
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmaceutics, Pulla Reddy Institute of Pharmacy, Dundigal, Hyderabad, Telangana, India, 502313
Liposome was derived from two Greek words “Lipos” meaning fat and “Soma” meaning body. Liposomes where spherical shaped vesicles and have diameter of 50-300 nm. consist of phospholipids and cholesterol vesicles which are under extensive investigation as drug carriers for improving the bioavailability and delivery of therapeutic agents. Liposomes have immense capability to prevent the degradation of drugs, reduce side effects and thus increasingly used for targeted drug delivery. The drugs can either incorporated inside aqueous space (hydrophilic drugs) or inside phospholipids bilayer (hydrophobic drugs). Liposomes can be manufactured in different lipid compositions it shows variation in particle size, Size distribution, surface, number of lamella and encapsulation efficacy. The present review paper explains about introductory, classification of liposomes, structural components of liposomes, methods of preparation and application of liposomes are explained detailed. They are recently used for various nanoscale drugs formulation. The possible use of liposomes in the therapy of cancer has also been investigated. Because liposomes are non-toxic, biodegradable, immunogenic and biocompatible. These liposomes are generally administrated by intra-venous route. The formulated liposome is evaluated by Zeta Sizer, entrapment efficiency. Research on liposome technology has progressed from conventional vesicles to ‘second- generation liposomes. Liposomes with modified surfaces have also been developed using several molecules such as glycolipids or sialic acid, antineoplastic agents, doxorubicin and daunorubicin and cytarabine are advanced stages of clinical testing in humans.
Liposomes are vesicular, colloidal structures made up of an equal number of aqueous compartments encircled by one or more lipid bilayers. The sphere-shaped shell contained a liquid which contains proteins, peptides, hormones, enzymes, antibiotics, antifungals and anti-cancer drugs. A free drug injected into the bloodstream usually reaches a therapeutic level for a brief period of time because of excretion and metabolism.1Liposomes are concentric, bilayered vesicles with a diameter of 0.01 to 5.0 µm. They can be composed of membrane proteins, long chains of fatty acids, glycolipids, cholesterol, sphingolipids and non-toxic surfactants.2When phospholipids are dispersed in water, vesicular structures known as liposomes. These structures have an inner aqueous phase is surrounded by phospholipid bilayer membranes. Encased in a sphere-shaped container, the liquid interior held materials such as proteins, enzymes, hormones, antimycotic and anticancer medicines and even plasmids.3 The majority of liposomes are made of biocompatible and biodegradable substances that can hold hydrophilic and hydrophobic molecules together on a single substrate. Current studies
concentrate more on the development of multifunctional liposomes with sophisticated in vitro characteristics and long-circulating stealth liposomes.4 The British hematologist Dr. Alec D. Bangham made the discovery of liposomes 54 years ago, which are now multifaceted tool used in biology, biochemistry and medicine. In 1960, liposomes were employed as a mean of transporting a variety of substances via their aqueous compartment. Variations in liposomes size, structure, charge and lamellarity can be produced and processed. Pharmaceutical companies currently sell liposomal antifungal and antitumor drugs.5
Advantages:6
• It offers specific passive targeting to tumor tissues.
• Improved efficacy and therapeutic index.
• Improved stability by encapsulation.
• Enhanced pharmacokinetic effects.
• Reduced toxicity of encapsulated compounds.
Disadvantages: 7
• Drugs that dissolve in water have poor entrapment efficiency.
• It can be difficult to remove organic solvent.
• Particle size is not controlled in large vesicles.
• Sterilization is required.
• Time consuming process.
• Low solubility
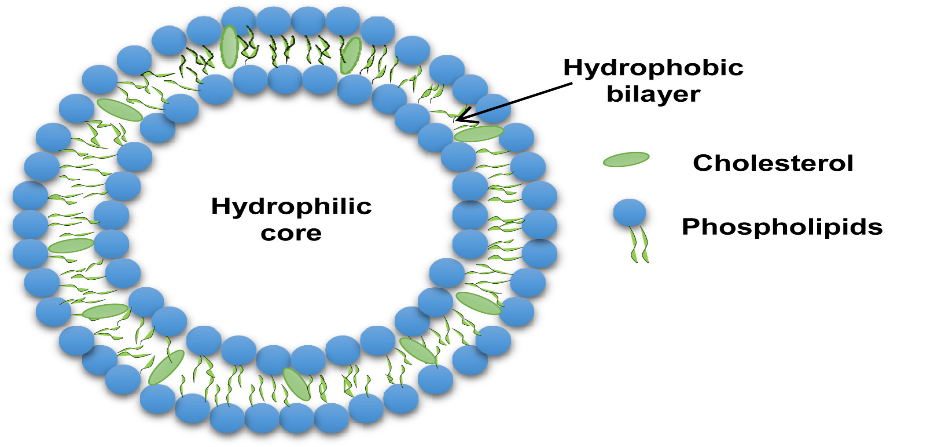
Figure 1: Structure of liposome
Classification of Liposomes:
Liposomes can range in size from 0.025µm to 2.5µm vesicles. Furthermore, the membrane of liposomes can be either single or bilayer. Liposome circulation half-life is mostly determined by size of vesicle and the quantity of medicine encapsulated in the liposomes is influenced by the size and number of bilayers. Liposomes can also be divided into two groups based on the size and quantity of bilayers they contain:8
1.Unilamellar vesicles
2. Multilamellar vesicles
Unilamellar vesicles can also be classified into two categories:
1.Large unilamellar vesicles
2. Small unilamellar vesicles
Unilamellar vesicles:
A single phospholipid bilayer sphere enclosing the aqueous solution is present in unilamellar liposomes.

Figure 2: Unilamellar and multilamellar liposomes
Multilamellar vesicles:(MLV)
Multi lamellar vesicles are made up of two or more bilayers and are larger than 0.1µm. Their formulation process, which involves hydrating lipids in excess of an organic solvent or using thin film hydration approach is straight forward and incredibly portable. When stored for a long time, they remain mechanically stable. Because of their size, the reticuloendothelial system (RES) cells can remove them quickly, which makes them useful for a variety of purposes that target the RES organs.9
Large unilamellar vesicles:(LMV)
The size of these liposomes is higher than 0.1µm and they are especially big unilamellar vesicles with a single bilayer. Since they can contain a huge volume of fluid in their cavity, they have a better encapsulation efficiency. They can be helpful for encasing hydrophilic medications due to their high trapped volume. The main benefit of LUV is that a greater amount of medication can be encapsulated with less fat. Because of their bigger size, RES cells remove them quickly, just as MLV. Many procedures including ether injection, detergent dialysis and reverse phase evaporation can be used to produce LUV. In addition to this technique, LUV can be prepared by slow swelling of lipids in non-electrolyte solution, dehydration/rehydration of SUV and freeze-thaw of liposomes.10
Small unilamellar vesicles:(SUV)
Small unilamellar vesicles have a single bilayer and they are smaller than MLV and LUV, measuring less than 0.1µm and they are distinguished by having a long circulation half-life and a low entrapped aqueous volume to lipid ratio. SUV can be made by lowering the size of MLV or LUV using sonication or extrusion processes in an inert atmosphere, such as nitrogen or argon or by employing solvent injection method. Either a bath or a probe type sonicator can be used to carry out sonication. SUV can also be accomplished by applying high pressure while passing MLV via a small opening at lower charge, these SUVs are prone to aggregation and fusion.11
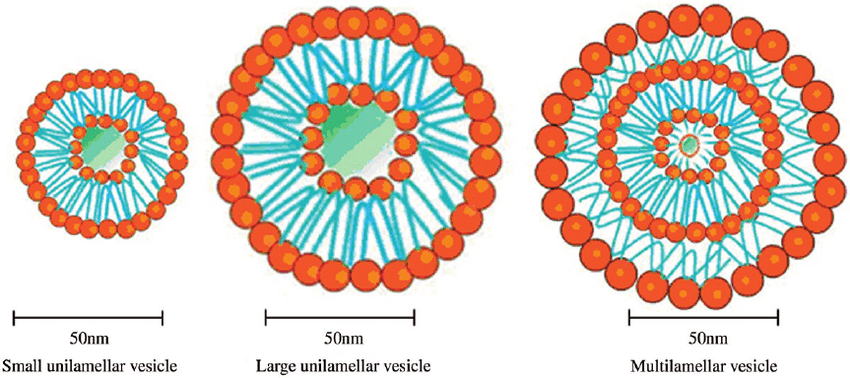
Figure 3: Different types of liposomes
Composition of Liposomes:
Phospholipids:
Phospholipids are primary building blocks of biomembrane called Phosphatidylcholine is most commonly used phospholipid in liposome composition.12 Phosphatidylcholine can be derived from naturally or produced using semi-synthetic and synthetic process. Phosphatidylcholines differ from other phospholipids in their bilayer sheet orientation in micellar compositions. Liposomes are made from several natural and semi synthetic phospholipids. Phosphatidylcholine molecules do not mix well with water. The researchers created a planar bilayer structure in aquatic conditions to reduce unwanted reactions with bulk water and non- esterified fatty acids. The sheets are folded into sealed capsules. 13,14
Cholesterol:
Generally, liposomes made up of phospholipids but have lack adequate rigidity due to low phase transition temperature or unsaturation in fatty alkyl chains, which results in problems in the cell membrane like packaging. The medicine is enclosed but it leaks out of the liposomes during packaging. Most liposomes formulations include one or two bilayer stabilizers to prevent leaking. Most commonly used are alpha tocopherol and cholesterol. Changes in phospholipid bilayer composition it affects the quality of liposome encapsulation and cholesterol is an essential part of natural lipid bilayers and it present in liposome bilayer causes a significant change in their properties and cholesterol incorporated into high amount of phospholipid membrane it doesn't form bilayer complex. Because of bilayer tight stacking, rigidity is increased, and water-soluble molecule permeability is decreased. Cholesterol makes the bilayer more organized and decreases fluidity above the phase transition temperature. (Tc). 15
Additional excipients:
Polyethylene glycol on the liposome surface promotes prolonged circulation, protects the captured medication from inactivation or metabolic degradation increases stability and improves intracellular intake.16 PEG has potential to form stealth liposomes and body reticuloendothelial system, and it cannot detect. PEG reduces particle agglomeration and increases storage stability. Liposomal technologies have evolved from traditional vesicles to "second-generation liposomes " and it release active pharmaceutical ingredients gradually. This can be accomplished by modifying the vesicle phospholipid composition, dimension and charge.
Liposome-Based carrier system:
Liposomes are drug carriers capable of delivering pharmaceuticals to specific target areas. 1
Table-1 –Different liposome carrier-based system 19-24
|
Vesicle type |
Characteristics |
|
Immunoliposomes |
Assist in focusing on the body active substances |
|
Virosomes or artificial viruses |
Made up of structurally reconstituted viral proteins |
|
Stealth liposomes |
Carrier surface is coated with hydrophilic chains. |
|
Archaeosomes |
Thermostable and impervious to oxidation, hydrolysis and enzymatic hydrolysis are archaeosomes |
|
Nanoliposomes |
Increased blood circulation duration and simple tissue penetration with long-lasting effects. |
|
Vesicular phospholipid gels |
These are semisolid vesicle-shaped phospholipid dispersions that are extremely concentrated. |
METHOD OF PREPARATION:
1.Passive loading technique
a. Mechanical dispersion
• Lipid film hydration method
• Micro emulsification
• Sonication
• French pressure cell
• Membrane extrusion
• Dried reconstituted vesicles
• Freeze- thawed liposomes
b. Solvent dispersion
• Ethanol injection method
• Ether injection method
1. Lipid Hydration method:
It is most well-known and extensively used method for MLVs.For its formulation, a round-bottomed flask might be used to create a thin coating, the lipophilic mixture is dehydrated, and the membrane is rehydrated with an aqueous buffer. The dispersion is then thoroughly mixed. The hydration stage occurs at a temperature higher than the lipid gel-liquid-crystalline (Lβ-Lα) transition temperatures or slightly higher than the transition temperature of the lipid mixtures maximal melting component. Depending on their solubility, the medicines kept into an aqueous buffer. The method has drawbacks such as low internal volume, lower encapsulation efficiency and varying sizes. However, diethyl ether can improve lipid encapsulation by hydrating them in immiscible organic solvents like petroleum ether, followed by sonication to emulsify the mixture. MLVs are developed by removing the organic layer with nitrogen.25,26
Figure 4: Lipid hydration method
2. Micro emulsification:
This method is used to create tiny lipid vesicles on a commercial scale utilizing a device known as micro fluidizer, small vesicles are created from concentrated lipid suspension. It is necessary to add the lipids to the fluidizer as a suspension of big MLVs then the fluid is pumped at extremely high pressure into a 5µm panel using this equipment.27 After then, two fluid streams collide at a straight angle at a very high velocity due to the force of a microchannel by controlling microemulsion can be created for biological use and speed ranging from 20 to 200.28

Figure 5: Micro emulsification
3. Sonication:
This method decreases the size of the vesicles and provides energy to lipid mixture. In this MLV can be exposed to ultrasonic radiation in order to achieve sonication in two ways:
Suspensions that require a lot of energy in a small volume are frequently treated using the probe sonicator.29When dealing with large amount of diluted lipids, the bath sonicator is employed. The main drawbacks of this method are poor internal encapsulation efficacy, phospholipid breakdown, removal of large molecules in particular metal contaminants from the probe tip and cohabitation of MLV and SUV. 30
4. French Pressure Cell method:
During this procedure MLV is extruded through a small hoe at 4°c at 20,000 pressures. This method has many advantages over sonication method. This method is easy, fast and repeatable, but it requires careful handling of dangerous ingredients. The disadvantages are difficulties in reaching the desired temperature and the working volumes are relatively smaller.31
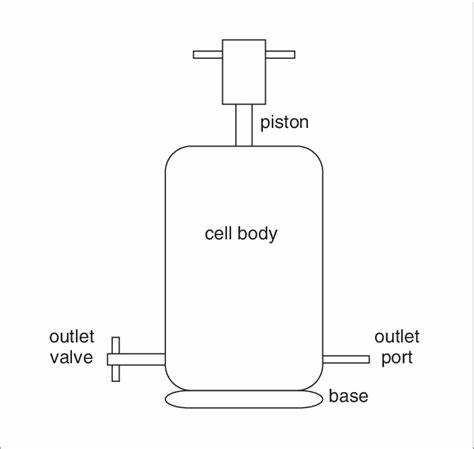
Figure 6: French pressure cell method
5. Membrane extrusion:
Processing of a heterogenous liposomal suspension involves a polymer sieve with a web like construction that creates a tortuous-path capillary hole, an interconnected network with a membrane atleast 100 microns thick and the size distribution of liposomes was tiny with a size of less than 0.4 micron. This method can be used for both MLVs and LUVs.32
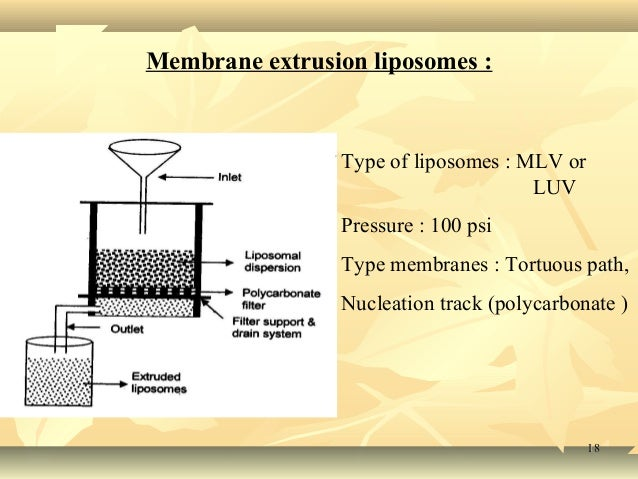
Figure 7: Membrane extrusion method
6.Dried reconstituted vesicles:
This method involves combining liposomes with lyophilized protein or exposing them to a pharmaceutical containing aqueous solution, followed by dehydration. 33
7.Freeze-Thaw method:
SUVs are frozen quickly and then thawed gradually throughout the process. By using sonication, aggregated chemicals are distributed to LUV.SUV17 is responsible for the creation of unilamellar vesicles during the freezing and thawing processes and increases ionic strength of the medium and the quantity of phospholipids greatly inhibits this type of fusion and increases entrapment efficiency. 34
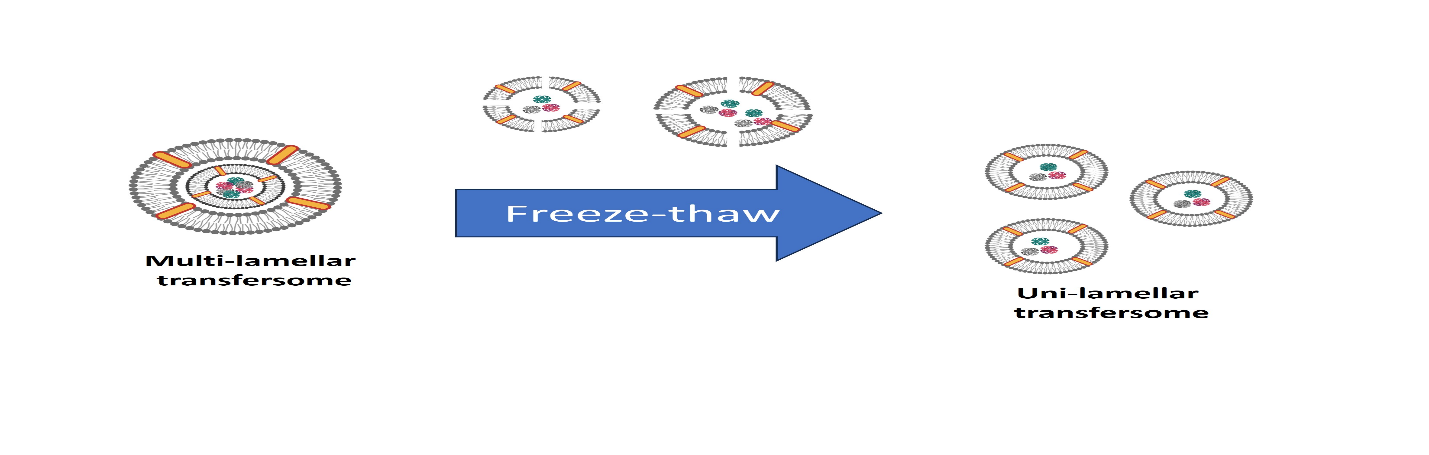
Figure 8: Freeze thaw method
8.Ethanol injection method:
This is a basic methodology, and it involves rapidly incorporating an ethanol lipid solution into an excess of saline or other aqueous solutions directly using a tiny needle.35 In water, phospholipid molecules are equally dispersed throughout the media and ethanol dissolves and this approach has primary drawback that the particles may vary widely in size (30-110nm). 36

Figure 9: Ethanol injection method
9.Double emulsification:
This method creates a primary emulsion by combining the medicines in an aqueous phase (w1), which is then further emulsified to create a primary w1/o emulsion in a solvent made up of organic polymers and then combine the emulsion with an emulsifier that contains an aqueous solution (w2) results in w1/o/w2 double emulsion and remaining solvent is eliminated from aqueous continuous phase is further separated by centrifuging and filtered.30
CHARACTERIZATION OF LIPOSOMES:
1.Visual appearance: Depending upon size and makeup of the particles, the liposomal suspension might have a clear or milky appearance, and the samples are homogenous if the turbidity is bluish, non- liposomal dispersion is flat and grey and typically distributed inverse hexagonal phase or dispersed micro crystallites observed under optical microscope can identify at >0.3µm.
2.Determination of liposomal size: When liposomes are administered parenterally or inhaled administration, the size distribution is crucial because it controls both liposomes and drug molecules in vivo and liposomes are measured using dynamic light scattering. Liposomes having a comparatively uniform size distribution are reliable for this method and it is possible to directly detect fully hydrodynamic radius using gel exclusion chromatography. Liposome separation is achieved by Sephacryl-S100 in 30-300 nm size range. Separating SUVs from micelles is possible using Sepharose columns-4B and 2B.
3.Determination of lamellarity: The lamellarity of a liposome determines its structure and their effectiveness in vivo. The quantity of lipid bilayers in the liposome has a significant impact on encapsulation and drug release kinetics. Lamellarity affects both intracellular destiny and liposome absorption and lamellarity of liposomes may be determined by using spectroscopic or electron microscopy method.
4.Stability of liposomes: Stability is crucial when employing liposomal drug delivery systems. A drug physical stability and chemical purity are influenced by its production and storage conditions and therapeutic function of drug is regulated by liposomal stability. It regulates size, distribution, composition and drug retention and chemical equilibrium is affected by phospholipid oxidation, hydrolysis and drug degradation. A safe liposomal pharmaceutical product possesses physical, chemical and microbiological stability and ensures product integrity during storage.
5.Entrapped volume: This is a crucial factor that controls the shape of vesicles and measured by total amount of solute trapped within liposomes, the entrapped liposome volume (in µL/mg phospholipids), guaranteeing that the solute concentration in the aqueous medium following isolation from untapped material. In two stages of preparation to get rid of organic solvent, water from the inner compartments may be lost during the drying process. A specific formula can be used to calculate this.
%%entrapment?efficiency=Entrapped?drug×?100Total?drug

6.Surface charge: The interaction between lipids and cells is determined by type and density of charge on the liposome surface. The charge on the vesicle surface is analyzed. Since liposomes are usually made up of incorporating charge to lipids. To analyze charge, two methods are typically used, Zeta potential and free flow electrophoresis.
7.Zeta potential: The zeta potential significantly impacts on cell absorption and targeted delivery. The laser Doppler electrophoresis and Zeta Sizer evaluates zeta potential of liposomal dispersion by applying an electrical field based on laser scattering on moving particles. There are several factors such as particle concentration, ionic strength and pH are some variables that affects zeta potential and overall surface charge and liposome blood circulation time are influenced by the liposome.
Table-2: Commercially available liposome formulation
|
Disease |
Drug |
Route of administration |
Method of preparation |
|
Fungal infection |
Amphotericin-B |
I.V infusion |
Conventional method |
|
Analgesic |
Morphine sulphate |
Epidural |
Depofoam technology |
|
Viral vaccines |
Hepatitis A |
IM/SC injection |
Detergent removal technique |
|
Cancer therapy |
Daunorubicin |
I.V injection |
Conventional method |
|
Asthma |
Terbutaline sulphate |
Subcutaneous injection |
Thin film hydration technique |
|
Keratitis |
Amphotericin-B |
Ocular |
Conventional method |
|
Breast neoplasms |
Doxorubicin |
I.V injection |
Stealth liposome technique |
|
Photodynamic therapy |
Verteporfin |
I.V injection |
Conventional technique |
|
Pseudomonas aeruginosa |
Fluoroquinolones |
Nebulized aerosol(pulmonary) |
Reverse phase evaporation |
|
Non-metastatic osteosarcoma |
Mifamurtide |
I.V infusion |
Non- PEGylated liposome technology |
APPLICATION OF LIPOSOMES:
1.Ophthalmic disorders: To treat eye problems such as dry eyes, keratitis and corneal transplant rejection medications can be applied topically as ointments, solutions or suspensions. These preparations have low bioavailability due to eye barriers and liposome compositions can overcome by developing liposome suspension to treat dry eyes and sprays that transport medication to the conjunctival sac. Ciprofloxacin/Ciprocin is commonly used eye drop that effectively combats both gram positive and gram negative. In comparison of ciprocin eye drops with liposomes in rabbits, liposome preparations have higher area under curve values, resulting improved ocular bioavailability and residence time.43
2. In Cancer chemotherapy: Cancer cells behave differently than normal cells and have a higher penetration rate and tumor cells have high permeability, allowing micro and macromolecules easily penetrate and cancer cells mainly include biomarkers (amino peptidase and integrin) that allows targeted drug administration to the tumor tissues and lysozyme enzyme is responsible for polymer breakdown at an acidic pH. Co-loading Doxorubicin (a breast cancer medication) with umbelliprenin into liposomes improves toxicity and homogenous distribution of liposomes at the targeted site.44
3.In Vaccines: Liposomes are employed as adjuvants in vaccine delivery and alters surface with peptide or virus antigens to enhance immunity and immunological responses. The membrane proximal external area is an HIV antigen on the virions surface that boosts immune responses despite its modest immunogenicity.45
4. In Gene Delivery: The most popular application for cationic liposomes is gene carriers. A commercially available cationic liposome for gene transfection is called Lofectamine 2000.Skin cancer is treated with curcumin loaded with STAT3 si-RNA, liposomes, which are made using the Bangham technique. Mainly to prevent development of B16F10 melanoma cells. Additionally, liposomes transport the CRISPR to cure various malignancies and genetic abnormalities. 46
5. Liposome for respiratory drug delivery: Liposomes are frequently utilized to treat a variety of respiratory conditions. It is possible to create liposomal aerosols with increased stability, decreased toxicity, avoid local irritation and sustained release. The composition, size, charge, drug/lipid ratio and this all techniques are used to prepare liposomes for lung distribution. Nebulization involves taking the liquid or dry form to inhale. Milling or spray drying are two methods used to create drug powder liposome.47
6. Liposomes in intracellular drug delivery: Drugs having intracellular receptors must pass through the plasma membrane in order to exhibit pharmacological activity, in which liposomes are used in intracellular drug delivery. Drugs that typically enter cells by pinocytosis can be delivered with greater efficacy. Because liposomes can hold higher drug concentrations than extracellular fluid. The mechanism of endocytosis, which is more effective than pinocytosis in which primarily absorb negatively charged liposomes. Liposomes can be used to improve the cytosolic administration of several medications that are typically not absorbed by cells.48
7. Liposomes in sustained release drug delivery: Sustained release drug delivery systems are necessary to reach and sustain the drug concentration and administered within the therapeutically effective range required for medication. As a result, the medication level fluctuates causing toxicity and ineffectiveness mainly to reduce this fluctuation, new drug delivery method has been created such as noisome and liposomes.49
8.Liposomes as protein drug delivery: They improve solubility of drugs.
9. Liposomes in Cosmetics: Liposomes are employed in cosmetics due to their comparable physiology to cell membranes and ability to release compounds into cells.50
Table-3: List of clinically approved liposomal drugs
|
Name |
Trade name |
Company |
|
Liposomal Amphotericin-B |
Abelcet |
Enzon |
|
Liposomal Amphotericin-B |
Ambisome |
Gilead Sciences |
|
Liposomal cytarabine |
Depocyt |
Pacira (Formerly skye Pharma) |
|
Liposomal Daunorubicin |
DaunoXome |
Gilead Sciences |
|
Liposomal Doxorubicin |
Myocet |
Zeneus |
|
Micellular estradiol |
Estrasorb |
Novovax |
|
Liposomal vaccine |
Epaxal |
Berna Biotech |
|
Liposomal vaccine |
Inflexal V |
Berna Biotech |
|
Liposomal morphine |
DepoDur |
Skye Pharma, Endo |
Table-4: MarketedLiposomal cosmetics formulations
|
S.No. |
Product |
Manufacturer |
|
1. |
Capture |
Cristian Dior |
|
2. |
Effect du Soleil |
L’Oreal |
|
3. |
Niosomes |
L’Oreal |
|
4. |
Nactosomes |
L’Oreal |
|
5. |
Symphatic 2000 |
Biopharma GmbH |
|
6. |
Natipide II |
Nattermann Pl |
|
7. |
Flawless finish |
Elizabeth Arden |
|
8. |
Inovita |
Pharm/Apotheke |
|
9. |
Eye perfector |
Avon |
|
10. |
Aquasome LA |
Nikko Chemical Co. |
CONCLUSION:
Liposomes have been used in a broad range of pharmaceutical applications. Liposomes are showing particular promise as intracellular delivery systems for anti-sense molecules, ribosomes, protein/peptides and DNA. Liposomes enhanced drug delivery to disease locations and longer residence times and achieve clinical acceptance. Liposomal drugs exhibit reduced toxicities and enhanced efficacy. Nowadays liposomes are used as carriers for a wide variety of drugs. Liposomes serve as versatile carriers.
REFERENCES






Anusha Rudroju*, Thummalapelli Sindhu, Kothakapu Gayathri, Rubana Rahat Shahi, Rabiul Hassan, Shaik Afroz, A Review on Formulation and Characterization Studies of Liposomes, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 5, 1116-1129. https://doi.org/10.5281/zenodo.15354875
 10.5281/zenodo.15354875
10.5281/zenodo.15354875