
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
K. K. College of Pharmacy
Keratitis, is an inflammatory condition of the cornea often caused by viral infection such as Herpex Simplex Virus, poses a substantial impact on your therapy due to poor drug bioavailability and rapid tears clearance. Usually, eye drops are affected with poor ocular retention time and limited corneal penetration, reducing therapeutic efficacy. Advanced drug delivery system such as cubosomes and insitu gel rose as a breakthrough approach to overcome the limitations. Hydrophilic and hydrophobic and amphiphilic drug can be encapsulated into a lipid-based nanovesicle called cubosomes which offers targeted controlled release and protection against drug degradation. In cubosomal insitu-gel, the drug loaded cubosome are further incorporated into insitu gel which undergoes phase transition upon contact with ocular physiological triggers (pH, temperature and ions) enhance drug retention and patient compliance by transforming into a viscous gel in the eye which exhibits improved corneal permeation and sustained drug release compared to conventional formulation.
Keratitis is the inflammation of the cornea, particularly caused by the Herpes Simplex Virus (HSV) which led to vision impairment or even blindness if left untreated. 1-3 The worldwide occurrence of herpetic keratitis is estimated to be around 1.5 million cases annually, with approximately 40,000 of these leading to severe vision loss due to corneal scarring and clouding. 4-5A variety of risk factors contribute to the development of keratitis. Infectious causes include microorganisms such as bacteria, fungi, viruses, and amoebae, while non-infectious causes include mechanical trauma, chemical or ultraviolet exposure, and improper contact lens use. Notably, viral keratitis has been identified as a leading cause of corneal opacity, significantly contributing to visual morbidity worldwide.6Antiviral drugs like Acyclovir, Valacyclovir, Ganciclovir have been used treat keratitis.7-8
Acyclovir, a purine nucleoside analog, remains the first line antiviral agent for managing HSV induced keratitis.9 However, topical eye drops of Acyclovir is often hampered by poor corneal penetration, low bioavailability, and the need for frequent administration due to rapid precorneal drug loss.10-11The drug which is used to treat HSK which is marketed in the form of eye drops and ointments.4
To overcome these limitations, nanocarrier-based drug delivery systems have gained considerable attention.12 Cubosomes, nanostructured liquid crystalline particles formed from the self-assembly of certain lipids in water and stabilized by surfactants, offer promising advantages. They possess a high internal surface area, biocompatibility, and the capacity to encapsulate hydrophilic, lipophilic, and amphiphilic drugs.13-14 When incorporated into an in-situ gel system, cubosomes can further improve ocular residence time and provide controlled drug release, enhancing therapeutic efficacy while reducing dosing frequency³.
1.1 Anatomy of Eye15
The structure of eye can be divided into two main parts: anterior segment and posterior segment. The anterior segment of the eye consists of the cornea, conjunctiva, aqueous humor, iris, ciliary body, and lens. The posterior segment mainly consists of the vitreous humor, retina, choroid, and optic nerve.
1.2 Topical Ocular Drug Delivery: Challenges and Advances
Medications are often applied to the surface of the eye for two main purposes: to treat external ocular conditions such as conjunctivitis, blepharitis, and keratitis sicca, or to deliver drugs intraocularly through the cornea for diseases like glaucoma and uveitis.16
Topical administration to the anterior segment of the eye primarily utilizes conventional dosage forms, including solutions (62.4%), suspensions (8.7%), and ointments (17.4%), which together account for approximately 90% of marketed ophthalmic formulations. However, despite their widespread use, these forms suffer from poor ocular bioavailability—typically around 0.5%—largely due to rapid clearance from lacrimal secretions and limited permeability across the corneal epithelium.17
A key requirement for any ocular drug delivery system is the absence of adverse ocular reactions, along with a goal to enhance drug bioavailability.18 To address the limitations of conventional formulations, researchers have focused on two primary strategies:
Among these, lipid-based nanocarriers I ncluding emulsions, liposomes, cubosomes, and niosomes offer several promising advantages for ocular delivery. These systems can enhance the bioavailability of poorly soluble drugs, enable targeted and sustained release, and minimize systemic side effects. The eye’s residence volume is typically 7–10 µL, and most topically applied solutions are eliminated within 15–30 seconds. Lipid components in nanocarriers can interact with the lipid layer of the tear film, allowing the formulation to remain longer in the conjunctival sac and function as a drug reservoir.20
1.3 Different types of keratitis:
1.3.1 Herpes Simplex Keratitis
Herpes Simplex Keratitis is a viral keratitis and it is caused by HSV and it can begin with conjunctival inflammation. The symptoms include Chronic corneal inflammation, neovascularization, scarring, glaucoma, vision loss. It can be treated by using Antiviral drugs like Acyclovir, Valacyclovir and Ganciclovir. 21-22
1.3.2 Bacterial Keratitis:
Bacterial keratitis is caused by wearing contact lenses for too long or not keeping them clean and some other pathogens like Staphylococcus aureus, Pseudomonas aeruginosa, Streptococcus pneumoniae, etc.The eye may feel painful, red, watery, and sensitive to light. This can be treated using antibiotics like ciprofloxacin, moxifloxacin, Levofloxacin, etc. 23-24
1.3.3 Acanthamebo Keratitis:
It can be caused by Acanthamoeba species and some other factors include Minimal corneal trauma, soft contact lens use, exposure to contaminated water. It can provide symptoms are Severe pain, redness, photophobia, tearing and can be treated by using Combination of anti-amoebic agents. 25
1.3.4 Photokeratitis:
Photokeratitis can be developed by Excess UV exposure (sunlight, welding arcs, snow reflection, tanning lamps) and symptoms produced are Delayed pain, redness, tearing, foreign body sensation. This can be prevented by Supportive care: Lubricants, cold compresses, avoiding further UV exposure. 26
1.3.5 Interstitial Keratitis:
Interstitial keratitis is a non-ulcerating inflammation of the corneal stroma can be caused by Infections (syphilis, herpes), autoimmune diseases (e.g., rheumatoid arthritis) and it will produce symptoms are Corneal cloudiness, reduced vision, inflammation within corneal layers. Drugs which are used to treat interstitial keratitis are Antibiotics, antivirals, or immunosuppressants. 27-28
1.3.6 Fungal Keratitis:
Fungal keratitis (keratomycosis) is a fungal infection of the cornea can be developed by species like Fusarium, Aspergillus, Candida and climatic changes which will produce symptoms are Pain, redness, photophobia, corneal ulceration. It can be treated by Antifungals: Polyenes (Amphotericin B, Natamycin), Azoles.29-31
1.4 Acyclovir 32-33
Acyclovir (ACV), a 2-deoxy guanosine derivative, is an exemplified drug of choice against herpes simplex virus (HSV), varicella zoster virus (VZV), cytomegalovirus (CMV), Ep stein bar virus (EBV) and human herpes virus 6 (HHV-6). The structure of acyclovir is shown in figure 1.
Acyclovir is a new antiviral drug that acts as a specific inhibitor of herpesvirus DNA polymerase. It shows good in vitro activity against herpes simplex and varicella-zoster viruses. The drug may be administered topically to the skin, intravenously, orally, or topically to the eye.
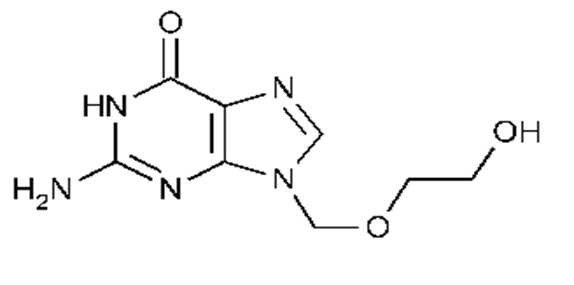
Figure 1 : Structure of acyclovir
1.4.1 Mechanism Of Action:
Acyclovir undergoes initial activation when the viral enzyme thymidine kinase converts it into acyclovir monophosphate, a process that selectively targets virus-infected cells. Subsequently, cellular enzymes transform it into its active form, acyclovir triphosphate. This active metabolite competes with the natural nucleotide deoxyguanosine triphosphate and is incorporated into viral DNA. Because it lacks the essential 3’-OH group, its incorporation leads to premature termination of DNA synthesis, effectively blocking viral replication. This specific mechanism enables acyclovir to suppress viral proliferation while minimizing harm to healthy cells.34-35 The mechanism of action of acyclovir is illustrated in Figure 2 , demonstrating its selective activation in virus-infected cells and subsequent inhibition of viral DNA replication.
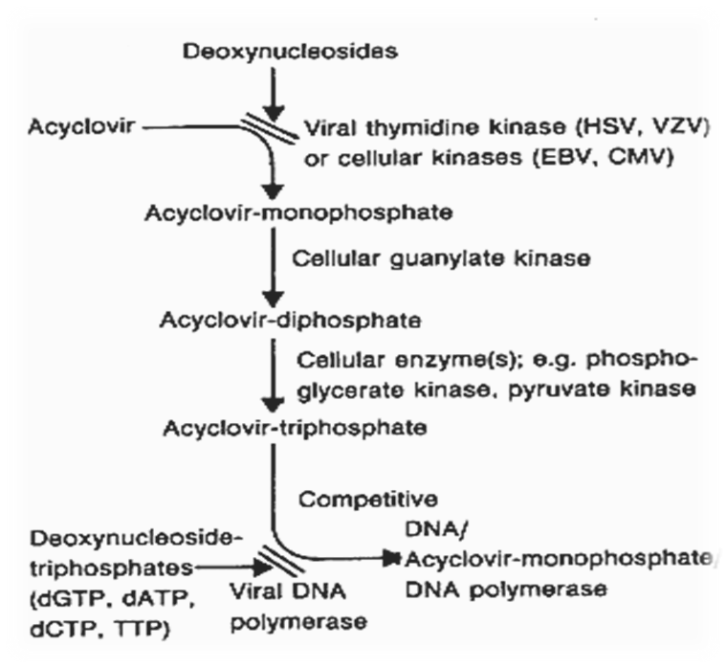
Figure 2 : Mechanism of Action of Acyclovir
This flowchart shows how acyclovir is activated by viral thymidine kinase and incorporated into viral DNA, causing premature chain termination and blocking viral replication.
1.5 Valacyclovir:
Valacyclovir is the l-valyl ester of ACV. It can be synthesized by the addition of an amino acid, L-valine, to the ACV molecule.36Valacyclovir was developed for the treatment of infections caused by herpes viruses, including herpes simplex virus (HSV) and varicella-zoster virus (VZV).37 The structure of acyclovir shown in the figure 3.
Valaciclovir is an effective treatment for herpes simplex keratitis.40 It provides effective treatment then acyclovir.38 Its oral bioavailability 3- to 5-fold higher than when given as acyclovir.39
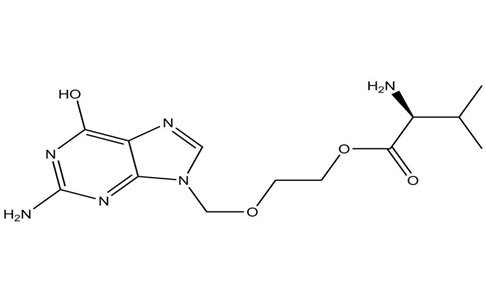
Figure 3 : Structure of Valacyclovir
1.5.1 Mechanism of action:41
Valacyclovir is a prodrug of acyclovir, meaning it is converted into acyclovir in the body to exert its antiviral effects.
1.6 Ganciclovir:
Ganciclovir is a synthetic nucleoside analogue of 2′-deoxyguanosine.43 It is an antiviral agent that is used to treat or cytomegalo virus (CMV) infections.42
Ganciclovir available in the form of 0.15% gel was more recently approved by the FDA in the United States for the topical treatment for herpes keratitis.44,45 Figure 4 shows the structure of ganciclovir.
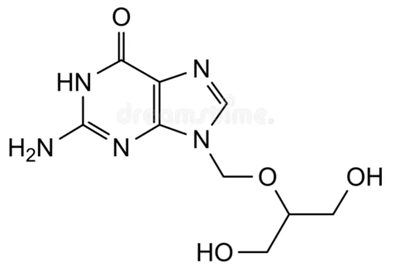
Figure 4: Structure of Ganciclovir
1.6.1 Mechanism of Action:
Ganciclovir is first phosphorylated to ganciclovir monophosphate after entered into infected cell. This initial phosphorylation is catalyzed by virus-encoded enzymes: for cytomegalovirus (CMV), it is the UL97 protein kinase; for herpes simplex viruses (HSV), it is the viral thymidine kinase.46 Then the monophosphate is then converted to ganciclovir diphosphate and subsequently to ganciclovir triphosphate by host cellular kinases.47
Ganciclovir triphosphate acts as a competitive substrate for viral DNA polymerase specifically, it competes with deoxyguanosine triphosphate (dGTP) and is preferentially incorporated into viral DNA. Once incorporated it inhibits further DNA elongation. Inhibition of viral replication is finally occurred.48
2. Overview of Cubosomes and Their Role in Drug Delivery49-54
The term “cubosome” was first introduced by Larsson, inspired by the resemblance of their internal structure to cubic molecular crystals, a configuration that was definitively confirmed in 1967. Cubosomes are dispersed colloidal particles derived from cubic phase liquid crystals and have garnered considerable attention in pharmaceutical research due to their potential as controlled-release drug delivery systems.
Cubosomes possess the unique ability to self-assemble in aqueous environments, forming nanoparticles typically ranging from 100 to 500 nanometers in size. Their internal structure resembles a honeycomb-like lattice with interconnected aqueous channels, known as bicontinuous cubic phases, which are stabilized at specific water concentrations. This well-defined architecture provides a large internal surface area and an ordered network that is ideal for drug encapsulation and delivery.
Primarily composed of amphiphilic lipids such as monoolein and phytantriol, cubosomes can simultaneously encapsulate both hydrophilic and lipophilic drugs. Hydrophilic drugs are sequestered within the aqueous channels or vesicles inside the cubosome, while lipophilic drugs are distributed within the lipid bilayer domains. This dual capacity for encapsulating diverse drug molecules makes cubosomes versatile and promising carriers for enhancing drug bioavailability, targeting, and controlled release in various therapeutic applications.
The structure of cubosome is illustrated in figure
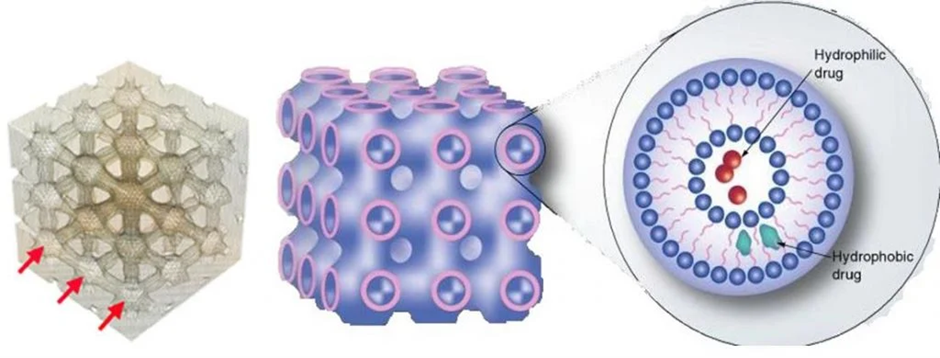
Figure 5 : Structure of Cubosome
This figure depicts the internal bicontinuous cubic phase structure of a cubosome, showing the honeycomb-like arrangement with interconnected aqueous channels. The amphiphilic lipids, such as monoolein and phytantriol, form the lipid bilayers that encapsulate hydrophilic drugs within the aqueous domains and lipophilic drugs within the lipid regions, enabling efficient drug delivery.
2.1 Components Of Cubosome
Table 1. Components of cubosomes along with its structure.
|
Component |
Description |
Image |
|
Amphiphilic Lipids |
||
|
Glycerol Monooleate (GMO) |
GMO is a glyceride of oleic acid and other fatty acids (C12–C22) that forms a cubic structure. It is biodegradable due to the ester bond and is classified as a Generally Regarded As Safe (GRAS) excipient 55. |
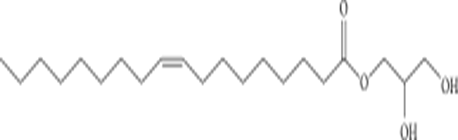
|
|
Phytantriol |
A polyhydroxy alcohol with three hydroxyl groups on one end and a long hydrocarbon chain on the other. It is stable in aqueous conditions because it lacks an ester bond 56 |
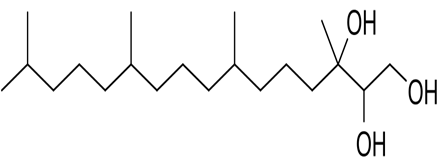
|
|
Monoelaidin |
Contains a trans double bond at positions 9 and 10 (C18:19). Its self-assembled water system undergoes lamellar to nonlamellar phase transitions, making it a suitable model for studying membrane fusion processes in cells 57 . |
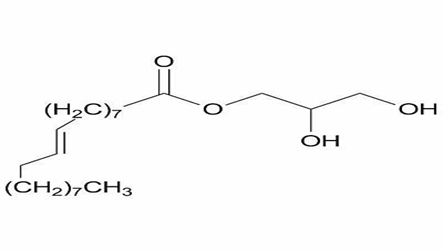
|
|
Stabilizers |
||
|
Pluronic F127 |
A self-assembling triblock copolymer composed of PEO-PPO-PEO blocks. PPO is hydrophobic and anchors to the particle surface, while PEO is hydrophilic and extends into the aqueous phase, providing steric or electrostatic stabilization 58. |
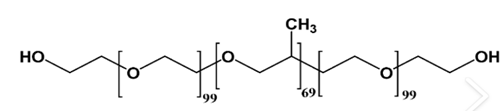
|
|
Poloxamine 908 |
Also known as Tetronic 908, it is a tetrafunctional PEG-PPO-ethylene diamine block copolymer used to stabilize cubosomal dispersions. 59-60 |
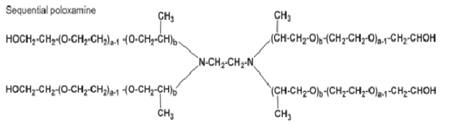
|
2.2 Preparation Of Cubosome:
Cubosomes can be prepared through various methods that utilize the unique self-assembling properties of amphiphilic lipids in aqueous environments. These methods aim to create stable nanostructured particles with internal cubic phase geometry, optimized for efficient drug delivery. The preparation techniques generally fall into two categories: top-down, which breaks down bulk cubic phases into nanoparticles using high energy, and bottom-up, which forms cubosomes from molecular precursors with minimal energy input. Additional advanced techniques such as fragmentation, thin film hydration, spray drying, and emulsification have also been developed to improve scalability, drug loading, and formulation stability
2.2.1 Top-down approach:61-63
The top-down approach is one of the most widely utilized techniques for the preparation of cubosomes. It involves two fundamental steps. Initially, a cubic phase gel is formed by blending cubosome-forming lipids, such as monoolein or phytantriol, with an appropriate stabilizer like Pluronic F127. This viscous gel represents the bulk cubic phase. Subsequently, high-energy dispersion techniques such as high-pressure homogenization or probe sonication are employed to fragment and disperse the bulk gel into nanoscale cubosomal particles in an aqueous medium. This method ensures the production of uniform, stable colloidal dispersions of cubosomes suitable for drug delivery applications.
2.2.2 Bottom-up approach:53
This approach, commonly known as the solvent dilution method, involves dispersing a mixture of cubosome-forming lipid, a stabilizer, and a hydrotrope into an excess of water. Upon dilution, the hydrotrope helps solubilize the lipid, allowing spontaneous self-assembly into cubosomes. This method requires minimal energy input and is suitable for temperature-sensitive drugs.
2.2.3 Fragmentation Method:64
In this method, glycerol monooleate and Poloxamer 407 are melted together at 60?°C in a hot water bath. The drug is then added and stirred until completely dissolved. Water is introduced dropwise with continuous vortex mixing to form a homogeneous solution. The resulting mixture undergoes probe sonication in cold water at 20?°C for 10 minutes, followed by high-pressure homogenization to yield a clear cubosomal dispersion.
2.2.4 Spray Drying:65-67
In the spray drying method, the lipid and stabilizer mixture is first dissolved in an organic solvent, commonly ethanol, although sometimes a mixture of solvents (binary solutions) is used. Meanwhile, a separate aqueous phase containing a hydrophilic solid carrier—such as sorbitol or dextran is prepared. These carriers help to stabilize the powder and improve its properties.The two mixtures are then combined to create a homogeneous solution. This mixture is atomized into fine droplets by spraying it into a chamber filled with hot air. The hot air causes rapid evaporation of the organic solvent from the droplets. As the solvent evaporates quickly, the lipid and carrier materials solidify, forming a dry powder composed of cubosomes.
This process allows the transformation of the lipid-surfactant solution into a dry, stable powder form, which is easier to handle, store, and transport. The use of hydrophilic carriers helps prevent clumping and enhances the dispersibility of the powder when it is later reconstituted. Spray drying is effective due to its speed and ability to produce powders with controlled particle size and morphology, making it suitable for pharmaceutical applications.
2.2.5 Emulsifying Method:68-69
In the emulsifying method, the lipids and stabilizers are melted together on a magnetic stirrer with a hot plate to form a molten mixture, which will become cubosomes. The drug substance is usually dissolved either in the aqueous phase or in ethanol that contains polyvinyl alcohol (PVA) or a surfactant, both of which help stabilize the final dispersion. Alternatively, the drug can be dispersed directly into the molten lipid-stabilizer mixture.
Once the lipid mixture is molten, it is injected into the pre-heated aqueous phase under continuous magnetic stirring. This mixture is then emulsified using a homogenizer, which helps create a stable dispersion of cubosomes by breaking down the lipid phase into fine particles within the aqueous medium. This process ensures efficient encapsulation of the drug and stable formation of cubosomes.
2.3 Advantages:70-71
2.4 Disadvantages:53,72
3. Insitu Gel:73-77
Gel systems offer better retention in the eye compared to conventional eye drops and are generally better tolerated by patients than inserts and ointments. An ideal in situ gelling system should be a low-viscosity, free-flowing liquid to allow easy and reproducible administration as eye drops. After administration, it should quickly undergo a phase transition to form a gel that is strong enough to withstand the shear forces in the eye’s cul-de-sac, ensuring prolonged residence time.
In situ gel-forming systems start as liquid aqueous solutions before application and transform into gels under physiological conditions. These systems can be easily instilled as eye drops and gel immediately upon contact with the eye, improving drug retention and effectiveness. This process is illustrated in Figure .
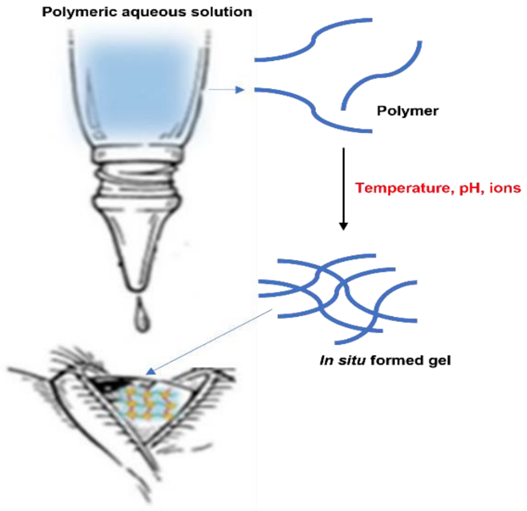
Figure 6 : In Situ Gel Formation of Polymeric Aqueous Solution upon Ocular Administration
Schematic representation of a polymeric aqueous solution instilled as eye drops that undergoes a phase transition to form an in-situ gel upon contact with the eye. This gelation increases the retention time of the formulation in the ocular cul-de-sac by transforming from a free-flowing liquid to a viscous gel.
3.1 Types Of In Situ Gel-Forming Systems Based On Phase Transition Triggers
In situ gel-forming systems undergo a sol-to-gel phase transition upon contact with the eye, triggered by specific physiological conditions.
Depending on the mechanism that causes this transition, three main types of systems are widely accepted:
In pH-triggered in situ gelation, the polymer swells and forms a gel in response to changes in the external pH. For polymers containing weak acidic (anionic) groups, swelling occurs as the pH increases. 78Conversely, for polymers with weak basic (cationic) groups, swelling happens when the pH decreases. Most commonly used anionic pH-sensitive polymers are based on polyacrylic acid (PAA), such as Carbopol or carbomer, and their derivatives. When the pH shifts from around 4.2 to the physiological pH of 7.4, a sol-to-gel phase transition takes place due to ionization changes in the polymer structure.83
Examples of polymers used in pH-triggered systems include cellulose acetate phthalate, latex, carbopol, polymethacrylic acid, polyethylene glycol (PEG), and pseudo-latexes.84
In pH-triggered in situ gelation, polymers swell and form gels in response to changes in external pH. Polymers with weak acidic (anionic) groups swell as the pH increases, while those with weak basic (cationic) groups swell when the pH decreases. The most commonly used anionic pH-sensitive polymers are based on polyacrylic acid (PAA), including Carbopol, carbomer, and their derivatives. A sol-to-gel transition occurs when the pH shifts from around 4.2 to the physiological pH of 7.4, driven by ionization changes in the polymer.79
Polymers frequently used in these systems include cellulose acetate phthalate, latex, carbopol, polymethacrylic acid, polyethylene glycol (PEG), and pseudo-latexes.85
Ion-activated in situ gels undergo a sol -to-gel phase transition triggered by the presence of ions in the surrounding environment. The formulation transforms from a liquid to a gel when exposed to an increase in ionic strength. For example, Gelrite, an anionic polysaccharide, forms a gel in the presence of mono- and divalent cations, particularly calcium ions (Ca²?). Other commonly used polymers in ion-activated systems include chitosan, gellan gum (Gelrite), and alginate.
3.2 Advantages:81
3.3 Disadvantages:82
CONCLUSION:
There are some challenges in the production of cubosomes, such as the complexity of large-scale manufacturing and the need for comprehensive long term safety studies. Therefore, strategies such as optimizing scalable manufacturing techniques exploring additional bio active compounds for incorporation under conducting extensive clinical trials to validate efficacy can be focused for the further research. Moreover, targeted Delivery and Controlled release profiles can be enhanced through the integration of multifunctional Stimuli response polymer and nanocarriers.
REFERENCES



Manojkumar Kathiravan, Shanthy Arumugam, Meena Ramaswamy, An Overview of Keratitis and Its Treatment with Antiviral Cubosomal Insitu Gel, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 9, 2628-2642. https://doi.org/10.5281/zenodo.17182813
 10.5281/zenodo.17182813
10.5281/zenodo.17182813