutral pH. The swollen fish skins were immersed in distilled water at 45oC for 12 hours with constant stirring to obtain gelatin from the skin material. The mixture undergoes centrifugation at 18,000 x g for 20 minutes at 10 °C. Gelatin obtained from the supernatant was freeze-dried with a freeze dryer. [18] (fig 1.4)

Figure 1.4 Flow Chart of synthesis of Gelatin from silver carp fishes [19]
The process involves cleaning beef bones, reducing their size, and degreasing, which entails eliminating dirt, fat, and meat remnants. To eliminate fat, impurities, and leftover meat from the bones, boil for 120 minutes, then strain. The subsequent step involves immersing the sample in a hydrolysis process, succeeded by demineralization, which is the elimination of calcium and other salts found in beef bones. Immersion lasted for 7 days utilizing different solvent concentrations (4, 5, 6, and 7) % for soaking. The solvents include sodium hydroxide, hydrochloric acid, sulfuric acid, acetic acid, and sodium bicarbonate. The soaking solution is replaced every 2 days. Following immersion, the osein solution was derived. The following step involved filtering and rinsing with running water until the pH of the solution reached 6–7. The subsequent phase involves extracting the resultant solution to yield gelatin. [20] The subsequent step involves the extraction of the resultant osein. The extraction procedure is performed in a water batch. Subsequently, it was removed in an oven set at 70 °C for varying durations of 4 hours and 6 hours, followed by filtration using Whatman paper. The filter outcomes are concentrated, and the concentration is performed using an evaporator until it reaches 25–30% at a temperature of 80 °C for about 2 hours. Following concentration, the sample was placed in a refrigerator at 40 °C until a gel developed. The gel-like sample was dried in an oven at 60 °C for 48 hours, resulting in a dry gelatin sample. This approach yields type B Gelatin. [21] (Fig 1.5)
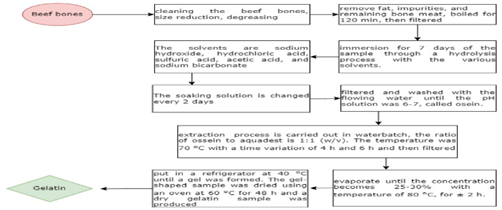
Figure 1.5 Flow chart of Synthesis of Gelatin from Bovine/Beef bones [22]
FUNCTION AND USES OF GELATIN
Gelatin has a wide range of uses in the pharmaceutical sector because of its distinct characteristics, such as the capability to form gels, enhance drug stability, improve drug delivery, and aid in the encapsulation of active pharmaceutical ingredients (APIs). Some typical pharmaceutical uses of gelatin include: [23-24]
Uses in accordance with their concentration: [25-26]
Gelatin is used various concentration in pharmaceutical industries. (Table 1.2):
Table 1.2 Uses of gelatin at different concentration
|
Concentration of gelatin |
Uses |
|
0.1% concentration |
In cell culture |
|
0.1% - 0.5% concentration |
For preparation of Frozen foods |
|
0.2%-1.0% concentration |
For preparation of yoghurt, cheese Cream |
|
1% -6% concentration |
For meat products such as corned beef, jellied beef |
|
1.5% concentration |
For production of marshmallows |
|
1.5%-2.6% concentration |
For preparation of desert |
HANDLING PRECAUTIONS
It is crucial to follow the instructions provided on the packaging of the gelatin product. Different types of gelatin may require different preparation methods, such as blooming in cold water or dissolving in hot water. Failure to follow the instructions may result in the gelatin not setting properly or producing undesirable textures in the final product. In pharmaceutical and cosmetic applications, gelatin may be used as a binder, coating, or encapsulating agent. It is important to ensure that the gelatin used meets the necessary quality standards and regulatory requirements for these applications. Additionally, proper hygiene and safety precautions should be followed when handling gelatin to prevent contamination and ensure product integrity. [27]According to the situation and the amount of material handled, take the standard measures. It is advised to wear gloves and eye protection. Gelatin needs to be stored away from heat sources and sources of ignition and handled in an area with adequate ventilation. It is best to evaporate the gelatin remnants beneath a fume hood because empty containers can catch fire. Fine gelatin powder can become airborne during handling; avoid inhaling it, especially in large quantities, as it may irritate the respiratory system Be mindful of temperature requirements, as overheating can denature gelatin and affect its gelling properties.[28]
STORAGE
Dry gelatin is stable in air. Aqueous gelatin solutions are also stable for long periods if stored under cool, sterile conditions. At temperatures above about 508OC, aqueous gelatin solutions may undergo slow depolymerization and a reduction in gel strength may occur on resetting. Depolymerization becomes more rapid at temperatures above 658C, and gel strength may be reduced by half when a solution is heated at 808OC for 1 hour. Keep gelatin away from direct sunlight and high temperatures, as excessive heat can cause it to degrade. [29] Store gelatin in airtight containers to avoid contamination and exposure to humidity. Gelatin is hygroscopic (absorbs moisture), so store it in a location with low humidity to avoid clumping or loss of functionality. Using desiccants like silica gel in the storage container can help absorb excess moisture. Store gelatin in a dark or shaded area to prevent damage from UV light, which can affect its structure and quality. [30]
INCOMPATIBILITIES
Gelatin is an amphoteric material and will react with both acids and bases. It is also a protein and thus exhibits chemical properties characteristic of such materials; for example, gelatin may be hydrolyzed by most proteolytic systems to yield its amino acid components. Gelatin will also react with aldehydes and aldehydic sugars, anionic and cationic polymers, electrolytes, metal ions, plasticizers, preservatives, and surfactants. [31] It is precipitated by alcohols, chloroform, ether, mercury salts, and tannic acid. Gels can be liquefied by bacteria unless preserved. Some of these interactions are exploited to favorably alter the physical properties of gelatin; for example, gelatin is mixed with a plasticizer, such as glycerin, to produce soft gelatin capsules and suppositories. [32]
ADVERSE EFFECT
Gelatin is commonly ingested in foods. It is likely safe when taken in quantities above the dietary amount for medicinal purposes, short-term. However, high doses of up to 15 grams daily could pose risks for side effects such as sore throat, gums swelling, and mouth ulcers. [33]
A specific type of gelatin, known as gelatin tannate, is possibly safe when administered orally as medication for up to 5 days. In children under 3 years old or that weighs less than 15 kg (33 lbs), taking 250 mg of gelatin tannate four times daily for up to 5 days appears to be safe. In children older than 3 years or weighing more than 15 kg, the repeated dose of 500 mg gelatin tannate four times daily for up to 5 days appears safe. Gelatin is rich in protein. Unless you are cautious, too much protein over time can lead to a variety of health issues, such as: bone disorders, kidney problems, heightened risk of cancer, liver disorders, disease. Rarely, a person with a gelatin allergy might develop an allergic reaction called allergic contact dermatitis. This occurs when the skin directly comes into contact with the substance, usually causing red, itchy, and inflamed skin. [34]
EVALUATION:
An assay is an investigative (analytic) procedure in laboratory medicine, mining, pharmacology, environmental biology, and molecular biology for qualitatively assessing or quantitatively measuring the presence, amount, or functional activity of a target entity. Gelatin has three assay methods: succinylated gelatin assay, biuret assay, hydroxyproline content assay.[34]
A succinylated gelatin assay is a spectrophotometric method that quantifies the catalytic activity of gelatinases. This assay utilizes succinylated gelatin as a substrate to evaluate the primary amines revealed after the gelatinases hydrolyze the substrate. The succinylated gelatin assay is founded on the treatment of gelatin with succinic anhydride to obstruct all free amino groups on the protein. The assay is specific and can assess the gelatinolytic activity of enzymes.[35]
The Lowry assay is a colorimetric method utilized to assess the total protein content of gelatin solutions. The assay detects the color change of a sample solution in correspondence to its protein concentration. In the present study, the variation in “protein color” development among gelatins in colorimetric total protein content measurements was examined at 540 nm using the Biuret assay and at 650 nm utilizing the Lowry assay, with bovine serum albumin as the reference protein. In both the Biuret and Lowry assays, the color response differed significantly among gelatins.[36] (table 1.3)
Table 1.3 Procedure and passing criteria of biuret assay
|
Procedure |
Passing criteria |
|
1. Dissolve 1 g of sample in purified water at about 55°C. Dilute to 100 ml with the same solvent and hold the solution at this temperature. 2. To 2 mL of solution add 0.05 mL of a 125-g/L solution of copper sulfate pentahydrate. Mix, and add 0.5 mL of an 85-g/L solution of sodium hydroxide. |
A violet color is produced |
The amino acid hydroxyproline is abundant in gelatin. Erlich's reagent (5% p-dimethylaminobenzaldehyde in n-propanol) is used to identify hydroxyproline after it has been released through acid hydrolysis and oxidized. [36] (table 1.4)
Table 1.4 Procedure and passing criteria of hydroxyproline content assay
|
Procedure |
Passing Criteria |
|
Dissolve 1.0 gram of material in 200 mL of water. Add 3 mL of this solution and 3mL of conc. HCl to an 18 x 150 mm test tube. Seal by melting the top in a Bunsen burner. Hydrolyze at 145°C for 1.5 hours in an agitated oil bath. Cool the hydrolysate, cut off the top of the tube, transfer contents to the volumetric flask and dilute to 500 mL. Transfer 1 mL to an 18 x 150 mm test tube. Add 1 mL 0.05N CuSO4, 1 mL 2.5N NaOH, and place in a 40°C water bath for 5 minutes. Add 1 mL 6% H2O2 and mix immediately. Keep the sample at 40°C for 10 minutes. Shake and rotate to remove all excess H2O2. Cool rapidly in an ice bath. Add 4 mL 3N H2SO4, mix, then add 2 mL Ehrlich’s reagent, mix, cover with aluminum foil and hold at room temperature for 15 minutes. |
The development of an intense red color shows the presence of hydroxyproline |
The pH is a numerical value that typically indicates the concentration of hydrogen ions in an aqueous solution. Using a pH meter and potentiometry, the pH of a 6.67 percent gelatine solution is measured at 55–60°C. As stated in the European Pharmacopoeia, the pH can also be measured on a 1% solution at 55°C. [37] (1.5)
Table 1.5 Procedure and passing criteria of pH
|
Procedure |
Passing Criteria |
|
Weigh 7.50 g of gelatin into a bloom jar or 150 mL beaker and add 105.g of deionized water, stirring often to suspend all gelatin particles. Cover and let stand 1 – 3 hours at room temperature. Note that the solution prepared for gel strength, viscosity and clarity may be used for this test. Dissolve the sample in a 65°C water bath for 10–15 minutes, stirring or swirling as required. Transfer the sample to the 60°C water bath and temper to 60ºC. Perform a two-point calibration on the pH meter, using pH 4 and pH 7 buffers, at 60°C. Determine the pH of the gelatin solution according the pH-meter instructions. Swirl the solution well using the pH probe to ensure the electrode is sufficiently saturated. |
pH 3.8–7.6 at 55°
|
A weighed sample of gelatin is maintained for 16 to 18 hours at 105°C and is then reweighed. The moisture content is defined as the percentage loss in weight of the sample.[37](table 1.6)
Table 1.6 Procedure and passing criteria of loss on drying
|
Procedure |
Passing Criteria |
|
Wash the evaporating dish very carefully in hot water. Place the dish in the drying oven at 105ºC for at least one hour. Cool dish in the desiccator until room temperature is reached. Weigh approximately 5.0 g of gelatin to the nearest milligram and note the weight of the test sample (m0) and the weight of the sample together with the evaporating dish (m1). Place the evaporating dish containing the sample in the drying oven at 105 ± 2ºC for 16 to 18 hours. Cool the dish in the desiccator until room temperature is reached and weigh to the nearest milligram (m2), weigh and calculate the percentage of residue. |
Not more than 15% |
A UV-vis spectrophotometer was used to characterize the amino acids that contribute to the specificity of gelatin in the 200–400 nm wavelength range. Characteristic peptide bond segments from each gelatin are indicated by chromophore groups that exhibit absorbance at 210–240 nm. Gelatin's optical characteristics and amino acid composition can be determined using its UV-Vis spectrum. The absorption of chromophore groups from amino acids such as glycine, proline, and arginine is visible in the UV spectra of gelatin in the 210–240 nm regions. [38]
Passing criteria: the UV absorbance of a gelatin solution is measured at specific wavelengths, often 214 nm, 257 nm, and 280 nm.
At 214 nm: The absorbance should not exceed a specified limit (e.g., ≤ 0.15 in some standard)
At 257 nm and 280 nm: The absorbance should typically be very low or meet the stated criteria (e.g., ≤ 0.01-0.05).
Gelatin is a protein that liquefies in the presence of gelatinase enzyme as the enzyme breaks down the complex structure of gelatin into monomeric amino acids. Gelatin hydrolysis test is also termed as the Gelatin Liquefaction test as it involves the liquefaction of gelatin in the presence of the gelatinase enzyme. Nutrient Gelatin Media is used for the demonstration of gelatin hydrolysis either by adding mercuric chloride or by the liquefaction of gelatin.[39] (table 1.7)
Table 1.7 procedure and passing criteria of gelatin hydrolysis test
|
Procedure |
Passing criteria |
|
Bacteria are inoculated into a nutrient medium that contains gelatin. The bacteria are incubated for a few days to a week. The test tubes are placed on ice or in the fridge to check for liquefaction. |
Positive result: The gelatin in the medium becomes liquefied. This indicates that the bacterium produces gelatinase and has hydrolyzed the gelatin. Negative result: The gelatin remains solid. This suggests the bacterium does not produce gelatinase and has not hydrolyzed the gelatin. |
DEV Gelatin Agar is used to detect microorganisms that liquefy gelatin in water and to measure the overall microbial count. The ability of an organism to create a proteolytic type enzyme (proteinase) is assessed by the digestion or liquefaction of gelatin, a protein derivative of bovine collagen that is added to a variety of media (1). Peptides, polypeptides, and amino acids are produced when gelatin is hydrolyzed by the enzyme gelatinase, also known as pepsin. In accordance with the German Drinking Water Regulations, 1990, gelatin DEV Agar is used to measure the overall microbial count and identify bacteria that liquefy gelatin in water. The medium is made up of nutrients such as meat extract, gelatin, and peptone, which give organisms carbon and nitrogen molecules to thrive. [40] (Table 1.8)
Table 1.8 procedure and passing criteria of gelatin DEV agar test
|
Procedure |
Passing criteria |
|
In 1000 milliliters of distilled water, suspend 50 grams. Stir well. Bring to a boil in order to fully dissolve the medium. Autoclave for 15 minutes at 121°C to sterilize. Avoid overheating. Reduce to 45–50°C. Pour into sterilized Petri dishes after thoroughly mixing. Apply the bacteria to the middle of the plate using an inoculating loop. The plate should be incubated upside down for a minimum of 48 hours. Pour a saturated ammonium sulfate solution onto the plate |
Clear zones are observed around gelatin-liquefying colonies. |
Gelatin agar alkalization is a technique for identifying gelatin deterioration in a media. It's a practical and considerate approach, but it might not work for every species. This technique detects degradation by Pseudomonas fluorescens with a high degree of sensitivity. [41] (Table 1.9)
Table 1.9 procedure and passing criteria of Alkalization of gelatin agar
|
Procedure |
Passing criteria |
|
25 grams of a sample, such as seafood or vegetables, should be weighed and then added to two flasks either blended or chopped into small bits. Fill one flask with 225 milliliters of Alkaline Peptone Water (M618), and another flask with 225 milliliters of Glucose Phosphate Broth (M070). Stir thoroughly. Incubate for 6 to 8 hours at 35° ± 2°C. One loopful should be added to the non-selective Gelatin Agar from each flask. |
After a few minutes of cooling, the typical foggy zone surrounding the colony, which is visible when V. cholerae is transparent, becomes more distinct. These colonies appear to be finely granular and iridescent green to bronze when viewed in oblique light.
|
MARKETED PRODUCTS
There are various manufacturing company of gelatin which produce gelatin capsule, gelatine sheet, gelatin powder, beef gelatin and many more. (Table 1.10)
Table 1.10 Marketed products of gelatin
|
Image of products |
Company name |
Description |
Price |

|
Nitta Gelatin india limited |
Nitta Gelatin, the global gelatin specialist, offers premium, 200 bloom, gold gelatin of international standard for the discerning. Good quality gelatin determines the excellent texture, melt-in-mouth feel, flavour retention and release of novel foods, desserts, entremets and confectioneries. Certified by premium Halal & Kosher certifying bodies, recognized worldwide |
?1495/- |

|
Foodchem |
Foodchem is a Chinese company specializing in the production and supply of food additives and ingredients, including gelatin. Their gelatin products are used in various industries, such as food, pharmaceuticals, and cosmetics, serving functions like gelling, thickening, and stabilizing. |
?700 |

|
Narmada gelatins ltd |
Narmada Gelatines Ltd is an Indian company specializing in the production of high-quality gelatine. Their gelatine is a highly versatile natural ingredient used in the food, pharmaceutical, and photographic industries for various purposes. |
?4569 |

|
PB gelatin |
PB Gelatins is a global manufacturer of gelatin and collagen peptides, offering a wide range of products for food, pharmaceutical, health, and nutrition applications. Their gelatins are known for their high quality and are used in products like confectioneries, dairy, desserts, and meat products. |
$356 |

|
Rousselot |
Rousselot is a leading global provider of gelatin and collagen solutions, offering a wide range of products for the food, pharmaceutical, and biomedical industries. Their gelatins are known for unique gelling, thickening, and stabilizing properties, making them suitable for various applications. Rousselot emphasizes clean-label ingredients, providing gelatins that are non-allergenic, cholesterol-free, purine-free, and fat-free |
Rp 418 |

|
Dr.oetker |
Dr. Oetker offers a range of gelatin products, including powdered gelatin and gelatin leaves, commonly used to stabilize cream cakes, jellies, and other desserts. Their gelatin is known for its quality and ease of use in various culinary applications |
?699/- |

|
Trobas pvt ltd |
Trobas Gelatine B.V., based in the Netherlands, produces high-quality edible and pharmaceutical gelatine in both granule and powder forms. Their product range includes pig skin gelatine (Type A) with gel strengths from 100 to 300 Bloom and bovine hide gelatine (Type B) with gel strengths from 100 to 250 Bloom. They offer customizable options to meet specific customer requirements. |
?12999 |

|
Butterfly |
Butterfly Veg Gelatine is a plant-based gelling agent, specifically carrageenan, used as a vegetarian alternative to traditional gelatin. It's suitable for various culinary applications, including desserts and confectioneries. |
?102/- |

|
Gelita Deutschland GmbH |
Gelita is a leading manufacturer of gelatin, providing a versatile ingredient used in numerous applications, including food, pharmaceuticals, and specialty industries. Their gelatin is essential in products like gummy bears, yogurts, and pharmaceutical capsules, offering unique gelling, thickening, and stabilizing propertie |
?7999 |

|
NDI neotea |
Neotea offers a range of gelatin powders suitable for various culinary and health applications. Their products are commonly used in making jellies, desserts, puddings, and other confections. Additionally, Neotea's gelatin is utilized in baking for items like candies, marshmallows, cakes, and ice creams |
?1400 |

|
Vital proteins |
Vital Proteins Beef Gelatin is sourced from pasture-raised, grass-fed bovine hides. This unflavored gelatin powder dissolves in hot liquids and gels upon cooling, making it ideal for recipes like gummies, custards, soups, and stews. It's also noted for supporting healthy hair, skin, nails, and joints |
$30 |

|
Great lakes gelatin co. |
Great Lakes Gelatin is a well-known brand offering high-quality, unflavored beef gelatin powder derived from grass-fed cattle. Their gelatin is versatile and commonly used in culinary applications such as making gummies, marshmallows, desserts, and as a thickening agent in recipes. Each serving provides approximately 12 grams of protein and is compatible with various dietary preferences, being gluten-free, kosher, keto-certified, and paleo-friendly |
$25.9 |
PATENTS:
Gelatin patents span diverse applications, including pharmaceutical capsules, edible films, biomedical devices, and adhesives. Innovations improve drug delivery, food stabilization, and encapsulation of oils. Other patents explore plant-based alternatives, gelatin hydrolysates, and nanoparticles. [42-43] (Table 1.11)
Table 1.11 Patents of gelatin
|
Patent number |
Inventor |
Invention |
Year of grant |
Year of expiry |
|
US2423773A |
Hart John Alfred Henry, Lee Edward William |
Purification of gelatin |
1947 |
1960 |
|
US1457466A |
Claude R Smith |
Process of purifying gelatin and glue |
1923 |
1940 |
|
US2191206A |
Schwartz Charles |
Process of purifying gelatin and casein |
1940 |
1957 |
|
EP0689570B1 |
Mats Lilja, Mats Larsson
|
Method for producing gelatin |
1998 |
2014 |
|
CN104231943A |
Reh lina |
Method for manufacturing gelatin |
2014 |
NA |
|
US20090143568A1 |
Robert C. ChangKari KivirikkoThomas, B. NeffDavid, R. OlsenJames ,W. Polarek |
Recombinant gelatins |
2009 |
NA |
|
US20080167447A1 |
Bhanu Manickavasagam |
Extraction of gelatins |
2008 |
NA |
|
US4176117A |
Oudem leon |
Process for obtaining gelatin |
1979 |
1997 |
|
US4780316A |
Werner brox |
Gelatin capsule |
1988 |
2005 |
|
US4369069A |
Wolfgang GraesserPeter, J. KoepffIvan Tomka |
Gelatin, method for producing it and its use |
1983 |
2000 |
CONCLUSION:
Gelatin’s functional properties make it a vital excipient in modern pharmaceutical applications. Its ability to form protective capsules, microencapsulate APIs, and act as a stabilizer has revolutionized drug delivery systems. Advancements in gelatin synthesis, including recombinant and plant-based alternatives, highlight its evolving role in addressing safety, sustainability, and efficiency concerns. As research progresses, gelatin will remain a cornerstone excipient, contributing to innovations in drug delivery, patient compliance, and sustainable pharmaceutical practices.
REFERENCE




Roshani Shah, Christina Viju, Ishika Puralkar, Shristi Singh, A Comprehensive Review on Gelatin: Synthesis, Functional Properties, and Pharmaceutical Uses, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 11, 4576-4592. https://doi.org/10.5281/zenodo.17747701
 10.5281/zenodo.17747701
10.5281/zenodo.17747701