
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmaceutics, Centre for Pharmaceutical Sciences, University College of Engineering, Science and Technology, JNTUH, Kukatpally, Hyderabad,500085.
Solid lipid nanoparticles have become a promising nano-carrier system for improving drug delivery and therapeutic efficacy. These SLNPs systems provide the advantages of both lipid formulations and polymeric nanoparticles by enhancing biocompatibility, controll drug release with an increase in stability. SLNPs are widely used for both hydrophilic and lipophilic drugs through different routes of administration. The preparation methods of solid lipid nanoparticles are high-pressure homogenisation, solvent evaporation, solvent injection method, double emulsion, etc. Characterisation parameters are zeta potential, SEM, TEM, FTIR, and entrapment efficiency, which plays an important role in determining their performance. This review article mainly focuses on SLNPs' structure, types of material used, method of preparation, advantages, disadvantages, characterisation, and applications of SLNPS.
Nanotechnology serves as a foundational technology that engages with objects sized in the nanometer range. Future developments in this field are expected to unfold at different levels: materials, devices, and systems. Presently, the materials level of nanotechnology is the most sophisticated, demonstrating both advanced scientific knowledge and extensive commercial applications. (1) Nanomaterials (NPs) can be sized from zero to three dimensions, contingent upon their shape. The size of nanoparticles is crucial in determining the features of the material. (2)
Long before nanotechnology became a recognised field, people were inadvertently interacting with various nanosized materials and utilising nano-scale methods. In ancient Egypt, the common practice of dyeing hair black was believed for a long time to be based on plant-derived substances like henna. However, recent studies of hair samples from ancient Egyptian burial sites have shown that the dye was actually created from a mixture of lime, lead oxide, and water. This dyeing technique resulted in the formation of galenite (lead sulfide, PbS) nanoparticles. The ancient Egyptians were adept at making the dyeing paste react with sulfur (a component of hair keratin), leading to the production of small PbS nanoparticles that allowed for uniform and consistent dyeing.
One of the most notable examples of ancient nanotechnology is the Lycurgus Cup (fourth century CE). This Roman cup possesses extraordinary optical properties; it changes colour based on the angle of the light source. In natural light, the cup appears green, but when lit from within (for instance, by a candle), it takes on a red hue. Recent examinations of this cup revealed that it contains 50–100 nm Au and Ag nanoparticle which contribute to the cup's distinctive coloration through plasmon excitation of electrons. The historical use of nanotechnology does not end here; there is also evidence of its early application in Mesopotamia, Ancient India, and among the Maya. (3)
In nanotechnology, there are numerous forms, such as liposomes, Niosomes, transferosomes, and solid lipid nanoparticles. In these solid lipid nano particles are mostly used nowadays,In lipid and lipid-based drug delivery systems, phospholipids are vital components owing to their amphiphilic nature, biocompatibility, and multifunctionality. However, systems like liposomes, lipospheres, and microsimulation carriers have significant limitations, including complex production methods, low entrapment efficiency (% EE), and challenges in large-scale manufacturing. This has led to the emergence of the SLN delivery system. SLNs are generally spherical, with diameters between 50 and 1000 nm. The essential components of SLN formulations include lipids that are solid at room temperature, emulsifiers, and sometimes a combination of both, along with active pharmaceutical ingredients (APIs) and an appropriate solvent system Nanocarrier-based drug delivery systems can be categorised in various ways, depending on the route of administration and the degree of degradability. The routes of administration include nanoparticles for parenteral, oral, ocular, and topical use, as well as nanoparticles for protein peptide delivery. Furthermore, nanocarrier systems can be classified based on their degradability. An ideal nanoparticulate drug delivery system should possess the following characteristics:
Structure of solid lipid nanoparticles
SLNs consist of a solid lipid core at room temperature, forming a well-structured lipid matrix. This matrix effectively shields the active ingredient and regulates its release with improved quality. An outer surfactant layer stabilizes SLNs, allowing the active ingredient to be contained within the lipid core, at the surfactant interface, or distributed throughout the entire nanostructure. Developed in the 1990s, SLNs were designed to combat the rapid degradation and drug stability problems in liposomes, as well as the toxicity associated with polymeric nanoparticles from the use of volatile organic solvents.[5]
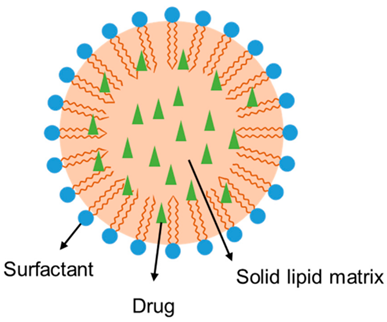
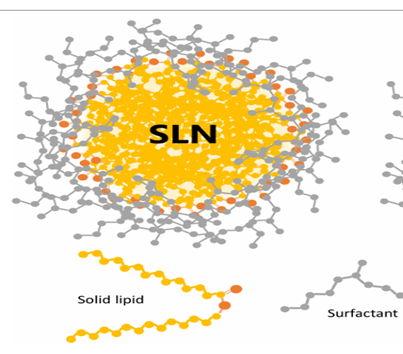
Fig 1,2: solid lipid nano particle [6,7]
Types of solid lipid nano particle:
TYPE 1: Homogeneous matrix model
The homogeneous matrix model can be achieved through the cold homogenization method, which incorporates lipophilic active molecules into the SLNs using the hot homogenization technique.
TYPE 2: Drug-enriched shell model
In this technique, a solid lipid core is formed once the recrystallization temperature of the lipid is attained, leading to the drug concentrating in the still-liquid outer shell of the SLN due to a decrease in dispersion temperature.
TYPE 3: Drug-enriched core model
In this method, the active compound precipitates initially, resulting in the shell having a notably lower drug concentration. This results in a membrane-controlled release that follows Fick's law of diffusion. [8]

Fig 3: Types of solid lipid nanoparticles
Advantages:
Disadvantages:
Methods of preparation:
1. Super critical fluid extraction method:
This method stands out as one of the most promising techniques for generating solid nanoparticles, based on the principle that lipid nanoparticles are formed from oil/water emulsions via supercritical fluid extraction (SCF).
The key advantage of this method over others is its operation at low temperatures (35°C) and the elimination of organic solvents in the nanoparticle fabrication process, which allows for solventless processing. Carbon dioxide (CO2) is commonly employed as the SCF, either on its own or with other solvents. The particles produced through this technique have smaller sizes and distributions, feature smooth surfaces, and are free-flowing, which supports the benefits of this method. Nonetheless, there are limitations to this approach, including costs, the low solvent strength of CO2, and the necessity for large quantities of CO2. SCF can act as a solvent, swelling and plasticizing agent, antisolvent, or solvent for polymerization in dispersed media during nanoparticle processing. The rapid expansion of supercritical CO2 solutions will yield solid lipid nanoparticles (SLNs). CO2 with a purity of 99.99% is an excellent solvent for preparing SLNs using this technique. Gas-saturated solutions (GSS), such as ammonia, chlorodifluoromethane (CHClF2), 1,1,1,2-tetrafluoroethane (CH2FCF3), and ethane, are the most effective SCFs and facilitate the melting of lipid materials, which then dissolve in the SCF under pressure along with the lipid melt and GSS. Spraying the saturated solution through an atomizer or nozzle causes it to expand and quickly release SCF, leaving fine dry lipid particles behind. The term “Supercritical fluid extraction of emulsions (SFEE)” refers to the method of producing lipid nanoparticles using emulsions through SCF technology.
The lipid component and the drug are dissolved in a suitable surfactant-containing organic solvent, such as chloroform, to create the organic phase. A high-pressure homogenizer is utilized to combine the organic solution with an aqueous solution that may also include a cosurfactant to form an o/w emulsion.
The supercritical fluid (maintained at a constant temperature and pressure) is currently supplied counter to the flow, while the o/w emulsion is delivered from one endpoint of the extraction unit (typically the top) at a steady flow rate. Continuous solvent extraction from o/w emulsions is being implemented. [11]
2. High-Pressure Homogenization:
In the production of drug-loaded Nano-Structured Lipid Carriers (NLCs), cold high-pressure mixing and hot high-pressure mixing—commonly referred to as cold HPH and hot HPH—are the most widely used techniques. Hot HPH is defined as the preparation of medications using a blend of melted liquid lipids and solid lipids. The combination of lipids and hydrophobic drugs under elevated temperatures is subsequently homogenized with a high-pressure solvent evaporator, undergoing 3–5 cycles to ensure thorough mixing with a heated water-based solution that includes both hydrophilic surfactants (like Tween) and amphiphilic surfactants (such as lecithin).
This method forces the hydrophobic phase into a confined area within the homogenizer, leading to the creation of tiny dispersed particles in the submicron size range.HPH offers numerous advantages, such as higher processing temperatures that contribute to smaller particle sizes due to the reduced viscosity of the lipid phase. Nevertheless, employing hot HPH for the preparation of drug-loaded NLCs presents certain challenges. These challenges include the drug's sensitivity to heat, low entrapment efficiency, and a reduced drug-loading capacity, which may occur if drug molecules migrate into the hydrophilic phase during lipid crystallization Hot HPH has its drawbacks. To address this issue, we can utilize cold high-pressure homogenization (HPH) at temperatures and pressures typically below room temperature. In this approach, the lipid is melted while the drug is frozen using liquid nitrogen or dry ice. The exploration of cold high-pressure homogenization has opened new avenues for the advancement of nanostructured lipid carriers, enhancing the potential to encapsulate thermos-labile drugs effectively and improving drug-loading capacity. This process involves heating the drug-lipid mixture until it melts and then rapidly cooling it by submerging it in dry ice or liquid nitrogen. This swift cooling accelerates the crystallisation of solid lipids (SL). [12]
3.Microemulsion:
Microemulsion is one of the most widely recognised, frequently used, and popular methods for producing nanostructured lipid carriers (NLCs). It is preferred for its cost efficiency and versatility in managing both polar and non-polar drugs (Table 1). An emulsion created with melted fat oil or any other modifications should be prepared by incorporating it into surfactants and co-surfactants dissolved in a water solution containing these compounds, such as sodium dodecyl sulphate. The proportions of the components will determine whether the emulsion is oil-in-water (o/w) or water-in-oil (w/o). This ensures thorough mixing until small sizes, like microns, are achieved [41–43]. Nanostructured lipid carriers (NLCs) are formed by mixing this tiny emulsion with a cold aqueous mixture, known as the hydrophilic phase, at a ratio of 1:25 to 1:50 with gentle stirring; this process leads to a reduction in particle size.[12]
4. Double emulsion technique: -
This method is extensively used for hydrophilic drugs, which are water-loving, and also for drugs that are sensitive to temperature. The double emulsion technique is applied to manufacture drug/biomolecule-loaded NLCs (like proteins and peptides)
This technique produces a water-in-oil (w/o) emulsion by combining a hydrophilic drug dissolved in an aqueous medium with a lipid melt containing lipophilic surfactants. The next step involves adding this primary emulsion to an aqueous solution containing a hydrophilic solvent, creating a double emulsion that is water-in-oil-in-water (w/o/w). The Nanostructured Lipid Carriers (NLCs) are then separated from the dispersion by ultrafiltration and solvent evaporation. The double emulsion technique has an advantage over the single emulsion method because it does not require a molten lipid phase. Once these methods optimise the preparation of NLCs, their practical applications across various drug delivery systems become clear.[12]
5. Solvent Injection Method:
Another term for this is the "solvent displacement technique".The rapid and easy formation of lipids from their dissolution in a water-miscible solvent is achieved through solvent injection. This solution is promptly injected through a needle into an aqueous system that contains surfactants. This method consists of mixing lipids and pharmaceuticals in a water-soluble solvent, such as methanol, ethanol, acetone, isopropanol, or a combination of these solvents. The aqueous phase (water phase) is generally obtained by dissolving an emulsifier or a mixture in a water/buffer solution. Thus, the needle continuously stirs the aqueous phase while simultaneously introducing the organic phase rapidly under mechanical agitation. This technique provides the advantages of straightforward preparation and avoids the requirement for high heat, excessive shear stress, or complex equipment. However, its drawbacks include the risk of organic solvent residue, which can result in high particle concentrations.[12]
6. Solvent Evaporation method:
The lipophilic compound is dissolved in an organic solvent that does not mix with water (such as cyclohexane) and is emulsified in an aqueous phase. When the solvent evaporates, nanoparticles are formed by the precipitation of the lipid in the aqueous medium, yielding nanoparticles with a mean size of 25 nm. The emulsification of the solution in the aqueous phase was achieved through high-pressure homogenization. The organic solvent was removed from the emulsion by evaporation under reduced pressure (40–60 mbar).[13]
7. Solvent emulsification diffusion method
This technique allows for the production of particles with average diameters ranging from 30-100 nm. The key advantage of this method is that it avoids heat generation during the preparation process. In this approach, lipids are generally dissolved in the organic phase within a water bath at 50 °C, and an acidic aqueous phase is utilised to modify the zeta potential, leading to the coacervation of SLN, which can then be easily separated by centrifugation. The SLN suspension is generated swiftly. The entire dispersed system can then be centrifuged and resuspended in distilled water.[13]
8. Spray drying:
An additional procedure to lyophilisation is available for the transformation of an aqueous dispersion into a drug. This method is more cost-efficient when compared to lyophilization. There is a risk of particle aggregation due to elevated temperatures, shear forces, and incomplete melting of the particles.[14]
9. Ultra Sonication:
SLN was also synthesised through high-speed stirring or sonication (Eldem et al., 1991). The tools required for this technique are standard in almost every laboratory. A key drawback of this method is the extensive particle size distribution that extends into the micrometre range, which is a leading cause of physical instability (Svilenov and Tzachev, 2009). Concerns such as particle growth during storage and the risk of metal decay are pressing issues in this approach. After extensive studies and thorough research, it has been confirmed that high-speed stirring and ultrasonication, when used together at high temperatures, produce a reliable formulation.[14]
10. Phase Inversion Temperature
Phase Inversion Temperature (PIT) Methodology. This methodology is founded on the capability of non-ionic surfactants to modify their affinity for water and oil based on temperature fluctuations. In this process, the oil and surfactant blend is melted and then introduced to water while being stirred continuously. The system is maintained at a heated state until it reaches the cloud point, followed by a rapid cooling phase. During the heating phase, the oil/water emulsion shifts to a water/oil configuration, and upon cooling, an oil/water emulsion is formed once more. With each phase inversion, the size of the particles diminishes, enabling the production of nanoparticles at the end of the procedure.[15]
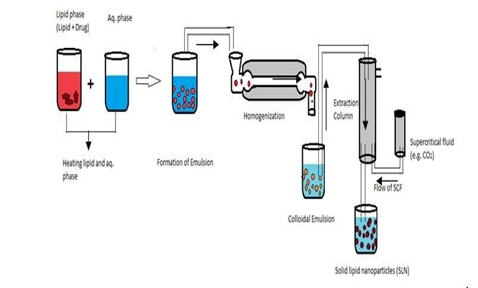
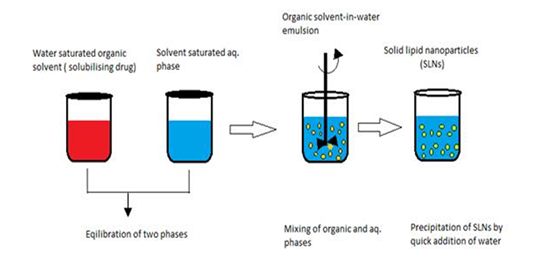
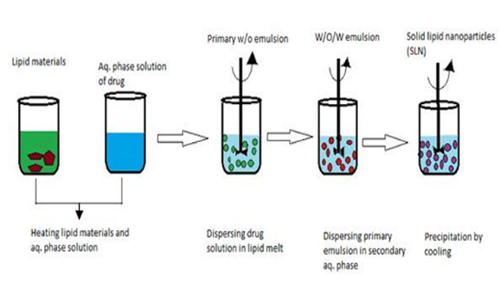
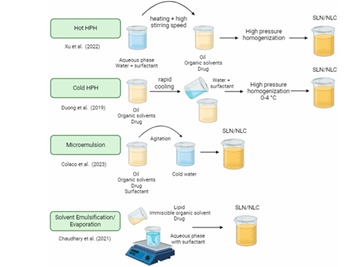
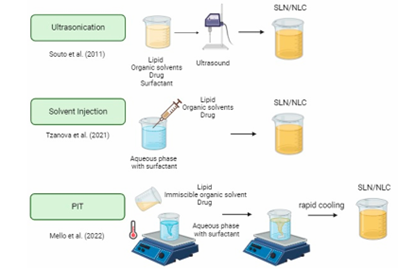
Fig 4, 5, 6,7&8. Figures explaining the process of Supercritical fluid extraction, Solvent emulsification diffusion method, double emulsion, hot and cold HPH, microemulsion, solvent evaporation, ultrasonication, solvent injection, and phase inversion temperature (PIT). [15,16]
DRUG LOADING AND RELEASE FROM SLN
The particle size and the type of drug entrapment model for SLNs play a crucial role in drug release, in line with the established theory of drug release from nanoparticles. Elements such as the drug solution and its interaction with the lipid matrix can affect the release of drugs. Temperature changes can influence the release profile of SLNs in response to both internal and external factors. Chen et al. studied the pH-sensitive release profile of cholesterol-PEG-coated SLNs loaded with doxorubicin and found that the drug released more quickly at pH 4.7 than at pH 7.4.
In general, SLNs displayed burst release. As the size of the particles increases, the burst release of the drug may reduce, resulting in a more extended release. Using tetracaine, etomidate, and prednisolone as model drugs, zur Mühllen et al. found that SLNs containing tetracaine and etomidate exhibited a burst drug release (100% release in under 1 minute) due to their significant surface area and drug enhancement in the outer shell. Conversely, prednisolone-loaded SLNs were shown to have a 5-week extended release. Burst (83.8%) and regulated releases (37.1%) were achieved due to the specific chemical activity of the lipid matrix, such as that of cholesterol and compritol.
According to Olbrich and Muller, a lipid interface is required for the activation of the enzymes that decompose the lipid matrix. It is recommended to modify the surface of SLNs with a hydrophilic carrier (such as PEG) to prevent lipase enzymes from easily recognizing them, thereby altering their release and enhancing their stability. Appropriate steric stabilizers and other surfactants should also be fine-tuned.
A recent review by Savla et al. indicates that drugs with a high melting point (not numerically defined) and a Log P value of 2 are typically poor candidates for lipid systems, especially for highly lipophilic drugs. [17]
Types of Materials used in Solid lipid Nanoparticles;
Table 1: Types of materials used in the preparation of solid lipid nanoparticles. [18,19]
|
Ingredients |
Examples |
|
Solid Lipid |
Glyceryl palmitostearate (Precirol ATO 5) Glycerol monostearate Decanoic acid Behenic acid |
|
Liquid Lipid |
MCT (Miglyol 812) |
|
Emulsifier, surfactants |
Poloxamer 188 Poloxamer 407 Tween 80 Span 80 Soybean lecithin, phosphatidylcholine Polysorbate 20 Polysorbate 80 Polysorbate 60 PEG-40 castor oil (Cremophor RH40) Sodium deoxycholate Sodium dodecyl sulfate poloxamer 182 Poloxamine 908 Tyloxapol |
|
Alcohols |
Ethanol Butanol |
|
Cryoprotectants |
Trehalose, mannose mannitol, polyvinyl, pyrolidone, glucose, maltose, lactose, glycine, gelatin, etc. |
|
Charge modifiers |
Stearylamine, diacetyl phosphate, dipalmitoyl phosphatidyl choline (DPPC), dimyristoyl phosphatidyl glycerol (DMPG) |
|
Stealthing agents |
Poloxamer, polyethylene glycol |
Characterisation of SLNPS:
1. Zeta potential:
Zeta potential measurement can be performed using a zeta potential analyser or zetasizer. Before measurement, SLN dispersions are diluted 50-fold with the original dispersion preparation medium, determining size and measuring zeta potential(Luo et al., 2006). A higher zeta potential value may result in the deaggregation of particles if there are no other complicating factors, such as steric stabilisers or hydrophilic surface appendages. Zeta potential measurements provide insights into the storage stability of colloidal dispersions.[20]
2. FTIR:
An FT-IR spectroscopic analysis was conducted to investigate the interaction between drug excipients. The distinct peaks of the drug within the IR spectrum of the mixture of drug and drug excipients were identified by absorption. Therefore, FTIR Spectroscopy was utilised to gather conformational data regarding the lipid molecules, and it was employed to explore the interactions among lipid, drug, and excipients, confirming that no significant interactions occurred between them, allowing the drug to remain unchanged in the formulation. [21]
3. SEM:
The SLNs suspension of SA-SLNs-SE and SA-SLNs-DE was treated with gold spray, and the surface morphology was observed using a scanning electron microscope (SEM) (S4800, Japan). The microscope operated at a voltage of 5 kV during the observation.[22]
4. TEM:
The morphology of the optimised SLNP was analysed using transmission electron microscopy (TEM) (JEM-2100F, Japan). The SLNs suspension was placed on a carbon-coated copper grid, followed by a drop of 2% phosphotungstic acid on the SLNs layer. After a 5-minute staining, excess dye was removed from the edges using filter paper. The sample was dried at room temperature for about half an hour and was then prepared for TEM investigation at 100 kV. [22]
5.Thermal behaviour:
Differential scanning calorimetric (DSC) analysis was executed to validate the drug-lipid association in nanoparticulate formulations and the crystallinity of the drug with the Mettler TASTARe system featuring a DSC 822e cell (Mettler Toledo, US). Samples comprising 40 mg of SLNs were weighed with precision in aluminium pans. DSC scans were recorded at a heating and cooling rate of 5º/min. The DSC analysis was performed on plain drug, blank SLNs, and drug-loaded SLNs. The SLNs were dried under vacuum at room temperature to remove moisture prior to the DSC study.
Differential scanning calorimetric (DSC) analysis was executed to validate the drug-lipid association in nanoparticulate formulations and the crystallinity of the drug with the Mettler TA STARe system featuring a DSC 822e cell (Mettler Toledo, US). Samples comprising 40 mg of SLNs were weighed with precision in aluminium pans. DSC scans were recorded at a heating and cooling rate of 5º/min. The DSC analysis was performed on plain drug, blank SLNs, and drug-loaded SLNs. The SLNs were dried under vacuum at room temperature to remove moisture prior to the DSC studies.[23]
6.Crystallinity analysis:
X-ray powder diffraction (pXRD) patterns were recorded on a Philips PW 17291 powder X-ray diffractometer (Philips, the Netherlands) utilizing Ni-filtered Cu-K radiation, with a voltage of 40 kV and a current of 25 mA. The scanning rate was maintained at 1° min−1 within the 10?40° 2θ range. pXRD diffraction patterns were documented for the plain drug, bulk lipids, and drug-loaded SLNs. The SLNs underwent vacuum drying at room temperature to remove water before the pXRD studies. [23]
7.In Vitro Drug Release
The drug release was analysed in 1× simulated gastric fluid (SGS) and 1× phosphate buffer saline (PBS) through the dialysis-bag method. A total of 2 mg of drug -SLN was placed in 1 mL of PBS within the dialysis bag and kept at 37 ± 1 ?C under magnetic stirring at 100 rpm in a 10 mL tube containing either SGS or PBS. At regular time intervals, aliquots of the dialysate were withdrawn, and the same volume of fresh medium was replenished. The samples were collected at various time points (i.e., 0, 10, 20, and 30 minutes, as well as 1, 2, 3, 4, 6, 7, 8, 9, 10, and 12 hours) for kinetics analysis, filtered through a 0.2 µm filter, and analysed by HPLC or UV at the required wave lengh, All experiments were conducted in triplicate, independently.[24]
8. Entrapment Efficiency:
Entrapment Efficiency: The efficiency of drug entrapment is determined by measuring the concentration of free drug in the dispersion medium.Ultracentrifugation was executed using Centrisart, which comprises a filter membrane (molecular weight cutoff 20,000 Da) at the bottom of the sample recovery chamber. The SLNs, along with the encapsulated drug, stay in the outer chamber while the aqueous phase flows into the sample recovery chamber. The concentration of the drug in the aqueous phase is assessed using HPLC or a UV spectrophotometer. [25]
% Entrapment efficiency = [(Initial drug weight–weight of free drug) / Weight of initial drug] × 100%
9. Stability Studies:
A stability study was performed on best formulation, which exhibits high entrapment efficiency. The formulation was stored at two distinct temperatures, 4 °C and 25±2 °C, and the drug content was assessed every 15 days to detect any variations in the entrapment efficiency of the SLNs [21].
Patents of Solid Lipid Nanoparticles:
Table 2: patents of solid lipid nanoparticles [26]
|
Patent title |
Patent number/ Application number/ Publication number |
|
Lipid-based nano pellets intended for oral drug delivery |
EP0167825 |
|
Drug delivery systems formulated using solid lipid particles |
WO9305768 |
|
Process for preparing solid lipid microspheres with controlled size distribution |
US5250236 |
|
Improved cyclosporine lipid formulation with enhanced stability and bioavailability |
US6551619 |
|
Lipid nanoparticles designed for nucleic acid delivery and their preparation method |
WO05120469 |
|
Lipid emulsions and nanoparticles used as carriers for genes or drugs |
WO0006120 |
|
Formulations for controlled release of fragrances and aromatic compounds |
CA2524589 |
|
Membrane reactor-based method for producing solid lipid particles |
WO07000531 |
|
SLN-based formulations for gene or drug delivery and their preparation |
KR2033665A |
|
Colloidal solid lipid systems for pharmaceutical applications |
US2006222716 |
|
Use of cholesteryl-based SLNs for drug delivery purposes |
WO06128888 |
|
Method for producing composite solid lipid drug particles |
US2006008531 |
|
Topical formulations containing lipid nanoparticles in oil-in-water emulsions |
DE19825856 |
|
Topical preparations using suspensions of solid lipid particles |
US5667800 |
|
Pharmaceutical formulations for treating eye-related diseases |
US2006024374 |
|
SLNs containing UV filters for sunscreen applications |
DE19952410 |
|
Incorporation of UV absorbers into SLNs for enhanced protection |
US7147841 |
|
Use of microemulsions as precursors for solid nanoparticles |
US7153525 |
|
Lipid-drug conjugate particles for controlled drug release |
US6770299 |
|
UV-protective agents using lipid nanoparticle systems for skin protection |
US6814959 |
|
Solid lipid nanoparticle formulations (general patent) |
430/DEL/2004 |
|
Ophthalmic pharmaceutical compositions using SLNs |
975/KOLNP/2005 |
Applications;
SLN as a potential new adjuvant for vaccines
Adjuvants are integral to vaccination as they amplify the immune response. The newer subunit vaccines, which are deemed safer, are often less effective in immunization, highlighting the need for effective adjuvants. Recent innovations in the adjuvant sector include emulsion systems, specifically oil-in-water emulsions that break down quickly in the body. The solid-state lipid components of SLNs, however, degrade more gradually, ensuring a longer exposure to the immune system.
Solid lipid nanoparticles in cancer chemotherapy
In the last two decades, various chemotherapeutic agents have been encapsulated in SLNs, with their efficacy evaluated both in vitro and in vivo. Findings from these studies have demonstrated an increase in the effectiveness of chemotherapeutic drugs while also reducing their side effects. Notable characteristics of SLNs include enhanced drug stability, the ability to encapsulate a range of chemotherapeutic agents with different physicochemical properties, improved drug efficacy, better pharmacokinetics, and lower in-vitro toxicity, making them a suitable vehicle for delivering chemotherapeutic drugs. Several challenges commonly faced by anticancer compounds, such as toxicity to normal tissues, poor specificity and stability, and a high occurrence of drug-resistant tumor cells, can be at least partially addressed through SLN delivery. A significant hurdle in targeting other tissues in the body, such as bone marrow and solid tumors, is the swift removal of colloidal particles by macrophages in the RES.
Solid lipid nanoparticles for ultrasonic drug and gene delivery
Drug delivery research utilizing micelles and nanoparticles has seen extensive application in ultrasonic drug and gene delivery in recent years. Notably, the use of these nano vehicles is of great interest as they selectively deliver high concentrations of cytotoxic drugs to diseased tissues, thereby minimizing the side effects of the agents on healthy parts of the body. Traditionally employed in diagnostic medicine, ultrasound is now being integrated into drug delivery systems in conjunction with these nanoparticles. Besides their non-invasive characteristics and ability to target specific tissues, acoustic waves are recognised for their role in releasing pharmacological agents from nanocarriers and enhancing the permeability of cell membranes. Ultrasonic drug delivery from micelles typically utilises polyether block copolymers and has proven effective in vivo for tumour treatment. The release of drugs from micelles via ultrasound is likely facilitated by shear stress and shock waves generated from the collapse of cavitation bubbles. Both liquid emulsions and solid nanoparticles are employed with ultrasound for gene delivery in vitro and in vivo. The compact size of nanoparticles enables them to extravasate into tumour tissues. The potential of ultrasonic drug and gene delivery from nanocarriers is immense due to the diverse range of drugs and genes that can be delivered to targeted tissues through relatively non-invasive methods.
Solid lipid nanoparticles for targeted brain drug delivery
The extremely small dimensions of solid lipid nanoparticles, which are under 50 nm, could be advantageous for drug targeting. Smaller carrier sizes generally lead to diminished uptake by the reticuloendothelial system. Furthermore, drug targeting may be facilitated through the surface modification of solid lipid nanoparticles. SLNs can enhance the drug's ability to traverse the blood-brain barrier, representing a promising drug targeting system for treating central nervous system disorders. In research aimed at overcoming the restricted access of the drug 5-fluoro-2’-deoxyuridine (FUdR) to the brain, 3’,5’-dioctanoyl-5-fluoro-2’-deoxyuridine (DO-FUdR) was synthesized and integrated into solid lipid nanoparticles (DO-FUdR-SLN).
The current state of surfactant-coated poly (alkylcyanoacrylate) nanoparticles specifically engineered for brain targeting is presented, emphasizing the transition of this technology to solid lipid matrices. The potential benefits of solid lipid nanoparticles compared to polymeric nanoparticles are based on lower cytotoxicity, enhanced drug loading capacity, and superior production scalability. The physicochemical characteristics of solid lipid nanoparticles are also particularly considered to tackle the critical challenges associated with developing suitable brain-targeting formulations.
Solid lipid nanoparticles in the fight against parasitic diseases
Parasitic diseases, such as malaria, leishmaniasis, and trypanosomiasis, are among the most pressing health challenges globally. Antiparasitic chemotherapy is currently the only treatment option for these infections, as they do not elicit a significant immune response, which complicates the development of effective vaccines. Solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs) are part of a new generation of colloidal carriers that have emerged as effective alternatives to liposomes, mainly due to their improved stability, scalability, commercialisation prospects, and cost efficiency. Additionally, SLNs and NLCs, due to their particulate nature and structural characteristics, show great promise in treating parasitic infections. Recent studies, including our own, have validated their effectiveness to some extent. However, there is an urgent need for extensive research on SLN and NLC matrices to enhance their versatility in encapsulation and targeting capabilities, ultimately leading to a more effective and cost-efficient approach for delivering antiparasitic therapies.
SLN applications for the enhanced delivery of antiretroviral drugs to the brain
The human immunodeficiency virus (HIV) can access the central nervous system during the initial stages of primary infection. Once inside the brain, the virus replicates vigorously, creating an independent viral reservoir that results in serious neurological complications, latent infections, and drug resistance. Current antiretroviral drugs (ARVs) often do not effectively diminish the HIV viral load in the brain. This is partly attributed to the poor transport of many ARVs, particularly protease inhibitors, across the blood-brain barrier (BBB) and the blood-cerebrospinal fluid barrier (BCSBF). Studies have demonstrated that nanocarriers, including polymeric nanoparticles, liposomes, solid lipid nanoparticles (SLN), and micelles, can enhance local drug concentration gradients, facilitate drug transport into the brain via endocytotic pathways, and inhibit the ATP-binding cassette (ABC) transporters located at the barrier sites. Delivering ARVs with nanocarriers is expected to significantly improve drug bioavailability to the brain. Recent research indicates that the specificity and efficiency of ARV delivery can be further optimized by using nanocarriers with specific brain-targeting capabilities, cell-penetrating ligands, or ABC transporter inhibitors. Future research should aim to achieve safe, efficient, and cost-effective delivery of ARVs to the brain.[27]
SLNs as cosmeceuticals:
The SLNs have been employed in the development of sunscreens and act as an active carrier for molecular sunscreens and UV blockers. A study conducted in vivo revealed that skin hydration could be enhanced by 31% after 4 weeks with the incorporation of 4% SLN into a conventional cream. Both SLNs and NLCs have been shown to be innovative occlusive topicals that allow for controlled release. Improved localization of vitamin A in the upper skin layers has been achieved with glyceryl behenate SLNs compared to standard formulations.
SLNs for potential agriculture application:
Essential oil extracted from Artemisia arboreseens L, when added to SLN, was capable of minimising rapid evaporation compared to emulsions, and these systems have been utilised in agriculture as effective carriers for ecologically safe pesticides. The SLNs were created here using Compritol 888 ATO as the lipid and poloxamer 188 or Miranol Ultra C32 as the surfactant.
Oral SLNs in antitubercular chemotherapy:
Antitubercular drugs, including rifampicin, isoniazid, and pyrazinamide-loaded SLN systems, have been effective in reducing the frequency of doses and improving patient adherence. The preparation of these solid lipid nanoparticles loaded with antitubercular drugs is achieved through the emulsion solvent diffusion technique. Moreover, nebulization in animals with the aforementioned drug in SLN has been reported to increase the drug's bioavailability.[28]
SLNs for Liver Targeting: Liver-targeting SLNs
SLNs that include the hepatoprotective drug, cucurbitacin B (Cuc B), were developed using a galactosylated lipid known as N-hexadecyl lactobionamide (N-HLBA). The preparation of the galactosyl-lipid N-HLBA was achieved through the lactone form intermediates of lactobionic acid, by anchoring galactose to hexadecylamine lipid. The Cuc B-loaded galactosylated SLNs and conventional SLNs were successfully produced using a high-pressure homogenization technique.
The encapsulation of Cuc B within SLNs resulted in enhanced cytotoxic activity, and the galactosyl ligand could further improve the cellular accumulation and cytotoxicity of Cuc B. The addition of N-HLBA into SLNs significantly enhanced the liver-targeting capability of Cuc B-loaded SLNs, suggesting that galactosylated SLNs have great potential as drug delivery systems for improved liver targeting. [25]
CONCLUSION
Solid lipid nanoparticles are a flexible and effective drug delivery system that holds considerable promise in contemporary pharmaceutical research. Their capacity to enhance drug stability, boost bioavailability, and facilitate controlled release renders them appropriate for a diverse array of therapeutic uses. Utilising physiological lipids leads to decreased toxicity and better patient adherence. Despite the existence of challenges such as polymorphic transitions, drug leakage, and restricted drug loading, ongoing progress in formulation techniques and nanotechnology is working to overcome these obstacles. Future investigations aimed at surface modification, targeting methods, and large-scale manufacturing are anticipated to further broaden the clinical use of SLNP. In summary, SLNs are poised to be a highly promising next-generation drug delivery platform for both research and industrial purposes.
REFERENCES



Gurajala Tejesh, Dr M. Sunitha Reddy, Dr K. Anie Vijetha, A Review on Solid Lipid Nano Particles: Design, Characterization & Therapeutic Applications, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 4, 658-673 https://doi.org/10.5281/zenodo.19414431
 10.5281/zenodo.19414431
10.5281/zenodo.19414431