
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmaceutics, Lisie College of Pharmacy, Ernakulam, Kerala, India – 682018
Acne vulgaris is a common inflammatory skin disorder that often requires long-term topical therapy; however, conventional formulations are limited by poor skin penetration, instability of active ingredients, and local irritation. Tea tree oil–loaded transethosomal gel can enhance topical anti-acne delivery. Transethosomes were prepared by the cold method using soya lecithin, cholesterol, ethanol, and Span 80, incorporating different concentrations of tea tree oil. The formulations were evaluated for vesicle size, organoleptic properties, drug content, pH, spreadability, in vitro permeation, and antibacterial activity against acne-causing microorganisms. The developed transethosomes showed nanosized vesicles with good uniformity, indicating enhanced deformability and suitability for dermal penetration. The transethosomal gel exhibited acceptable pH, good homogeneity, satisfactory spreadability, and efficient washability. In vitro permeation studies demonstrated improved drug diffusion compared to conventional gel systems, while antibacterial studies showed notable inhibitory activity against, Propionibacterium acnes confirming enhanced antimicrobial efficacy of the formulation. Overall, the results suggest that tea tree oil–loaded transethosomal gel is a promising, non-invasive delivery system that improves stability, skin permeation, and therapeutic potential of herbal anti-acne agents. This novel formulation strategy offers an effective alternative for topical acne management with improved efficacy and patient compliance.
To improve the delivery of medications through the skin, scientists have developed highly deformable lipid vesicles. These specialized vesicles make it easier for drugs to pass through the skin barrier. There are several types of these vesicles, including ethosomes, transferosomes, and transethosomes, which are commonly used in both cosmetic and pharmaceutical applications. 1 Transferosomes are flexible vesicular carriers made up of a lipid bilayer along with a substance called an edge activator. However, one major limitation of transferosomes is their difficulty in loading hydrophobic (water-repelling) drugs without losing their elastic nature. Ethosomes are another type of vesicular carrier composed of phospholipids and a high concentration of alcohol, making them hydro-alcoholic systems. But ethosomes also have a disadvantage when applied to the skin in non-occlusive conditions (without covering), the alcohol tends to evaporate, which can cause skin dryness. 1 To overcome the limitations of both transferosomes and ethosomes, scientists developed transethosomes, a hybrid system that combines the properties of both. Transethosomes are designed to maintain high flexibility, improve drug loading, and enhance skin penetration while minimizing issues like alcohol evaporation and loss of elasticity.1
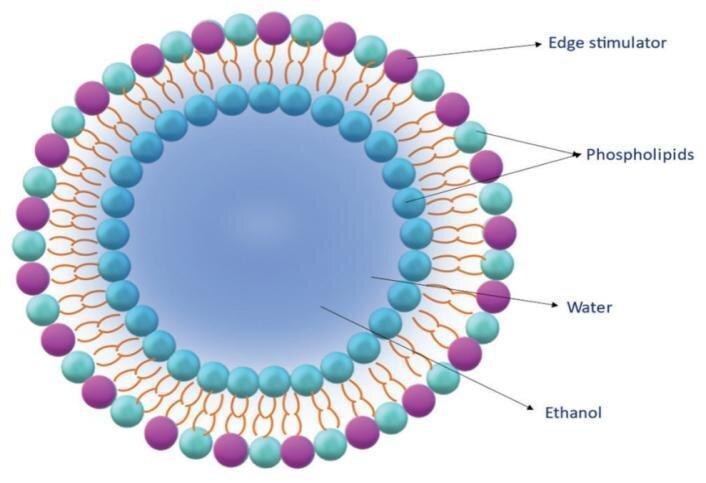
Fig 1: Structure of transethosomes 2
1.1 Introduction to transethosomal gel
Transethosomal gel is an advanced drug delivery system designed to help medicines pass through the skin more effectively. It is made up of phospholipids, ethanol, and an edge activator, which work together to improve how well the drug can penetrate the skin and stay in the body for a longer time. This leads to better therapeutic results.3 They have very small vesicle sizes, which allows them to easily change shape and move through the skin layers efficiently. This makes them a promising option for delivering drugs through the skin in a more effective and controlled way.3 Gels are defined as semi rigid systems in which the movement of the dispersing medium is restricted by an interacting three-dimensional network of particles or solvated macromolecules of the dispersed phase. Gel-forming polymers can produce materials with a wide range of consistencies, starting from a sol and gradually increasing in rigidity through mucilage and jelly to gels and hydrogels.4
1.2 Advantages of transethosomes 3
1.3 Disadvantages of transethosomes 5
2 ACNE 6
Acne vulgaris is a common skin disorder that mainly affects adolescents and young adults. It develops in the sebaceous (oil) follicles of the skin and can vary from mild blackheads and whiteheads to severe, inflamed cystic acne on the face, chest, and back.
Conventional topical formulations like creams, gels, and lotions often face limitations such as poor skin penetration, instability of active ingredients, and irritation. To overcome these issues, transethosomes, a novel vesicular delivery system, have been developed to enhance skin permeation and therapeutic efficacy. Tea tree oil (Melaleuca alternifolia) is well-known for its antimicrobial, anti-inflammatory, and anti-acne properties. However, its volatility and potential skin irritation limit its direct application. Incorporating tea tree oil into a transethosomes gel formulation can improve its stability, skin penetration, and effectiveness in acne management.
2.1 Natural drugs used for acne
3. MATERIALS AND METHODS
Table 1: Ingredients in anti-acne transethosomal gel and its uses
|
SI.no |
Chemicals/Reagents |
Use |
|
1 |
Tea Tree Oil |
Treat acne |
|
2 |
Soya lecithin |
Emulsifier |
|
3 |
Cholesterol |
Stabilizer |
|
4 |
Span 80 |
Surfactant |
|
5 |
Ethanol |
Solvent |
|
6 |
Coconut Oil |
Diluent for tea tree oil |
|
7 |
Propylene glycol |
Humectant |
|
8 |
Carbopol 934 |
Gelling agent |
|
9 |
Propyl paraben |
Preservative |
|
10 |
Distilled water |
Solvent |
3.1 Preformulation studies
3.1.1 Identification of tea tree oil
3.1.1.1 Organoleptic characteristics 10
The colour, odour and texture were checked.
3.1.1.2 Chemical test 11
3.1.1.3 pH determination 12
A small quantity of tea tree oil was directly spread on pH paper and the colour change was compared with the standard pH colour chart.
3.1.1.4 Determination of Thin Layer Chromatography 13
Analyses were performed on silica gel 60 F254 coated plates. Sample solutions (10 µL) were applied as spots and the plates were developed with 50 mL of mobile phase (toluene: ethyl acetate 95:5 v/v). Terpenes were detected by spraying with vanillin-sulfuric acid reagent, followed by heating at 105 °C for 10 minutes.
3.1.1.5 Determination of fatty oils in essential oils 13
One drop of tea tree oil sample has been allowed to fall onto filter paper. The filter paper must be observed after 24 hours.
3.2 Methodology
3.2.1 Cold Method 3
The cold method is preferred for drugs that are sensitive to heat, such as peptides, proteins, or certain plant-based compounds. In this method, the lipids are first dissolved in ethanol at room temperature, and then slowly hydrated with an aqueous solution that contains the drug, allowing vesicles to form gently without exposing the drug to heat. In this process phospholipids and cholesterol are first dissolved in an organic solvent like ethanol. At the same time the drug and edge activators are dissolved in distilled water. Maintain the same temperature, the aqueous phase is slowly added to the alcoholic phase while stirring continuously for 5 minutes. This helps the components mix evenly and promotes the spontaneous formation of liposomes, as the lipid molecules naturally arrange themselves into bilayer structures that can trap the drug inside.
To make the mixture more uniform and to reduce the size of the transethosomes, the preparation is then sonicated for 20 minutes using an ultra sonicator. The cold method is preferred because it is easy to perform, avoids heat related degradation and result in stable, uniform transethosomes for effective drug delivery.

Fig 2: Schematic representation of transethosome preparation by cold method
3.2.2 Formulation of transethosomes
Table 2: Formula of transethosomes
|
SI.no |
Ingredients |
F1 |
F2 |
F3 |
F4 |
|
1 |
Tea tree oil |
2.5% |
5% |
7.5% |
10% |
|
2 |
Soya lecithin |
1 g |
1 g |
1 g |
1 g |
|
3 |
Cholesterol |
0.58 g |
0.58 g |
0.58 g |
0.58 g |
|
4 |
Ethanol |
10 ml |
10 ml |
10 ml |
10 ml |
|
5 |
Span 80 |
3 drops |
3 drops |
3 drops |
3 drops |
|
6 |
Propylene glycol |
1.5 ml |
1.5 ml |
1.5 ml |
1.5 ml |
|
7 |
Coconut oil |
5 ml |
5 ml |
5 ml |
5 ml |
|
8 |
Distilled water |
3 ml |
3 ml |
3 ml |
3 ml |
Weighed 1 g of soya lecithin and 0.58 g of cholesterol, along with 3 drops of Span 80, were dissolved in 10 ml of ethanol and stirred using a magnetic stirrer until a clear ethanolic phase was obtained. Separately, the oil phase was prepared by mixing 1.5 ml of propylene glycol, 5 ml of coconut oil, 3 ml of distilled water, and various concentrations of tea tree oil. This mixture was also stirred using a magnetic stirrer.
The aqueous phase was then slowly added to the ethanolic phase with continuous magnetic stirring. The resulting mixture was further processed by sonication using an ultrasonicator to reduce vesicle size and obtain a uniform transethosomal dispersion.
3.2.3 Formulation of transethosomal gel
3.2.3.1 Preparation of gel 14
A precisely weighed amount of Carbopol 934 was taken in a beaker and dispersed in 85 ml of distilled water. The mixture was allowed to stand for 30 minutes to let the Carbopol swell. After swelling, it was stirred using a homogenizer at 1200 rpm for 30 minutes.
3.2.3.2 Incorporation of transethosomes into gel 15
The prepared transethosomal dispersion was carefully incorporated into the previously prepared Carbopol gel base to achieve a final drug concentration. The mixture was stirred gently to ensure uniform distribution of the vesicular dispersion throughout the gel matrix. The entire mixture was then thoroughly homogenized using a homogenizer until a uniform, smooth, and consistent gel was obtained.
The final formulation exhibited desirable consistency and spreadability, making it suitable for topical application. The prepared transethosomal gel was stored in a well-closed container for further characterization and evaluation.
3.3 Evaluation of transethosomes
3.3.1 Organoleptic characterization
The colour, odour and shape were checked.
3.3.2 Determination of vesicle size 16
Vesicle size was determined using dynamic light scattering technique with a LitesizerTM 500 at 25 °C. Samples were diluted 10,000-fold with distilled water to avoid multiple scattering effects. Parameters such as the hydrodynamic diameter and mean intensity were evaluated to assess the uniformity of vesicle size distribution.
3.3.3 Determination of Thin Layer Chromatography 13
Analyses were performed on silica gel 60 F254 coated plates. Sample solutions (10 µL) of four different concentrations were applied as spots and the plates were developed with 50 mL of mobile phase (toluene: ethyl acetate 95:5 v/v). Terpenes were detected by spraying with vanillin-sulfuric acid reagent, followed by heating at 105 °C for 10 minutes.
3.4 Evaluation of transethosomal gel
3.4.1 Organoleptic characterization 17
About 1 g of drug samples was placed in watch glass and was observed for appearance, colour, any peculiar odour.
3.4.2 pH determination 17
The pH of the transethosomal gel formulation was determined by using pH paper.
3.4.3 Homogeneity 17
All the developed transethosomal gels were evaluated for homogeneity by visual inspection after setting in their respective containers. The gels were examined for uniform appearance and checked for the presence of any visible aggregates or inconsistencies.
3.4.4 Spreadability 18
The most common method for measuring the spreadability is the parallel- plate method. For the parallel-plate method, 1 g of the sample is placed between two glass plates measuring 20×20 cm. A weight of 200g (within the range of 50-500g) is then applied on the upper plate for 1 minute. After this period, the diameter of the sample spread between the plates is measured.
Spreadability is calculated using the formula: Spreadability= d2 ×π4/w
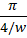
3.4.5 Washability 19
A glass slide was taken and about 1 gm of the transethosomal gel was spread evenly over the surface using a spatula to form a thin layer of approximately 1mm thickness. The applied gel was allowed to stand at room temperature for 1minutes. The slide was then gently rinsed under lukewarm running water for 60 seconds. Finally, the slide was patted dry using tissue paper and the ease of removal was observed.
3.4.6 Drug Content 20
Accurately weigh 1 gm of transethosomal gel and transfer it into a 10 mL volumetric flask. Add a suitable solvent such as ethanol, and sonicate the solution for 15 minutes to ensure a complete extraction of the drug from the gel matrix. After sonication, make up the volume to 10 ml with the same solvent. Filter the solution using filter paper to remove any undissolved particles. Dilute the filtrate appropriately and measure the absorbance at 222 nm using a UV- Visible spectrophotometer, using the corresponding solvent as a blank. Determine the drug concentration from the calibration curve. Calculate the percentage drug content using the formula:
% Drug content =Measured?concentrationLabelled?concentration×100

3.4.7 Occlusivity test 21
Take a glass beaker and fill it with 60 ml of distilled water. Cover the opening with filter paper or a cellophane membrane and record the initial weight of the setup. Spread the gel evenly over the membrane surface. Keep the beaker for 48 hours. After 48 hours, reweigh the beaker to determine the loss of water. Run a control in the same manner but without applying the gel to obtain value A. The water loss in the presence of gel is recorded as B.
Calculate the occlusion factor using the formula: Occlusion Factor (OF) = [(A-B) / A] * 100
3.4.8 Moisture Content 22
Accurately weighed about 1 gm of transethosomal gel and transferred it into a previously tarred, china dish. Place the dish in a hot air oven maintained at 105 0C and dry the sample for 3 hours until a constant weight is obtained. After drying calculated the percentage moisture content using the formula: % Moisture= [(Initial weight- Dried weight)]/ Initial weight * 100
3.4.9 Anti - Bacterial study 23
The antibacterial activity of the test samples was evaluated using the agar well diffusion method on Mueller–Hinton agar. Propionibacterium acnes were cultured and adjusted to a standardized inoculum, then uniformly swabbed onto sterile agar plates. Wells of 6 mm diameter were aseptically punched and filled with 100 µL of TTO (5%), TTO (100 µL), and ciprofloxacin (100 µ1/10 µg) as the positive control. Plates were incubated under incubator 37°C, appropriate conditions, and the zones of inhibition were measured in millimetres after incubation to assess antibacterial efficacy.
3.4.10 Invitro permeation Study 24
In vitro studies were performed using an egg membrane mounted on a Franz diffusion cell to evaluate drug permeation. The receptor compartment contained phosphate-buffered saline (PBS) at pH 5, and the formulation was applied to the donor compartment. Samples were withdrawn at fixed time intervals while maintaining sink conditions. The collected samples were analysed at 222 nm using a UV- Visible spectrophotometer to determine drug diffusion.
4. RESULT
4.1 Identification of tea tree oil
4.1.1 Organoleptic characteristics
Table 3: Organoleptic characteristics of tea tree oil
|
SI.no |
Characteristics |
Observation |
|
1 |
Colour |
Colourless to light yellow liquid |
|
2 |
Odour |
Aromatic |
|
3 |
Texture |
Thin, watery, non-greasy |
4.1.2 Chemical test
Chemical test was performed as per the procedure. The result was found to be:


Fig :3Salkowski test (positive) Fig 4: Liebermann-Burchard test (positi
4.1.3 pH determination

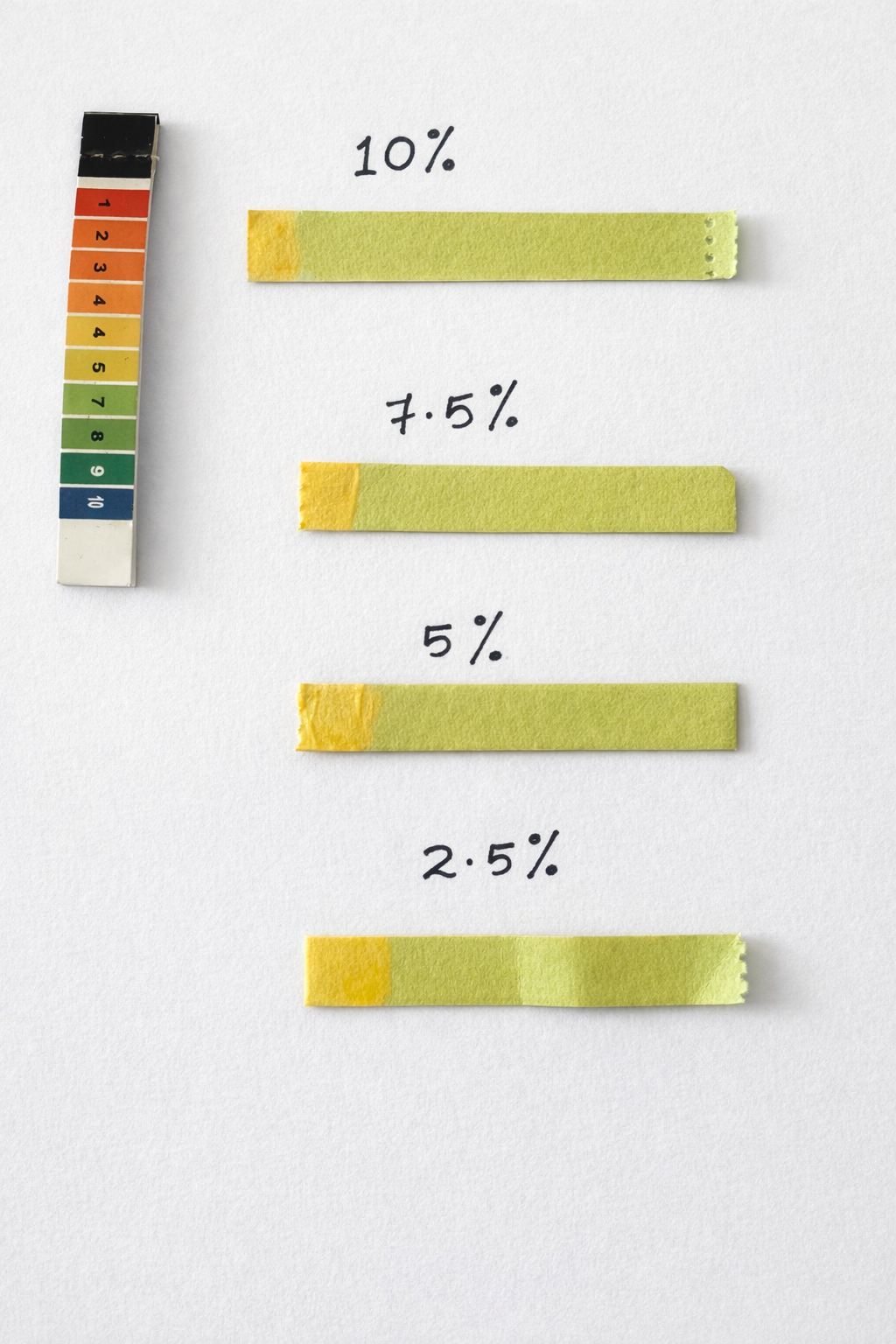
Fig 5: pH of tea tree oil
4.1.4 Determination of Thin Layer Chromatography
The Thin Layer Chromatography was performed as per the procedure mentioned in the methodology. The Rf value was found to be 0.7513.
Fig 6: Thin Layer Chromatography of tea tree oil
4.1.5 Determination of fatty oils in essential oils
The detection of translucent spot reveals the presence of fatty oils.

Fig 7: Translucent spot by Tea tree oil
4.2 Evaluation of transethosomes
4.2.1 Organoleptic characteristics
Table 4: Organoleptic characteristics of transethosomes
|
SI.no |
Characteristics |
Observation |
|
1 |
Colour |
Pale yellow to light yellow |
|
2 |
Odour |
Aromatic |
|
3 |
Shape |
Spherical |
4.2.2 Determination of Vesicle size
The average particle size distribution of the prepared formulations (F1 – F4) ranged from 219.1 nm – 245.7 nm.
Table 5: Determination of vesicle size
|
SI.no |
Formulations |
Particle size distribution (nm) |
Hydrodynamic diameter (nm) |
Mean Intensity (kcounts/s) |
|
1 |
F1 |
243.6 |
316.9 |
319.3 |
|
2 |
F2 |
219.1 |
306.4 |
168.4 |
|
3 |
F3 |
244.5 |
307.4 |
316.9 |
|
4 |
F4 |
245.7 |
349.1 |
210.8 |
4.2.3 Determination of Thin Layer chromatography
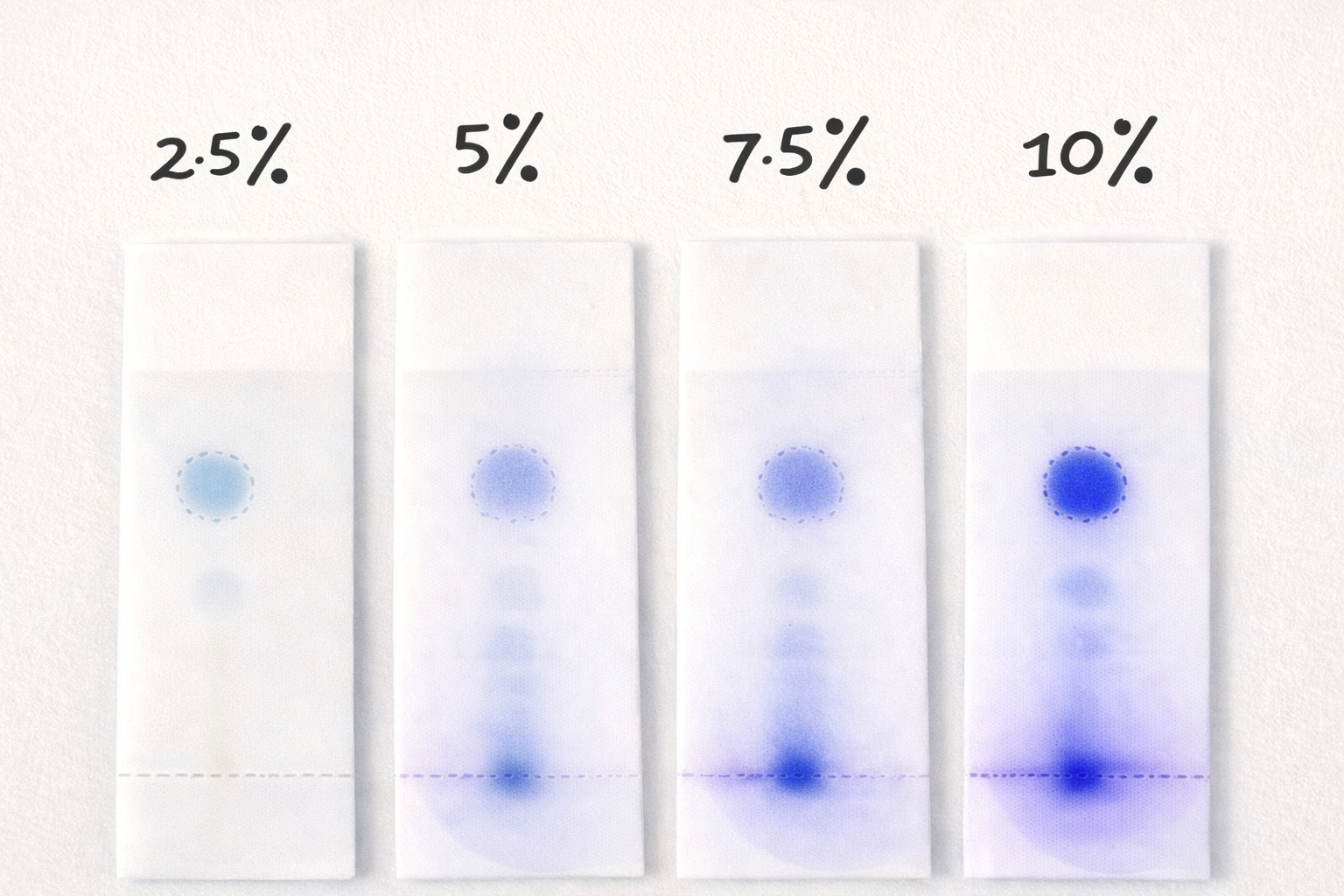
Fig 8: TLC plates for transethosomes (F1-F4)
4.3 Evaluation of anti-acne transethosomal gel
4.3.1 Organoleptic evaluation
The colour, odour and texture of transethosomal gel was found to be,
Table 6: Organoleptic characteristics of transethosomal gel
|
SI.no |
Characteristics |
Observation |
|
1 |
Colour |
Pale yellow |
|
2 |
Odour |
Aromatic |
|
3 |
Texture |
Smooth, gel like |
4.3.2 pH determination
The average pH of prepared transethosomal gel ranged from 4-5.
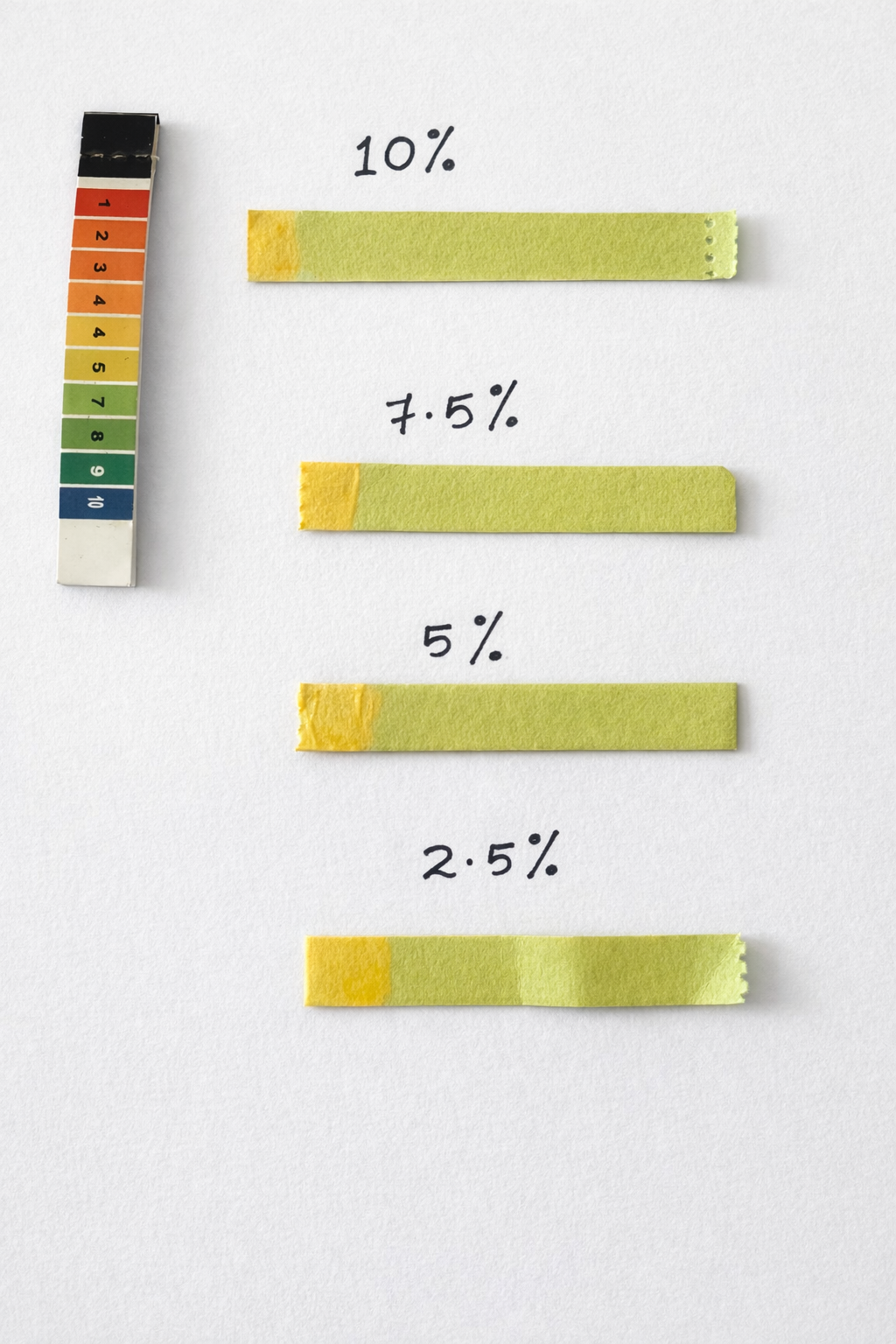
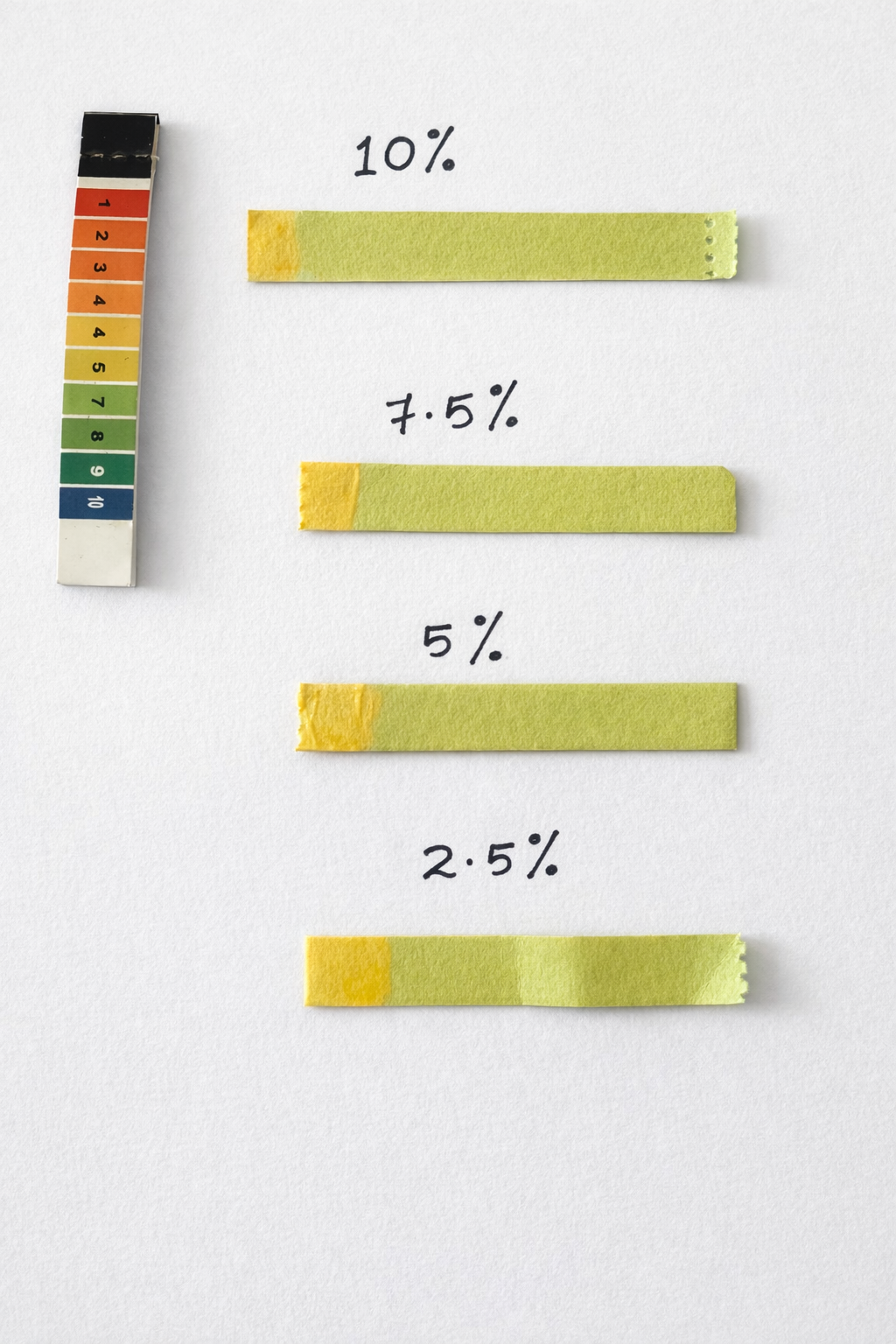
Fig 9: pH determination of transethosomal gel (F1-F4)
4.3.3 Homogeneity
All four formulations (F1-F4) showed good homogeneity with uniform appearance and no visible lumps or phase separation.
Table 7: Homogeneity
|
Formulations |
Observation |
|
|
1. |
F1 |
Moderate |
|
2. |
F2 |
Good |
|
3. |
F3 |
Good |
|
4. |
F4 |
Moderate to good |
4.3.4 Spreadability
The average spreadability values of formulations (F1-F4) ranged from
0.949 cm to 1.130 cm.
Table 8: Spreadability
|
SI. no |
Formulations |
Spreadability (cm) |
|
1 |
F1 |
1.130 |
|
2 |
F2 |
1.326 |
|
3 |
F3 |
1.326 |
|
4 |
F4 |
0.949 |
4.3.5 Washability
All formulations showed good washability indicating suitable for prolonged skin contact.
Table 9: Washability
|
SI.no |
Formulations |
Washability |
|
1. |
F1 |
Easily washable with water |
|
2. |
F2 |
Washable with slight rubbing |
|
3. |
F3 |
Easily removable |
|
4. |
F4 |
Moderately to easily washable |
4.3.6 Drug content
The average drug content of the prepared formulations (F1-F4) was ranged between 4 mg – 5.3 mg.
Table 10: Drug content
|
SI.no |
Formulations |
Drug content(mg) |
|
1 |
F1 |
4.0 |
|
2 |
F2 |
4.9 |
|
3 |
F3 |
4.1 |
|
4 |
F4 |
5.3 |
4.3.7 Occlusivity test
The average occlusivity of the prepared formulations (F1-F4) was ranged from 50% - 55%.
Table 11: Occlusivity
|
SI.no |
Formulations |
Occlusivity (%) |
|
1 |
F1 |
50% |
|
2 |
F2 |
55% |
|
3 |
F3 |
53% |
|
4 |
F4 |
50% |
4.3.8 Moisture Content
The average moisture content of the prepared formulations (F1-F4) after 3 hours, ranged from 80.2% - 93.5%.
Table 12: Moisture content
|
SI.no |
Formulations |
Moisture content (%) |
|
1 |
F1 |
93.5% |
|
2 |
F2 |
80.2% |
|
3 |
F3 |
93.5% |
|
4 |
F4 |
84.9% |
4.3.9 Permeability test
Above 90% drug release was seen in tenth minute in all formulations (F1-F4). The average drug release of prepared formulations (F1-F4) was ranged between 90.2% - 91.2%.
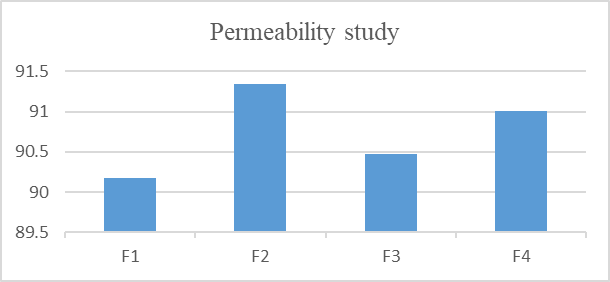
Fig 10: Permeability study (F1-F4)
4.3.10 Anti-bacterial study
The antibacterial activity of F2 was conducted against Propionibacterium acnes and compared to the standard ciprofloxacin (Cip). The results demonstrated that F2 exhibited a measurable zone of inhibition (16 mm) indicating moderate antibacterial activity against Propionibacterium acnes. The standard drug ciprofloxacin produced a larger inhibition zone (26 mm), reflecting its higher antibacterial potency compared to the test formulation. Although the antibacterial activity of F2 was lower than that of the standard antibiotic, the observed inhibition suggests that F2 possesses appreciable activity against P. acnes.
Overall, the findings indicate that formulation F2 has potential as a topical antibacterial agent against P. acnes , particularly for adjunct or alternative therapy for infections caused by P. acnes. Though further optimization may be required to achieve efficacy comparable to conventional antibiotics.
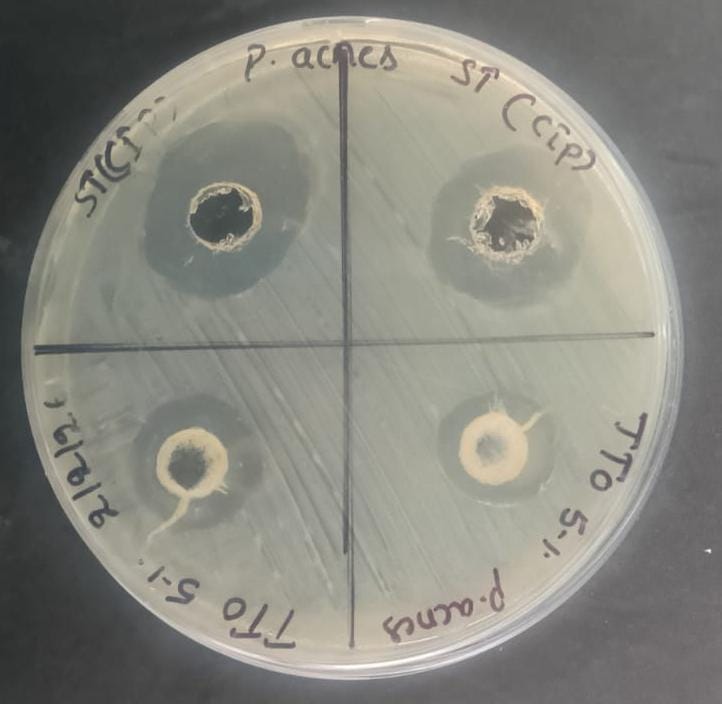
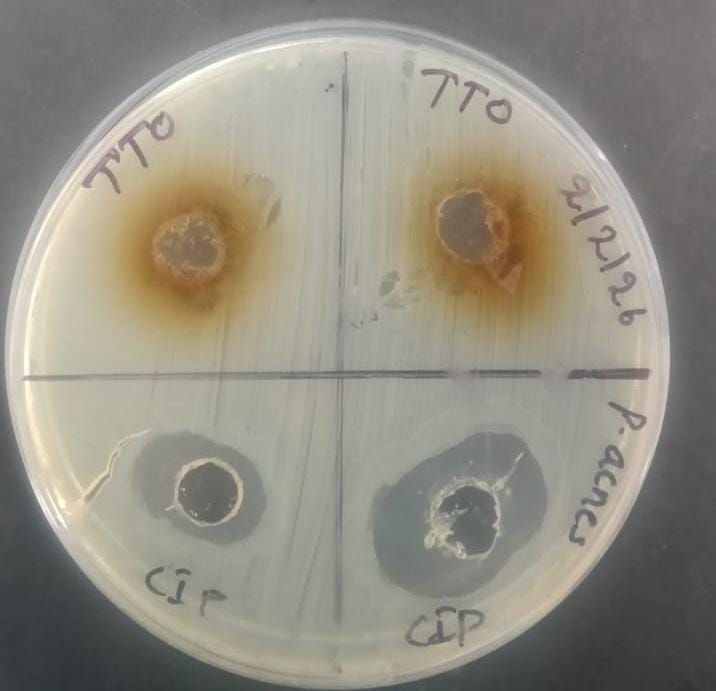
Fig 11: Antibacterial study of F2
Table 13: Antibacterial activity of F2 with zone of inhibition in diameters (mm)
|
Microorganism |
F2 (100 µl) |
TTO (100 µl) |
Ciprofloxacin 100 µl/10 µg) |
|
Propionibacterium acnes |
16 mm |
0 mm |
26 mm |
DISCUSSION
Among the four formulations developed (F1-F4), F2 was selected as the best formulation because it showed the most balanced and satisfactory results overall. A major concern of the study was to prepare formulations within the normal skin pH range of 4.7 to 5.7 to avoid irritation and ensure safety, and all the formulation were successfully maintained within this range. Compared to the others, F2 demonstrated an optimal vesicle size that supports better skin penetration, along with good homogeneity and a smooth gel texture. It also showed better spreadability, making it easier to apply on the skin. The drug content was uniform, indicating effective encapsulation of tea tree oil, while acceptable occlusivity and moisture content supported prolonged skin contact. In addition, F2 showed better antibacterial activity against acne-causing microorganisms, which may be due to the optimal concentration of tea tree oil and excipients used. Overall, these findings indicate that F2 is the most suitable formulation for topical anti-acne application.
SUMMARY AND CONCLUSION
Transethosomal gel as a thoughtful and practical advancement in topical drug delivery, bringing together the flexibility of transferosomes and the penetration-enhancing ability of ethosomes to overcome the natural barrier of the skin. By combining phospholipids, ethanol, and edge activators, transethosomes form ultra-deformable vesicles capable of carrying both hydrophilic and lipophilic drugs deep into the skin, while their incorporation into a gel base improves stability, skin retention, and patient comfort. The book clearly shows how this system is especially relevant for acne management, where conventional formulations often fail due to poor penetration, irritation, or instability of active ingredients. The formulation and evaluation of a tea tree oil–loaded transethosomal gel demonstrate how a natural, volatile anti-acne agent can be stabilized, delivered effectively, and potentially made more patient-friendly through this approach. Overall, transethosomal gels emerge as a promising, non-invasive, and versatile delivery system that bridges the gap between efficacy and safety, offering strong potential for future dermatological and transdermal therapies when supported by further
ACKNOWLEDMENT: We acknowledged to teaching and non-teaching staff of Lisie College of Pharmacy, Ernakulam, for providing all the support.
CONFLICT OF INTEREST: No conflict of interest
REFERENCES
[Cited on 23/10/2025]
[Cited on 23/10/2025]




Ronald Peter, Alna Sebastian, Anaswara Paul, Aneesa V. A., Formulation And Evaluation of Tea Tree Oil Infused Transethosomal Gel., Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 4099-4108. https://doi.org/10.5281/zenodo.18786864
 10.5281/zenodo.18786864
10.5281/zenodo.18786864