Methylprednisolone is a medication for the management and treatment of allergic conditions, arthritis, asthma exacerbations, long term asthma maintenance and as an anti-inflammatory and immunosuppressive agent. A systematic DoE approach was utilized for the development and validation of a Stability indicating GREEN-HPTLC method for estimation of Methylprednisolone in bulk and formulation. The Box-Behnken design was utilized for the optimization of chromatographic conditions in HPTLC. ANOVA results and Contour plot indicates that Glacial acetic acid and Cyclohexane had more impact than Ethyl acetate on retardation factor. The method utilized aluminium HPTLC plate precoated with silica gel G60 F254 as a stationary phase along with Ethyl acetate: Cyclohexane: Glacial acetic acid (7.9: 1.9: 0.2 v/v/v) as a mobile phase. The retardation factor was found to be 0.50. The densitometric analysis was carried out on the absorbance mode 243 nm. The linearity was found to be in the range of 200-1200 ng/band with a correlation coefficient 0.995. The detection and quantification limit were found to be 1.794 and 5.436 ng/band. No interference was observed from excipients and mobile phase solvents in the formulation, indicating specificity of the method. The proposed method was validated as per ICH Q2 (R1) guidelines. Forced degradation study was carried out using acidic, basic, oxidative, thermal and photolytic condition. HPTLC method was found to be simple, accurate, and precise according to acceptance criteria given in ICH guidelines. The results indicate that the DoE approach is effectively applied to optimize the retardation factor, and the method is appropriate for routine laboratory analysis.
Methylprednisolone, GREEN-HPTLC, DoE, Box-Behnken design Stability indicating, ICH guidelines
1.1 Introduction to Analytical Chemistry
The branch of chemistry known as analytical chemistry is said to be responsible for describing the composition of matter, both qualitatively (what's there) and quantitatively (how much is present) [1].
1.2 Introduction to Analytical Method Development
Analytical method development and validation plays an important role in the discovery, development, and manufacture of pharmaceuticals. Method development is defined as development, revision of the validated, standardized, and official method of analysis [2].
1.3 Introduction to Chromatography
Chromatography is a method used for the separation of the components of a sample, in which the components are distributed between two phases, one of which is stationary phase while the other is mobile phase. The stationary phase may be a solid or a liquid supported on a solid or a gel, and may be packed in column, spread as a layer or distributed as a film. The mobile phase may be gaseous or liquid. High Performance Thin Layer Chromatography: As HPTLC uses smaller particles and has a 100 µm thicker sorbent layer than TLC, its efficiency is much higher. It is a separation technique that breaks down the mixture into its constituent parts with the assistance of the stationary phase and mobile phase. HPTLC can be used to identify chemicals, determine their purity, and identify the components in a mixture [3-9].
1.4 Introduction to Analytical Method Validation
As per the ICH Q2 (R1)20 guidelines, the objective of validation of an analytical procedure is to demonstrate that it is suitable for its intended purpose. Typical validation characteristics which should be considered are - Specificity, Accuracy, Precision (Repeatability, Intermediate Precision, Reproducibility), Linearity, Range, Limit of Detection, Limit of Quantitation and Robustness [10-13].
1.5 Design of Experiment (DOE)
Design of Experiment (DoE) is part of AQbD used for optimization of the method. By using DoE different factors during method development & their interactions are assessed with maximum efficiency. The effects of these factors are mostly measured at varying levels & results can be applied to a broad range of conditions [14-16].
1.6 Introduction to Stability Study
The term "Drug Stability" refers to "the extent to which a drug substance or product retains, within specified limits throughout its period of storage and use, the same properties and characteristics that is possessed at the time of its manufacture [7, 8]. Performed stressed degradation studies through the effects of acid, base, H2O2, light and thermal conditions [9-12].
1.7 Introduction to Green TLC/HPTLC Method
Nowadays pharmaceutical analysis of medicines presupposes the use of chemical substances, solvents and reagents, which, of course, have a negative impact on both the laboratory personnel and the environment. An important issue of choosing a solvent is the assessment of its environmental friendliness. Water, Ethyl acetate, Ethanol, n-butanol, n-butyl acetate, Ethylene Glycol etc., are the green solvents that can be used as a solvent in TLC.
1.8 Drug Profile
Methylprednisolone belongs to the category of corticosteroids, used to reduce inflammation in many conditions. These include endocrine diseases, severe allergies, ulcerative colitis, and multiple sclerosis. It’s also used to treat rheumatic diseases, such as rheumatoid arthritis and osteoarthritis. Methylprednisolone belongs to a class of drugs called glucocorticoids. Methylprednisolone works by decreasing inflammation and changing your body’s immune response [17-21].
- MATERIALS AND METHODS
- Materials
Methylprednisolone was obtained from reputed pharmaceutical industry, Gujarat, India. The AR grade of solvents like Methanol, Ethyl acetate, Glacial acetic acid and cyclohexane obtained from LOBA chemicals, Mumbai, Maharashtra. Medrol tablet was purchased from local market and each tablet of Medrol contains 4 mg of Methylprednisolone.
-
- Instruments
The samples were applied in the form of a band of width 6 mm with a Hamilton 100 μL sample syringe using Camag Linomat 5 (CAMAG, Switzerland) sample applicator on precoated silica gel aluminum plate 60 F254 (10 cm x 10 cm with 0.2 mm thickness, Merck, Germany). Camag TLC scanner was used for the densitometric scanning of the developed chromatogram. HPTLC system was controlled using Camag winCATS software, Version 1.4.8.
-
- Preparation of standard stock solution
10 mg of Methylprednisolone was accurately weighed and was transformed into a 10 ml clean, dried volumetric flask, the volume was made up to the mark with methanol to obtain a concentration of 1000 μg/ml Methylprednisolone. 1ml of Methylprednisolone standard stock solution (1000 µg/ml) was transferred to 10ml volumetric flask and the volume was made up to the mark with methanol to obtain concentration of 100 µg/ml Methylprednisolone.
-
- Sample application
Application of standard and sample solution of Methylprednisolone was done on TLC plate in form of narrow bands of 6 mm length, 15 mm of bottom and left edge, with 10 mm distance between two bands. Along with this, sample continues observed dry stream of nitrogen gas.
-
- Mobile phase development
Plates were developed using a mobile phase contain ethyl acetate: cyclohexane: glacial acetic acid (8:2:0.2 v/v/v). Linear ascending preparation was carried out in a twin-trough glass chamber saturated with the mobile phase vapors for 20 min at 25 ± 2ºC. 10 mL of the mobile phase (5 mL in the trough containing the plate and 5 mL in the other trough) was used for each development and was allowed to migrate at distance of 70 mm. After development, the HPTLC plates were dried with hair drier.
-
- Densitometric analysis
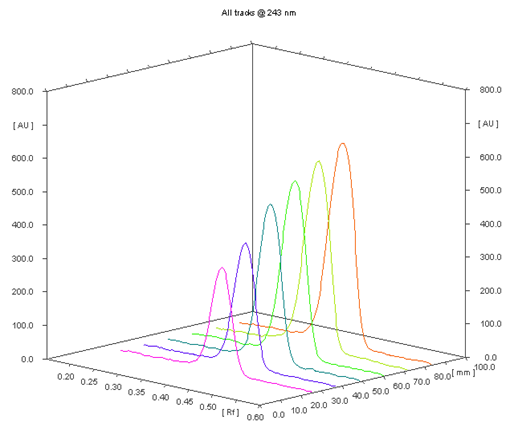
Densitometry scanning was performed in the absorbance mode under control by winCATS planar chromatography 1.4.8 software. Deuterium lamp was source of radiation, and bands were scanned at 243 nm. The slit dimensions were 4 mm length and 0.30 mm width, with a scanning rate of 20 mm/s. Concentrations of the compound chromatographed were determined from the intensity of diffusely reflected light and evaluated as peak areas against concentrations using a linear regression equation (Figure 1).
-
- Validation of developed HPTLC method
Validation of the developed HPTLC method was carried out according to International Council for Harmonization (ICH) of Technical Requirements for Pharmaceuticals for Human Use guidelines Q2 (R1) for the specificity, sensitivity, accuracy, precision, repeatability and robustness.
-
-
- Specificity
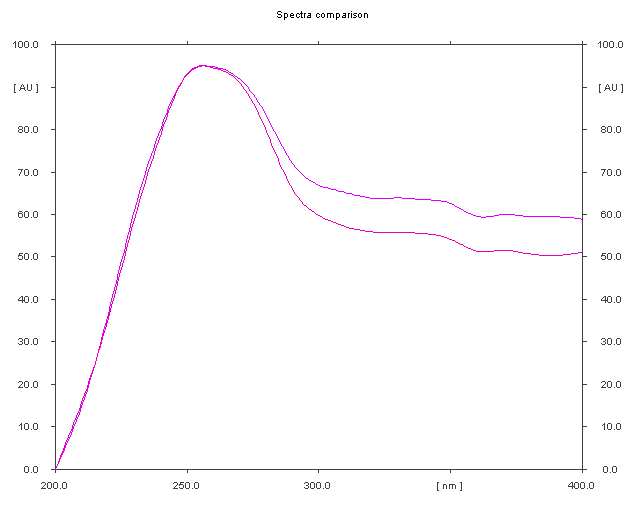
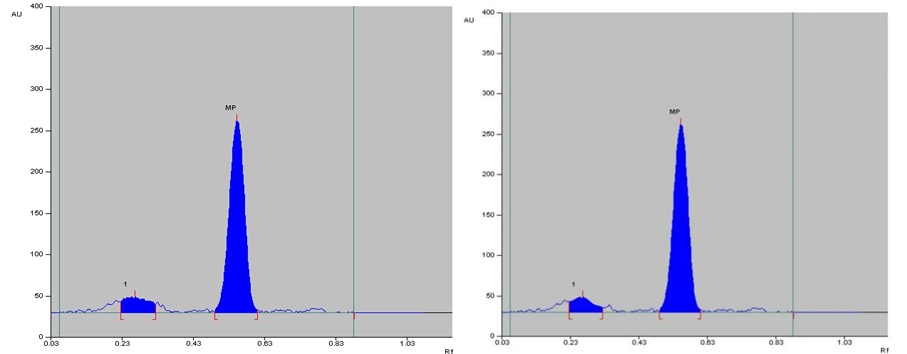
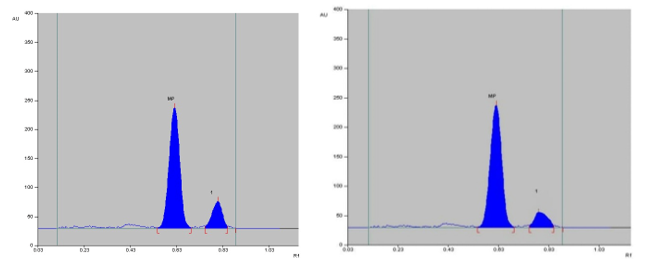
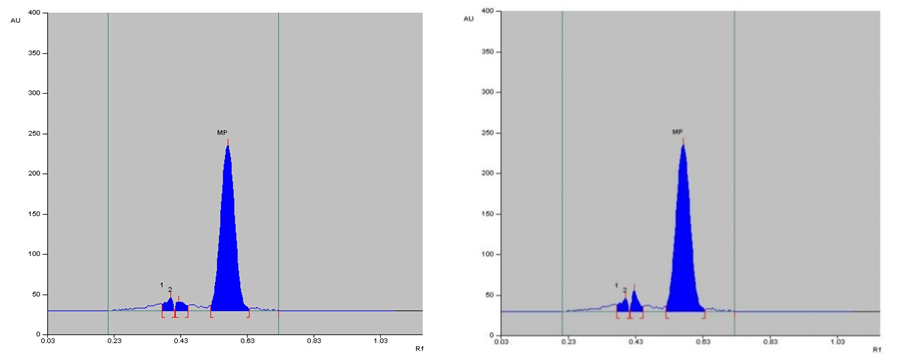
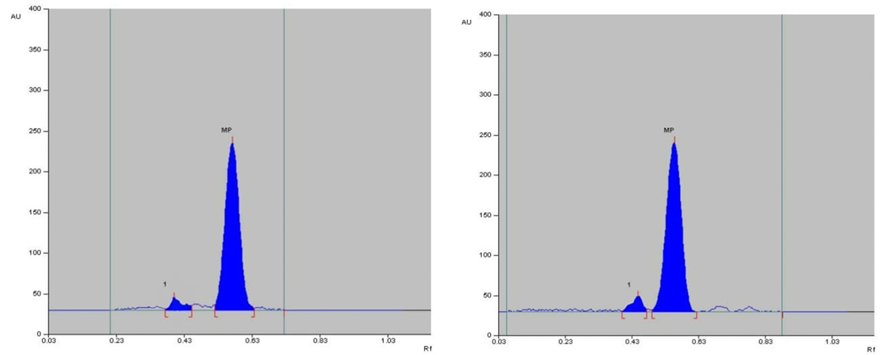
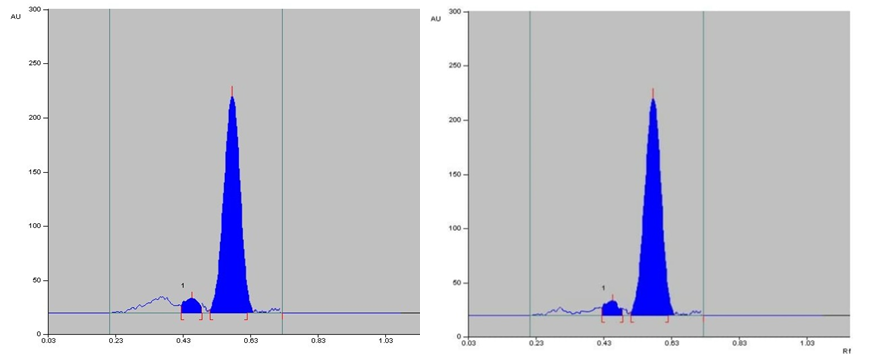
The specificity of the method was established by analyzing Perindopril erbumine, Indapamide and Amlodipine besylate in presence of excipients in marketed tablet formulation. The band of Methylprednisolone were confirmed by comparing Rf values, area and respective spectra of sample with those of standards.
-
-
- Linearity and range
Linearity of the method was evaluated by constructing curves at seven concentration levels over a range of 200– 1200 ng/band for Methylprednisolone from stock solution have been applied on pre coated TLC plate using sample applicator. The calibration curves were developed by plotting peak area versus concentration (n=6) with the help of the winCATS 1.4.8 software.
-
-
- Limit of detection and Limit of quantification
The sensitivity of the method was determined with respect to LOD and LOQ. These two parameters were determined from linearity study by a linear regression model calculated from the slope (a) and the intercept (b) SD of calibration curves. LOD was calculated as 3 times the noise level and LOQ was calculated 10 times the noise level
-
-
- Precision
Precision was evaluated in terms of intraday and interday precision. Working standard solution of 400 ng/band of Methylprednisolone were prepared and used for the precision study. Intraday precision was determined by analyzing solution of Methylprednisolone at three levels converting lower, medium and higher proposing of the calibration curve three times on the same day. Interday precision was determined by analyzing sample solution of Methylprednisolone at three levels covering lower, medium and higher concentration period of three days. The peak area obtain were used to calculate mean and %RSD value
-
-
- Accuracy
The accuracy of the method was determined by calculating recoveries of Methylprednisolone by method of standard addition at 80, 100 and 120% levels. Known amount of Methylprednisolone (0, 600, 800, 1000 ng/band) were taken from working standard of 400 ng/band. It was added to pre quantified sample, and the amount of Methylprednisolone were estimated by measuring the peak area and by fitting this value to the straight line equation of calibration curve.
-
-
- Robustness
Small deliberate changes in the chamber saturation time, change in temperatures, change in wavelength and mobile phase composition were introduced and the effects on the results were examined. Robustness of the method was determined in triplicate at a concentration level of 400 ng/band for Methylprednisolone. The mean and %RSD of peak area were calculated.
-
- Analysis of marketed formulation
Twenty tablets were weighed and finely powdered. The powder equivalent to 10 mg of methylprednisolone was accurately weighed and transferred into a 10 mL volumetric flask. 5 mL of methanol added to the above flask and sonicated for 5 min. The volume was adjusted to the mark with the methanol. An aliquot of 1 mL was transferred into a 10 mL volumetric flask and the volume was made up to the mark with methanol to obtain 100μg/mL of Methylprednisolone. 6μL of solutions were applied to precoated TLC plates with the help of Hamilton syringe under a continuous stream of nitrogen gas to obtain a spot containing 600 ng/band for Methylprednisolone and analyzed it by the proposed method. The amount of Methylprednisolone computed using regression equation.
-
- Stability of API Methylprednisolone
- Acid degradation
Accurately weighed 5mg of Methylprednisolone was transferred in 10ml volumetric flask and dissolved in methanol to obtain 500µg/ml. 2ml aliquot from this solution was taken in 10ml volumetric flask and mixed with 2ml of 0.01N HCl and refluxed for 5 hours at 80ºC. The flask solution was then neutralized with 0.01 N NaOH and then made up to the mark with methanol. A chromatogram was obtained using 6µl of the resultant solution (600ng/band) and recorded at 243 nm. The quantity of drug affected by acid was calculated.
-
-
- Base degradation
Accurately weighed 5mg of Methylprednisolone was transferred in 10ml volumetric flask and dissolved in methanol to obtain 500µg/ml. 2ml aliquot from this solution was taken in 10ml volumetric flask and mixed with 2ml of 0.01N NaOH and refluxed for 2 hours at 80ºC. The flask solution was then neutralized with 0.01 N HCl and then made up to the mark with methanol. A chromatogram was obtained using 6µl of the resultant solution (600ng/band) and recorded at 243 nm. The quantity of drug affected by base was calculated.
-
-
- Oxidative degradation
Accurately weighed 10mg of drug was transferred into 10ml volumetric flask and dissolved in methanol to obtain 1000µg/ml, from this 2ml of aliquot was taken in 10ml volumetric flask and 2ml of 3% H2O2 solution was added and allowed to set for 6hrs at room temperature. After the completion of 6hrs, the flask was made up to the mark and the chromatogram was obtained by spotting 6µl solution from the flask at 243 nm. The quantity of drug affected by the peroxide was calculated.
-
-
- Photolytic degradation
Accurately weighed 10mg API was allowed to degrade under UV light at short wavelength for 24 hours. After completion of 24 hours, 1mg of drug was dissolved using methanol in 10ml volumetric flask (100µg/ml). From this solution chromatogram was obtained by spotting 6µl at 253 nm. The quantity of drug affected by UV light was calculated.
-
-
- Thermal degradation
Accurately weighed 10mg API was allowed to degrade in hot air oven at 80oC for 24 hours. After completion of 24 hours, 1mg of drug was dissolved using methanol in 10ml volumetric flask (100µg/ml). From this solution chromatogram was taken by spotting 6µl at 243 nm. The quantity of drug affected by temperature was calculated.
-
- Stability of Formulation
- Acid degradation
The powdered tablet was equivalently weighed 5mg and transferred in 10ml volumetric flask and dissolved in methanol to obtain 500µg/ml. The solution then sonicated for 15 minutes and filtered using Whatmann filter paper (0.45µm). 2ml aliquot from this solution was taken in 10ml volumetric flasks and mixed with 2ml of 0.01N HCl and refluxed for 5 hours at 80oC. The flask solution was then neutralized with 0.01N NaOH and made up to the mark. A chromatogram was obtained using 6 µl of the resultant solution (600ng/band) and recorded at 243 nm. The quantity of drug affected by acid was calculated.
-
-
- Base degradation
The powdered tablet was equivalently weighed 5mg and transferred in 10ml volumetric flask and dissolved in methanol to obtain 500µg/ml. The solution then sonicated for 15 minutes and filtered using Whatmann filter paper (0.45µm). 2ml aliquot from this solution was taken in 10ml volumetric flask and mixed with 2ml of 0.01N NaOH and refluxed for 2 hours at 80o C. The flask solution was then neutralized with 0.01N HCl and made up to the mark with methanol. A chromatogram was created using 6 µl of the resultant solution (600ng/band) and recorded at 243 nm. The quantity of drug affected by base was calculated.
-
-
- Oxidative degradation
The powdered tablet was equivalently weighed 5mg and transferred in 10ml volumetric flask and dissolved in methanol to obtain 500µg/ml. The solution then sonicated for 15 mins and filtered using Whatmann filter paper (0.45µm). 2ml aliquot from this solution was taken in 10ml volumetric flasks and 2ml of 3% H2O2 solution was added in the flask and allowed to set for 6hrs at room temperature. After the completion of 6hrs, the flask was made up to the mark with methanol and chromatogram was obtained by spotting 6µl solution from each flask at 243 nm. The quantity of drug affected by the peroxide was calculated.
-
-
- Photolytic degradation
The powdered tablet was equivalently weighed 5mg and allowed to degrade under UV light at short wavelength for 24 hours. After completion of 24 hours, 1mg of drug was dissolved in methanol in 10ml volumetric flask and made up to the mark (100µg/ml). From this solution chromatogram was taken by spotting 6µl at 243 nm. The quantity of drug affected by UV light was calculated.
-
-
- Thermal degradation
The powdered tablet was weighed equivalent to 5mg and allowed to degrade in hot air oven at 80oC for 24 hours. After completion of 24 hours, 1mg of drug was dissolved in methanol in 10ml volumetric flask and made up to the mark (100µg/ml). From this solution chromatogram was taken by spotting 6µl at 243 nm. The quantity of drug affected by temperature was calculated.
RESULTS AND DISCUSSION
-
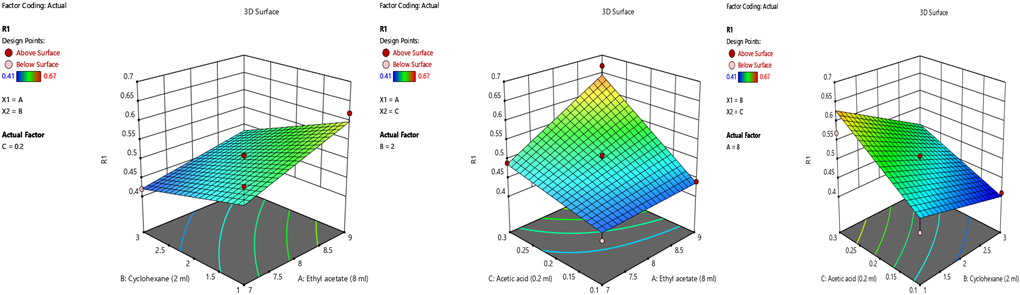
- Optimization of mobile phase using DoE approach
Selection of the mobile phase was carried out on the basis of polarity. A mobile phase that would give a dense and compact band with an appropriate Rf value for Methylprednisolone was desired Various mobile phases such as methanol: toluene, toluene: ethyl acetate, methanol: ethyl acetate, methanol: toluene: ethyl acetate: ammonia, dichloromethane: toluene, dichloromethane: methanol: toluene was tried in different proportions. A mobile phase consisting of Ethyl acetate: cyclohexane: Glacial acetic acid (8:2:0.2 v/v/v) gave good separation of Methylprednisolone. These chromatographic conditions produced a well-defined, compact band of Methylprednisolone with optimum migration at Rf 0.50 ± 0.05, respectively. The method was optimized using the DoE approach and the design selected is Box-Behnken design with the 16 DoE based trials, which increases the design space of the working area. The sensitivity of HPTLC method that uses UV detection depends upon proper selection of detection wavelength. An ideal wavelength is the one that gives good response for the drugs that are to be detected. In the present study individual drug solution of 10μg/ml was prepared in methanol. The drug solutions were then scanned in the UV region of 200 – 400 nm and the overlay spectra were recorded. All three graphs are shown appreciable absorption at 243 nm (shown in figure 1).


 Bhakti Chapla*
Bhakti Chapla*
 Dr. Vaishali Karkhanis
Dr. Vaishali Karkhanis
 Yash Pokiya
Yash Pokiya
 Payal Panchal
Payal Panchal
 10.5281/zenodo.14916905
10.5281/zenodo.14916905