
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Govindrao Nikam College of Pharmacy, Mumbai university, Sawarde, India 415606
Phytochemicals, a diverse group of plant-derived secondary metabolites, have attracted considerable interest due to their wide range of therapeutic properties. Compounds such as curcumin, quercetin, and silymarin exhibit potent antioxidant, anti-inflammatory, hepatoprotective, and anticancer activities. Despite their promising pharmacological potential, clinical translation of phytochemicals remains limited because of intrinsic challenges including poor aqueous solubility, low stability under physiological conditions, rapid metabolism, and poor oral bioavailability. In recent years, nanocarrier-based drug delivery systems have emerged as a transformative strategy to overcome these limitations. This review highlights recent advances in nanocarriers developed for phytochemical delivery, focusing on five major systems: phytosomes, liposomes, niosomes, ethosomes, and solid lipid nanoparticles (SLNs). Each system is critically evaluated in terms of structural characteristics, drug loading capacity, release kinetics, stability, and transmembrane absorption mechanisms. Comparative analysis reveals that phospholipid-based carriers offer notable advantages, with phytosomes demonstrating superior oral bioavailability and liposomes providing versatility for targeted drug delivery. Surfactant-based niosomes exhibit enhanced stability and cost-effectiveness, while ethanol-rich ethosomes are particularly effective for dermal and transdermal delivery. SLNs are discussed for their controlled release behavior, physical stability, and potential for lymphatic uptake. The review further emphasizes application studies and pharmacokinetic evidence showing multiple-fold increases in bioavailability of phytochemicals using advanced nanocarriers, along with their translational relevance in human studies. Overall, this review integrates mechanistic insights with comparative performance data to propose a rational strategy for selecting optimal nanocarrier systems based on phytochemical physicochemical properties, underscoring nanocarriers as a crucial bridge between laboratory research and clinical application of phytochemical-based therapies.
Herbal medicine has been a source of treatment for several thousands of years, and the World Health Organization recommends it for healthcare needs. A number of these products are being studied today for their potential therapeutic benefits on various diseases, including infectious diseases, metabolic syndrome, neurological disorders and cancer.(1) The increase in the use of herbal remedies can be attributed to some important factors including 1) lack of effective cure for all human pathologies by modern drugs, 2) growing concern and interest over the guarantee and safety of synthetic drugs, and 3) several natural products have exhibited effectiveness superior to standard drugs without side effects.(2) Herbal preparations are extracts comprising a variety of pharmacologically/biologically active phytochemicals including terpenoids, phenolics and alkaloids.(1) They are secondary metabolites that the biosynthetic pathways of plants generate as a response to stress and pathogens microorganisms, in human have an important relevance for preventing these diseases and pathological conditions. Terpenoids, phytosterols, carotenoids, alkaloids, capsacinoids and phenolic compounds are some of the most representative families.(3) Phytochemicals are bioactive compounds in edible plants (fruits, vegetables, seeds, nuts and cereals) which have been studied for their potential health promoting properties including antioxidant, antimicrobial, anti-inflammatory and chemo-preventive effects.(4) Many active substances of phytomedicine are hydrophobic and poorly solubilized, liable to instability in gastric and colonic conditions; this limits its clinical application.(1) In order to circumvent these limitations, Nanotechnology based drug delivery systems work as carriers for drugs and hold promising potential in overcoming different obstacles which limit the efficacy of herbal medicines, including enhancing phytochemical bioavailability and bioactivity. The nanotechnology approach may be a promising novel technology for phytochemical components of the plant and improve the efficiency of phytotherapy with herbal medicine.(5)
This review provides a comprehensive comparative analysis prominent nanocarrier systems-Phytosomes, Liposomes, Niosomes, Ethosomes, and Solid lipid nanoparticles focusing on their composition, mechanism, comparative advantages of phytochemical encapsulation, and enhancement of therapeutic efficacy.
TYPES OF NANOCARRIERS:-
Phytosomes are nanotechnology-based drug delivery systems in which plant extracts or phytoconstituents are complexed with phospholipids. This complexation enhances solubility, stability, and bioavailability. Phytosomes have been demonstrated to improve the pharmacokinetic performance of phytoactive compounds, enabling improved targeting and sustained drug release.(6) The word “phyto” is plant, and “some” means of a cell. It is also named as herbosomes.(7) Phytosome enhances the absorption and bioavailability of phytoconstituent through phospholipid standardised plant extracts. Thus, stoichiometric amount of phospholipid (phosphatidylcholine) when reacted with standardized extract in a nonpolar solvent. Phytosomes act as a linkage between conventional delivery system and modern novel delivery system They are also called as phytolipid delivery system.(8)
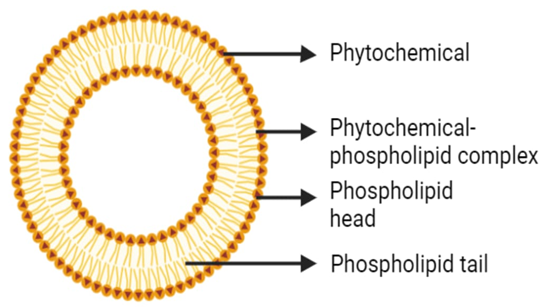
Fig. 1 Structure of Phytosome (9)
Liposomes are spherical, concentric lipid bilayers were first introduced by combining the Greek words, which is how the term "liposome" was created; lipo meaning fat and soma meaning body. In 1961, when Bangham accidentally dispensed the phosphatidylcholine molecule in water, He discovered that it had assumed a closed bilayer form in which an aqueous space was trapped between two layers of lipid. Thus, liposomes were first described.(10) Because liposomes may be easily modified to fulfil a range of delivery requirements, their versatile physicochemical and biophysical properties make them an appealing delivery option. Liposomes are concentric, bilayered vesicles ranging in diameter from 0.01 to 5.0 μm. Their composition may include membrane proteins, fatty acids, glycolipids, long chains of fatty acids, non-toxic surfactants, and sphingolipids.(11) Liposomes are small colloidal particles that consist of one or more phospholipid layers enclosing aqueous compartments. They are of great interest because they can encapsulate both hydrophilic and hydrophobic solutes, and so have been developed as a drug carrier for delivery systems (DDS). The term "liposome" may also refer to synthetic, microvesicles whose interior contains one or more phospholipid layers and a fluid compartment. The interior of the sphere-like aqueous core can encapsulate various substances, such as peptides, proteins, hormones, enzymes, antibiotics, antifungals and anticancer drugs. Liposomes are tiny, spherical artificial vesicles with a size ranging from nanometers to micrometers, and both hydrophilic and hydrophobic parts that may be derived from cholesterol or natural non-toxic phospholipid. They are used as carriers for adjuvant therapy; health and medicine. Their characteristics are: 1) lipophilic tail; 2) hydrophilic head. (12,13)
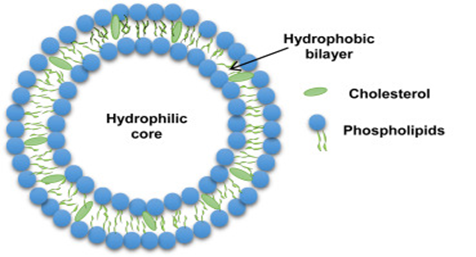
Fig.2 Structure of Liposome(14)
Niosomes are novel drug delivery system, involves encapsulating the drug in a vesicle. Niosomes get their name from the fact that the vesicle is made up of a bilayer of non-ionic surface active substances and hence the name niosomes. The niosomes are miniscule, and extremely tiny. When synthetic non-ionic surfactant are hydrated with or without the addition of cholesterol and other lipids, non-ionic surfactant vesicles known as niosomes.
They are vesicular systems that resembles liposomes that can employed to transport both amphiphilic and lipophilic drugs. Because, niosome are non-ionic, they are less toxic and increase the medicinal value of drugs by limiting their effect to target cells, making them prospective drug delivery vehicle.(15)
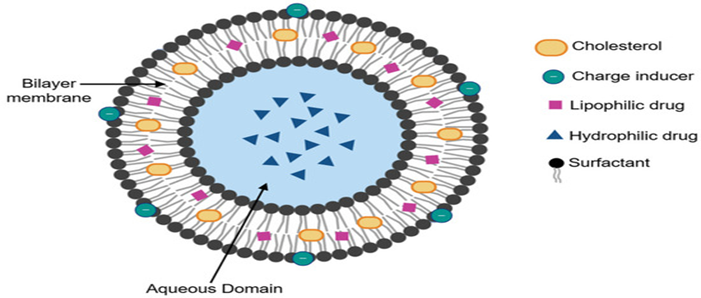
Fig.3 Structure of Niosome (16)
Ethanolic vesicles are referred to as “Ethosomes”. This term was coined by Touitou, who developed an innovative vesicular system incorporating ethanol into its structure.(17) Presently, the vesicular system represents one of the most extensively studied approaches for delivering medications transdermally. Ethosomes serve as non-invasive delivery vehicles that facilitate the entry of drugs into systemic circulation or enable deep penetration through skin layers. They make transdermal drug delivery possible due to their unique properties. The remarkable elasticity of human skin allows these vesicles to traverse it without causing damage. Typically, ethosomes consist primarily of a combination of alcohol in relatively high concentrations (20–45%), water, and phospholipids, resulting in soft and flexible lipid vesicles. Touitou and her team first introduced ethosomes in 1997. These versatile vesicles can vary in size from a few tens of nanometers to several microns. Ethosomes exhibit significantly enhanced transdermal flow and penetrate epidermal layers more rapidly than traditional methods. Besides facilitating medication delivery to deeper skin layers, ethosomes meet crucial criteria for the safe and effective administration of both hydrophilic and lipophilic drugs. They have the capability to encapsulate various types of molecules, including those that are hydrophilic, lipophilic, or have high molecular weights. Ethosomes can effectively transport medications through the skin under both occlusive and non-occlusive conditions.(18)
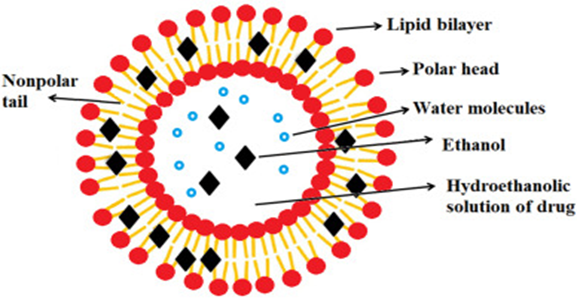
Fig.4 Structure of Ethosome (19)
Solid lipid nanoparticles (SLN), first in 1991, are a feasible alternative to traditional colloidal carriers such as polymeric micro- and nanoparticles, liposomes, and emulsions. These solid lipid-based nanoparticles have sparked interest as novel colloidal medication carriers for intravenous use. SLNs, or sub-micron colloidal carriers, are composed of physiological lipids dispersed in water or an aqueous surfactant solution and range in size from 50 to 1000 nm. Small-sized nanoparticles (SLN) have unique properties such as a large surface area, high drug loading capacity, and phase interaction at the interface, making them promising for improving medicinal efficacy.
SLN are commonly used to increase a drug's solubility and bioavailability. Because of their tiny size (50 to 1000 nm), these delivery systems have increased permeability, allowing them to successfully cross physiological barriers. Reducing particle size causes a significant increase in the surface area of insoluble drug particles after oral delivery. This leads to improved absorption through the monolayer cells of the gastrointestinal system. SLNs have been demonstrated to improve lymphatic movement via microfold cells (M cells) and are more easily absorbed by cells. Improved lymphatic conveyance causes a reduction in early medication metabolism, which enhances drug bioavailability. The stratum corneum's resistance to a wide range of drugs at adequate concentrations is the primary impediment to topical distribution in the dermal system. Furthermore, due to the tiny size of the particles, SLN can improve the bioavailability of drugs that penetrate healthy skin. Because of their near proximity to the stratum corneum, the amount of encapsulated medication that reaches the desired site of action increases.(20)
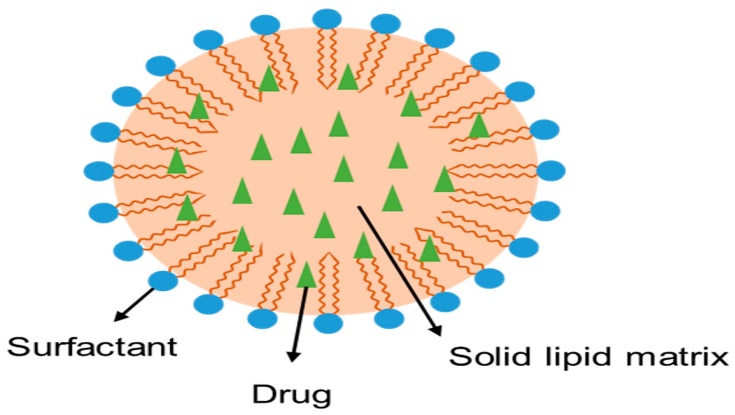
Fig.5 Structure of Solid Lipid Nanoparticles(21)
COMPARISON BETWEEN THE NANOCARRIER FEATURES:-
The following table1. gives an idea about the difference between Phytosome, Liposome, Niosome, Ethosome, Solid lipid nanoparticles
Table1. Comparison between the nanocarrier features
|
Feature |
Phytosome |
Liposome |
Niosome |
Ethosome |
Solid Lipid Nanoparticles |
|
Basic composition |
Phytochemicals + Phospholipid (Mainly phosphatidylcholine) formed molecular complex. (2) |
Phospholipid (Phosphatidylcholine) + Cholesterol (stabilizer) + Aqueous medium. (14) |
Non-ionic surfactant+ Cholesterol (stabilizer)+ Aqueous medium. (15) |
Phospholipid (e.g. Phosphatidylcholine) + High Ethanol (20-45%) + Aqueous medium. (22) |
Solid Lipids + Surfactants + Active Ingredient. (20) |
|
Structure |
A well-defined molecular complex, The phospholipid chemically bound to the phytochemicals. Phosphatidylcholine’s charged phosphate head forms hydrogen bond with polar functional group of lipophilic substances (e.g. polyphenols). (2) |
A bilayer vesicle with an aqueous core. The drug is entrapped in the core (hydrophilic drugs) or bilayer (lipophilic drugs). (14) |
An aqueous core in a bilayer vesicles. Structurally similar to liposomes but uses non-ionic surfactants. (15) |
A highly deformable bilayer vesicle with an aqueous core. The high ethanol content makes it soft and flexible. (22) |
SLNs are spherical particles that range in size from 50 to 1000 nm. The drug can be uniformly dispersed in the lipid matrix or concentrated in the core or shell. (20) |
|
Mechanism of action |
Molecular complexation enhances absorption by mimicking body’s natural absorption of fatty acids. The complex can be absorbed directly through cell membrane.(23) |
The vesicle fuses with or is endocytosed by cells, releasing its cargo inside.(24) They can be formulated for targeted delivery. (25) |
Similar to liposomes, they fuse with cell membranes or are taken up by cells. (15) |
The stratum corneum lipids are fluidised by the high ethanol concentration, which enables the flexible ethosomes to enter the skin deeply and deliver their cargo. (22) |
Enhanced Bioavailability: The small particle size increases the surface area for drug dissolution and absorption, particularly for poorly soluble drugs. SLNs provide sustained drug release as well as targeted drug delivery.(20) |
|
Stability |
Showed High stability: The Phospholipid and Phytoconstituent formed strong chemical bonding resulting in higher stability. (26) |
Chemically unstable because phospholipids are prone to oxidation. Can be stabilized with cholesterol but has a lower shelf-life than niosomes.(27) |
Having more stability than liposomes. Non-ionic surfactants are less prone to oxidation and hydrolysis. (28) |
Because of its high ethanol content which inhibit fusion and aggregation. It is generally stable.(22) |
SLNs offer high physical stable compared to liposomes. The solid lipid core protects the drug from chemical degradation. (20) |
|
Advantages |
Direct absorption provides superior bioavailability for phytochemicals. High stability and site specific delivery to the target tissues.(6,26) |
Highly biocompatible and biodegradable. They can encapsulate both hydrophilic and lipophilic drugs.(29) |
Low production costs, good chemical stability, and extended shelf-life, Non-immunogenic. (28) |
Superior transdermal penetration distribution to the deepest layers of the skin. Suitable for systemic and topical medication administration through the skin. (18) |
Both biodegradable and biocompatible. |
|
Limitations |
Only applicable to phytochemicals those having an active hydrogen atom (-COOH, -OH, -NH2, -NH, etc.) that can form a complex with phospholipids.(2) |
Phospholipids are expensive and have low shelf-life due to instability. Prone to leakage and aggregation.(12) |
Physical instability, which leads to aggregation and fusion, drug leakage and hydrolysis of encapsulated drug, which reduces shelf life. (28) |
A drying effect or skin irritation could result from the high ethanol content. Not as well suited for oral delivery. (22,30) |
Having low drug loading capacity, and the potential for drug expulsion Possessing a high water content. (20) |
|
Best applications |
Enhancing oral bioavailability of herbal extracts like Quercetin, silymarin, and curcumin. Nutraceuticals and dietary supplements.(2,31) |
Drug delivery in oncology and vaccine development. (32,33) Encapsulation of variety of drugs e.g., Doxil® (PEGylated liposomal doxorubicin) for treatment of cancer.(32) |
Topical/ dermal delivery, cosmetic applications, and sustained-release formulations. (34,35) |
Transdermal drug delivery for pain management, hormone replacement, and cosmetic applications.(22) |
Used in oral, cutaneous, ophthalmic, and intravenous drug delivery to enhance therapeutic efficacy. It also improves anticancer drug delivery, cosmetic stability and skin penetration, and antigen delivery in vaccination. (20,36–38) |
Application of Nanocarriers for the encapsulation of phytochemical
Curcumin is yellow polyphenolic molecule, and the primary accomplished bioactive phytochemical extracted from turmeric (Curcuma longa). It serves as a powerful antioxidant and anti-inflammatory agent due to the natural symmetry of its organic substance; diferuloylmethane. Over decades, curcumin has been extensively studied for its usage as an antioxidant, anti-inflammatory and cancer suppressor, metabolic diseases, and neurodegenerative disorders. however, its pharmacokinetic properties limit the potential for clinical application. Such poor bioavailability arises from low aqueous solubility, rapid metabolism, and quick systemic elimination from the body. Scientists are actively developing novel delivery techniques to improve absorption and stability.(39,40)
Table 2. Comparison of features between curcumin nanocarriers
|
|
Curcumin Phytosome |
Curcumin Liposome |
Curcumin Niosomes |
Curcumin Ethosomes |
Curcumin SLNs |
|
Basic Composition |
The Curcuminoid extract (Curcumin, Demethoxycurcumin, Bisdemethoxycurcumin) is complexed with a natural phospholipid, typically Phosphatidylcholine (PC) used, in a stoichiometric ratio. (41,42) |
Curcumin is integrated into lipid Bilayer a structure mostly composed of phospholipids (e.g., soy phosphatidylcholine, egg yolk phosphatidylcholine, hydrogenated soya PC) and cholesterol to provide structural integrity and modulate membrane fluidity. (43) |
Curcumin Niosomal formulation comprised of bilayer Components such as Non-ionic surfactants (e.g., Span, Tween) and cholesterol, where the cholesterol act as Stabilizer to give rigidity, reducing bilayer fluidity, and preventing the leakage of curcumin which is encapsulated within a surfactant bilayer. (44) |
Curcumin ethosome composed of Lipid Phase including phospholipids (e.g., Phosphatidylcholine).an aqueous phase: water or buffer (e.g. pH 7.4); and a key component a high concentration ethanol (generally 20% to 45% v/v), providing flexibility and act as a penetration enhancer. (45) |
Curcumin SLNs consist of solid core made up of Solid Lipids with a high melting point, e.g. tristearin, glyceryl monostearate, And a Surfactant like Tween 80, poloxamer 188 used as an emulsifier to stabilize the particles. (46,47) |
|
Interaction with curcumin |
Chemical Complexation: Curcumin forms a stable, lipid-compatible molecular complex with the polar head of Phosphatidylcholine via hydrogen bonds and hydrophobic interactions. (42) |
Encapsulation and Protection: Liposome encapsulate and protect curcumin by forming a spherical closed bilayer around it. This technique solubilizes the hydrophobic curcumin in an aqueous environment. (48) |
Entrapment in Bilayer: Niosomes encapsulate hydrophobic curcumin predominantly within the surfactant bilayer’s hydrophobic core. Where the vesicle structure solubilize the curcumin in aqueous environment while also it protecting it from chemical degradation (especially rapid breakdown at alkaline pH) and light. (44) |
Entrapment and Enhanced permeation: The flexible lipid bilayer encapsulates curcumin. The high ethanol content disrupts the tightly packed lipid structure of the skin's stratum corneum, allowing the flexible ethosomes to squeeze through the intercellular spaces and deliver the curcumin deep into the skin layers and potentially the systemic circulation. (49) |
Curcumin is a fat-soluble drug, is readily dissolved in the melted solid lipid during the preparation procedure. As the lipid solidifies, it confines the curcumin molecules within its matrix. This encapsulation protect curcumin from degradation by light, heat, and the harsh pH of the GIT. (50) |
|
Bioavailability |
Significantly enhanced: The oral bioavailability of curcumin is significantly higher than that of unformulated curcumin. Studies show an increase in plasma curcumin levels by 29 times. (e.g. Meriva) (40,51) |
Significantly enhanced: Encapsulation significantly improves oral bioavailability of curcumin. (reported to increase the total exposure (AUC) and plasma concentration (Cmax) by 8-20 fold compared to standard curcumin suspension. (52,53)
|
The niosomes prolong the residence time of drug in the body, achieved the controlled release effect and improved the bioavailability in In vivo studies conducted on rabbits. (54) |
Significantly Enhanced Transdermal Bioavailability: provide a major increase a drug deposition in the skin layers and improves systemic exposure via the topical route, overcoming the poor oral bioavailability of curcumin. Mechanism: This enhancement is due to the dual effect of the ethanol (fluidizing the skin lipids) and the deformable nature of the ethosome. (49,55) |
Significantly increase: The encapsulation in SLNs improve curcumin's solubility and absorption. The lipid-based nature of the nanoparticles facilitates absorption via the intestinal lymphatic system, bypassing first-pass metabolism in the liver. This causes a larger concentration of curcumin in the blood and longer retention duration. (56–58) In rat model, >12 fold increase in bioavailability compared to curcumin solution. (59) |
|
stability |
Improved: The curcumin is stabilized by the chemical complexation; Which protect against rapid metabolism/ conjugation in liver and degradation (such as hydrolysis and chemical instability at alkaline PH in the gut). (42) |
Improved chemical stability: The liposomal environment protects curcumin from premature degradation caused by light and the severe pH environment of the gastrointestinal tract.(60,61) Physical stability: generally considered stable, but requires careful formulation (e.g., use of cholesterol and specialised preparation procedure) and storage (e.g., low temperature, 4?C) to avoid aggregation or drug leakage. (43) |
High Chemical Stability: Because the chemical structure of Non-ionic surfactant makes them more stable than liposome. Physical Stability: Generally exhibits good stability when kept at optimal temperatures (e.g., 4?C), with less susceptibility to oxidative degradation and hydrolysis compared to liposomes. (44,62) |
Good Physical Stability: Ethosomal formulations can maintain good physical stability (e.g., small, uniform size and high entrapment efficiency) over several months when kept appropriately (45,63). |
High stability: The solid lipid core of SLNs shields curcumin from environmental influences like light, oxygen, and hydrolysis, considerably reducing its degradation and maintaining its chemical stability. (64,65) |
|
Advantages |
1. High Bioavailability: Greatly improved oral absorption and systemic availability of curcumin. (42,51) 2. Improved Physicochemical Properties: transforms curcumin, which is weakly soluble in water, into an amphiphilic molecule that is more readily soluble and more tolerable. (40) |
1. High Drug Loading & Enhanced Solubility: Allows delivery of effective curcumin doses. (53) 2. Reduced Toxicity: Targeted delivery to cancer cells can reduce systemic exposure and toxicity to healthy cells. (66) 3. Targeting: Offers passive targeting (EPR effect in tumors) or active targeting (by surface modification, e.g., PEGylation to prolong circulation time). (67) |
1. Low cost: Non-ionic surfactants are frequently less expensive than the phospholipids used in liposomes. 2. High Biocompatibility: Non-ionic surfactants are often non-immunogenic and well-tolerability. (44) 3.Versatile Delivery: Can be used for oral, topical (dermal), and parenteral administration. (68,69)
|
1. Non-Invasive Delivery: Drugs can be delivery to deeper skin layers or systemic circulation without injection. 2. High Penetration: Due to the presence of ethanol, leading to deeper drug penetration. 3. High Patient Compliance: They are simple for self-application. 4. Can Incorporate Large Molecules: Suitable for delivering both small and large therapeutic molecules. (49) |
1. Enhanced bioavailability : Focus on the major limitations of curcumin’s low oral absorption. (70) 2. Controlled Drug release: The solid matrix enables for a sustained release of curcumin, resulting in prolonged therapeutic effect. (71) 3. Biocompatible and safe: Made from physiologically compatible and biodegradable lipids, reducing the risk of toxicity. (21) 4. Targeting Potential: surface-functionalized SLN can target specific cells or tissues, such as cancer cells. (72) |
|
Limitations |
1. Fixed Ratio & High Lipid Content: Requires a specific stoichiometric ratio, which frequently result in a high phospholipid content for given dose of curcumin, increasing the total mass of the dose. (41,73)
|
1. Physical Instability: Susceptible to aggregation, fusion, or drug leakage under non-optimal conditions. (43,74) 2. Rapid RES Clearance: liposomes can be quickly cleared by the Reticuloendothelial System (RES), requiring modifications like PEGylation to achieve long circulation times. (75,76) |
Risk of curcumin hydrolysis. Time consuming preparation process. Sometimes low drug loading. Opposite surface charges may cause fusion of vesicles. (44) |
1. Only suitable for Topical/ Transdermal Use: Ethosomes are only suitable for skin delivery of curcumin; the high ethanol content makes them unsuitable for many other routes of administration. 2. Potential Skin Irritation: The high concentration of ethanol can potentially cause skin dryness or irritation in sensitive individuals, requiring careful formulation. (49)
|
1. Potential for Drug Expulsion: The crystalline structure of some solid lipids might cause the expulsion of curcumin from the core upon storage, affecting stability and release profile. (50,77)
|
|
Applications |
Due to its enhanced bioavailability, it is widely used in: 1. Anti-inflammatory support: Osteoarthritis, rheumatoid arthritis. (42,78) 2.Neuroprotection: Supports cognitive function, Alzheimer’ prevention. (79,80) 3.Hepatoprotection: Liver support and management of liver disorders. (81) 4. Cardiovascular Health: Atherosclerosis Reducing inflammation and improving endothelial function. (82) |
1. Oncology: because of its anti-cancer and chemopreventive properties Targeted delivery for various cancers (e.g., colon, pancreatic, breast, liver). (83) 2. Anti-inflammatory/Antioxidant: Treatment of chronic inflammatory diseases and conditions related to oxidative stress. (84) 3. Hepatoprotection: Delivery to the liver for conditions like Non-Alcoholic Fatty Liver Disease (NAFLD). (85) |
1. Cancer Therapy: Give targeted delivery to tumors (passive targeting via the EPR effect) for enhanced cytotoxic activity. (44) 2. Inflammatory Diseases: Systemic or topical administration for conditions like arthritis and inflammatory skin disorders: psoriasis (due to anti-inflammatory properties). (86,87) |
1. Dermatology: Used for Topical treatment for various skin conditions like psoriasis, acne, fungal infections, and skin cancer (e.g., melanoma). (88–90) 2. Anti-inflammatory Therapy: Transdermal administration for localized relief in conditions like arthritis and chronic inflammation. (91)
|
1. Oral therapy: Curcumin is administered orally for treating conditions such as chronic inflammation, cancer, arthritis, and metabolic syndrome. (50,92,93) 2. Topical delivery: used for wound healing, skin inflammation, and other dermatological conditions. (94,95) 3. Targeted Cancer Therapy: Curcumin-loaded SLNs are now being studied for targeted cancer therapy. (96,97) |
Silymarin is a complex mixture, it's not a single compound; and it is a standardized extract from the seeds of milk thistle plant (Silybum marianum). It has a complex of flavonolignans, of which silybin (also known as silibinin) is the most biologically active and amounting to 50-70% of the extract. Other major constituents are isosilybin, silychristin and silydianin. Silymarin known for its potent hepatoprotective (liver protecting) effects. Silymarin also has antioxidant, anti-inflammatory, antiviral, anticancer properties. A major challenge in its clinical use is its poor oral bioavailability due to lower water solubility and rapid metabolism; several efforts have been done in order to improve absorption through different delivery systems. With advances in novel drug delivery systems (NDDS), various nanocarrier-based approaches have been introduced to enhance the solubility, stability, and absorption of silymarin, thereby improving its oral bioavailability.(98,99)
Table 3. Comparison of features between silymarin nanocarriers
|
|
Silymarin phytosome |
Silymarin Liposome |
Silymarin niosomes |
Silymarin Ethosomes |
Silymarin SLNs |
|
Basic Composition
|
The silymarin phytosome form a stoichiometric complex (e.g., 1:1 or 1:2 molar ratio between the silymarin (predominantly Silybin) and Phospholipid (e.g., Soya Phosphatidylcholine - PC). (100).(101) |
Active Drug: Silymarin (primary component: Silybin/ Silibinin). Lipid Components: Phospholipids (e.g., Soy Lecithin or Phosphatidylcholine), Cholesterol (for membrane rigidity). Aqueous Phase: Water or buffer solution. Optional Additives: Bile salts, PEG (Polyethylene Glycol) for stabilization or stealth properties. (102)
|
Active Drug: Silymarin (primarily Silybin/ Silibinin). Vesicle-Forming Components: Non-ionic Surfactants (e.g., Span series like Span 60, Tween series) with a suitable Hydrophilic-Lipophilic Balance (HLB). Membrane Stabilizer: Cholesterol (used to provide rigidity and reduce membrane permeability). Aqueous Phase: Hydration medium (water or buffer solution). (103) |
Phospholipid: (e.g., Soya Phosphatidylcholine, Hydrogenated Phosphatidylcholine) Ethanol/ Alcohol: High concentration (typically 20-45% w/w) Water: Hydration medium Drug: Silymarin (Silybin, etc.) Ethanol acts as a permeation enhancer and provides flexibility to the vesicle membrane, aiding deep skin penetration. Phospholipids form the core bilayer structure. (104) |
Solid Lipids: A solid core composed of lipids that are solid at body temperature, such as palmitic acid, or glyceryl behenate. Surfactant/ Emulsifier: A stabilizing agent like Tween 80 to form a stable dispersion in water. Silymarin: The active drug, which is a complex of lipophilic flavonoids (silybin, silychristin, silydianin), is encapsulated within the lipid matrix. (105–107) |
|
Interaction with silymarin |
Silymarin molecules (the polar, poorly-absorbed phytoconstituent) are chemically bonded (primarily via hydrogen bonding) to the polar head of the phosphatidylcholine. This interaction effectively transforms the poorly lipid-soluble Silymarin into a lipid-compatible compound, making it miscible with oils and allowing it to easily cross the lipid-rich intestinal cell membranes. (100,108,109) |
Silymarin, being a highly lipophilic (fat-soluble) compound, is primarily encapsulated within the lipid bilayer of the liposome. (110) |
Silymarin, being a highly lipophilic compound, is encapsulated within the hydrophobic core/ bilayer of the niosome membrane, formed by the tail-to-tail arrangement of non-ionic surfactants. The interaction stabilizes the drug within the nanocarrier. The encapsulation efficiency for silymarin in niosomes has been reported to be high (∼90% or more in some studies). (103) |
Silymarin, being a lipophilic (fat-soluble) compound, is entrapped within the lipid bilayer of the ethosome. The high concentration of ethanol in the formulation helps to increase the solubility and high loading of the lipophilic Silymarin within the vesicular system. (111) |
Silymarin, being highly lipophilic, is easily dissolved or dispersed within the molten lipid matrix during the preparation process. As the lipid cools and solidifies, it forms a core that effectively entraps and protects the silymarin molecules. This prevents the drug from precipitating in the gastrointestinal tract and shields it from enzymatic degradation. (105,107,112) |
|
Bioavailability |
Markedly Enhanced oral bioavailability (e.g. 6-fold increase in systemic absorption compared to free Silymarin). e.g. Siliphos®. The complex mimics a physiological absorption pathway. The new lipid-compatible molecule is better absorbed via passive diffusion across the gastrointestinal tract (GIT) membrane. It also resists degradation in the gut. (113,114) |
Conventional silymarin has poor oral bioavailability (∼20%−50%) due to low water solubility and high first-pass metabolism.(115) Liposomal encapsulation: 1. Increases Solubility in the gastrointestinal tract. 2. Protects the drug from degradation by gastric enzymes. 3. Enhances absorption across the intestinal membrane (e.g., via lymphatic uptake). Bioavailability has been reported to be multiple times higher (e.g., 4.8 times) than that of non-complexed silymarin. (102) |
Significantly Enhanced. Similar to liposomes, niosomes drastically improve silymarin's oral bioavailability by: 1. Increasing Aqueous Solubility/ Dissolution Rate. 2. Protecting the drug from degradation in the acidic gastric environment. 3. Enhancing Intestinal Absorption across the epithelial cells, leading to higher plasma and targeted organ (liver) concentrations (103,116). |
Significantly Enhanced (especially for transdermal/ systemic absorption). Studies show an increased systemic exposure (e.g., in blood and tissues) compared to free Silymarin. Ethosomes facilitate the drug's passage through the skin's stratum corneum barrier, and for oral routes, they protect the drug from GI degradation, overcoming its poor solubility (BCS Class IV). (116–118) |
Significantly Enhanced. The lipid-based nature of the SLNs facilitates their absorption through the intestinal lymphatic system, bypassing the first pass metabolism in the liver.(119) This leads to higher oral bioavailability of silymarin SLNs (2.79 fold) compared to silymarin suspension. (120) |
|
Stability |
The main interaction that occurs between phospholipid and the active substance is the formation of hydrogen bond between the polar region of phospholipid and polar region of phytoconstituents. This interaction allows a formulation with better stability compared to liposomes.(109) |
Silymarin liposomes exhibit enhanced stability compared to free silymarin, with factors like composition, storage temperature (4?C), and preparation method being critical. (121) Liposomes can suffer from physical and chemical instability (aggregation, leakage, oxidation, hydrolysis). Strategies for stability enhancement often include: Adding Cholesterol (to increase membrane rigidity) or preparing as a Proliposome (a dry powder that forms liposomes upon hydration) for long-term storage stability. (116,122)
|
Generally superior to conventional Liposomes. Niosomes are known to offer: - Higher Chemical Stability: Non-ionic surfactants are less prone to oxidative degradation (compared to phospholipids in liposomes). (123) - Higher Osmotic Stability: The surfactant structure provides greater stability against osmotic shock. - Good Shelf-life Stability: Niosome formulations, especially those with cholesterol, maintain their size and encapsulation efficiency well over time. (124) |
Generally considered stable with a good shelf-life when stored under appropriate conditions (e.g., refrigeration, incorporated into a gel). (125) |
High stability: The solid lipid matrix provides a robust physical barrier that prevents drug leakage and protects silymarin from environmental factors like oxidation. SLNs have a longer shelf-life and greater stability during storage. (112) |
|
Advantages |
1. High Absorption: Dramatically overcomes Silymarin's low water solubility (BCS Class IV). (113) 2. Synergism: The phospholipid carrier (Phosphatidylcholine) itself offers hepatoprotective benefits, providing a synergistic therapeutic effect. (113,126) |
1. Maximized Therapeutic Efficacy due to higher drug concentration in the target organ (liver). 2. Reduced Dosing Frequency/Amount due to enhanced absorption. 3. Potential for Targeted Delivery to the liver via the enhanced permeability and retention (EPR) effect or surface modification. 4. Biocompatible and Biodegradable components (phospholipids). (127) |
1. Lower Cost: Non-ionic surfactants are often less expensive and more readily available than phospholipids. 2. High Stability: Better chemical and physical stability, allowing for easier storage and scale-up. 3. Flexibility: Can entrap both lipophilic (Silymarin) and hydrophilic drugs. 4. Non-Immunogenic: Surfactants are generally well-tolerated. 5. Sustained Release: Capable of offering a prolonged and controlled drug release profile. (123,128) |
1. Enhanced Permeation: Superior delivery into and across the skin due to the ethanol content. 2. Increased Solubility & Encapsulation: High ethanol content solubilizes the drug, leading to high drug loading. 3. Non-Invasive Delivery: Enables systemic absorption via the skin (transdermal) or topical action. (125) |
1. Increased Bioavailability: Overcomes silymarin's inherent poor solubility and low absorption. (120) 2. Hepatoprotective Targeting: The lymphatic uptake mechanism of SLNs makes them particularly effective for delivering drugs to the liver, which is the primary target organ for silymarin's action. (120) 3. Controlled and Sustained Release: The slow degradation of the solid lipid core ensures a prolonged release of silymarin, maintaining therapeutic levels over an extended period. (112) |
|
Best Application |
1. Hepatoprotection (Oral): Treatment and management of chronic liver diseases (cirrhosis, hepatitis, fatty liver disease). (129) 2. Systemic Effects: Leveraging its enhanced antioxidant and anti-inflammatory properties for general systemic health, dermatology, and neuroprotection. (130) |
1.Treatment of liver disorders: Hepatitis, Cirrhosis, Non-Alcoholic Fatty Liver Disease (NAFLD). 2.As an antioxidant and anti-inflammatory agent in cosmetics, food supplements, and drug delivery for other conditions like cancer or neuroprotection. (116,131,132) |
Primary: Oral delivery for treating liver diseases (Hepatitis, Cirrhosis, NAFLD) where targeted liver uptake and enhanced efficacy are needed. (103) Other: Used in transdermal delivery systems and potentially for topical and cosmetic applications due to the nature of the surfactants. (128) |
1.Topical/Transdermal Delivery: Treatment of skin conditions (e.g., Psoriasis, skin cancer prevention). (125) |
1. Oral Therapy: Primarily used to improve the efficacy of silymarin in the treatment and management of various liver diseases( cirrhosis, Hepatitis) (133) 2. Targeted Drug Delivery: Potential for targeted delivery of silymarin to specific liver cells by modifying the SLN surface. (134) |
|
Limitations |
1. Dosage/ Molar Ratio: The ratio of drug to phospholipid is fixed, meaning large amounts of phospholipid are required for high-dose herbal extracts. (135) 2. Limited Load: The technology is best suited for low molecular weight, poorly lipid-soluble compounds (like Silymarin). |
1. Low Stability: Prone to chemical instability (oxidation, hydrolysis of phospholipids) and physical instability (aggregation, fusion, leakage) over long periods or at high temperatures. (136) 2. High Cost: High production cost for industrial scale-up and requires specialized storage (e.g., refrigeration). (121)
|
1.Purity/ Toxicity of Surfactant: Potential for toxicity or unwanted effects if the non-ionic surfactant is not biocompatible, or if residual organic solvent remains. (137) 2. Low Skin Penetration: Similar to liposomes, niosomes can be restricted to the upper layers of the skin for topical applications (unless modified). (138) 3. Leakage: Susceptible to leakage of the encapsulated drug. (139) |
1. Ethanol-Related Issues: High ethanol content can potentially cause skin irritation (for topical use) and may lead to vesicle leakage or precipitation if concentration is not optimized. (140) |
1. Potential Drug Expulsion: As the lipid core crystallizes, silymarin can be expelled from the matrix, leading to reduced drug loading capacity and a faster release profile than desired. (141) 2. Low Drug Loading: The amount of silymarin that can be loaded into the SLN core is often limited by the ordered crystalline structure of the lipids. (142) |
Quercetin is a prominent flavonoid, a type of plant pigment, that’s found in many fruits and vegetables like onions, garlic, apples, berries and cherries. Its chemical structure, a flavonol compound that contains five hydroxyl groups and responsible for strong biological activities. Quercetin is a powerful antioxidant that can help protect the body from oxidative stress and resultant cellular damage caused by free radicals. It also has strong anti-inflammatory, antiviral, anticancer, cardioprotective and neuroprotective effects. Quercetin also suffers from poor bioavailability and low water-solubility and poor chemical stability like other phytochemicals which restricts it to be absorption by the body for therapeutic purpose. To address these challenges, researchers have developed novel drug delivery system to enhance its solubility, stability, and absorption, thereby improving its therapeutic efficacy and bioavailability.(143,144)
Table 4. Comparison of features between Quercetin nanocarriers
|
|
Quercetin Phytosome |
Quercetin Liposome |
Quercetin Niosome |
Quercetin Ethosome |
Quercetin SLNs |
|
Basic composition |
Active Ingredient: Quercetin (a flavonoid, poorly soluble). Carrier: Phospholipids, typically Lecithin (e.g., from soy or sunflower, mainly phosphatidylcholine). (145,146) Structure: A molecular complex, The Quercetin molecule is complexed/bound to the phospholipid molecule, where the phospholipid's polar head binds to the polar hydroxyl groups of Quercetin. (147) |
Lipids (Primary): Phospholipids (e.g., Phosphatidylcholine (PC), DPPC, DMPC) forming the core bilayer structure. (148,149) Stabilizer: Cholesterol is often included to increase the mechanical strength, reduce membrane fluidity, and decrease the leakage of the encapsulated drug. Drug Component: Quercetin (a hydrophobic/ amphiphilic flavonoid), which is primarily inserted within the hydrophobic lipid bilayer. Aqueous Phase: Water or buffer (e.g., Phosphate Buffered Saline). (148) |
Active Ingredient: Quercetin (a poorly soluble, lipophilic flavonoid). Membrane Formers: Non-ionic Surfactants (e.g., Span 60, Tween 80). These molecules self-assemble into a bilayer structure. Stabilizer: Cholesterol (or its derivatives) is included in the bilayer to enhance membrane rigidity, integrity, and stability. Structure: Spherical vesicles with an aqueous core and a bilayer shell composed of non-ionic surfactants and cholesterol. (150,151) |
Active Ingredient: Quercetin (a lipophilic flavonoid with poor water solubility and low permeability). Lipid Component: Phospholipids (e.g., Phosphatidylcholine, Egg Phosphatidylcholine). High Concentration of Alcohol: Ethanol (20–50% w/w) or a combination of ethanol and other alcohols like propylene glycol. This makes the vesicles highly deformable and enhances skin penetration. Solvent: Water (Aqueous phase). (18,152,153) |
Solid Lipids: A core matrix made of lipids that are solid at room and body temperature (e.g., stearic acid). Surfactant: An emulsifier (e.g., Poloxamer 407, Span 60) to stabilize the nanoparticles in the aqueous phase. Co-surfactant/ Stabilizer: Sometimes used to further improve stability. Quercetin: The active drug is loaded into the solid lipid core. (154) |
|
Interaction with Quercetin |
Chemical/ Molecular Bonding: Quercetin forms a stoichiometric complex with the phospholipid molecule, usually via hydrogen bonding between the phenolic hydroxyl groups of Quercetin and the phosphate and choline groups of the phospholipid.(147) Conversion to Lipophilic Form: This molecular binding effectively converts the poorly soluble Quercetin into a lipid-compatible (lipophilic) complex, allowing it to mix better with gut lipids and easily cross the lipophilic cell membranes. (146) |
Encapsulation & Protection: Quercetin, being largely lipophilic with hydroxyl groups, partitions primarily into the lipid bilayer of the liposome, sometimes anchoring near the hydrophilic head groups. (155) Stabilization: Encapsulation protects Quercetin from degradation factors like oxidation, hydrolysis, and rapid metabolism in the gut and liver. (156) |
Encapsulation: Quercetin, being largely lipophilic, is primarily entrapped within the lipophilic bilayer of the niosome membrane. Physical Interaction: The hydroxyl (OH) groups of Quercetin can interact with the polar head groups of the non-ionic surfactant via hydrogen bonding, further stabilizing the structure. (150) Increased Solubility: The encapsulation solubilizes Quercetin, which has extremely low aqueous solubility ( 1 μg/mL) (150,157) |
Encapsulation: Quercetin, being a lipophilic molecule, is entrapped within the phospholipid bilayer of the ethosome. Solubilization: The high ethanol content helps solubilize the poorly water-soluble Quercetin within the system, increasing the total drug loading. Stabilization: Encapsulation protects Quercetin from chemical degradation (e.g., oxidation) compared to a free solution. (144,158) |
Quercetin, being a lipophilic molecule, is dissolved or dispersed within the solid lipid core. This encapsulation protects it from degradation, such as in the acidic gastric environment, and improves its solubility in the gastrointestinal tract. The lipid matrix acts as a carrier, shielding the quercetin molecules. (159) |
|
Bioavailability |
Dramatically Enhanced: Oral absorption and systemic bioavailability are vastly improved, often reported to be up to 20 times greater than unformulated Quercetin in human studies. (146) Mechanism: The phospholipid complex mimics the cell membrane structure, facilitating its passage through the gut's lipid-rich enterocyte membranes and subsequent absorption into the bloodstream. (146) |
Greatly Enhanced Bioavailability: Liposomal encapsulation dramatically increases both the solubility and absorption of Quercetin. (160) Mechanism: Liposomes can be absorbed via endocytosis or fuse with the cell membrane, allowing the payload to bypass normal gastrointestinal absorption barriers and first-pass metabolism, resulting in higher plasma concentrations and improved tissue distribution. (161) |
Observed Increase: Studies show a notable increase in systemic absorption and bioavailability compared to free Quercetin solution. The nanosize also facilitates cellular uptake. Sustained Release: Can provide a prolonged release profile, leading to sustained therapeutic plasma concentrations. (162,163) |
Significantly Enhanced: Ethosomes drastically increase the bioavailability of Quercetin, especially via the transdermal route. Observed Increase (Transdermal): Studies show a significantly higher maximum concentration (Cmax?) and area under the curve (AUC) in rats following transdermal administration of Quercetin ethosomal gel compared to non-ethosomal gel or oral suspension. (164,165) |
Significantly increased compared to free quercetin. (e.g. >5 fold in rats for QT-SLNs).(166) The small size of SLNs (typically 50–1000 nm) and their lipid composition allow for enhanced absorption through the lymphatic system and Peyer's patches, bypassing first-pass metabolism in the liver. This leads to a higher concentration of quercetin reaching systemic circulation (167,168) |
|
Stability |
Enhanced Stability: Complexation with phospholipids protects Quercetin from degradation (e.g., oxidation, enzymatic degradation) in the gastrointestinal tract. (147) Physical Stability: Generally exhibits good physical stability. The formation of a complex, tends to offer better stability against leakage and breakdown. (169) |
Enhanced Stability: Offers better protection against chemical changes (oxidation, light) and thermal degradation compared to free Quercetin. Physical Stability Concerns: Liposomes can be susceptible to aggregation, fusion, or drug leakage upon prolonged storage, especially at higher temperatures. Use of stabilizing agents (like cholesterol or PEGylation) and proper storage (e.g., 4?C) is critical. (156,170) |
Chemical Stability: Encapsulation protects Quercetin from degradation factors like oxidation, hydrolysis, and enzymatic breakdown in the GI tract. Storage: Stable over periods of cold storage (e.g., up to 30 days) with minimal change in particle size and entrapment efficiency. (151,171) |
Dependent on Formulation: Stability over time (e.g., 3 months) in terms of particle size and drug leakage is highly dependent on the type and concentration of the phospholipid and alcohol used. (164) Good Short-Term Stability: Optimized formulations typically exhibit good physical stability and maintain a high entrapment efficiency. (171) Protection: The vesicular structure offers a degree of protection against chemical degradation (oxidation, hydrolysis). (164) |
High stability due to the solid lipid core. The crystalline structure of the lipid protects quercetin from chemical degradation (e.g., oxidation). SLNs also prevent aggregation and drug leakage, providing a longer shelf-life compared to liquid lipid-based systems like liposomes. (172) |
|
Advantages |
1. Superior Bioavailability: The key benefit, enabling effective therapeutic blood levels from lower doses.(146) 2. Efficacy: Maximizes Quercetin's inherent therapeutic effects (antioxidant, anti-inflammatory, immune support). (173–175) 3. Safety/Tolerability: Uses food-grade, endogenous phospholipids (lecithin), ensuring high biocompatibility and safety. (146,175) |
1. Increased Efficacy: The enhanced bioavailability translates to better therapeutic outcomes at lower doses, including superior antioxidant, anti-inflammatory, and anticancer activities. (176) 2. Targeting Potential: Can be surface-modified (e.g., with PEG or glucose for brain targeting) to achieve passive or active targeting to specific tissues (like tumor sites). (177,178) 3. Biocompatible: Composed of naturally occurring phospholipids, making them generally non-toxic and biodegradable. (179) |
1. Low Cost/High Stability: Niosomes use non-ionic surfactants, which are often cheaper and more chemically stable than the phospholipids used in liposomes. (180) 2. High Biocompatibility: Non-ionic surfactants are generally non-toxic and biodegradable. (144,181) 3. Versatile Delivery: Suitable for various routes of administration (oral, transdermal, parenteral, nasal), with potential for targeted delivery. (144,180) |
1. Enhanced Skin Penetration: The primary advantage; high ethanol content creates highly deformable vesicles that can penetrate the deep layers of the skin, facilitating systemic delivery via the transdermal route. (182) 2. High Entrapment Efficiency: Can efficiently encapsulate both lipophilic (like Quercetin) and moderately hydrophilic drugs. (183) 3. Biocompatibility: Composed of biocompatible and non-toxic ingredients (phospholipids, ethanol, water). (184) |
1. Enhanced Bioavailability: Overcomes the major limitation of quercetin's poor oral absorption. (166) 2. Controlled Release: The solid matrix allows for sustained release of quercetin, maintaining a therapeutic concentration over a prolonged period. (154) 3. Biocompatible: SLNs show greater biocompatibility, they are well-tolerated with low toxicity. (172) |
|
Limitations |
1. Scale-up: The manufacturing process, involving solvent evaporation and complex formation, can be more complex than simple mixing. (185) 2. Dosing: May still require careful consideration due to possible interactions with certain medications (e.g., those metabolized by Cytochrome P450 enzymes). (186) |
1. Physical Instability: Prone to leakage and degradation during manufacturing, lyophilization, or long-term storage if not properly formulated. (187,188) 2. Complex Pharmacokinetics: Interactions with blood components and clearance by the Reticuloendothelial System (RES) can lead to a short blood circulation time unless modified (e.g., PEGylated liposomes). (144) |
1. Physical Stability Concerns: Niosomes can still suffer from issues like aggregation, fusion, and drug leakage over long-term storage, which is dependent on formulation (surfactant type, cholesterol content). (137) 2. Entrapment Efficiency: The efficiency of encapsulating the drug is highly dependent on the formulation parameters and may be suboptimal if not carefully optimized. (144) |
1. High Ethanol Content: The presence of a large amount of ethanol may cause irritation or a drying effect upon topical application in some individuals. (153)
|
1. Potential for Drug Expulsion: If the lipid matrix crystallizes too perfectly, quercetin molecules can be expelled from the core, leading to drug leakage. (172)
2. Low Drug Loading: The crystalline nature of the solid lipids can limit the amount of quercetin that can be incorporated. (172,189)
3. Scale-up Challenges: Achieving consistent particle size and stability during large-scale manufacturing can be difficult. (190)
|
|
Applications |
1. Dietary Supplements: Widely used as a highly bioavailable oral supplement for general wellness. (146) 2. Immune Support/ Allergy: To support a balanced immune response and help stabilize mast cells to reduce histamine release. (175) 3. Metabolic Health: Potential use in managing conditions like hyperuricemia (high uric acid) and metabolic syndrome. (191) |
1. Oncology: Targeted delivery for colorectal cancer cells to enhance anticancer activity. (192) 2. Neuroprotection: Delivery across the blood-brain barrier for treating neurodegenerative diseases. (177) 3. Dermatology: Topical formulations for skin conditions involving inflammation/oxidative stress associated with pre-cancerous/ cancerous skin lesions. (193) |
1. Oral Supplements: As a dosage form for higher and more consistent systemic absorption of Quercetin. (194) 2. Topical/ Transdermal Delivery: Used for enhanced skin penetration and localized action (e.g., anti-aging, antioxidant effects in the skin). (195) 3. Targeted Drug Delivery: Explored for targeting specific organs (e.g., liver, brain via intranasal route) due to their nanocarrier properties, such as in the management of Parkinson's disease. (196) |
1. Transdermal Delivery: Localized or systemic delivery of Quercetin through the skin (e.g., anti-inflammatory, antioxidant action). (197) 2. Topical Treatment: Incorporation into gels or creams for skin conditions like melanoma or general skin care. (184) 3. Enhanced Systemic Delivery: Alternative route (to oral) for systemic delivery when enhanced absorption is required. (183) |
1. Oral Delivery: Used for dietary supplements and drugs containing quercetin for conditions like inflammation, osteoprotective activity. (198) 2. Topical Formulations: Incorporated into creams and gels to improve the local delivery of quercetin for skin diseases or antioxidant protection. (199) 3. Ocular Delivery: Used for treating eye diseases, as SLNs can improve drug penetration into ocular tissues. (200,201) |
DISCUSSION
The encapsulation of phytochemicals into nanocarriers has emerged as a promising strategy to address the limitations of conventional herbal formulations, particularly low solubility, instability, and poor bioavailability. Among the systems explored,
Phytosomes: offer a unique advantage by forming molecular complexes where the phytochemical is chemically bonded to a phospholipid like phosphatidylcholine. This unique structure makes the phytochemical itself lipid-compatible, enhancing absorption. For curcumin, phytosome significantly boost oral absorption by forming a lipid compatible complex, improving uptake and stability. In studies, showing an increase in plasma curcumin levels by 29 times compared to unformulated curcumin. (e.g. Meriva®). For silymarin, Silymarin's active components (like silybin) are chemically bonded to the polar head of phosphatidylcholine. Silymarin benefits from the synergistic effect of phosphatidylcholine, because Phosphatidylcholine has its own hepatoprotective activity. In studies oral bioavailability of silymarin phytosome (e.g. Siliphos®) is significantly enhanced 6 fold compared to free silymarin. For Quercetin, Quercetin's hydroxyl groups form a molecular complex with the polar head of the phospholipid carrier. Quercetin Phytosome Offers dramatically enhanced oral absorption, reported to be up to 20 times greater than unformulated quercetin in human studies. For maximum oral bioavailability, Phytosomes consistently perform best due to their unique molecular complexation, making them ideal for oral supplements of curcumin, silymarin and quercetin. Certain limitations include applicability only to phytochemicals with polar functional group amenable to complexation.
Liposomes: Liposomes are spherical vesicles made of a phospholipid bilayer surrounding an aqueous core. They can encapsulate both water-soluble (in the core) and fat-soluble (in the bilayer) drugs. In Curcumin, The hydrophobic curcumin molecule is encapsulated within the liposome's lipid bilayer. Encapsulation significantly improves bioavailability up to 8-20 fold increase compared to curcumin suspension. Liposomes allows for high drug loading and can be modified for targeted delivery to tumors (passive targeting via the EPR effect). But face challenges like physical instability, rapid clearance from the body, which often requires modifications like PEGylation. For silymarin, The lipophilic silymarin is encapsulated within the lipid bilayer. Silymarin liposomes improve oral bioavailability up to 4.8 times higher than un-complexed silymarin by protecting it from gastric enzymes and enhancing intestinal absorption. Still, stability enhancement (e.g. cholesterol addition) is needed to address rapid degradation or proliposome form. Quercetin liposomes can be surface-modified for active targeting to specific tissues, such as the brain for neuroprotection or tumors for oncology, but can suffer from physical instability (leakage) and rapid clearance if not properly formulated. Liposomes remain versatile and are particularly valuable when active drug targeting is required (e.g., in oncology), despite their higher cost and lower stability.
Niosomes: Niosomes use non-ionic surfactants to form bilayer vesicles similar to liposomes but offer better chemical stability, cost effectiveness, and longer shelf-life. Curcumin is entrapped within the hydrophobic core of the non-ionic surfactant bilayer, improving bioavailability and chemical protection. Silymarin niosome enhance chemical stability and are cost effective with moderate clinical validation. Quercetin niosomes are noted for stability and affordability, supporting sustained release formulations. Niosomes offers versatility, Suitable for various routes of administration (oral, transdermal, parenteral, nasal). Niosome show less clinical use compared to liposomes but present a promising alternative.
Ethosomes: These are highly flexible and deformable lipid vesicles containing a high concentration of ethanol (20-45%). The ethanol disrupts the skin's lipid barrier, making ethosomes exceptionally effective for deep skin penetration and transdermal delivery. Curcumin ethosomes excel in transdermal delivery by disturbing skin lipids to deliver drug deeply, bypassing first-pass metabolism but are not suitable for oral use due to ethanol irritation. Silymarin ethosomes are utilized for transdermal therapeutic application but require caution due to ethanol’s skin irritation. Quercetin ethosomes perform strongly in transdermal pathway offering deep skin delivery with limitations of irritation potential. For transdermal and deep skin delivery, Ethosomes are unparalleled due to the penetration-enhancing effect of their high ethanol content.
Solid Lipid Nanoparticles (SLNs): SLNs consist of a solid lipid core that encapsulates lipophilic drugs, providing high physical and chemical stability and controlled sustained release. Curcumin SLNs offer significant protection against oxidation and degradation, extending shelf life and providing controlled release. The bioavailability of curcumin SLNs significantly increased. (e.g. 12 fold increase in rat model). The lipid nature promotes absorption via the intestinal lymphatic system, bypassing first-pass liver metabolism and leading to higher blood concentrations. Silymarin SLNs improve bioavailability and hepatoprotective targeting by bypassing first pass metabolism and ensuring prolonged systemic circulation but face issues with drug expulsion and low loading capacity. The lipophilic Quercetin is dissolved and incorporated into the solid lipid core. The bioavailability of Quercetin SLNs is significantly increased (e.g., >5-fold in rats). SLNs suitable for oral, topical and also ocular drug delivery. While SLNs provide enhanced bioavailability, their crystalline lipid matrix limits drug loading and cause drug expulsion upon lipid polymorphic transitions during storage. SLNs are scalable and biocompatible but formulation optimization is necessary to maximize drug payload.
Overall, while all nanocarriers enhance phytochemical performance, the choice of carrier depends on the physicochemical nature of the phytochemical, the desired route of administration, and the therapeutic target.
CONCLUSION
Nanocarrier formulations have emerged as a major advancement in the phytochemical therapeutic formulations and successfully overcome long-existing issues associated with poor solubility, stability, and bioavailability of compounds including curcumin, silymarin and quercetin. Among the various nanocarriers, Phytosomes consistently stand out as the most effective oral delivery system for the discussed phytochemicals. Their unique mechanism of molecular complexation with phospholipids (e.g., phosphatidylcholine) makes the phytochemical itself lipid-compatible, resulting in dramatic bioavailability enhancement, although their applicability is limited to phytochemicals with polar functional group. Liposomes offer high versatility, capable of encapsulating both water- and fat-soluble compounds and are particularly valuable when active drug targeting (e.g., to tumors via surface modification) is required. However, they face challenges with physical instability and rapid clearance, often necessitating modifications like PEGylation. Niosomes offer a promising, more cost-effective alternative to liposomes by using non-ionic surfactants, providing better chemical stability and a longer shelf-life, offers versatility, Suitable for various routes of administration (oral, transdermal, parenteral, nasal), though they have less clinical validation. Ethosomes are unparalleled for transdermal delivery due to their high ethanol content (20–45%), which effectively disrupts the skin barrier for deep drug penetration, completely bypassing first-pass metabolism. Their use, however, is limited to topical applications due to potential irritation. SLNs provide high physical and chemical stability and a controlled-release profile. Their solid lipid core promotes absorption via the intestinal lymphatic system, effectively bypassing first-pass liver metabolism, which is beneficial for hepatoprotective agents like silymarin, despite limitations in drug loading capacity. The comparative study of curcumin, silymarin, and quercetin shows that the use of these nanocarriers not only improves the pharmacokinetic profile of plant-based compounds but also expand their therapeutic applications, ranging from cancer therapeutics and metabolic management to hepatoprotection and skin protection. However, major challenges still exist such as high cost and scale-up difficulty for large production, and limitations in the drug loading content. Therefore, future research must therefore focus on creating hybrid, multifunctional nanocarriers, advancing promising formulations into clinical trials, and establishing uniform regulatory procedures. With continued advancement nanocarriers are poised to revolutionize phytotherapy by merging the most valuable aspects of traditional medicine with modern pharmaceutical technology. Overall, nanocarriers play vital role in the integration of herbal medicine into mainstream healthcare, offering more effective and reliable natural therapeutic options.
AUTHOR CONTRIBUTIONS:
Conceptualization, Writing original Draft preparation, Data Curation, Writing review and editing by Sanika Kadam; Validation, Investigation, Supervision, by Bharati Tare.
FUNDING: ‘Not relevant’
ACKNOWLEDGMENT:
My sincerest thanks to my professor for providing supervision, mentorship, continuous support, and encouragement received throughout this review preparation as a student. We also thank our librarian, for his dedicated assistance in plagiarism checking and maintaining the academic integrity of this manuscript. Finally, we are grateful to the faculty members of our institute for providing a constructive and supportive environment essential for the completion of this article.
CONFLICT OF INTEREST:
The authors declare no conflict of interest.
REFERENCES


Sanika Kadam, Bharati Tare, Advances in Nanocarriers for the Encapsulation of Phytochemicals: A Comparative Review, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 3083-3120. https://doi.org/10.5281/zenodo.18700610
 10.5281/zenodo.18700610
10.5281/zenodo.18700610