Abstract
In order to determine the amounts of cilnidipine and lisinopril dihydrate, the Dual Wavelength Spectroscopic approach that was proposed was successfully utilized. The method that was created was verified in accordance with ICH Q2 (R1) without any deviations, and based on the findings, it was determined that the current method has the potential to be utilized for the routine examination of raw materials as well as in the formulation of pharmaceuticals. According to the findings of the linearity investigation, the suggested approach was able to produce linear results throughout the concentration range of 4–24 and 10–60 ?g/mL for lisinopril dihydrate and cilnidipine, respectively. It was determined that the proposed method was accurate based on the findings of the precision data and the low levels of the relative standard deviation (RSD). Both the LOD and LOQ values for cilnidipine and lisinopril dihydrate were determined and established. It was discovered that the recovery for lisinopril dihydrate was 98.82%, and the recovery for cilnidipine was 100.24%. The low values of the relative standard deviation (RSD) demonstrate that the procedure was accurate within the range that was defined. The suggested approach was found to be linear, sensitive, exact, and accurate for the simultaneous estimation of lisinopril dihydrate and cilnidipine in bulk and pharmaceutical formulations. This conclusion was reached as a consequence of the successful completion of the validation research and the findings that were discovered.
Keywords
cilnidipine and lisinopril dihydrate, Calcium Channel Blocker, Development and Validation of Analytical Methods.
Introduction
Hypertension is the leading preventable risk factor for cardiovascular disease (CVD) and all-cause mortality worldwide.1,2 In 2024, 31.1% of the global adult population (1.39 billion people) had hypertension, defined as systolic BP ?140 mmHg and/or diastolic BP ?90 mmHg.3 The prevalence of hypertension is rising globally owing to ageing of the population and increases in exposure to lifestyle risk factors including unhealthy diets (i.e. high sodium and low potassium intake and lack of physical activity.3 However, changes in hypertension prevalence are not uniform worldwide. In the past two decades, high-income countries (HICs) experienced a modest decrease in hypertension prevalence, while low and middle-income countries (LMICs) experienced significant increases.3 These disparities in hypertension prevalence trends suggest that health care systems in LMICs could be facing a rapidly increasing burden of hypertension and BP-related cardiovascular diseases, in some cases in addition to a substantial burden of infectious diseases. In this Review, we discuss estimates and trends in mean BP levels and hypertension prevalence, awareness, treatment, and control worldwide. We also examine risk factors for hypertension, strategies for hypertension control, and evidence of the financial burden of hypertension. We conclude by discussing the consequences of current trends in hypertension and areas where more research is needed.
Early knowledge and measurements
In the Yellow Emperor’s Classic of Medicine (first written approximately 200–400 BCE), the Yellow Emperor of China (approximately 2600–2700 BCE) was believed to have talked about the so-called ‘hard pulse disease’, claiming that ‘if too much salt is used in food, the pulse hardens’ and suggested the use of venesection for treatment15,16. Physicians in ancient Egypt (approximately 1500 BCE) and India (approximately 150 BCE) also noted the relationship between pulse quality and the development of afflictions of the heart and brain17. Pulse also had an essential role in ancient Greek medicine, and its relationship with environment and disease was discussed at length by physicians including Hippocrates (460–370 BCE), Erasistratus (304–250 BCE) and Galen (130–210 CE)16,17,18,19. However, these physicians did not note the connection between apoplexy and high blood pressure or hardening of the pulse16.
In 1628, William Harvey described the process of blood flowing out of the heart and then returning to the heart via arteries, peripheries and veins17,20. Nearly 300 years later, blood pressure was discovered, and a reliable method for its measurement was devised. Even before this technology was developed, the work of a few physicians, including Richard Bright and Frederick Akbar Mahomed, led to the first description of essential hypertension in the nineteenth century, for example, hypertension in the absence of renal disease16,17,21. The first accurate, direct measurement of human blood pressure was performed by the surgeon Faivre with the use of a mercury manometer during a limb amputation in 1856, with a reported arterial blood pressure of 115–120?mmHg (refs22,23). Devices for the indirect measurement of blood pressure (that is, to measure the counter-pressure needed to stop the blood flow in an artery) evolved from the first sphygmograph to visualize pulse waves, invented by Karl Vierordt in 1855, to Samuel Siegfried Ritter von Basch’s sphygmomanometer in 1880 (ref.23). In 1896, Scipione Riva Rocci invented an inflatable cuff that compressed around the whole circumference of the arm to apply uniform pressure. The cuff size was later changed to 12?cm in 1901 from the original 5?cm (refs22,23), and it became the prototype of cuffs that continue to be used in modern devices. In 1905, Nikolai Korotkoff, a Russian surgeon, reported a method that uses the tapping sounds detected through a stethoscope at different phases during the deflation of the cuff to determine the pressure at which blood flow was completely blocked, that is, SBP, and the pressure at which blood flow was no longer restrained, that is, DBP22,23. Together, Korotkoff’s auscultatory technique and Rocci’s cuff formed the basis of modern blood pressure measurement devices.
Blood pressure as a risk factor
The quantitative connection between high blood pressure and mortality was first revealed in studies with the use of insurance data at the beginning of the twentieth century24,25. These data also revealed that blood pressure rises with age and is higher in those who have higher weight for their height26. The Framingham Heart Study27 showed a greater risk of coronary heart disease in men and women with hypertension (defined as SBP ?160?mmHg or DBP ?95?mmHg) than in individuals with SBP <140 href="https://www.nature.com/articles/s41569-021-00559-8#ref-CR28" xss=removed title="Vasan, R. S. et al. Impact of high-normal blood pressure on the risk of cardiovascular disease. N. Engl. J. Med. 345, 1291–1297 (2001).">28). The Prospective Studies Collaboration pooled 61 prospective observational studies with 1 million participants in Asia, Australasia, Canada, Europe and the USA and found a doubling of the risk of ischaemic heart disease and stroke with every 20?mmHg and 10?mmHg increase in SBP and DBP, respectively, starting from as low as 115?mmHg for SBP and 75?mmHg for DBP. The Asia Pacific Cohort Studies Collaboration found similar associations in Asian and Australasian populations. On the basis of observational studies, each 10?mmHg increase in SBP is associated with a 45% higher risk of ischaemic heart disease and about a 65% higher risk of ischaemic or haemorrhagic stroke in those aged 55–64 years. The relative risk is inversely associated with age. The observational results were confirmed by data from clinical trials that lowered blood pressure. These trials included the VA Cooperative Trials, Multiple Risk Factor Intervention Trial (MRFIT), and those included in the Blood Pressure Lowering Treatment Trialists’ Collaboration. Other trials, including the Systolic Blood Pressure Intervention Trial (SPRINT), and meta-analyses of trials further showed reductions in CVD events but mixed results for cardiovascular and all-cause mortality with intensive blood pressure lowering to levels below the conventional cut-off for hypertension of 140/90?mmHg, for example, SBP of 120–130?mmHg. However, most of the trials have been performed in Western populations, and trial evidence is especially scarce for Africa, Latin America and South Asia.
Global burden of hypertension
In 2019, the global age-standardised prevalence of hypertension in adults aged 30–79 years was 32% (95% CrI 30–34) in women and 34% (32–37) in men, similar to 1990 levels of 32% (30–35) in women and 32% (30–35) in men (fig,1 ). The stable global prevalence was a net effect of a decrease in high-income countries, and for women also in central and eastern Europe, and an increase in some low-income and middle-income countries. The decline was greater than 12 percentage points in women in several high-income countries (posterior probability [PP] of the observed decline being a true decline >0·98 for all country and sex combinations; fig,1). By contrast, age-standardised prevalence increased, or at best remained unchanged, in most low-income and middle-income countries (fig,1). The increase was 10–15 percentage points among men in three countries and among women in four countries (PP 0·85–0·99).
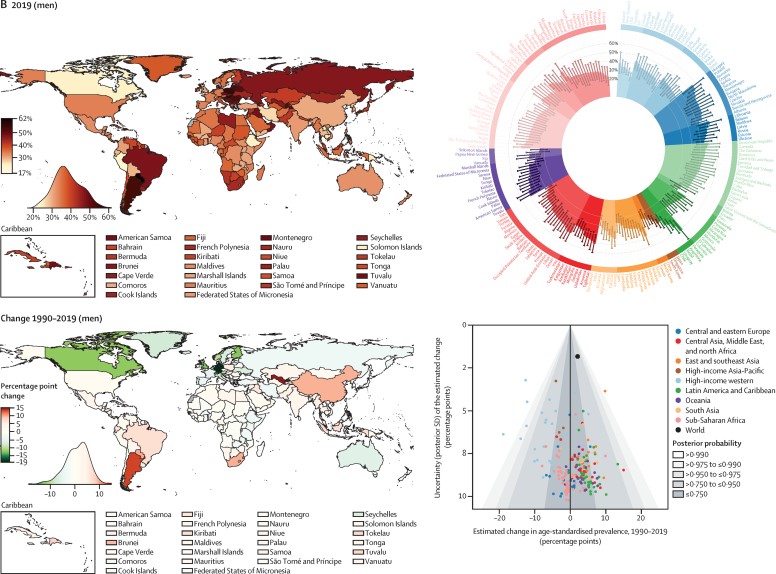
Fig: 1 Both women (A) and men (B) have a higher prevalence of hypertension in 2019, and this prevalence has changed from 1990 to 2019. The density plot that is located next to each map illustrates the distribution of estimations regarding the various countries. In every panel, the graph located in the upper right corner displays the data that are arranged among regions and super-regions with credible intervals of 95%. Between the years 1990 and 2019, the graph in the bottom right corner of each panel illustrates the change in the prevalence of hypertension in proportion to the uncertainty of the change as assessed by posterior standard deviation. The posterior probability of an estimated rise or drop being an actual increase or decrease is represented by the shaded areas in the graph. Each point represents a different nation. For the numerical results, please refer to the appendix (pages 33–46). Nationally, prevalence of hypertension in 2019 was lowest in Canada and Peru for both men and women; in Taiwan, South Korea, Japan, and some countries in western Europe for women; and in some low-income and middle-income countries for men. Age-standardized prevalence in all of these countries was less than 24% for women and less than 25% for men in 2019. Hypertension prevalence was highest throughout central and eastern Europe, central Asia, Oceania, southern Africa, and some countries in Latin America and the Caribbean fig,1. For women in two countries and men in nine countries, age-standardized prevalence surpassed 50% fig,1.
Arterial Stiffness
Arterial stiffness refers to a reduction in elasticity and distensibility of arteries, and pulse wave velocity (PWV) is often used to represent the degree of stiffness in large arteries. An increase in PWV indicates severe arterial stiffness and impaired in arterial dilatation capacity (8). Arterial stiffness has been closely associated with an increased risk of essential hypertension (9, 10), especially the isolated systolic hypertension (11). Vice versa, systolic BP is also associated with a clinically significant progression of arterial stiffness (12). It is still a “chicken and egg question” that elevated blood pressure and arterial stiffness which come first. Arterial stiffness can be classified into functional arterial stiffness and structural arterial stiffness (13). Functional arterial stiffness is mainly related to the contractile function of vascular smooth muscle cells (VSMCs) which is influenced by a variety of factors (fig, 2). Among them, an increase in intracellular calcium ion (CA2+) concentration can directly influence VSMCs. And calcium channel blockers (CCBs), which are widely used in clinical settings, are to reduce intracellular calcium concentration in SMCs (14) and thus controlling BP. The nitric oxide (NO)-nitric oxide-sensitive guanylate cyclase (NOsGC)-cGMP pathway is also a well-studied pathway that is closely related to the contractile function of VSMCs. NO-NOsGC-cGMP pathway begins in vascular endothelial cells and regulates VSMCs contraction through a series of signaling (15–17). Injectable antihypertensive drugs, such as sodium nitroprusside and nitrates, all exert their vasodilatory effects through the NO-NOsGC-cGMP pathway.
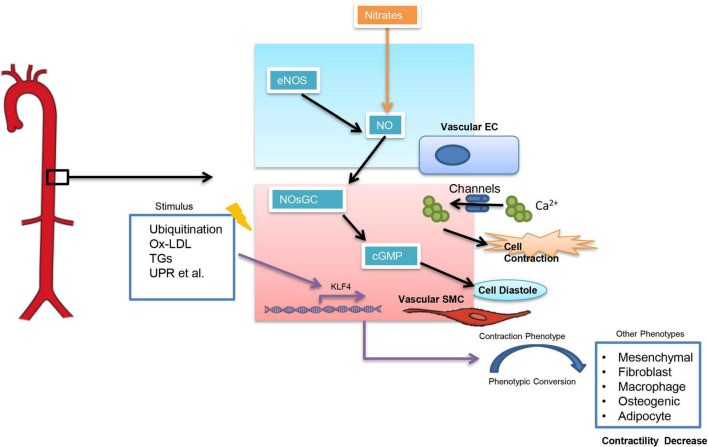
Fig: 2 The contractile action of smooth muscle cells is modified by a number of different factors. KLF4 stands for Kruppel-Like Factor 4, TGs stands for triglycerides, and UPR stands for unfolded protein response. Ox-LDL stands for oxidised low-density lipoprotein.
Water-sodium retention
Water-sodium retention is a key cause of abnormal increases in intravascular fluid volume. Diuretics (especially thiazide diuretics) are important in the control of hypertension caused by water-sodium retention (24). Except secondary hypertension resulted from renal dysfunction, there is also a group of hypertensive patients related to water-sodium retention in essential hypertension, namely salt-sensitive hypertension. High-salt intake is an important trigger in essential hypertension caused by water-sodium retention. Not all people will develop increased BP after consuming excessive salt. According to the blood pressure reactivity to salt-intake, patients are called salt-sensitive and salt-resistant, respectively (25). Multiple factors may contribute to the development of salt-sensitive hypertension, including age, obesity, genetic background, and maternal conditions during fetal life etc. (26), but the underlying mechanisms of salt-sensitive hypertension are not fully understood. Studies showed that low potassium activates (turns on) the system by hyperpolarizing the membrane, thereby driving Cl– out of the cell and off the inhibitory binding site on WNK (with no lysine [K]) kinases. Once disinhibited, WNK kinases phosphoactivate SPAK (STE20/SPS1-related proline/alanine–rich kinase), which in turn phosphoactivates nine–rich kinase co-transporter (NCC), and this called the Potassium Switch theory (27–29), which is one of the important theories on the pathogenesis of salt-sensitive hypertension. Recently, increasing evidence showed that intestinal flora is closely associated with salt-sensitive hypertension (30, 31) (fig, 3). The fecal microbiota of healthy rats could significantly lower BP in high-salt diet induced hypertensive (hSIH) rats, whereas the fecal microbiota of hSIH rats had opposite effects (32). Adoptive transfer of fecal material from conventionally housed high-salt diet–fed mice to germ-free mice predisposed them to increased inflammation and hypertension, the reason for this result may associated with an increase in Firmicutes, Proteobacteria, and genus Prevotella bacteria (33).
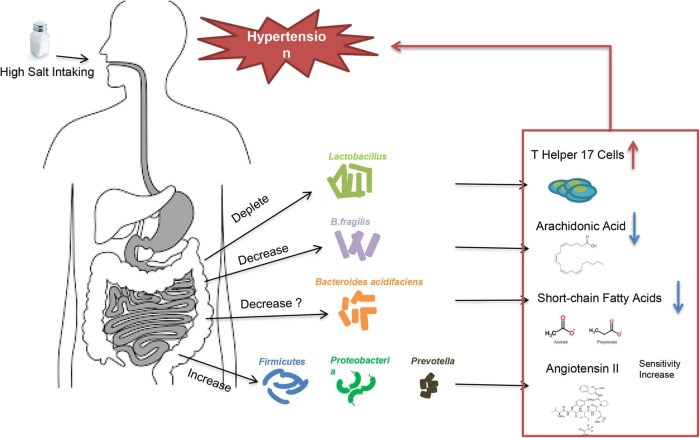
Fig: 3 When a person consumes a lot of salt, the gut flora causes them to have high blood pressure.
The underlying mechanisms for intestinal flora to lead to increased BP are still under investigation. There is evidence that high-salt intake depleted Lactobacillus to induce T helper 17 cells and to promote hypertension (34). Another study demonstrated that high-salt dietry reduced the levels of B. fragilis and arachidonic acid in the intestine, which increased intestinal-derived corticosterone production and corticosterone levels in serum and intestine, thereby promoting BP elevation (32). In deoxycorticosterone acetate (DOCA)–salt mice model of hypertension, short-chain fatty acids released by the fermentation of fiber from the intestinal flora are associated with lower BP levels, and this may be closely related to the increase in Bacteroides acidifaciens (35). Notably, the intestinal flora is not only involved in salt sensitivity, it also participate in other underlying mechanisms of hypertension (36), including RAAS (37–39), vascular endothelium (40), and renal dysfunction (41) etc. A recent large intestinal flora sequencing study demonstrated the role of intestinal flora in human was extremely complex (42). Intestinal flora also has the potential to be an independent mechanism of essential hypertension.
Angiotensin-converting enzyme inhibitors and angiotensin II type I receptor blockers
ACEIs and ARBs have been the cornerstone of RAAS inhibition for years and are key therapeutic options in patients with hypertension (HTN), reducing cardiovascular morbidity and mortality and improving renal outcomes. In the HOPE (Heart Outcomes Prevention Evaluation) 5, MICRO-HOPE (The Microalbuminuria, Cardiovascular, and Renal Outcomes in HOPE) 6, EUROPA (European Trial on Reduction of Cardiac Events with Perindopril in Stable Coronary Artery Disease) 7, SOLVD (Studies of Left Ventricular Dysfunction) 8, and Captopril Prevention Project 9 studies, ACEIs were beneficial in reducing rates of death, myocardial infarction, stroke, coronary revascularization, cardiac arrest, and heart failure, complications related to diabetes and heart failure. Both the RENAAL 10 and IDNT 11 trials demonstrated a renoprotective effect of RAAS inhibition in nephropathy due to diabetes. ACEIs and ARBs are considered to be equally beneficial on the basis of studies such as ONTARGET 12, which compared telmisartan and ramipril, and DETAIL 13, which compared telmisartan with enalapril and found no difference in progression of diabetic nephropathy. Azilsartan, a novel ARB, has been shown to have superior efficacy in reducing blood pressure (BP) when compared with olmesartan or valsartan 14– 16. Azilsartan 80 mg reduces BP more efficaciously than the optimal and highest tolerated dosage of both olmesartan (40 mg) and valsartan (320 mg) using 24-hour ambulatory BP monitoring and without increase in adverse effects 14. A recent analysis of a German registry confirmed these results; among 3,849 patients with essential HTN, 61% of patients who initiated therapy with azilsartan achieved a BP target of less than 140/90 mmHg compared with 56% of those who were started on an ACEI 17. Azilsartan may be more effective at lowering BP because of its more potent ability to block AT 1 receptors 18. However, only a prospective, randomized, dose-escalation study can truly test whether azilsartan is superior to other ARBs at lowering BP.
Direct renin inhibitors
Renin secretion, a rate-limiting step, is the first step of the RAAS cascade. Renin has a unique specificity for its substrate angiotensin. Inhibition of renin provides an attractive option to inhibit the RAAS from its origin. The development of DRI started more than 30 years ago 19, but there were issues with potency, bioavailability, and cost. Currently, aliskiren is the only approved DRI for use in HTN 20, and a significant BP reduction has been demonstrated in patients with essential HTN 21, 22. Aliskiren is well tolerated and has a similar dose-dependent BP reduction in hypertensive patients as ARBs and a safety profile similar to that of placebo 23, 24. However, several recent studies have shown either no benefit or even harmful effects of aliskiren in certain populations. The ALTITUDE (Aliskiren Trial in Type 2 Diabetes Using Cardio-Renal Endpoints) 25 trial randomly assigned patients with type 2 diabetes and chronic kidney disease (CKD) or with cardiovascular disease (or with all three) already receiving an ACEI or ARB to aliskiren or placebo. Although there was a lower BP in the aliskiren arm, there was no reduction in the primary composite outcome, which included cardiovascular and renal events and mortality (hazard ratio [HR] 1.08, 95% confidence interval [CI] 0.98–1.20). In the ATMOSPHERE 26 trial (Aliskiren Trial to Minimize Outcomes in Patients with Heart failure), the addition of aliskiren to enalapril in patients with chronic heart failure was not associated with reduction in adverse outcomes (see “Recent clinical trials” section for details). Similarly, no improvement in coronary atherosclerosis in pre-hypertensive patients (AQUARIUS) 27 or improvement in cardiovascular outcomes in patients hospitalized with heart failure (ASTRONAUT) 28 was seen with aliskiren compared with placebo. Given the lack of demonstrated benefit and increased rates of adverse events such as hyperkalemia, hypotension, and renal impairment as seen in ASTRONAUT when combined with ACEIs or ARBs, the current use of aliskiren is limited 28.
Mineralocorticoid receptor antagonists
MRAs competitively inhibit mineralocorticoid receptors and decrease the number of epithelial sodium channels in the distal renal tubule 29. Spironolactone, an MRA, has long been used for the treatment of HTN; however, its non-specificity for mineralocorticoid receptors manifests as anti-androgenic and progestational effects 30, 31. Spironolactone was found to be the most effective add-on anti-hypertensive drug when compared with doxazosin and bisoprolol in treating resistant HTN in the PATHWAY-2 trial 32. This trial supports the important role of sodium retention in resistant HTN. Eplerenone, an MRA with lower affinity to progesterone and androgen receptors than spironolactone 29, has been shown to be efficacious even using ambulatory monitoring studies 33 and safe in the management of HTN. When compared with enalapril 34, losartan 35, and amlodipine 36, eplerenone monotherapy was as efficacious in treating HTN. Currently, third- and fourth-generation MRAs are being developed to have the potency of spironolactone and the selectivity of eplerenone 37. Finerenone, a novel non-steroidal MRA, has a greater affinity to the mineralocorticoid receptor than does eplerenone 38 and greater selectivity than does spironolactone 39. The Mineralocorticoid Receptor Antagonist Tolerability Study (ARTS) showed that finerenone (2.5–10 mg per day), in patients with CKD and albuminuria, decreased albuminuria with lower rates of hyperkalemia compared with spironolactone 40. With the recent development of patiromer, a non-absorbed potassium binder, a safe option for the treatment of chronic hyperkalemia is available, maintaining and using RAAS inhibitors including spironolactone, in CKD and heart failure 41. Patiromer has also been shown to decrease potassium and aldosterone levels in patients with CKD and hyperkalemia on RAAS inhibitors independent of renin activity 42. The recent ARTS-DN (Mineralocorticoid Receptor Antagonist Tolerability Study–Diabetic Nephropathy) study demonstrated greater reduction in albuminuria with the addition of finerenone to ACEI or ARB in patients with diabetic nephropathy compared with placebo.43, 44.
Aldosterone synthase inhibitors
Another way of blocking the effects of mineralocorticoid receptor activation is to inhibit aldosterone formation. LCI699 is a potent first-in-class aldosterone synthase inhibitor. In patients with primary aldosteronism, LCI699 (up to 1.0 mg twice a day) caused modest reduction in 24-hour systolic BP (SBP) and office SBP compared with placebo 45. One mg of LCI699 was not superior to eplerenone 50 mg in reducing ambulatory SBP in patients with stage 1 or 2 HTN 46. LCI699 significantly lowered office and ambulatory BP in patients with primary HTN, but 20% of the patients on LCI699 developed blunted cortisol release 46. Because of this non-specificity, the development of LCI699 has been stopped in favor of developing more specific inhibitors.
Aminopeptidase A inhibitor
Functional RAAS may be present in the brain 47, and several animal models have demonstrated that hyperactivity of brain RAAS may contribute to HTN 48, 49. Brain aminopeptidase A (APA) converts angiotensin II to angiotensin III. APA inhibition by EC33 has been shown to be a novel anti-hypertensive agent in animals 50– 52. QGC001, an orally active prodrug of EC33, was tolerated in healthy normotensive human males but with no changes in renin, aldosterone, BP, or heart rate 53– 56. This study demonstrates that QGC001, the first drug in its class, can be safely administered to humans, but further studies are required to assess the safety and efficacy of QGC001 in patients with HTN. Additional research on novel ways to block the RAAS, including development of prorenin blockade, gene- and vaccine-based strategies, and targeting ACE2, is being developed but has not yet translated into the clinic.
CONCLUSION
Although there is regional variability in the outlook for hypertension over the next 5 to 10 years, It is clear overall that the prevalence of hypertension and, therefore, the associated global burden attributable to hypertension, will increase. Global population growth and aging will largely contribute to this increase – 1.5 billion people are expected to be affected by 2025– which will be focused in low and middle income countries. However, these adverse trends in disease burden will be variably offset by improvements in prevention, awareness and treatment. The size of improvements in each of these 3 areas will vary from non-existent (-hypertension prevention could even worsen in some parts of the world, as exposure to factors that promote raised BP increases) to substantially large and important elsewhere in the world. Overall, prevention will probably contribute least to any improvement in BP-associated disease burden. This is because 80% of the world is in the process of developing, which hitherto has inevitably been associated with increased exposure to the main environmental determinants of raised BP such as excess intake of calories, alcohol and salt. Food and drink industries, governments and education systems would be required to cooperate in order to reverse this pattern. Implementation of preventive strategies has largely been limited to high income countries. Despite reasonably compelling evidence to the contrary, recommendations that the general population should restrict salt intake have been questioned on the basis of largely suboptimal observational data Such confusion worsens an already very difficult public health challenge. Data show that only approximately half of people with hypertension are aware of their condition, and the Lancet Commission on hypertension identified improving awareness of hypertension is a critical action needed to improve the current disease burden. The global BP awareness campaign promoted by the International Society of Hypertension whereby World Hypertension Day was extended to become May Measurement Month (MMM) in 2017 could contribute substantially to improving rates of routine BP screening around the world. Over 1.20 million adults (?18 years) from >100 countries were screened as part of MMM and the ensuing data allied to health-economic analyses will be used to persuade policy makers in each country that enhanced local BP screening and treatment facilities are wise financial investments.
REFERENCES
- Luft FC Twins in Cardiovascular Genetic Research. Hypertension 37, 350–356 (2001).
- Fagard R et al. Heritability of Conventional and Ambulatory Blood Pressures?: A Study in Twins. Hypertension 26, 919–924 (1995).
- Surendran P et al. Trans-ancestry meta-analyses identify rare and common variants associated with blood pressure and hypertension. Nat. Genet 48, 1151–1161 (2016).
- Ehret GB et al. The genetics of blood pressure regulation and its target organs from association studies in 342,415 individuals. Nat. Genet 48, 1171–1184 (2016).
- Liu C et al. Meta-analysis identifies common and rare variants influencing blood pressure and overlapping with metabolic trait loci. Nat. Genet 48, 1162–1170 (2016).
- Dominiczak A, Delles C & Padmanabhan S Genomics and Precision Medicine for Clinicians and Scientists in Hypertension. Hypertension 69, e10–e13 (2017).
- Lifton RP, Gharavi AG & Geller DS Molecular Mechanisms of Human Hypertension. Cell 104, 545–556 (2001).
- Ehret GB & Caulfield MJ Genes for blood pressure: an opportunity to understand hypertension. Eur. Heart J 34, 951–961 (2013).
- Maass PG et al. PDE3A mutations cause autosomal dominant hypertension with brachydactyly. Nat. Genet 47, 647–653 (2015).
- Forouzanfar MH et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388, 1659–1724 (2016).
- Blood Pressure Lowering Treatment Trialists’ Collaboration. Blood pressure-lowering treatment based on cardiovascular risk: a meta-analysis of individual patient data. Lancet 384, 591–598 (2014).
- Page LB, Damon A & Moellering RC Antecedents of cardiovascular disease in six Solomon Islands societies. Circulation 49, 1132–46 (1974).
- Poulter NR, Prabhakaran D & Caulfield M Hypertension. Lancet 386, 801–812 (2015).
- Rose G & Day S The population mean predicts the number of deviant individuals. BMJ 301, 1031–4 (1990).
- Mills KT et al. Global Disparities of Hypertension Prevalence and Control: A Systematic Analysis of Population-based Studies from 90 Countries. Circulation 134, 441–450 (2016).
- Forouzanfar MH et al. Global Burden of Hypertension and Systolic Blood Pressure of at Least 110 to 115 mm Hg, 1990–2015. JAMA 317, 165 (2017)
- Lewington S et al. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet (London, England) 360, 1903–13 (2002).
- Rapsomaniki E et al. Blood pressure and incidence of twelve cardiovascular diseases: lifetime risks, healthy life-years lost, and age-specific associations in 1·25 million people. Lancet 383, 1899–1911 (2014).
- Stamler J, Stamler R & Neaton JD Blood pressure, systolic and diastolic, and cardiovascular risks. US population data. Arch. Intern. Med 153, 598–615 (1993).
- Klag MJ et al. Blood pressure and end-stage renal disease in men. N. Engl. J. Med 334, 13–8 (1996).
- Goff DC et al. 2013 ACC/AHA Guideline on the Assessment of Cardiovascular Risk. J. Am. Coll. Cardiol 63, 2935–2959 (2014).
- Hall ME & Hall JE Pathogenesis of Hypertension. Hypertension: A Companion to Braunwald’s Heart Disease 33–51 (2018). doi: 10.1016/b978-0-323-42973-3.00005-6
- Gangwisch JE A review of evidence for the link between sleep duration and hypertension. Am. J. Hypertens 27, 1235–42 (2014).
- Palagini L et al. Sleep loss and hypertension: a systematic review. Curr. Pharm. Des 19, 2409–19 (2013).
- Mikael L. de R. et al. Vascular Aging and Arterial Stiffness. Arq. Bras. Cardiol 109, 253–258 (2017).
- Sindler AL et al. Nitrite supplementation reverses vascular endothelial dysfunction and large elastic artery stiffness with aging. Aging Cell 10, 429–37 (2011).
- Steppan J, Barodka V, Berkowitz DE & Nyhan D Vascular stiffness and increased pulse pressure in the aging cardiovascular system. Cardiol. Res. Pract 2011, 263585 (2011).
- Page IH Pathogenesis of Arterial Hypertension. Jama J. Am. Med. Assoc 140, 451 (1949).
- Harrison DG The Mosaic Theory revisited: common molecular mechanisms coordinating diverse organ and cellular events in hypertension. J. Am. Soc. Hypertens 7, 68–74 (2013).
- Feng W, Dell’Italia LJ & Sanders PW Novel Paradigms of Salt and Hypertension. J. Am. Soc. Nephrol 28, 1362–1369 (2017).
- Wilck N et al. Salt-responsive gut commensal modulates TH17 axis and disease. Nature 551, 585 (2017).
- Singh A & Williams GH Textbook of nephro-endocrinology
- Varagic J, Ahmad S, Nagata S & Ferrario CM ACE2: angiotensin II/angiotensin-(1–7) balance in cardiac and renal injury. Curr. Hypertens. Rep 16, 420 (2014).
- Ferrario CM ACE2: more of Ang-(1–7) or less Ang II? Curr. Opin. Nephrol. Hypertens 20, 1–6 (2011).
- Zimmerman D & Burns KD Angiotensin-(1–7) in kidney disease: a review of the controversies. Clin. Sci 123, 333–346 (2012).
- Zhou ZH & Bubien JK Nongenomic regulation of ENaC by aldosterone. Am. J. Physiol. Cell Physiol 281, C1118–30 (2001).
- McCurley A & Jaffe IZ Mineralocorticoid receptors in vascular function and disease. Mol. Cell. Endocrinol 350, 256–265 (2012).
- Kerkelä R, Ulvila J & Magga J Natriuretic Peptides in the Regulation of Cardiovascular Physiology and Metabolic Events. J. Am. Heart Assoc 4, e002423 (2015).
- Woodard GE & Rosado JA Chapter 3 Natriuretic Peptides in Vascular Physiology and Pathology. in International review of cell and molecular biology 268, 59–93 (2008).
- Curry F-RE Atrial natriuretic peptide: an essential physiological regulator of transvascular fluid, protein transport, and plasma volume. J. Clin. Invest 115, 1458–1461 (2005).
- Armaly Z, Assady S & Abassi Z Corin: a new player in the regulation of salt-water balance and blood pressure. Curr. Opin. Nephrol. Hypertens 22, 713–722 (2013).
- Schlueter N et al. Metabolic actions of natriuretic peptides and therapeutic potential in the metabolic syndrome. Pharmacol. Ther 144, 12–27 (2014).
- Khaddaj Mallat R, Mathew John C, Kendrick DJ & Braun AP The vascular endothelium: A regulator of arterial tone and interface for the immune system. Crit. Rev. Clin. Lab. Sci 54, 458–470 (2017).
- Sandoo A, van Zanten JJCSV, Metsios GS, Carroll D & Kitas GD The endothelium and its role in regulating vascular tone. Open Cardiovasc. Med. J 4, 302–12 (2010).
- Spieker LE, Flammer AJ & Lüscher TF The Vascular Endothelium in Hypertension. The Vascular Endothelium II 249–283 doi: 10.1007/3-540-36028-x_8
- Ayub T, Khan SN, Ayub SG, Dar R & Andrabi KI Reduced nitrate level in individuals with hypertension and diabetes. J. Cardiovasc. Dis. Res 2, 172–176 (2011).
- Panza JA, Casino PR, Badar DM & Quyyumi AA Effect of increased availability of endothelium-derived nitric oxide precursor on endothelium-dependent vascular relaxation in normal subjects and in patients with essential hypertension. Circulation 87, 1475–1481 (1993).
- Kohan DE & Barton M Endothelin and endothelin antagonists in chronic kidney disease. Kidney Int 86, 896–904 (2014).
- Serrano-Ponz M et al. Temporal profiles of blood pressure, circulating nitric oxide, and adrenomedullin as predictors of clinical outcome in acute ischemic stroke patients. Mol. Med. Rep 13, 3724–34 (2016).
- Vendégh Z et al. Calcitonin gene-related peptide, substance P, nitric oxide and epinephrine modulate bone marrow micro circulation of the rabbit tibia and femur. Clin. Hemorheol. Microcirc 45, 9–17 (2010).
- Yu M et al. Antihypertensive effect of glucagon-like peptide 1 in Dahl salt-sensitive rats. J. Hypertens 21, 1125–1135 (2003).
- Popolo A, Autore G, Pinto A & Marzocco S Oxidative stress in patients with cardiovascular disease and chronic renal failure. Free Radic. Res 47, 346–356 (2013).
- Lazich I & Bakris GL Endothelin Antagonism in Patients with Resistant Hypertension and Hypertension Nephropathy. Contributions to Nephrology 223–234 (2011). doi: 10.1159/000328988
- Dharmashankar K & Widlansky ME Vascular Endothelial Function and Hypertension: Insights and Directions. Curr. Hypertens. Rep 12, 448–455 (2010).
- HEYMANS C & DELAUNOIS AL Fundamental role of the tone and resistance to stretch of the carotid sinus arteries in the reflex regulation of blood pressure. Science 114, 546–7 (1951). [PubMed] [Google Scholar]
- Pijacka W et al. Carotid sinus denervation ameliorates renovascular hypertension in adult Wistar rats. J. Physiol 594, 6255–6266 (2016).
- de Leeuw PW et al. Sustained Reduction of Blood Pressure With Baroreceptor Activation TherapyNovelty and Significance. Hypertension 69, 836–843 (2017).
- Grassi G et al. Excessive Sympathetic Activation in Heart Failure With Obesity and Metabolic Syndrome: Characteristics and Mechanisms. Hypertension 49, 535–541 (2007).
- Mancia G & Grassi G The Autonomic Nervous System and Hypertension. Circ. Res 114, 1804–1814 (2014).
- Augustyniak RA et al. Sympathetic nerves and the progression of chronic kidney disease during 5/6 nephrectomy: Studies in sympathectomized rats. Clin. Exp. Pharmacol. Physiol 37, 12–18 (2010).
- Augustyniak RA, Tuncel M, Zhang W, Toto RD & Victor RG Sympathetic overactivity as a cause of hypertension in chronic renal failure. J. Hypertens 20, 3–9 (2002).
- DiBona GF Sympathetic Nervous System and Hypertension. Hypertension 61, 556–560 (2013).
- Grassi G, Mark A & Esler M The Sympathetic Nervous System Alterations in Human Hypertension. Circ. Res 116, 976–990 (2015).
- Grassi G, Cattaneo BM, Seravalle G, Lanfranchi A & Mancia G Baroreflex Control of Sympathetic Nerve Activity in Essential and Secondary Hypertension. Hypertension 31, 68–72 (1998).
- Smith PA; Graham LN; Mackintosh AF; Stoker JB; Mary D Relationship between central sympathetic activity and stages of human hypertension. Am. J. Hypertens 17, 217–222 (2004).
- Fujita T Mechanism of Salt-Sensitive Hypertension: Focus on Adrenal and Sympathetic Nervous Systems. J. Am. Soc. Nephrol 25, 1148–1155 (2014).
- Mu S et al. Epigenetic modulation of the renal ?-adrenergic–WNK4 pathway in salt-sensitive hypertension. Nat. Med 17, 573–580 (2011).
- Harrison DG & Bernstein KE Inflammation and Immunity in Hypertension. Hypertension: A Companion to Braunwald’s Heart Disease 60–69 (2018). doi: 10.1016/b978-0-323-42973-3.00007-x
- Devallière J & Charreau B The adaptor Lnk (SH2B3): An emerging regulator in vascular cells and a link between immune and inflammatory signaling. Biochem. Pharmacol 82, 1391–1402 (2011).
- Rodriguez-Iturbe B Autoimmunity in the Pathogenesis of Hypertension. Hypertens. (Dallas, Tex. 1979) 67, 477–83 (2016).
- Mattson DL et al. Genetic mutation of recombination activating gene 1 in Dahl salt-sensitive rats attenuates hypertension and renal damage. AJP Regul. Integr. Comp. Physiol 304, R407–R414 (2013).
- Roush GC et al. Prognostic impact from clinic, daytime, and night-time systolic blood pressure in nine cohorts of 13?844 patients with hypertension. J. Hypertens 32, 2332–2340 (2014).
- Stergiou GS et al. Methodology and technology for peripheral and central blood pressure and blood pressure variability measurement. J. Hypertens 34, 1665–1677 (2016).
- Parati G et al. European Society of Hypertension Practice Guidelines for home blood pressure monitoring. J. Hum. Hypertens 24, 779–785 (2010).
- O’Brien E et al. European Society of Hypertension Position Paper on Ambulatory Blood Pressure Monitoring. J. Hypertens 31, 1731–1768 (2013)
- Muntner P & Whelton PK Using Predicted Cardiovascular Disease Risk in Conjunction With Blood Pressure to Guide Antihypertensive Medication Treatment. J. Am. Coll. Cardiol 69, 2446–2456 (2017).
- Mancia G et al. 2013 ESH/ESC Guidelines for the management of arterial hypertension. J. Hypertens 31, 1281–1357 (2013).
- Pickering TG Recommendations for Blood Pressure Measurement in Humans and Experimental Animals: Part 1: Blood Pressure Measurement in Humans: A Statement for Professionals From the Subcommittee of Professional and Public Education of the American Heart Association Cou. Circulation 111, 697–716 (2005).
- Whelton PK The elusiveness of population-wide high blood pressure control. Annu. Rev. Public Health 36, 109–30 (2015).
- Primatesta P & Poulter NR Improvement in hypertension management in England: results from the Health Survey for England 2003. J. Hypertens 24, 1187–1192 (2006).
- Ashworth M, Medina J & Morgan M Effect of social deprivation on blood pressure monitoring and control in England: a survey of data from the quality and outcomes framework. BMJ 337, a2030–a2030 (2008).
- Serumaga B et al. Effect of pay for performance on the management and outcomes of hypertension in the United Kingdom: interrupted time series study. BMJ 342, d108–d108 (2011).
- Poulter NR & Lackland DT May Measurement Month: a global blood pressure screening campaign. Lancet 389, 1678–1680 (2017).
- He J et al. Migration, blood pressure pattern, and hypertension: the Yi Migrant Study. Am. J. Epidemiol 134, 1085–101 (1991).
- Poulter NR et al. The Kenyan Luo migration study: observations on the initiation of a rise in blood pressure. BMJ 300, 967–72 (1990).
- Rosenthal T The effect of migration on hypertension and other cardiovascular risk factors: A review. J. Am. Soc. Hypertens 8, 171–191 (2014).
- Klag MJ et al. The contribution of urinary cations to the blood pressure differences associated with migration. Am. J. Epidemiol 142, 295–303 (1995).
- The effects of nonpharmacologic interventions on blood pressure of persons with high normal levels. Results of the Trials of Hypertension Prevention, Phase I. JAMA 267, 1213–20 (1992).
- Whelton PK et al. Efficacy of nonpharmacologic interventions in adults with high-normal blood pressure: results from phase 1 of the Trials of Hypertension Prevention. Trials of Hypertension Prevention Collaborative Research Group. Am. J. Clin. Nutr 65, 652S–660S (1997).
- Effects of weight loss and sodium reduction intervention on blood pressure and hypertension incidence in overweight people with high-normal blood pressure. The Trials of Hypertension Prevention, phase II. The Trials of Hypertension Prevention Collaborative Research Group. Arch. Intern. Med 157, 657–67 (1997).
- Sacks FM et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N. Engl. J. Med 344, 3–10 (2001).
- Whelton PK et al. Effects of oral potassium on blood pressure. Meta-analysis of randomized controlled clinical trials. JAMA 277, 1624–32 (1997).
- Aburto NJ et al. Effect of increased potassium intake on cardiovascular risk factors and disease: systematic review and meta-analyses. BMJ 346, f1378–f1378 (2013).
- Whelton SP, Chin A, Xin X & He J Effect of aerobic exercise on blood pressure: a meta-analysis of randomized, controlled trials. Ann. Intern. Med 136, 493–503 (2002).
- Xin X et al. Effects of alcohol reduction on blood pressure: a meta-analysis of randomized controlled trials. Hypertens. (Dallas, Tex. 1979) 38, 1112–7 (2001).
- Roerecke M et al. The effect of a reduction in alcohol consumption on blood pressure: a systematic review and meta-analysis. Lancet Public Heal 2, e108–e120 (2017).
- Appel LJ et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N. Engl. J. Med 336, 1117–24 (1997).
- Whelton PK Hypertension curriculum review: epidemiology and the prevention of hypertension. J. Clin. Hypertens. (Greenwich) 6, 636–42 (2004).
- Whelton PK et al. Primary prevention of hypertension: clinical and public health advisory from The National High Blood Pressure Education Program. JAMA 288, 1882–8 (2002).
- Whelton PK et al. Sodium Reduction and Weight Loss in the Treatment of Hypertension in Older Persons. JAMA 279, 839 (1998)


 Rohit Kumar Dwivedi*
Rohit Kumar Dwivedi*



 10.5281/zenodo.14586971
10.5281/zenodo.14586971