
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Shri Laxmanrao Mankar Institute of Pharmacy, Amgaon-441902
Flame photometer has a high resolution for the detection of alkali and alkaline earth metal ions such as sodium, potassium, lithium, and calcium. Flame photometry separates the characteristic spectra of an element while also measuring emission by atomizing a solution sample into a flame. Alkali and alkaline earth metals, such as Na, K, Ca, and Mg, play important functions in the human body. They are crucial in physiology, industry, and pharmaceuticals as well. In a wide range of applications, routine, accurate, and reliable qualitative and quantitative assessment of these metals is critical. Since the beginning of time, flame photometry has been the most widely utilised technology for this purpose. This resurrected paper discusses the numerous applications of flame photometry that have been carried out to date.
Flame photometry is an indispensable tool in the field of analytical chemistry. Flame atomic emission spectroscopy (FAES) is another name for it. “The first flame photometer was developed and became available commercially in the 1940s”.(Barnes et al.) Bunsen and Kirchhoff were the first to investigate the idea of using distinctive emission from energetic electrons of the atom in quantitative element analysis for analytical chemical research.(Banerjee and Prasad) The concentration of metal ions of group- I and group-II such as sodium, potassium, lithium, calcium, and caesium can be determined using a flame photometer .In flame photometer spectra the metal ions are used in the form of atoms. When alkali (Group I) and alkaline earth metal (Group II) compounds are exposed to flame, they disintegrate into atoms. Some of these atoms are subsequently excited, reaching even higher levels of excitation. However, at higher levels, these atoms are unstable. Hence, these atoms emit radiations when returning back to the ground state. These radiations generally lie in the visible region of the spectrum. Each of the alkali and alkaline earth metals has a specific wavelength The intensity of the emission is directly proportional to the number of atoms returning to the ground state and the light emitted is in turn proportional to the concentration of the sample.
Flame photometry is based on the measurement of intensity of the light emitted when a metal is introduced into a flame. The wavelength of colour tells us what the element is and the colour intensity tells us how much of the element present. Flame photometry is also named as flame emission spectroscopy because of the use of a flame to provide the energy of excitation to atoms introduced into the flame. Flame photometry, coupled with simple read out devices provides high sensitivity and high reliability for the determination of elements in the first two columns of the periodic table. Among the elements are sodium, potassium, lithium, calcium, magnesium, strontium and barium. The measurement of these elements is very useful in medicine, agriculture and plant science. Flame photometry is also successful in determining certain transition elements such as copper, iron and manganese. By making a wavelength scan of the emission spectrum, it is also possible to do qualitative analysis by employing flame photometer, but the application is severely limited. In addition to the determination metals, it can be applied to non-metal analysis by utilizing the infrared region of the spectrum. Flame photometry as a sample, rapid method for the routine determination of elements that can be easily excited.
Flame Photometry is a branch of spectroscopy in which the species are examined in the spectrometer are in the form of atoms. A Flame Photometer is an instrument used in inorganic chemical analysis to determine the concentration of certain metal ions (mainly are sodium, potassium, calcium, lithium).The flame photometry equipment is composed of an atomizer that disperses the sample generating a cloud that when in contact with the flame generates a signal, this signal that leaves the atomizer contains the information of the analytic and other radiations produced that pass to a monochromator . The monochromatic eliminates the other radiations produced decreasing the noise in the analysed sample, the signal that leaves the monochromator is an electromagnetic analytical signal that is translated into an electric current in a phototube, this electric current is translated into a signal processor and recorded in a meter obtaining the concentration measurement of the analyte studied, for the case of sodium these emissions are generated at a wavelength of 589 nm .
Flame photometry is based on measurement of intensity of light emitted when a metal is introduced into the flame. The wavelength of the colour tells what the metal is flame. The potential of atomic spectroscopy in both the qualitative as well as quantitative analysis were then well established.
Qualitative:
The colours intensity tells us how much element present.
Quantitative:
Flame Photometry is also named as Flame Emission Spectroscopy because of the use of a flame to provide the energy of excitation to atoms introduced into the flame
Table1: colour of flame and characteristics emission wavelength of same element.
|
Element |
Colour of Flame |
Emission of wavelength (nm) |
|
Na |
Yellow |
589 |
|
K |
Violet |
766 |
|
Ba |
Lime green |
544 |
|
CA |
Orange |
622 |
|
LI |
RED |
670 |
Sodium (Na) is the major extracellular cation and it plays a role in body fluid distribution. Concentration of sodium ions inside the plasma (extracellular) is 130-145 mmol/l. Higher and lower concentrations are referred to as hypernatremia and hypernatremia, respectively. When a solution containing cations of sodium and potassium is spayed into flame, the solvent evaporates and ions are converted into atomic state. In the heat of the flame (temperature about 1800ºC), small fraction of the atoms is excited. Relaxation of the excited atoms to the lower energy level is accompanied by emission of light (photons).
Potassium (K) is the major cation found inside of cells. The proper level of potassium is essential for normal cell function. An abnormal increase of potassium (hyperkalaemia) decrease of potassium (hypokalaemia) can profoundly affect the nervous system and heart, an when extreme can profoundly affect the nervous system and heart, and when extreme, can be fatal. The normal blood potassium level is 3.5 - 5.0 mill moles/litter (m moll/l).
PRINCIPLE:
Flame photometry is a type of atomic spectroscopy where atomic emission is measured using a spectrophotometer when a metallic space is introduced in the flame. The metal salt is burnt emitting certain a colour wavelength and this instrument is based on measurement of intensity of colour generated by different element each metal give characteristics colour and the intensity of colour depict the amount of quantity of metal present we identify the metal present in the unknown sample with respect to the colour developed . It was first developed Nelson A in 1955 and he receive a patent for the flame photometry in 1958. Liquid sample containing salt solution is introduced into the flame Solvent is first vaporized, leaving the particles of solid salt which are then vaporised into gaseous state the gaseous molecules are dissociated to give free neutral atoms or radicals which can be excited by thermal energy of flame . The unstable excited atoms emit photons while returning to lower energy state. The measurement of emitted photons-radiation forms the basis of flame.
If E2 andE1 represent the energy of the higher and lower energy levels concerned, the radiation emitted during the jump may be defined by equation.
E2 - E1 = hv
Where h is the Planck’s constant, and v the frequency of emitted light which is defined as follows:
v = c/λ
On combining equations, we get
E2 - E1 = hc / λ
[or] λ = hc /E2 – E1
From equation can calculate the wavelength of the emitted radiation which is characteristic of the atoms of the wavelength of the emitted radiation which is characteristic of the atoms of the particular element from which it was emitted.
The intensity of radiation emitted depends upon the of thermally excited atom which depend upon the temperature of flame
Fraction of free atom of thermally excited
=n /No = Ae –E/ kt
Were,
The temperature of fuel is controlled by the type of fuel and oxidant used. Some flame temperatures are given in Table.
|
Fuel |
Oxidant |
Flame temperature |
|
propane |
Air |
1900 |
|
propane |
Oxygen |
2800 |
|
Water |
Air |
2100 |
|
Water |
Air |
2800 |
The energy available in the flame for exciting atoms is dependent on the flame temperature as defined by the Boltzmann distribution. Flames have only limited amounts of energy available when compared to excitation sources such as emission spectrographs or plasma emission torches (ICP). For this reason, flames are most useful only for elements that require low amounts of energy to become excited. This means that the method is most useful for the determination of the alkali metals and alkaline earth metals. They are not the method of choice for transition metals and most of the other metals in the periodic table. These metals itch generally require significantly more energy in order to become excited. They are most often done by emission spectrographs or plasma emission where much higher energy is available. However, it must also be stated that when we try to analysis group I and group II elements in these high-energy sources, we nun the risk not only of exciting them but of ionizing them. Although the Boltzmann distribution would lead us to believe that the emission intensity would be greatly increased in plasma emission for the group I and group II elements, in practice it is found not to be so because the atoms are ionized and this results in a loss of atoms and the generation of ions. Ionization causes a complete change of energy levels and therefore the entire emission spectrum, with a reduction in the intensity of the atomic spectra.
THEORY
flame photometry, a branch of atomic spectroscopy is used for inorganic chemical analysis for determining the concentration of certain metal ions such as sodium, potassium, lithium, calcium, Caesium, etc. In flame photometry the species (metal ions) used in the spectrum are in the form of atoms. The International Union of Pure and Applied Chemistry (IUPAC) Committee on Spectroscopic Nomenclature has recommended it as flame atomic emission spectrometry (FAES). The basis of flame photometric working is that, the species of alkali metals (Group 1) and alkaline earth metals (Group II) metals are dissociated due to the thermal energy provided by the flame source. Due to this thermal excitation, some of the atoms are excited to a higher energy level where they are not stable. The absorbance of light due to the electrons excitation can be measured by using the direct absorption techniques. The subsequent loss of energy will result in the movement of excited atoms to the low energy ground state with emission of some radiations, which can be visualized in the visible region of the spectrum.
INSTRUMENTATION OF FLAME PHOTOMETRY
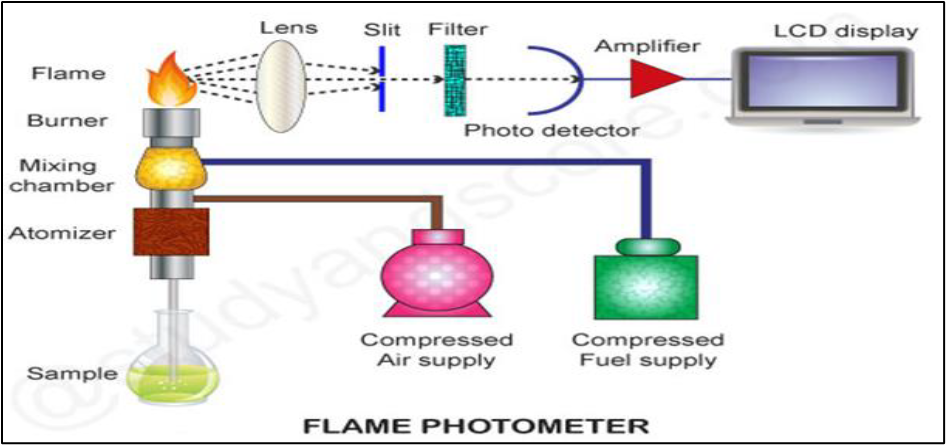
There are Three components for introducing the liquid sample.
Nebulizer: it breaks up the liquid into small droplets. Nebulization is a conversion of sample to a mist of finely divided droplets using a jet of compressed gas. a flaw carries the sample into atomization region.
Nebuliser is a device used for sample introduction into the flame. The process is called nebulisation and consists of thermal vaporization and dissociation of aerosol particles at high temperatures producing small particle size with high residence time.
A number of nebulisation methods are available. A few are listed below.
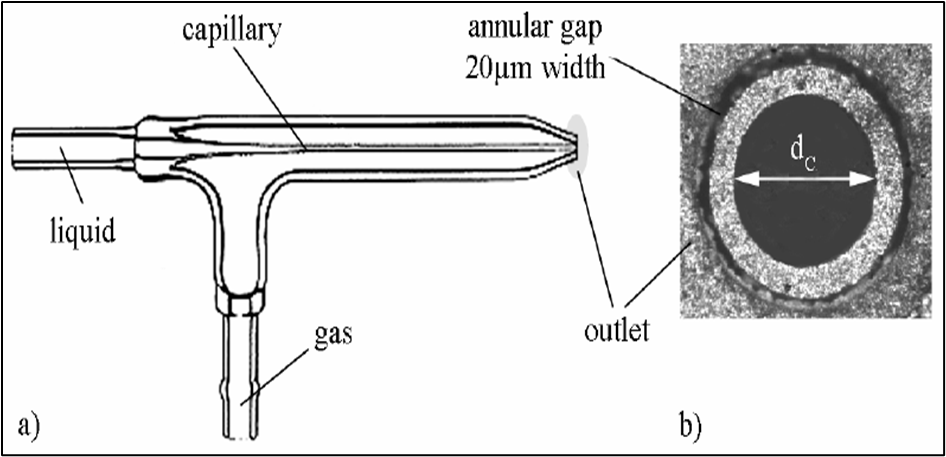
However, we would discuss about the pneumatic nebulisation only. It is the most commonly employed nebulisation method in flame photometers. Pneumatic nebuliser is the most commonly used nebuliser for introducing aqueous/ liquid samples. In this the sample solution is fed or aspirated into the nebuliser which converts liquid into a fine mist, or aerosol which is then fed into the flame. A common type of pneumatic nebuliser is called concentric pneumatic nebuliser, as shown in Fig. 7.9. The concentric pneumatic nebuliser consists of a fine capillary surrounded by concentric tube with a small orifice near one end of the capillary. The capillary is dipped into a solution of the analyte while the outer tube is connected to a high pressure gas supply. The analyte is sucked into the capillary by the high pressure gas stream flowing around the tip of the capillary using the Bernoulli effect. The process is called aspiration. The high velocity gas breaks up the liquid into various sized fine droplets. The other types of the pneumatic nebulisers also work on the same principle.
(b) Aerosol modifier: it removes large droplets from the stream and allow only smaller droplets.
(c) Flame or Atomizer: it converts the analyte into free atoms.
The flame used in the flame photometer must possess the following functions: The flame should possess the ability to evaporate the liquid droplets from the sample solution resulting in the formation of solid residue. The flame should decompose the compounds in the solid residue formed in step resulting the formation of atoms. The flame must have the capability to excite the atoms formed in step and cause them radiant energy. For analytical purposes, it becomes essential that emission intensity should be over reasonable periods of time (1-2 min). The temperature of the flame, which is primarily responsible for the occurrence of the above mention processes is controlled by several factors which are summarised as follows: Type of fuel and oxidant and fuel-to-oxidant ratio Type of solvent for preparing the sample solution Amount of solvent which is entering into the flame Type of burner employed in flame photometer The particular region in flame which is to be focussed into the entrance slit of the spectral isolation unit.
Structure of flame: Flames are not uniform in composition, length or cross section. The structure of a premixed flame, supported as a laminar flow is shown in Figure:
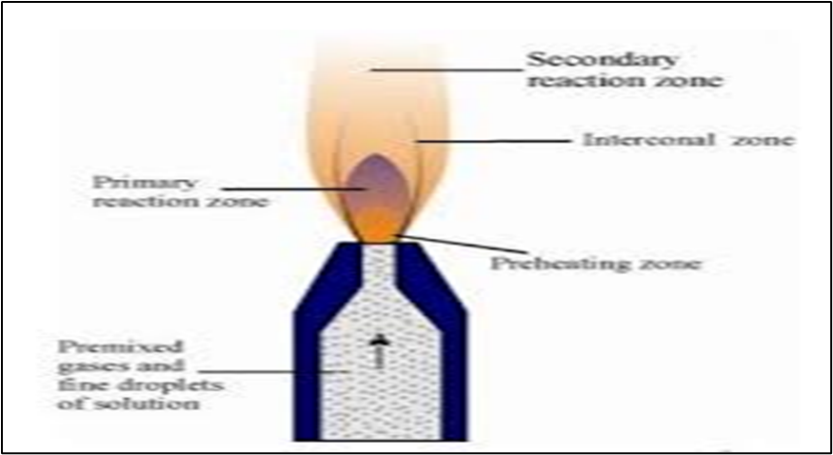
Schematic structure of a laminar flow flame showing various zones
As seen in the figure, the flame may be divided into the following regions or zones.
Types of Burners
A. Meeker burner
This was the primitive type of burner used in flame photometry and was used earlier. It generally works with aid of natural gas and oxygen as fuel and oxidant. The temperature so produced in the flame was relatively low resulting in low excitation energy. Now a days it is not used but it was best suited for alkali metal.

B. Total consumption burner
Due to the high pressure of fuel and oxidant the sample solution is aspirated through capillary. As soon as liquid sample drawn into the base of flame oxygen aspirates sample solution leaving solid residue. Atomization and excitation then occurs. Hydrogen and oxygen are generally employed as fuel and oxidant.
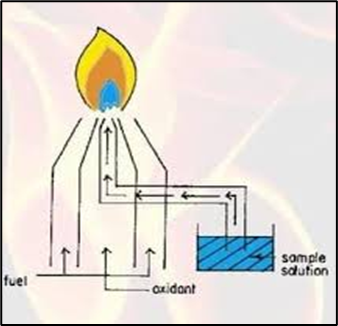
C. Premix burner
In this type of burner the sample and fuel oxidant and thoroughly mixed aspiration and reaching to the flame the advantage type of burner is uniformity flame produced however it has a disadvantage the hevy loss of mixture up to 90% occur during the process.
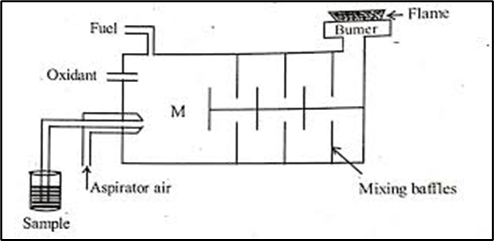
D. Lundengraps burner
In this particular burner the sample are aid of mixed in a chamber and a mixed composition is sent to be fuel nozzle where it is atomized here sample reaches to the flame is only about 5% of the total content.
E. Shielded burner
In this burner shielded from the ambient atmosphere by a system of inert gas celling is required to better analytical sensitivity during the measurement process the following result obtained with sale did.
F. nitrous oxide burner.
These flame are superior to other flames for effectively producing free atom metal with very reflective oxides such as aluminium are titanium are analysed by this burner it has drawback that high temperature reduces its use fullness for the determination of alkali metal as the easily ionized. Also it is produced intense background emission which make measurement of metal emission difficult .the flame is main source which is the responsible for creates the process of atomization and subsequent measurement.
The radiation from the flame is emitted in all direction in space. In order to maximize the amount of radiation used in the analysis, a mirror is located behind the burner to reflect the radiation back to the entrance slit of the monochromator. This mirror is concave and covers as wide angle from the flame as possible.
With the best equipment, entrance and exit slits are used before and after the dispersion elements. The entrance slit cuts out most of the radiation from the surroundings and allows only the radiation from the flame and the mirrored reflection of the flame to enter the optical system. The exit slit is placed after the monochromator and allows only a selected wavelength range to pass through to the detector. For many purposes it is essential that this wavelength range be narrow that is of the order of few Nanometres. This is necessary if emission lines from other components in the flame have wavelength similar to those of the emission lines of the elements being determined. The slit must prevent such and interfering lines from reaching the detector.
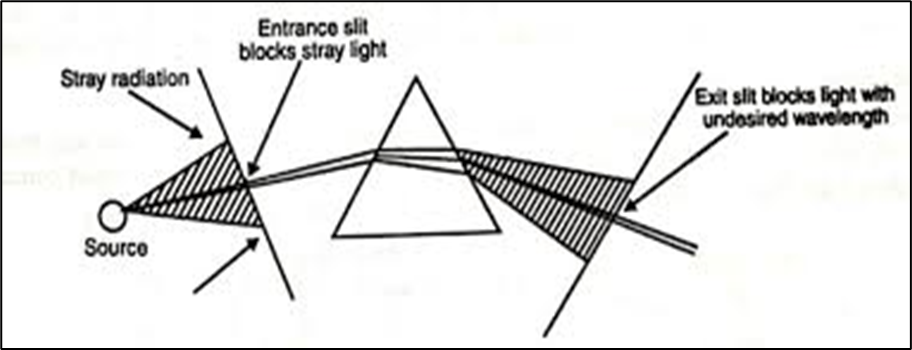
In flame photometry wavelength and intensity of the readfition emitted by the element are monitored hence a filter monochromator is to be used instrument. A simple flame photometer contain, a filter when containing al for contning like sodium, potassium, calcium, lithium, and when a particular element has to be analysed a specific choosen .In simple flame photometer, the monochromator is the prism but in expensive models the grafting monochromator are used. Quartz is the materials most commonly used for making prisms even though its dispersive power is less than that of glass. The reasons for this is that quartz is transparent over the region. The grafting monochromator employs a grafting which is essentially a series of parallel straight lines into a plane.
In some elements, the emission spectrum contains a few lines. In such cases wide wavelength ranges will be allowed to enter the detector without causing any serious error. In such a situation an optical filter may be used in place of the slit and monochromator system. The filter is made from a material which is transparent over a narrow spectral range. When a filter is kept between the radiation of the desired wavelength from the flame will be entering the detector and measured. The remaining undesired wavelength will be absorbed by the filter and not measured. The flame photometers which use filter monochromators are very convenient for simple repetitive analysis. However, such instruments can be used for a small number of elements. The reason for ha is that a large number of filters are employed. The radiation coming from the optical system is allowed to fall on the detector where radiation of the appr measures the intensity of radiation falling on it. The detector should be sensitive to radiation all wavelengths that may be examined. In good flame photometers, the photomultiplier detectors are employed which produce an electrical signal from the radiation falling on them.
The radiation coming from the optical system is allowed to fall on the detector which measure the intensity of radiation falling on it. The detector should be sensitive to radiation of all wavelengths that may be examined. In good flame photometer, the photomultiplier detectors are employed which produce an electrical signal from the radiation falling on them.
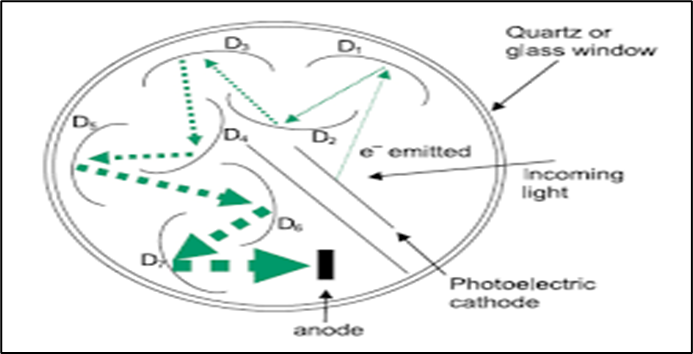
Fig. photomultiplier tube
Photomultiplier tube (PMT) detector is most sensitive of all detector available and expensive. The principle employed in the detector or multiplication photoelectron by second any emission of electron. This multiplication is achevied by using photocathode and series of anode called by dynodes. The photomultiplier tube can use upto 10 dynodes these dynodes are maintained at 70 -100 v higher than the preceding dynodes. After each stage electron emission is multiplied by factor of 4-5.the photomultiplier tube can detect very weak signals.
The photovoltaic cell as thin metallic a layer coated with a silver or gold which app says an electrode also has a metal base plate which is acts as another electrode. The two layer are separated by semiconductor layer of selenium. When light radiation fall in selenium layer is crates potential difference between two electrode and causes flow of current. The flow of current causes deflection of galvanometer needle which depend on the wavelength of intensity of radiation. It has disadvantage that the amplification of the signal is not possible because the resistance of the flow external circuit has to be low fatigue affects of lesser response of the detector with light other than blue and red light. It is very inexpensive instrument.
Read out device- the from the signal from the detector is shown as a response to the digital reader device the digital reading are displaced as an arbitrary scale.
INTERFERENCES
The Plame photometry has several (imitations choice Heeds critical attention. The temperature of measurement is hot high enough to excite transition metals. Hence, it is only Selective toxoids detecting cocaine earth metals The relatively low energy available in the flame leads to relatively 1ow intensity of the radiation arising From the metal atoms: 37 Low temperature renders to cause interference and the stability of the Flame and aspiration conditions and affected. Higher chances of unavoidable interference by other elements. out of the several limitations 0l6 Flame photometry, the venous types of interferences needs sufficient knowledge in order to minimise their incident and to know the proper Couse behind the interference The interferences can be of three types. They are 1 spectral interference.
Other spectral line Interference example:
a) Na and K mixtures interfere with each other.
Remedy:
b) Aluminium interferes with emission line of Ca and Mg
c) Orange band of Ca – 543 – 622 nm interference with Na doublet 589 and 589.6nm and Ba line at 553.6nm
4. Ionization Interference: In some cases, some of the metal atoms may ionize in high temperature flame. e.g., Sodium ion possesses an emission spectrum of its own, with different frequencies from those of the atomic 14 spectrum of sodium. This can be overcome by adding a large quantity of potassium salt to all the solution-unknown and standard. The addition of K prevents the ionization of Na.
5. Cation-Cation Interference: This decreases the signal intensity of the element present in the sample. Ex= Al interference with Ca and Mg. Also Na and K have interference on one another.
6. Cation-Anion Interference: Anion such as oxalate, phosphate, sulphate, may affect the intensity of radiation emitted by element. For example, Ca in the presence of phosphate ion apparently forms a stable substance, so the calcium signal is depressed because it will not decompose easily.
7. Oxide Formation Interference: It arises due to the formation of stable oxide with free metal atoms if oxygen is present in the flame. This can be overcome by either using very high temp flames for dissociation or using oxygen free environ.
8. Electro- thermal vaporizes An elect thermal vaporizers contains an evaporator present in closed chamber through which an intent gas carries the vaporise Sample into the atomiser.
ADVANTAGE:
DISADVANTAGE:
APPLICATION:
Used for the determination of alkali and the alkaline earth metals in samples which are easily prepared as aqueous solution.
Example – sodium produces yellow flame.
Non radiating elements such as carbon, hydrogen and halides cannot be detected.
The concentration of various alkali and alkaline earth metal is important in determining the suitability of the soli for cultivation.
Used for determination of the concentration of sodium and potassium ions in body fluids since their controls the action of muscles including the heart.
Analysis of water from various sources is carried out to determine its suitability for drinking, washing, agriculture and industrial proposes.
Used for determination of lead in petrol.
Analysis of soft drinks fruit juices and alcoholic beverages can also be analysed by using flame photometry.
CONCLUSION:
The Flame photometry coupled simple read out device provides high sensitivity and high reliability for the determination of element in the first two columns of the periodic table.
REFERENCES



Khushal Tirele, Rani Bhagat, Tulsidas Nimbekar, Application of Flame Photometry in the Detection of Main Group Metals (Na, K, Li & Ca) in Pharmaceutical Industry, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 12, 2667-2680. https://doi.org/10.5281/zenodo.17959128
 10.5281/zenodo.17959128
10.5281/zenodo.17959128