
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Ideal Institute of Pharmacy, Posheri, Wada Palghar.
Ficus racemosa Linn. (Family; Moraceae) popularly known as the cluster fig tree or Gular, is famous medicinal plant in India, which is used in traditional system of medicine for a long time, for the treatment of various disorders like diabetes, liver disorders, diarrhoea, inflammatory conditions, haemorrhoids, respiratory and urinary diseases. This herb is mentioned in all ancient scriptures of Ayurveda, Siddha, Unani and Homeopathy. Various parts of these plants—such as roots, bark, latex, leaves, fruit pulp, and the fruits themselves—are rich in biologically active constituents.These compounds include flavonoids (like flavanols, flavones, flavonols, isoflavones, chalcones, and anthocyanins), phenolic acids (such as hydroxycinnamic and hydroxybenzoic acids), phytosterols, a wide range of terpenes (triterpenes, tetraterpenes, diterpenes, sesquiterpenes, and monoterpenes), along with coumarins, hydroxybenzoates, phenylpropanoids, chlorins, pheophytins, megastigmanes, chitinases, organic acids, fatty acids, amino acids, alkaloids, and glycosides. Collectively, these compounds support over 30 traditional ethnomedicinal uses. It is a good remedy for excessive appetite. The extract of fruit is used in diabetes, leucoderma, refrigerant, antiasthmatic, hepatoprotective, antioxidant, antiulcer and menorrhagia. It is used locally to relieve inflammation of skin wounds, lymphadenitis, in sprains and fibrositis. The present review is therefore, an effort to give a detailed study in Pharmacogonstical, phytochemical & Pharmacological properties.This first part of the review summarizes findings from various published studies (including in vitro, in vivo, and clinical research) on the antimicrobial, antifungal, antiviral, antihelminthic, hypoglycemic, hypolipidemic, hepatoprotective, anti-inflammatory, analgesic, and antipyretic activities of the plant, along with its potential adverse and toxicological effects.
The genus Ficus comprises a significant group of trees known for their diverse chemical compounds with notable medicinal properties. Revered in both Hindu and Buddhist traditions, this genus includes four key species that form the group called “Nalpamaram.” These are Ficus racemosa, Ficus microcarpa, Ficus benghalensis, and Ficus religiosa, traditionally referred to as Athi, Ithi, Peral, and Arayal, respectively.1 Ficus racemosa Lin. is a large deciduous tree commonly found across India, especially in evergreen forests and humid regions. Various parts of the plant including the root bark, leaves, fruits, and galls are utilized for their medicinal benefits. Traditionally, the bark, leaves, and unripe fruits have been recognized for their carminative, astringent, digestive, and anthelmintic (vermifuge) properties.According to descriptions in Ayurvedic texts such as the Nighantus, a decoction prepared from the bark, fruit, and leaves is described as cooling, sweet, and astringent in nature.2 Chemically, the bark contains compounds like gluanol acetate, beta-sitosterol, and leucocyanidin. The leaves are rich in beta-amyrin, beta-sitosterol, and tannins, while the fruits contain lupeol acetate, glucose, sterols, and gluanol acetate. India, with its abundant biodiversity, is often referred to as the botanical garden of the world.3 A wide range of medicinal plants found here serve as vital sources for new drug discoveries and have long been used in traditional healing systems. Ficus racemosa is also known by several names, including Udumbara, Yajnanga, Yajniya, Ajnayoga, Yajnyasara, Gular, Cluster Fig Tree, and Country Fig Tree. The tree has cultural and religious significance and is often used in ritual sacrifices. It belongs to the group of trees known as Ksiri Vriksha, which are characterized by the exudation of milky latex when leaves are cut.4 It is among the plants included in a group known as Pancavalkala, which refers to the thick barks of five herbs: udumbara, vata, asvattha, parisa, and plaksa. A decoction made from Pancavalkala is administered orally or as an enema to treat bleeding from the rectum or vagina. According to Maharishi Charaka, udumbara is classified as mutra sangrahaniya, meaning it acts as an anti-diuretic herb.2
Africa, Asia, Australia, Bangladesh, Cambodia, China, East Africa,East Timor, Egypt, Ethiopia, Hawaii, Himalayas, India, Indochina, Indonesia, Laos, Malaysia, Myanmar, Nepal, North Africa, Northeastern India, Pacific, Pakistan, Papua New Guinea, PNG, Southeast Asia,Sikkim, Singapore, Sri Lanka, Taiwan, Thailand, Timor-Leste, USA, Vietnam.64
Ficus racemosa is an attractive cluster-fig tree with a curved trunk and a wide, spreading crown. Unlike banyan trees, it does not develop aerial roots. Its most striking feature is the small, red figs that grow in clusters directly on the trunk. Each tree produces hundreds of figs, and their flowers are pollinated by tiny wasps that enter through the fig’s opening to find a suitable site for laying eggs. These wasps are essential for the tree’s reproduction, as fig trees cannot produce seeds without them.65 In return, the flowers offer a safe habitat and nourishment for the wasps’ offspring. Ficus racemosa is commonly found across villages, towns, and cities.

Fig. 1 : Fruits, leaves, trunk of Ficus racemosa lin.(75)
Table 1: Taxonomic Classification of Ficus racemosa Lin.(5)
|
Kingdom |
Plantae |
|
Division |
Magnoliophyta |
|
Class |
Magnolipsida |
|
Order |
Urticales |
|
Family |
Moraceae |
|
Genus |
Ficus |
|
Species |
racemosa |
Table 2 : Vernacular Names of Ficus racemosa Lin.(10,11)
|
Sr.No. |
Language |
Names |
|
1 |
Sanskrit |
Yajnayoga, Sadaphalah, Brahanvrkisha, Shitavalkah, Sutah, Udumbara, Gular, Mashakin, Jantukaphalah, Jantuphalah, Krmiphalah, Vasudrumah, Saumya, Hemadugdhaka, Jantumati, Yagniyah, Audumbara |
|
2 |
Hindi |
Pushp-hina, Pani Bhuj, Dumar, Goolar, Umari, Yajnyadumbur, Udumbara, Jantu Phal, Dharma Patra, Goolar |
|
3 |
English |
Gular fig, Cluster Fig, Country Fig |
|
4 |
Bengali |
Udumbara |
|
5 |
Telugu |
Brahmamamidi, Atti, Bodda |
|
6 |
Gujrati |
Goolar, Umbaro |
|
7 |
Manipuri |
Heibong |
|
8 |
Malayalam |
Atthi, Atthi Al, Aththi, Atthi-al, Udumbaram, Jantuphalam |
|
9 |
Marathi |
Umbar, Udumbar |
|
10 |
Urdu |
Dumar |
|
11 |
Others |
Goolar, Atthi, Atteeka, Athi, Crattock, Country Fig, Dumrii, Cluster Fig, Vellaiatthi, Gular Fig, Indian Fig, Redwood Fig, Rumbodo |
Table 3: Behavior of the Ficus racemosa Lin. bark powder with different chemical reagents.(12)
|
Sr. No. |
Reagent |
Colour/Precitate |
Result |
|
1 |
Picirc acid |
No precipitation |
Alkaloids absent |
|
2 |
Ammonia solution |
No change |
Anthraquinone glycoside absent |
|
3 |
5% Aq.KOH |
No change |
Anthraquinone glycoside absent |
|
4 |
Mayer’s reagent |
No precipitation |
Alkaloids absent |
|
5 |
Spot test |
Stain observed |
Fixed oil present |
|
6 |
Aq. AgNO3 |
Precipitation Observed |
Proteins present |
|
7 |
Aq. NaOH |
Reddish brown |
Flavonoids present |
|
8 |
MG-HCL |
Magenta |
Flavonoids present |
|
9 |
Dragendroff’s Reagnt |
No precipitation |
Alkaloids absent |
|
10 |
Aq. Lead Acetate |
White precipitate |
Tannins presents |
|
11 |
Libermann Burchard test |
Redidsh green |
Steroids / Triterpinoids present |
|
12 |
Conc. H2SO4 |
Reddish brown |
Steroids / Triterpinoids present |
|
13 |
Aq. FeCl3 |
Greenish black |
Tannins, flavonoids present |
|
14 |
Iodine Solution |
Blue |
Starch |
*NaOH- Sodium Hydroxide, KOH- Potassium Hydroxide, Mg-HCl- Magnisium metal-hydrochloric acid, FeCl3- Ferric chloride, H2SO4 - Sulphuric acid, AgNO3- Silver Nitrate.
2. Plant authentication details
Leaves of Ficus racemosa Linn. (Moraceae) were collected in March 2022 from Banitabla, located in the Uluberia region of Howrah district, West Bengal, India. The specimen was authenticated by K. Karthigeyan, Scientist-D at the Central National Herbarium, Botanical Survey of India, Howrah (PIN 711103), and a voucher sample was deposited at the Central National Herbarium, Shibpur, Howrah, West Bengal. The collected leaves were shade-dried, ground into a fine powder using a mechanical grinder, and stored in a sealed container for future use.58
3. Cultivation details
Ficus racemosa Linn. is a widely occurring and ecologically important tree in lowland tropical rainforests, thriving both as a canopy and understory species. While most fig species prefer per-humid woodlands, many can also grow in monsoon climates and teak forests, including areas where the soil experiences seasonal drying. Fig trees have a unique reproductive system, relying on a highly specialized wasp species that is entirely dependent on that fig genus for reproduction. The trees produce three types of flowers—male, long-styled female, and short-styled female (also called gall flowers)—all of which are contained within the fig fruit structure. Female fig wasps enter the fig to lay eggs on the short-styled female flowers, while simultaneously pollinating the long-styled female flowers. Wingless male wasps emerge first, fertilize the females, and create exit tunnels for them. The fertilized female wasps then collect pollen from the male flowers and fly out in search of figs with receptive female flowers.(59) To maintain a stable population of its pollinators, Ficus species must flower asynchronously, and the tree population must reach a certain minimum density so that at any given time, some plants are producing figs with overlapping periods of wasp activity.(60-61)
4. Habit And Habitat
The plant is widely distributed across India, thriving in forests and hilly regions. It is commonly seen near water streams and is also cultivated in various areas. The tree reaches a medium height of 10–16 meters, with dense green foliage that offers ample shade. Its bark is reddish-grey and often appears cracked.6 Ficus racemosa is widely cultivated throughout India and also grows naturally in numerous forests and hilly regions. It is commonly found near water sources such as ponds and riverbanks and can be grown artificially as well. The tree is medium to tall, reaching up to 12 meters in height, with lush green foliage that provides ample shade. Its bark is reddish-grey and has a cracked or fissured texture.62-63
5. Morphological Characteristics
5.1. Macroscopical Characteristics
( Plant Description) :This plant is native to Australia, Southeast Asia, and the Indian subcontinent. It grows widely across India, especially in forests and hilly areas.7 It is commonly seen near streams and is also grown in villages for its shade and edible fruits.8 The tree grows to a medium height of about 18 meters and has lush green leaves that offer excellent shade. The leaves are dark green, around 7.5 to 10 cm long, oval or elliptical in shape, and usually appear in large clusters on the older parts of the main trunk.The fruit structures are pear-shaped, measuring 2 to 5 cm in diameter, and grow in large bunches directly from the main trunk or big branches. The fruits look like figs; they are green when unripe and turn orange, dull red, or dark crimson as they ripen.9 The seeds inside are very small, numerous, and look like grains. The roots are long, brown, have a distinct smell, and taste slightly bitter.
5.2. Microscopical Characteristics
The cork consists of polygonal or rectangular cells. The phellogen comprises one or two layers of thin-walled cells. The phelloderm is a distinct, compact tissue primarily made up of parenchymatous cells, often containing isolated or small clusters of sclereids, especially towards the inner side. These sclereids are lignified and exhibit simple pits. Several parenchyma cells contain either a single prismatic crystal of calcium oxalate or a brownish substance. The cortex is broad and contains numerous sclereids, while some cortical cells have a resinous material. Prismatic calcium oxalate crystals are also found in certain cells.6Sclereids are thick-walled, pitted, and either rectangular or roughly equal in dimensions (isodiametric). The phloem is composed of sieve tubes, companion cells, phloem parenchyma, sclereids, phloem fibers, and medullary rays. Starch grains are found in oval to spherical forms. The phloem region also contains laticiferous vessels filled with light brown, granular material. The cambium consists of 2 to 3 layers of thin-walled cells that are elongated in a tangential direction. The surface of the figs is smooth, although it may occasionally have fine, soft hairs.(13,14)
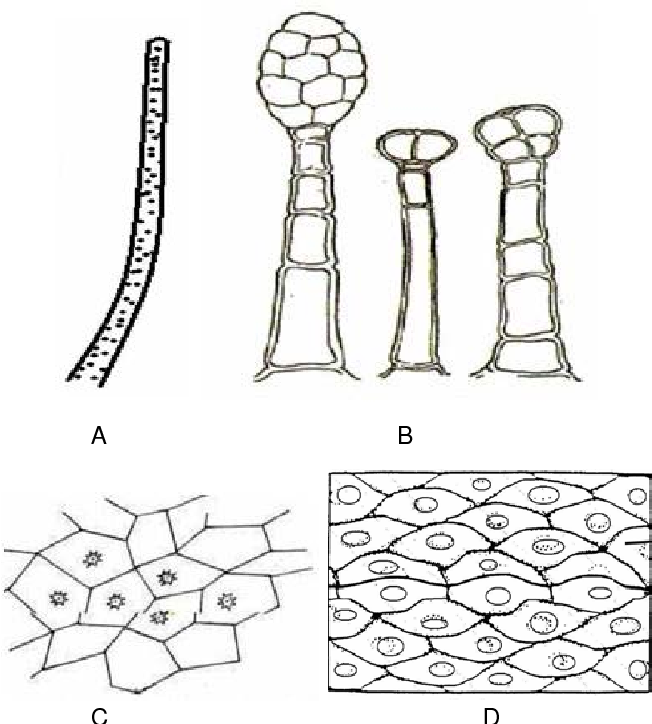
Fig. 2 : Microscopical character of F. racemosa {A-Fiber; B- Glandular Trichoems; C- Calcium Oxlate; D-Stone Cells }(6,12)
Sclereids are thick-walled, pitted, and either rectangular or roughly equal in dimensions (isodiametric). The phloem is composed of sieve tubes, companion cells, phloem parenchyma, sclereids, phloem fibers, and medullary rays. Starch grains are found in oval to spherical forms. The phloem region also contains laticiferous vessels filled with light brown, granular material. The cambium consists of 2 to 3 layers of thin-walled cells that are elongated in a tangential direction. The surface of the figs is smooth, although it may occasionally have fine, soft hairs.(13,14)
5.2.1. Cork
It consists of cells that are either polygonal or rectangular in shape. Cork tissue contains both polygonal and rectangular cells. The phellogen comprises one or two layers of thin-walled cells.
5.2.2. Phelloderm
The phelloderm is lignified and contains simple pits. It consists of a compact tissue made up of parenchyma cells or small groups of sclereids. Many parenchymatous cells enclose a single, reddish-colored prism of calcium oxalate.
5.2.3. Cortex
The cortex is composed of several sclereids that are either rectangular or isodiametric, having extremely thick and pitted walls. The cortical cells contain a resin-like substance. Some cells also possess prismatic crystals of calcium oxalate. The phloem includes sieve tubes, companion cells, phloem parenchyma, sclerenchymatous fibers, and medullary rays. The cambium is made up of 2 to 3 layers of tangentially elongated, thin-walled cells.(66)
5.2.4. Leaf
The leaf exhibits dorsoventral characteristics and contains a single layer of palisade cells beneath the upper epidermis. Both the upper and lower epidermis show the presence of numerous trichomes, which are often uniseriate, unicellular, and thin-walled, emerging from the upper epidermal cells.
5.2.5. Mesophyll
The mesophyll is the middle layer of the leaf, located between the upper and lower epidermis. It contains sclerenchymatous cells that surround the vascular bundles. Collenchymatous cells are also present in the region between the vascular bundles and the lower epidermis.(67,68)
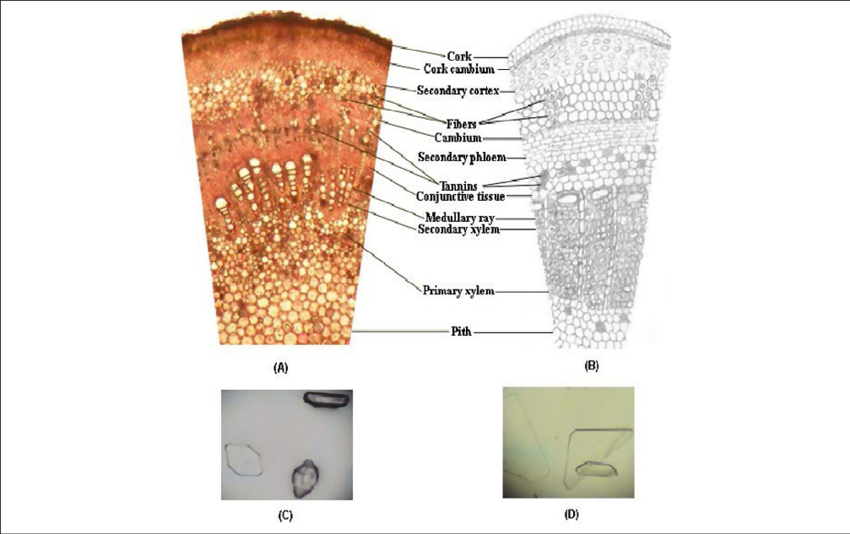
Fig. 3 - (A)- T.S. of the young stem containing the bark.12
(B)- Schematic diagram of the T.S. (C) -Rhomboidal crystals. (D)- Prismatic crystals.

Fig. 4,5: Ficus racemosa lin. (Fruits and Leaf) (76-77)
6. Physical Constants
The sample contains approximately 2% foreign matter, 14% total ash, and 1% acid-insoluble ash. The alcohol-soluble extractive is 7%, while the water-soluble extractive is 9%.15
7. Active Principles
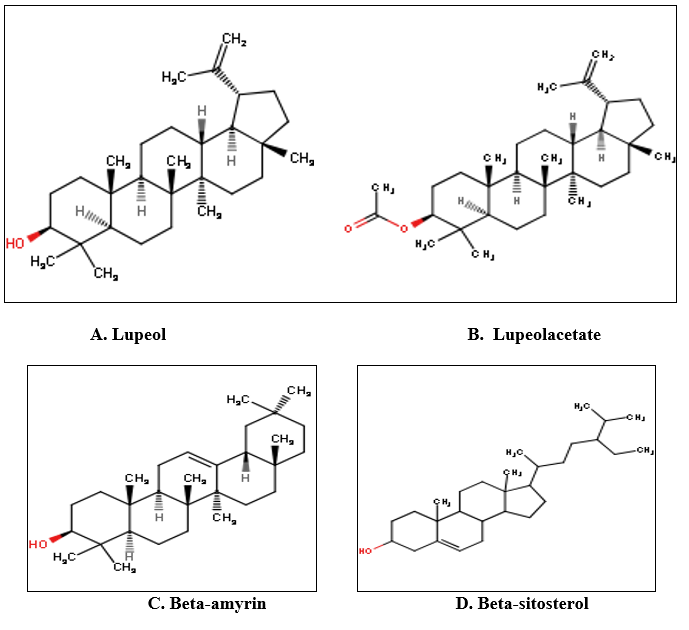
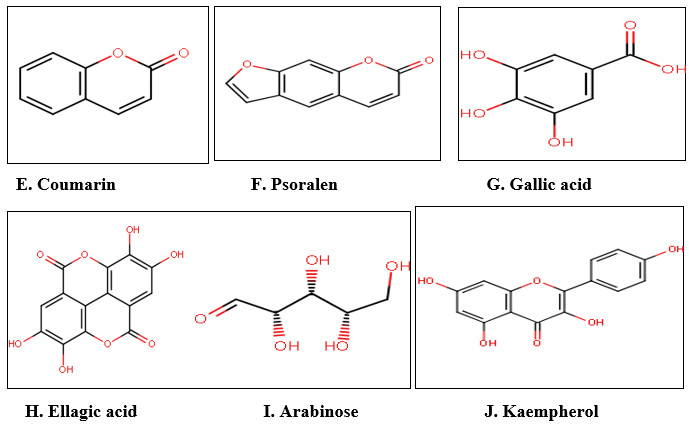
Fig. 6 : Structures of phytochemicals identified and isolated from various parts of Ficus racemosa Lin.(18)
8. TRADITIONAL USES
Ficus racemosa Linn has been widely utilized in traditional medicine to treat various health conditions. Different parts of the plant—including the bark, fruits, leaves, roots, latex, and seeds—are employed in medicinal preparations, either alone or in combination with other herbal ingredients.19
8.1. Bark
The bark is highly effective in managing threatened abortion and is also prescribed for conditions such as urinary disorders, diabetes, hiccups, leprosy, dysentery, and hemorrhoids.20,2122
8.2. Leaves
The leaves serve as an effective wash for wounds and ulcers and are beneficial in treating dysentery and diarrhea. An infusion made from the bark and leaves is used as a mouthwash for spongy gums and taken internally for dysentery and excessive menstrual bleeding. It is also considered a useful remedy for glandular swellings, abscesses, chronic wounds, cervical lymph node inflammation, and coughing up blood (hemoptysis).23-24
8.3. Fruits
The fruits possess astringent, stomachic, and cooling properties. They are beneficial in treating dry cough, hoarseness, and ailments related to the kidneys and spleen. Acting as a bowel astringent, styptic, and general tonic, the fruits are used in managing conditions such as leucorrhoea, blood disorders, burning sensations, fatigue, urinary issues, leprosy, intestinal parasites, and also serve as a carminative. Additionally, they are effective in cases of miscarriage, excessive menstrual bleeding (menorrhagia), involuntary seminal discharge (spermatorrhoea), cancer, scabies, hemoptysis, and internal obstructions.22,25,26
8.4. Roots
The roots are used in the treatment of dysentery, chest-related ailments, and diabetes. They are also applied externally for mumps, various inflammatory glandular swellings, and hydrophobia (rabies).21,22,23
8.5. Latex
The latex possesses aphrodisiac properties and is used in the treatment of haemorrhoids, diarrhoea, diabetes, boils, traumatic swellings, toothaches, and vaginal ailments.27
8.6. Root sap
The root sap is employed in the management of diabetes.28 It is also commonly used as a traditional remedy for mumps and various inflammatory swellings.29-30
9.1. Antidiurectic
The bark decoction of Ficus racemosa exhibited antidiuretic activity at doses of 250, 500, and 1000 mg/kg body weight. The effect began quickly (within an hour), reached its maximum at 3 hours, and persisted throughout the 5-hour observation period. It also led to a decrease in urinary sodium levels and the sodium-to-potassium ratio, along with an increase in urinary osmolarity, suggesting involvement of multiple mechanisms.31
9.2. Antiulcer
A 50% ethanol extract of Ficus racemosa fruits was evaluated using various gastric ulcer models in rats, including pylorus ligation, ethanol-induced, and cold restraint stress-induced ulcers. The extract was administered orally at doses of 50, 100, and 200 mg/kg body weight twice daily for five days. It produced a dose-dependent reduction in the ulcer index across all three models.32,33
9.3. Anti-inflammatory
The anti-inflammatory effect of Ficus racemosa extract was tested using rat hind paw edema models induced by carrageenan, serotonin, histamine, and dextran. At a dose of 400 mg/kg, the extract showed maximum inhibition of inflammation by 30.4%, 32.2%, 33.9%, and 32.0% in the respective models. In a chronic inflammation model, the same dose resulted in a 41.5% reduction in granuloma weight, showing effectiveness comparable to that of phenylbutazone.34
Bioassay-guided fractionation of the ethanol leaf extract led to the isolation of racemosic acid, which demonstrated strong inhibitory effects on COX-1 and 5-LOX enzymes in vitro, with IC?? values of 90 µM and 18 µM, respectively.35
The ethanol extract of the stem bark also showed COX-1 inhibitory activity, with an IC?? value of 100 ng/ml, supporting its potential use in the treatment of inflammatory conditions.36
9.4. Antibacterial
The hydroalcoholic extract of the leaves showed effectiveness against Actinomyces viscosus, with a minimum inhibitory concentration (MIC) of 0.08 mg/mL.37
9.5. Antitussive
The methanolic extract of the stem bark was evaluated for its antitussive activity using a sulphur dioxide-induced cough model in mice. At an oral dose of 200 mg/kg, the extract showed a maximum cough suppression of 56.9% after 90 minutes of administration.38
9.6. Antipyrectic
The methanolic extract of the stem bark demonstrated a significant, dose-dependent decrease in both normal body temperature and yeast-induced fever in albino rats, lasting up to 5 hours after oral administration at doses of 100, 200, and 300 mg/kg body weight. Its antipyretic activity was found to be comparable to that of paracetamol.39
9.7. Antifilarial
Both alcoholic and aqueous extracts inhibited the spontaneous motility of the whole worm and nerve-muscle preparation of Setaria cervi, marked by an increase in the amplitude and tone of contractions. Additionally, both extracts demonstrated in vitro lethality against microfilariae. The LC?? and LC?? values for the alcoholic extract were 21 ng/mL and 35 ng/mL, respectively, while for the aqueous extract, they were 27 ng/mL and 42 ng/mL.40
9.8. Analgesic
The ethanol extracts of the bark and leaves demonstrated dose-dependent analgesic effects when tested using an analgesiometer at doses of 100, 300, and 500 mg/kg, indicating significant pain-relieving properties.41
9.9. Anthelmintic
The crude bark extracts were tested for anthelmintic activity using adult earthworms and showed a dose-dependent reduction in spontaneous movement (paralysis) and response to pin-prick stimulation. The effects were comparable to those produced by 3% piperazine citrate. Notably, worms treated with the aqueous extract did not recover, indicating a wormicidal effect.42
9.10. Wound healing
The ethanol extract of the stem bark exhibited wound healing activity in both excision and incision wound models in rats.43
9.11. Hypoglycemic
The methanolic extract of the stem bark, at doses of 200 and 400 mg/kg orally, significantly reduced blood glucose levels in both normal and alloxan-induced diabetic rats. Its hypoglycemic effect was comparable to that of the standard antidiabetic drug glibenclamide (10 mg/kg), supporting its traditional use as an antidiabetic remedy.44-45 The hypoglycemic studies on Ficus racemosa in the post-absorptive state indicated that the drug exhibited enhanced hypoglycemic activity after absorption, suggesting a stronger effect following its uptake into the system.46 The ethanolic extract, administered orally at a dose of 250 mg/kg/day, significantly reduced blood glucose levels within two weeks in alloxan-induced diabetic albino rats, confirming its hypoglycemic potential.47 β-sitosterol isolated from the stem bark, demonstrated significantly stronger hypoglycemic activity compared to the other isolated compounds.47The methanolic extract of powdered fruits, administered at doses of 1, 2, 3, and 4 g/kg, effectively lowered blood glucose levels in both normal and alloxan-induced diabetic rabbits.48 The ethanolic leaf extract, given at a dose of 100 mg/kg body weight, reduced blood glucose levels by 18.4% at 5 hours and 17.0% at 24 hours in streptozotocin-induced diabetic rats challenged with sucrose, indicating its antihyperglycemic effect.49
9.12. Hypolipidemic
A significant hypocholesterolemic effect was observed when the dietary fiber from the fruits was included in the rats' diet, as it enhanced the fecal elimination of both cholesterol and bile acids.50 The hypolipidemic effects of the ethanolic bark extract were evaluated in alloxan-induced diabetic rats at doses ranging from 100 to 500 mg/kg body weight. The results revealed that the extract exhibited strong antidiabetic and hypolipidemic properties, comparable to those of the standard drug, glibenclamide.45
9.13. Radio protective/antioxidant
Ethanol and water extracts were evaluated for their free radical scavenging potential using both steady-state and time-resolved techniques. Among them, the ethanol extract showed markedly higher antioxidant activity in the steady-state method. It also demonstrated concentration-dependent scavenging of DPPH, ABTS, hydroxyl, and superoxide radicals, along with inhibition of lipid peroxidation, when compared with standard compounds.6 The in vitro radioprotective potential was evaluated using the micronucleus assay in irradiated Chinese hamster lung fibroblast cells (V79). Pre-treatment with various doses one hour before exposure to 2 Gy γ-radiation significantly reduced the percentage of micronucleated binuclear V79 cells, indicating its effectiveness as a radioprotective agent.51 The methanolic extract of the stem bark exhibited significantly stronger in vitro antioxidant activity compared to the methanolic extract of the roots.52 The ethanol extract of the fruits demonstrated notable antioxidant activity in the DPPH free radical scavenging assay. Additionally, 3-O-(E)-Caffeoyl quinate exhibited strong antioxidant potential.53
9.14. Renal anticarcinogenic
Ficus racemosa extract, administered at doses of 200 mg/kg and 400 mg/kg body weight, significantly reduced levels of xanthine oxidase, lipid peroxidation, γ-glutamyl transpeptidase, and hydrogen peroxide. It also led to a marked restoration of renal glutathione levels and antioxidant enzymes, along with a decrease in elevated renal ornithine decarboxylase activity, DNA synthesis, blood urea nitrogen, and serum creatinine. Comparable effects were observed in models using ferric nitrilotriacetate (Fe-NTA) as a renal carcinogen.54 Both sets of results confirmed that the extract possesses strong chemopreventive properties.
9.15. Memory Enhancing Activity
An aqueous extract of Ficus racemosa Linn. (Moraceae) bark was examined for its effects on memory and brain chemistry in rats. When administered at doses of 250?mg/kg and 500?mg/kg, the extract significantly elevated acetylcholine (ACh) levels in the hippocampus—by 22% and 38%, respectively—relative to control animals. In parallel, both doses significantly reduced transfer latency (time taken to move through the maze) in the elevated plus-maze test, an exteroceptive behavioral assessment of memory. These results suggest that the F.?racemosa bark extract may hold promise for Alzheimer’s disease management.55
9.16. Cardioprotective Activity
The standardized extract of Ficus racemosa Linn. stem bark demonstrated cardioprotective activity against doxorubicin-induced toxicity. It notably reduced thiobarbituric acid reactive substances (TBARS) and enhanced glutathione levels in both serum and cardiac tissues.56
9.17. Diabetic Complications
Administration of the tannin fraction from Ficus racemosa Linn. effectively reversed elevated levels of blood glucose, total cholesterol, triglycerides, and low-density lipoprotein (LDL), while significantly improving insulin and high-density lipoprotein (HDL) levels in the serum. Furthermore, the tannin fraction restored the activity of key antioxidant enzymes like superoxide dismutase and catalase, reduced levels of glutathione peroxidase and glutathione, and helped normalize the overall antioxidant status of the organs.57
9.18. Larvicidal
The larvicidal potential of hexane, ethyl acetate, petroleum ether, acetone, and methanol extracts from the leaf and bark of Ficus racemosa was assessed against early fourth-instar larvae of Culex quinquefasciatus.18 After 24 hours of treatment, all extracts showed moderate toxicity, with the bark extract in acetone causing the highest larval death.
9.19. Hepatoprotective
An ethanolic extract of Ficus racemosa leaves exhibited hepatoprotective effects in rats with chronic liver damage induced by subcutaneous injections of 50% v/v carbon tetrachloride in liquid paraffin at a dose of 3 ml/kg, administered on alternate days for four weeks. Liver function was assessed using biochemical markers such as SGOT, SGPT, serum bilirubin, and alkaline phosphatase.69 In another study, methanolic extracts of Ficus racemosa stem bark at doses of 250 and 500 mg/kg were evaluated for their protective effect against carbon tetrachloride-induced liver injury in rats, using silymarin as the reference drug. The treatment significantly restored all altered serum, liver, and kidney biochemical parameters toward normal levels compared to the carbon tetrachloride-treated group.70
9.20. Antidiarrhoeal
The ethanolic extract of Ficus racemosa stem bark was evaluated for its antidiarrheal activity in rats using various experimental models of diarrhea. The extract significantly reduced castor oil-induced diarrhea and PGE?-induced enteropooling, demonstrating its potential as an effective antidiarrheal agent.71
9.21. Antifungal
Ficus racemosa exhibited antifungal activity by suppressing the growth of six fungal species, including Trichophyton mentagrophytes, Trichophyton rubrum, Trichophyton soudanense, Candida albicans, Candida krusei, and Torulopsis glabrata.
9.22. Anti-cancer Activity
Oral administration of Ficus racemosa extract at doses of 200 and 400 mg/kg significantly suppressed lipid peroxidation, xanthine oxidase activity, glutamyl transpeptidase, topoisomerase-II, and hydrogen peroxide (H?O?) production. Additionally, it mitigated the depletion of renal glutathione and antioxidant enzymes caused by potassium bromate (KBrO?), a known nephrotoxic agent that induces kidney cancer in rats. The treatment also led to a marked recovery in renal glutathione levels and antioxidant enzyme activity. These results indicate the chemo-preventive potential of Ficus racemosa extract against KBrO?-induced nephrotoxicity. Furthermore, lupeol, a compound found in Ficus racemosa, exhibited anti-tumor promoting effects by modulating critical signaling pathways involved in cancer development, including nuclear factor kappa B (NF-κB) and phosphatidylinositol 3-kinase (PI3K)/Akt pathways.

Beta-stitosterol (steroidal strcture)

Inhibitory effect against DNA topoisomerase- II reduse lipid peroxidation, xanthine oxidase
Fig. 7 : Anticancer mechanism of extract of Ficus.(58)
10. Quality control test and pharmacokinetic study
A quality control evaluation should be conducted on various parts of Ficus racemosa Linn., including the root, stem, fruit, and bark. This assessment should include nutritional and mineral profiling, phytochemical screening, and toxic element analysis, among other relevant tests. Racemosic acid, an ethanol-extracted compound from Ficus racemosa leaves, demonstrated strong in vitro anti-inflammatory activity by effectively inhibiting COX-1 and 5-LOX enzymes, with IC?? values of 90 μM and 18 μM, respectively. The use of the plant in treating inflammatory conditions is supported by the ethanolic extract of the stem bark, which was found to inhibit COX-1 activity with an IC?? value of 100 ng/mL.(74)
REFERENCES



Harshali Thakare, Vaishnavi Patil*, Dr. Sonali Uppalwar, A Multifaceted Medicinal Tree: A Review on the Traditional and Modern Uses of Ficus Racemosa, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 10, 2205-2221 https://doi.org/10.5281/zenodo.17398105
 10.5281/zenodo.17398105
10.5281/zenodo.17398105