
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
1,3 Delhi Institute of Pharmaceutical Sciences and Research, Delhi Pharmaceutical Sciences and Research University, New Delhi 110017, India
2,4 Indian Pharmacopoeia Commission, Ministry of Health & Family Welfare, Government of India, Raj Nagar, Ghaziabad 201002, India
Archaeologists suggest that the Ginkgo biloba is one of the most ancient trees existing on earth. Several pharmacopoeias list the plant, which is well-known in Chinese and Japanese traditions for its medicinal benefits. The plant is rich in diverse phytochemicals and is known as a cognitive enhancer and for other therapeutic properties. The current review article has critically assessed several research studies and reviews on the plant in the existing databases such as ScienceDirect, Sci-finder, Google Scholar, PubMed Central, and pharmacopoeias (IP, BP, USP, Chinese, Japanese, etc.). In this article, we will look at all of the monographs on Ginkgo biloba from the Indian Pharmacopoeia, the US Pharmacopoeia, the European Pharmacopoeia, the British Pharmacopoeia, and the Chinese Pharmacopoeia. We will use Thin Layer Chromatography (TLC) and High-Performance Liquid Chromatography (HPLC) to identify Ginkgo biloba. The article also highlights the therapeutic scope of Ginkgo biloba, bioactive compounds, and their mechanisms of action behind their therapeutic uses. Finally, the paper provides a comprehensive understanding of phytochemical studies and uses of Ginkgo biloba in traditional therapies and current pharmaceutical applications. The information obtained from this review might help researchers, scientists, and health professionals in their journey to investigate the medicinal properties of Ginkgo biloba—consolidated information on the above-mentioned plant.
Ginkgo biloba, confusingly called the "silver almond tree," is from China and apparently one of the rare species that survived even through the last Ice Age. According to fossil records, this tree species is one of the oldest living trees, dating back some 280 million years. For thousands of years, Chinese people have utilized seeds and leaves as medicine to treat respiratory and cardiovascular diseases. It was brought to China and codified in Chinese traditional medicine, where it has become part of the official pharmacopeia of China (He Shan - An, 1997). This is the spiciest plant in the world. Moreover, G. biloba is conceded as a versatile therapeutic plant by which it can be used in a number of illnesses or disease states (Noor-E-Tabassum, 2022). Research has demonstrated that it can lessen memory loss, improve brain function, and moderate the deteriorating effects of Alzheimer's disease (Stefan Weinmann, 2010).
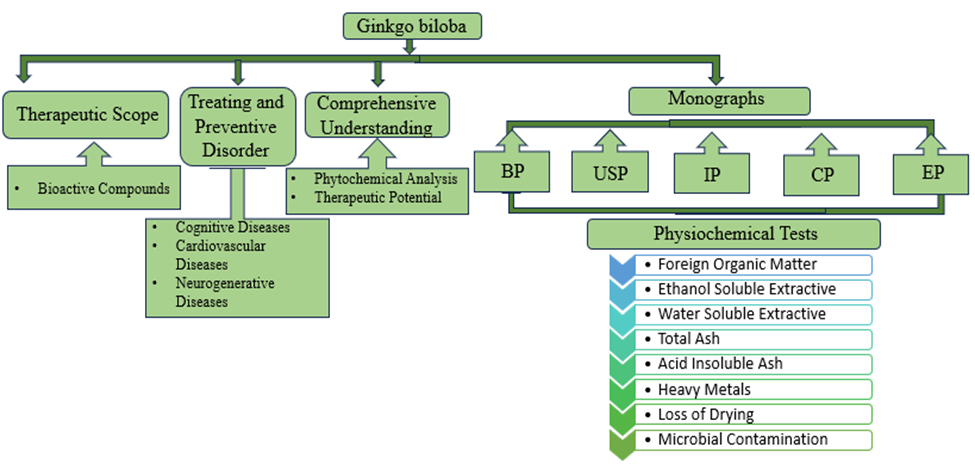
Figure 1 Graphical Presentation of Ginkgo biloba Characteristics
The Ginkgo biloba Extract (GbE), which was separated from the dehydrated leaves of G. biloba, saw as demonstrated to have a variation of advantageous properties, like anti-inflammatory, anticancer, antibacterial, and neuroprotective effects (Noor-E-Tabassum, 2022). Ginkgo biloba is widely used herbal supplements in the globe (Tran Nguyen, 2023). Compared to synthetic drugs, EGb761 has much fewer side effects related to mood stabilizers, antipsychotics, and mood disorders. Clinical treatment for neurological and cardiovascular conditions, including dementia and Alzheimer's disease, has made extensive use of EGb (Anna Nowak, 2021). Ginkgo biloba extract is the most studied and widely utilized herbal medicinal product (HMP). Ginkgo biloba, a popularly known herb with potential circulatory and cognitive effects, is currently one of the most researched and highly profitable medicinal plant preparations on the market. Its popularity in modern as well as traditional herbal medicine is highlighted by its wide appeal and continuous scientific interest (Noor-E-Tabassum, 2022).

Figure 2: Ginkgo biloba

Figure 3: Ginkgo leaves
Applications of Ginkgobiloba-
Ginkgo biloba have various therapeutic uses. It is widely used in different types of diseases. Various therapeutic applications of Ginkgo biloba are follows-
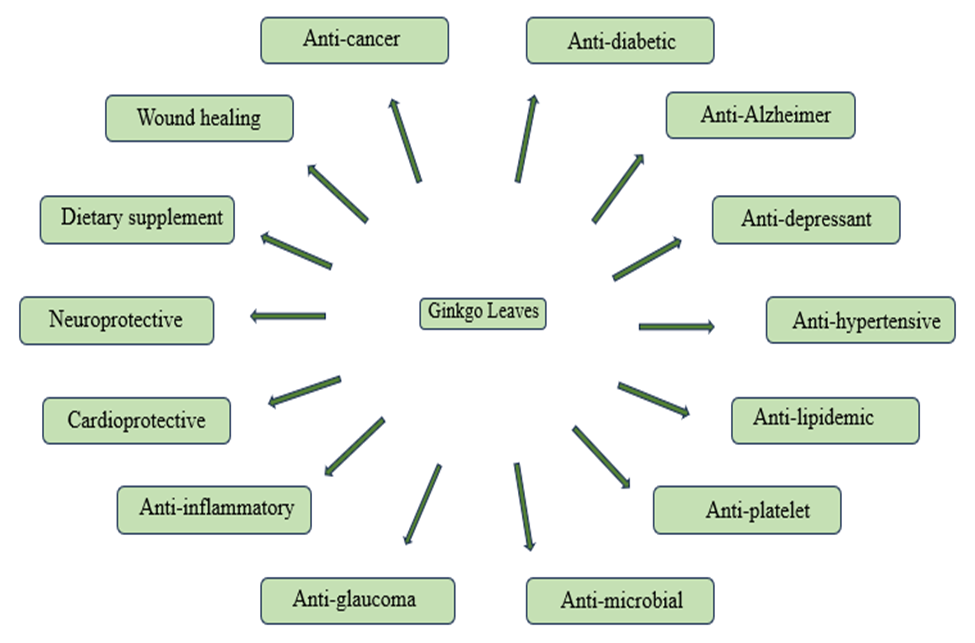
Figure 4: Applications of Ginkgo biloba
Ginkgo biloba has potential anticancer properties due to its rich content of flavonoids and terpenoids, which have antioxidant and anti-inflammatory effects. Several in vitro and in vivo studies suggest that Ginkgo biloba extract (particularly EGb761) may inhibit the proliferation of cancer cells, induce apoptosis, and enhance the efficacy of chemotherapeutic agents. For example, research has shown that EGb761 can suppress tumor growth in breast, colon, and liver cancer models by modulating pathways such as PI3K/Akt and MAPK, reducing oxidative stress, and enhancing immune responses (Gauthier & Mendy, 2018).
Ginkgo biloba has shown its antidiabetic effects in both experimental and clinical studies. The extract, EGb761, is used to improve insulin sensitivity, enhance glucose uptake, reduce oxidative stress, and protect pancreatic β-cells. In animal models of diabetes, Ginkgo biloba supplementation led to reduced blood glucose levels, improved lipid profiles, and modulation of key signaling pathways involved in glucose metabolism (Mohamed et al., 2018). Clinical studies have also suggested benefits when used alongside standard antidiabetic therapies (El-Abhar & Schaalan, 2014).
Ginkgo biloba has potential neuroprotective effects, particularly in the context of Alzheimer's disease (AD). The standardized extract EGb761 has shown benefits in improving cognitive function, slowing cognitive decline, and enhancing memory in patients with mild to moderate Alzheimer’s disease. (Snitz et al., 2009).
Ginkgo biloba extract (EGb 761) has shown antidepressant effects by improving mood, reducing anxiety, and enhancing the action of neurotransmitters like serotonin and dopamine. It is especsially beneficial in older adults and for those having anxiety-related symptoms (Woelk, Arnoldt, Kieser, & Hoerr, 2007).
Ginkgo biloba may help reduce blood pressure by improving endothelial function, enhancing nitric oxide production, and increasing blood vessel dilation. Its antioxidant effects support cardiovascular health, especially in older adults with mild hypertension (Cieza, Maier, Köpfli, & Schläppi, 2003).
Ginkgo biloba has shown potential in improving lipid profiles by lowering total cholesterol, triglycerides, and LDL levels while increasing HDL. These effects are primarily due to its antioxidant compounds. Studies in animal models have supported its use as a natural agent for managing hyperlipidemia (Choi, Choi, Park, & Kang, 2011).
Ginkgo biloba shows antiplatelet effects by inhibiting platelet-activating factor (PAF), which plays a key role in platelet aggregation and thrombus formation. The standardized extract EGb 761 contains ginkgolides, particularly ginkgolide B, known for their PAF antagonist activity. This makes Ginkgo biloba is potentially useful in reducing the risk of thrombosis and improving circulation, exhibits antiplatelet effects (Kubota, Tanaka, Yamazaki, & Kishimoto, 2004).
Ginkgo biloba exhibits antimicrobial activity, primarily due to its bioactive compounds such as flavonoids, terpenoids, and ginkgolic acids. These compounds can disrupt microbial cell walls, inhibit enzyme systems, and prevent DNA replication. In vitro studies have shown that Ginkgo biloba extract exhibits antibacterial effects against both Gram-positive and Gram-negative bacteria, including Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa, as well as antifungal activity against Candida albicans. This supports its potential role as a natural antimicrobial agent. (Sati & Joshi, 2011; Ryu et al., 2009).
Ginkgo biloba shows promise as an adjunctive treatment for glaucoma, particularly normal-tension glaucoma (NTG). Its neuroprotective and vasodilatory effects help improve ocular blood flow and reduce oxidative damage to retinal ganglion cells. The extract EGb 761 has been associated with improved visual field performance in patients with normal tension glaucoma (Quaranta et al., 2003).
Ginkgo biloba have anti-inflammatory properties, largely due to its rich content of flavonoids and terpenoids. These compounds inhibit the production of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6, and reduce the activity of enzymes like cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS). Extract EGb 761, has shown effectiveness in reducing inflammation in models of arthritis, neuroinflammation, and cardiovascular diseases (Kim, Kang, Lee, Kim, & Kim, 2009).
Ginkgo biloba has cardioprotective effects. It improves blood circulation, reducing oxidative stress, and preventing platelet aggregation. Its active compounds support heart health by lowering blood pressure, improving lipid profiles, and protecting against ischemic injury (Smith & Luo, 2004).
Ginkgo biloba has neuroprotective effects. It improves cerebral blood flow, reduces oxidative stress, and inhibits neuronal apoptosis. The extract EGb 761 has been shown to enhance cognitive function and protect against neurodegenerative diseases such as Alzheimer's and Parkinson's (Mancuso, Santangelo, & Barone, 2007).
Ginkgo biloba is widely used as a dietary supplement to enhance cognitive function, improve memory, and boost mental clarity. It is also used to reduce anxiety, improve circulation, and support cardiovascular health. The extract EGb 761 is the most commonly used form in supplements (Schmidt, Willner, & Schaefer, 2015).
Ginkgo biloba has been found to promote wound healing through its antioxidant, anti-inflammatory, and vasodilatory properties. It accelerates tissue repair by enhancing blood circulation, reducing oxidative stress, and stimulating collagen production, making it a useful natural agent in treating chronic wounds and skin injuries (Hossain, Rahman, & Yoon, 2014).
Ginkgo biloba Monographs:
Table 1 Monograph followed by IP (Indian Pharmacopoeia)
|
Tests |
Ginkgo Leaf |
Ginkgo Dry Extract |
Ginkgo Tablet |
|
|
Category |
Brain tonic |
Brain tonic |
Brain tonic |
|
|
Identification by TLC |
Mobile phase |
7.5 amounts of acetic acid, 67.5 amounts of ethyl acetate, 17.5 amounts of water, and 7.5 amounts of formic acid in anhydrous state. ((IPC), 2022). |
Combine 67.5 volumes of ethyl acetate, 17.5 volumes of water, 7.5 amounts of acetic acid, and 7.5 amounts of aqueous formic acid ((IPC), 2022). |
Combine 67.5 volumes of ethyl acetate, 17.5 volumes of water, 7.5 amounts of acetic acid, and 7.5 amounts of aqueous formic acid ((IPC), 2022). |
|
Test solution |
Reflux 2 grams of the coarsely ground substance under investigation for 10 minutes with 10 milliliters of methanol. After cooling, filter the mixture. Then, use an additional 20 milliliters of methanol to rinse and filter the remaining residue. Combine all the filtrates and use a vacuum to concentrate the solution until it reaches a final volume of 10 milliliters ((IPC), 2022). |
10 mL of methanol was utilized to dissolve 100 milligrams of the extract, which was then sonicated for 10 minutes ((IPC), 2022). |
10 milliliters of methanol have been used to dissolve 100 milligrams of the extract, and it has been sonicated for 10 ((IPC), 2022). |
|
|
Reference solution |
After 10 minutes of refluxing 2 grams of the Ginkgo IPBRS in 10 milliliters of methanol, let cool and filter. After adding twenty milliliters of methanol to the residue again, cool it down and filter it. Put all of the filtrates together and vacuum-concentrate to 10 milliliters ((IPC), 2022). |
After 10 minutes of refluxing 2 g of Ginkgo IPBRS in ten milliliters of methanol, let cool and filter. After adding 20 milliliters of methanol to the residue again, cool it down and filter it. Put all of the filtrates together and vacuum-concentrate to 10 milliliters ((IPC), 2022). |
After 10 minutes of refluxing 2 g of Ginkgo IPBRS in 10 milliliters of methanol, let it cool and filter. After adding 20 milliliters of methanol to the residue again, cool it down and filter it. Put all of the filtrates together and vacuum-concentrate to 10 milliliters ((IPC), 2022). |
|
|
Application |
10 µl as band ((IPC), 2022). |
10 µl as band ((IPC), 2022). |
10 µl as band ((IPC), 2022). |
|
|
Development |
Across an 8 cm path ((IPC), 2022). |
Across an 8 cm path ((IPC), 2022). |
Across an 8 cm path ((IPC), 2022). |
|
|
Drying |
At 105° ((IPC), 2022). |
At 105° ((IPC), 2022). |
At 105° C ((IPC), 2022). |
|
|
Detection |
At 254 nm and 366 nm ((IPC), 2022). |
At 254 nm and 366 nm ((IPC), 2022). |
At 254 nm and 366 nm ((IPC), 2022). |
|
|
Assay by HPLC
|
Test solution |
Reflux 2.5 grams of the coarsely ground material under investigation in 50 milliliters of methanol for 20 minutes. Once the reflux process completes, let the mixture cool to room temperature. Once cooled, filter the solution to remove any solid particles. Collect and combine all the filtrates, then concentrate the mixture until it is fully dried. Next, add 20 milliliters of methanol and 20 milliliters of 1.5 M of HCL to the dried residue. Sonicate the mixture for 10 minutes to ensure proper dissolution, then dilute it to a total volume of 50 milliliters using methanol. Centrifuge the solution for 10 minutes to separate the phases. Carefully remove 10 mL of the supernatant liquid and transfer it into a brown glass vial 10-milliliters. Use an aluminum cap and rubber stopper to seal the vial, then heat it in a water bath for 25 minutes. After the heating process is finished, let the vial cool to room temperature ((IPC), 2022). |
Reflux approximately 0.300 grams of the extract under investigation in 100 milliliters of an extraction solvent composed of 25 amounts of methyl alcohol, 3 amounts of concentrated hydrochloric acid, and 12 amounts of water. Carry out the reflux process in a water bath for 135 minutes. Once the reflux process is finished, let the solution cool to room temperature, then add methanol to bring the final volume to 100 ml ((IPC), 2022). |
Weigh and grind twenty tablets. Precisely weigh out 50 mg of powdered flavone glycosides (about) in a volumetric flask (50.0 ml). Following a three-minute sonication, add 20 milliliters of methanol. For 135 minutes, add 20 milliliters of 1.5 M hydrochloride acid to 12 volumes of water in a bath of water. Let it cool to room temperature, then add methanol to bring the final volume to 50 milliliters ((IPC), 2022). |
|
Reference solution |
The Reference solution (a). After dissolving about 10 milligrams of quercetin IPRS in 25 milliliters of methanol and ten minutes of sonication, dilute the contents to 50 milliliters ((IPC), 2022). The Reference solution (b). Dissolve approximately 10 milligrams of kaempferol IPRS and 2.5 milligrams of isorhamnetin IPRS in 25 milliliters of methanol. After complete dissolution, filter the solution to remove any impurities ((IPC), 2022). |
The Reference solution (a). Mix 12.5 milligram of quercetin IPRS in 50 milliliters of methanol. Sonicate the solution for 10 minutes to ensure thorough dissolution. Afterward, dilute the mixture to a total volume of 100 milliliters with methanol, then filter to remove any impurities ((IPC), 2022). The Reference solution (b). To prepare the solution, 12.5 mg of kaempferol IPRS and 3 mg of isorhamnetin IPRS were mixed in 50 milliliters of methanol and subjected to sonication for 10 minutes. After that, added methanol to the solution to dilute it to a final amount of 100.0 milliliters and subsequently filtered ((IPC), 2022). |
To prepare the solution, approximately 2 mg of quercetin IPRS, 2 mg of kaempferol IPRS, and 0.5 milligram of isorhamnetin IPRS were dissolved in 5 ml of methanol. After sonicating the mixture for 10 minutes in order to ensure complete dissolution, it was diluted with methanol to a final quantity of 10 ml and closely filtered to remove any impurities ((IPC), 2022). |
|
|
Column |
A 15 cm by 4.6 mm stainless steel column stuffed with octadecylsilane bound to porous silica (5µm) ((IPC), 2022). |
A stainless-steel column, measuring 15 cm in length and 4.6 millimeter in inner width, packed with octadecylsilane (ODS) associated with 5 µm porous silica particles, was used ((IPC), 2022). |
A stainless-steel column, 15 centimeter in length and 4.6 millimeter in inner width, packed with octadecylsilane bonded to 5 µm porous silica particles, was used for the analysis ((IPC), 2022). |
|
|
Elution solvent |
Elution solvent A: Water and orthophosphoric acid, Adjust the pH 2 with orthophosphoric acid and water. Elution solvent B: Methanol ((IPC), 2022). |
Elution solvent A: Water and orthophosphoric acid, Adjust the pH 2 with orthophosphoric acid and water. Elution solvent B: Methanol ((IPC), 2022). |
Elution solvent A: Water and orthophosphoric acid, Adjust the pH 2 with orthophosphoric acid and water. Elution solvent B: Methanol ((IPC), 2022). |
|
|
Gradient Program |
0 min A 60% ; 1 min A 60% ; 20 min A 45% ; 21 min A 0% ; 25 min A 60% ; 27 min A 60 % ((IPC), 2022). |
0 min A 60% ; 1 min A 60% ; 20 min A 45% ; 21 min A 0% ; 25 min A 60% ; 27 min A 60 % ((IPC), 2022). |
0 min A 60% ; 1 min A 60% ; 20 min A 45% ; 21 min A 0% ; 25 min A 60% ; 27 min A 60 % ((IPC), 2022). |
|
|
Flow rate |
1.0 ml per minute ((IPC), 2022). |
1.0 ml per minute ((IPC), 2022). |
1.0 ml per minute ((IPC), 2022). |
|
|
Detection |
Spectro photo-meter at 370 nm ((IPC), 2022). |
Spectro photo-meter at 370 nm ((IPC), 2022). |
Spectro photo-meter at 370 nm ((IPC), 2022). |
|
|
Injection |
10µl. ((IPC), 2022). |
10µl ((IPC), 2022). |
10µl ((IPC), 2022). |
|
Table 2 Monograph followed by USP (United State Pharmacopoeia)
|
Tests |
Ginkgo (leaf) |
Ginkgo Capsule |
Powdered Ginkgo Extract |
Ginkgo Tablet |
|
|
Identification by TLC |
Standard |
In methanol, USP Quercetin RS (0.2 mg/mL), 0.6 milligram per milliliter of USP Rutin RS and 0.2 milligram per milliliter of USP Chlorogenic Acid RS ((USP), 2023). |
- |
In methyl alcohol, USP Quercetin RS (0.2 mg/mL), 0.6 milligram per milliliter of USP Rutin RS and 0.2 milligram per milliliter of USP Chlorogenic Acid RS ((USP), 2023). |
- |
|
Sample |
Put 10 milliliter of methyl alcohol into a 50-milliliter RBF with a reflux condenser, add 1 g of finely powdered ginkgo, and then reflux on a hot bath of water for 10 minutes. Once it gets to ambient temperature, filter it out ((USP), 2023). |
- |
A 4:1 methanol and water mixture containing 5 mg/mL of the powdered extract ((USP), 2023). |
-
|
|
|
Elution solvent |
Aqueous formylic acid, anhydrous ethanoic acid, ethyl ethanoate, and water (11:11:100:26) ((USP), 2023). |
- |
Aqueous formylic acid, anhydrous ethanoic acid, ethyl ethanoate, and water (11:11:100:26) ((USP), 2023). |
- |
|
|
Derivatization |
Derivatization reagent A: 5 milligram per milliliter of (2-aminoethoxy) diphenyl borane in methanol Derivatization reagent B: 50 milligram per milliliter of polyethylene oxide (PEO) 400 in alcohol ((USP), 2023).
|
- |
Derivatization reagent A: 5 milligram per milliliter of (2-aminoethoxy) diphenyl borane in methanol Derivatization reagent B: 50 milligram per milliliter of polyethylene oxide (PEO) 400 in alcohol ((USP), 2023). |
-
|
|
|
Assay by HPLC - 1 |
Standard |
Standard solution A: methanol containing 0.02 milligram/milliliter of USP Quercetin RS Standard solution B: USP Kaempferol RS 0.02 mg per milliliter in methyl alcohol Standard mixture C: 0.005 milligram per milliliter of USP Isorhamnetin RS ((USP), 2023). |
0.05 milligram/milliliter of USP Isorhamnetin RS, 0.2 milligram/milliliter of USP Kaempferol RS, and 0.2 milligram/milliliter of USP Quercetin RS in methyl alcohol ((USP), 2023).
|
Standard mixture A: methanol with 0.125 milligram/milliliter of USP Quercetin RS Standard mixture B: methanol with 0.125 milligram/milliliter of USP Kaempferol RS Standard mixture C: methanol with 0.03 milligram/milliliter of USP Isorhamnetin RS ((USP), 2023). |
Methanol with 0.2 milligram/milliliter of USP Quercetin RS, 0.2 milligram/milliliter of USP Kaempferol RS, and 0.05 milligram/milliliter of USP Isorhamnetin RS ((USP), 2023). |
|
Sample |
To a 250-mililiter flask fitted with a reflux condenser assembly, add about 1.0 g of ginkgo powder. For 135 minutes, reflux the mixture in a water bath., after adding 78 mL of the extraction solvent. Let cool until room temperature is achieved. Once the filter residue has been drained, The rinsate should be put into a 100-milliliter volumetric flask, diluted with methanol, and mixed. Wash the filter residue with a little bit of methanol ((USP), 2023).
|
Weigh and grind the NLT 20 capsules into a fine powder. Fill a 50-milliliter volumetric flask with a precisely weighed amount of the powder, or around 50 mg of flavanol glycosides. After three minutes of sonication, add 20 mL of methanol. After adding 20 milliliters of 1.5 N hydrochloride acid, sonicate for an additional 10 minutes. Let it cool to room temperature before adding methanol to dilute it to volume. After centrifuging, transfer an amount of the clear supernatant into a glass vial that's low-actinic and has a rubber stopper. After 25 minutes of heating in a steam bath, To room temperature, cool in an ice bath ((USP), 2023). |
The powdered extract must be added to a 250-milliliter flask that has a reflux condenser set up accurately, weighing 0.3 grams. Add 78 milliliters of the extraction solvent, then warm the mixture in a hot bath of water and reflux for 135 minutes. Let the mixture reach room temperature. Once the contents have cooled, proceed them into a 100-milliliter volumetric flask, diluted with water to the desired amounts, and then thoroughly mix ((USP), 2023). |
Weigh and grind the NLT 20 tablets into a fine powder. Fill a 50-milliliter volumetric flask with a precisely weighed amount of the powder, or around 50 mg of flavanol glycosides. After three minutes of sonication, add 20 mL of methanol. After adding 20 milliliters of 1.5 N hydrochloride acid, sonicate for an additional 10 minutes. Let it cool to room temperature before adding methanol to dilute it to volume. After centrifuging, pour some of the transparent condensate into a glass vial in a low actinic content with a rubber stopper. After 25 minutes of heating in a steam bath, To room temperature, cool in an ice bath ((USP), 2023). |
|
|
Mobile phase |
Water, phosphoric acid, and methanol (100: 1:100) ((USP), 2023). |
Phosphoric acid, methanol and water (1:100:100) ((USP), 2023). |
Phosphoric acid, water and methanol (1:100:100) ((USP), 2023). |
Phosphoric acid, water and methanol (1:100:100) ((USP), 2023) |
|
|
Diluents |
- |
- |
- |
- |
|
|
Detection wavelength |
UV 370 nm ((USP), 2023). |
UV 370 nm ((USP), 2023). |
UV 370 nm ((USP), 2023). |
UV 370 nm ((USP), 2023). |
|
|
Gradient Program |
-
|
- |
- |
- |
|
|
Assay by HPLC 2 |
Standard |
Make five USP Ginkgo terpene lactones RS dilution solutions for each of the relevant terpene lactones based on the labeled content of each individual lactone; if necessary, dissolve the analytes using sonication; filter through a 0.45-µm or smaller pore ((USP), 2023).
|
Make five preparations of USP Ginkgo Terpene Lactone RS in diluent using the specified content of each separate terpene lactone. For each relevant terpene lactone, prepare five solutions (between 5 and 500 µg/mL). If needed, to dissolve the analytes, utilize sonication. Apply a filter. that has pores that are at least 0.45 µm in size ((USP), 2023). |
Five USP Ginkgo Terpene Lactones RS solutions need to be prepared in diluent, with each relevant terpene lactone's labeled content taken into account. The range for each solution should be between 5 and 500 µg/mL. If necessary, employ the sonication for dissolving the analytes. Go through a filter with pore sizes of at least 0.45 µm ((USP), 2023) |
In the diluent, five preparations of USP Ginkgo Terpene Lactones RS must be prepared. The concentrations of each terpene lactone must be modified based on its labeled content, ranging from 5 to 500 µg/mL. To dissolve the analytes, use sonication if required. Transform via a Filter with pores as small as 0.45 µm ((USP), 2023). |
|
|
Sample |
Weigh 2.5 grams of ginkgo precisely, then transfer it to a glass centrifuge tube that is thirty milliliters in size and has a screw cap and PTFE gasket. Seal the tube tightly, then use a vortex mixer to mix it well in 10 mL of solvent. For thirty minutes, the mixture gets heated to 90°C in a water bath. After cooling and centrifuging, pour the supernatant into a flask with a circular bottom and reserve the residue in a glass tube. Two more extractions should be performed, each using 10 milliliters of solvent. Mix the extracts and vacuum-dry until completely dry on a bath of water kept at 50°C. Sonicate for five minutes after adding 10 ml a buffer solution to the residue. Move the mixture quantitatively to a glass chromatographic tube that can hold 20 milliliters of the aqueous phase and is filled with silica earth. After using two 5-milliliter portions of the buffer solution to rinse the beaker, move the rinsates to the column. Permit the column to absorb the buffer solution. After 15 minutes, use 100 ml of ethyl acetate to elute the column. Gather the eluate and vacuum-evaluate it until it is completely dry on a bath of water kept at 50°. Use 10 milliliters of diluent to dissolve the residue ((USP), 2023). |
Weigh and grind the NLT 20 capsule contents into a fine powder. Spoon into a 25-ml beaker a precisely calculated amount of the powder, around 120 milligrams of ginkgo extract powder. After adding 10 milliliters of the buffer solution, sonicate for ten minutes. Transfer the solution quantitatively to a glass chromatographic tube that can hold 20 milliliters of the aqueous phase and is filled with chromatographic silica earth. Transfer the wash solution to the column after washing out the beaker with 5-milliliter components of the buffer solution. Permit the column to absorb the buffer solution. Purify column containing 100 milliliters of ethyl acetate after 15 minutes, collect the resultant solution, and vacuum-evade the substance until it is completely dry in a bath of water kept at 50 degrees. Add 20 milliliters of diluent to the residue and dissolve. Empty the first few milliliters of the filtrates after passing an quantity of the solution via a filter with pores that are at least 0.45 µm in size. ((USP), 2023).
|
Fill a 25-milliliter beaker with approximately 120 mg of precisely weighed powdered extract. Sonicate the residue for five minutes after adding 10 milliliters of the buffer solution. In a glass chromatographic tube with a capacity to hold 20 milliliters of aqueous phase, qualitatively transfer the solution to the tube stuffed with chromatographic silica earth. Move the washings to the column after rinsing. the buffer solution in two 5-milliliter parts in the beaker. In the chromatographic tube, do not use more than 20 milliliter of the overall water phase or its potential for storage. Let the buffer solution soak into the column. Later fifteen minutes, elute the column with one hundred milliliters of ethyl ethanoate, gather the ethyl ethanoate solution, and vacuum-save it until it is completely dry in a bath of water kept at fifty degrees. Add 20 milliliters of diluent to the residue and dissolve ((USP), 2023). |
Weigh and grind NLT 20 tablets to a fine powder. Fill a 25-mL beaker with a precisely measured amount of the powder, or roughly 120 milligrams of ginkgo extract in powder form. After 10 minutes of sonication, add 10 milliliters of the buffer solution. Transfer the mixture quantitatively to a glass chromatographic tube that can hold 20 milliliters of the aqueous phase and is stuffed with chromatographic silica earth. Transfer the washings to the column after washing the beaker with 5-milliliter parts of the buffer solution. Let the column absorbed the buffer solution. Purify the column with 100 milliliters of ethyl ethanoate after 15 minutes and collect the solution and vacuum-evade the material until it is completely dry in a bath of water kept at 50°C. In 20 milliliters of diluent, dissolve the residue. After putting some of the fluid through the filter, empty the initial few milliliters via a filter with pores that are 0.45 µm or less ((USP), 2023). |
|
Mobile phase |
Solution A: Water Solution B: Methanol ((USP), 2023). |
Solution A: Water Solution B: Methanol ((USP), 2023). |
Solution A: Water Solution: Methanol ((USP), 2023). |
Solution A: Water Solution B: Methanol ((USP), 2023). |
|
|
Diluent |
Water with methanol (1:1) ((USP), 2023). |
Water with methanol (1:1) ((USP), 2023). |
Water with methanol (1:1) ((USP), 2023). |
Water with methanol (1:1) ((USP), 2023). |
|
|
Detection wavelength |
Evaporative light-scattering detector ((USP), 2023). |
Evaporative light-scattering detector ((USP), 2023). |
Evaporative light-scattering detector ((USP), 2023). |
Evaporative light scattering detector ((USP), 2023). |
|
|
Gradient program |
0 min A 75% ; 23 min A 52% ; 28 min A 52% ; 30 min A 25% ; 35 min A 10% ; 40 min A 75% ; 50 min A 75% ((USP), 2023). |
0 min A 75% ; 23 min A 52% ; 28 min A 52% ; 30 min A 25% ; 35 min A 10% ; 40 min A 75% ; 50 min A 75% ((USP), 2023). |
0 min A 75% ; 23 min A 52% ; 28 min A 52% ; 30 min A 25% ; 35 min A 10% ; 40 min A 75% ; 50 min A 75% ((USP), 2023). |
0 min A 75% ; 23 min A 52% ; 28 min A 52% ; 30 min A 25% ; 35 min A 10% ; 40 min A 75% ; 50 min A 75% ((USP), 2023). |
|
Table 3 Monograph followed by EUP (European Pharmacopoeia)
|
Tests |
Ginkgo Dry Extract |
Ginkgo Leaf |
||||||||||||||||||||||||||||||
|
Definition |
Flavonoids, stated as the flavone glycosides (Mr 756.7): 22–27 percent (dry extract); -The dry extract contains bilobalide in a concentration ranging from 2.6% to 3.2%, while the ginkgolides A, B, and C are present at levels between 2.8% and 3.4%. Additionally, the amount of ginkgolic acids in the extract is strictly limited to no more than 5 parts per million (ppm) (Commission E. P., European Pharmacopoeia, 2023). |
The dried drug contains 0.5% or more of flavonoids, represented as flavone glycosides with a molecular weight of 757 (Commission E. P., European Pharmacopoeia, 2023) |
||||||||||||||||||||||||||||||
|
Identification By TLC |
Test solution |
20 milligrams of the extract to be tested should be dissolved in 10 milliliters of a solution that contains 2 and 8 portions of each water R and methanol R (Commission E. P., European Pharmacopoeia, 2023). |
Mix 10 milliliter of methyl alcohol R to 2 grams of the herbal powdered medication (710). Warmth in a water bath at 65°C for 10 minutes, shaking constantly. After allowing it to reach room temperature, filter. (Commission E. P., European Pharmacopoeia, 2023). |
|||||||||||||||||||||||||||||
|
Reference solution |
Mix 20 milliliters of methanol R with 1 milligram of Caffeoyl quinic acid R and 3 milligrams of rutoside trihydrate R (Commission E. P., European Pharmacopoeia, 2023). |
Mix 20 milliliters of methanol R with 1 milligram of Caffeoyl quinic acid R and 3 milligrams of rutoside trihydrate R (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Plate |
Plate R for TLC silica gel (5-40 µm) or Plate R for TLC silica gel (2–10 µm)] (Commission E. P., European Pharmacopoeia, 2023). |
TLC silica gel plate R (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Elution solvent |
7.5 amounts of anhydrous formic acid R, 7.5 amounts of glacial acetic acid R, 17.5 amounts of water R and 67.5 amounts of ethyl ethanoate R (Commission E. P., European Pharmacopoeia, 2023). |
7.5 amounts of glacial acetic acid R, 7.5 amounts of anhydrous formic acid R, 17.5 amounts of water R and 67.5 amounts of ethyl ethanoate R (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Application |
20 µL or 5 µL, in bands (Commission E. P., European Pharmacopoeia, 2023). |
20 µL as bands (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Development |
Over a path of 17 cm (Commission E. P., European Pharmacopoeia, 2023). |
Over a path of 17 cm (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Drying |
at 100-105 °C (Commission E. P., European Pharmacopoeia, 2023) |
at 100-105°C (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Detection |
While the plate is warm, dissolve 10 grams per liter of 2-aminoethyl diphenyl borate R in alcohol R. Next, prepare a solution of 50 grams per liter of macrogel 400 R in methanol R, and apply it. Examine the plate with an UV lamp set at 365 nm after letting it air dry for around 30 minutes. (Commission E. P., European Pharmacopoeia, 2023). |
Begin by applying a 10 grams per liter solution of 2-Aminoethyl diphenyl borate in methanol R to the pre-warmed plate. Next, make a solution of fifty grams per liter of macrogel 400 R in methyl alcohol R. Let the plate to air dry for roughly 30 minutes, and afterward, inspect it under a UV light source calibrated to 365 nm (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Assay by HPLC-1 |
Analytical solution |
Mix the extract (0.200 grams) for analysis in 20 milliliters of methanol R. Pour in 15 milliliters of methanol R. Transfer ten milliliters of this liquid into a glass vial that is brown with a capacity of 10mL. Use a strong rubber membrane stopper to close the vial, then fasten it with an aluminum crimped cap. 25 minutes of heating on a water bath. Let it get down to 20 °C (Commission E. P., European Pharmacopoeia, 2023). |
2.500 grams of the powdered herbal medication (710) (2.9.12) is heated for 30 minutes in 50 milliliter of a 60% V/V acetone R solution using a reflux condenser. Gather the filtrate after filtering. The drug residue was extracted a second time using 40 milliliters of a sixty percent V/V acetone R solution and a filter in the same way. Gather filtrates and dilute with a 60% V/V acetone R solution to 100mL. To remove the acetone, permeate 50.0 milliliters of the solution, then move to a 50-milliliter vial and rinse with thirty milliliters of methanol R. Put 4.4 milliliters of HCL RI, diluted with water R, to a volume of 50 milliliter, and centrifuge. Take a 10-milliliter brown glass vial, fill it with 10 mL of the supernatant, seal it with an aluminum cap and rubber seal, and warm it in a water bath for 25 minutes. Then, let it to reach to room temperature (Commission E. P., European Pharmacopoeia, 2023). |
|||||||||||||||||||||||||||||
|
Reference solution |
Mix 20 milliliters of methanol R with 10 milligrams of quercetin dihydrate CRS. Combine 15 milliliter of diluted HCL R with 5 milliliter of water R, and dilute with methyl alcohol R to make 50 mL (Commission E. P., European Pharmacopoeia, 2023). |
Mix 20 milliliters of methanol R with 10 milligrams of quercetin dihydrate R. Mix 5 milliliter of water and 15 milliliters of diluted HCL, then dilute with 50 mL of methanol (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Column |
Dimensions: l = 0.125 m, ø = 4 mm; temperature: 25°C; octadecyl silyl gel of silica for chromatography R (5µm) as the stationary phase (Commission E. P., European Pharmacopoeia, 2023). |
Measurements: l = 0.125 m, ø = 4 mm; static phase: chromatography R (5 µm) octadecyl silyl gel of silica; temperature: 25 °C (Commission E. P., European Pharmacopoeia, 2023).
|
||||||||||||||||||||||||||||||
|
Mobile phase |
Elution solvent A: a 0.3 gram per liter phosphoric acid solutions (phosphoric acid R), modified to a pH of 2;
(Commission E. P., European Pharmacopoeia, 2023). |
Elution solvent A: 0.3 gram per liter of ortho-phosphoric acid R solution modified to pH 2; Elution solvent B: methyl alcohol R.
(Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Flow rate |
1 mL/min (Commission E. P., European Pharmacopoeia, 2023). |
1 mL/min (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Detector |
Spectrophotometer at 370 nm (Commission E. P., European Pharmacopoeia, 2023). |
Spectrophotometer at 370 nm (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Injection |
10 µL (Commission E. P., European Pharmacopoeia, 2023). |
10 µL (Commission E. P., European Pharmacopoeia, 2023). |
||||||||||||||||||||||||||||||
|
Assay by HPLC-2 |
Solution for the test |
Transfer 0.120 gram of the test extract into a 25-milliliter beaker and mix with 10 milliliter of phosphate buffer solution R (pH 5.8) to dissolve it. After adding 15 grams of kieselguhr for chromatography R to a 0.15 mm long and 30 mm internal diameter chromatography column, transfer the solution to it. Next, wash the beaker twice with 5 millimeters of PH 5.8 phosphate- buffer solution each, and then move the wash solutions to the chromatographic column. Let it stand for fifteen minutes. Use 100 milliliters of ethyl ethanoate R to elute. Simmer the eluate in a bath of water at 50°C until it evaporates completely, maintaining a pressure of no more than 4 kPa. Air currents are used to remove the solvent residue. Empty the remains into 2.5 milliliters of the mobile phase (Commission E. P., European Pharmacopoeia, 2023). |
|
|||||||||||||||||||||||||||||
|
Reference solution |
The reference solution (a): Weigh out 30 milligrams of benzenemethanol CRS, Dissolve the mixture to 100 milliliters after dissolving it in the elution solvent. The reference solution (b): After putting 0.120 grams of the ginkgo dry extract for peak-identifying CRS in a 25 millilitre beaker and stirring in 10 milliliter of phosphate buffer solution, dissolve it to proceed as instructed for the test solution. R = pH 5.8 |
- |
||||||||||||||||||||||||||||||
|
Column |
The size is l = 0.25 m, and the temperature is 25°C. The stationary phase is octyl silyl gel of silica chromatography R (5 µm). |
|
||||||||||||||||||||||||||||||
|
Elution solvent |
Methanol R, water R, tetrahydrofuran R (20:75:10 V/V/V) |
|
||||||||||||||||||||||||||||||
|
Rate of flow |
1 milliliter per minute. |
|
||||||||||||||||||||||||||||||
|
The detection |
Refractometer kept at 35 degrees Celsius. |
|
||||||||||||||||||||||||||||||
|
The injection |
100 microliters. |
- |
||||||||||||||||||||||||||||||
|
Assay by HPLC-3 |
The test solution |
Use 8 milliliter of methyl alcohol R to dissolve 500 grams of the powder extract for testing (sonicate if required), and then use the same solvent to dilute to 10 milliliters. Centrifuge if necessary. |
- |
|||||||||||||||||||||||||||||
|
Reference solution |
In 8 milliliter of methanol R, dissolve 10 milligrams of ginkgolic acids CRS (sonicate if needed), and then dilute to 10 milliliter using the same solvent. This solution should be diluted with methanol R to yield 10.0 mL from 2.0 mL |
|
||||||||||||||||||||||||||||||
|
Column |
-size: l = o.25 m, -Phase of stationary: octyl silyl Gel of silica for chromatography R (5µm); -temperature: 35°C |
- |
||||||||||||||||||||||||||||||
|
Elution solvent |
Trifluoroacetic acid R (0.1 milliliters) must be diluted with 1000 milliliters of water (R) in elution solvent A and with acetonitrile (R) in elution solvent B.
|
- |
||||||||||||||||||||||||||||||
|
Rate of flow |
1 milliliter per minute |
- |
||||||||||||||||||||||||||||||
|
The detection |
Spectrophotometer at 210 nanometer. |
- |
||||||||||||||||||||||||||||||
|
The injection |
50 microliters |
|
||||||||||||||||||||||||||||||
Table 4 Monograph followed by BP (British Pharmacopoeia)
|
Tests |
Ginkgo Leaf |
Ginkgo Dry Extract |
||||||||||||||||||||||||||||||
|
Definition |
NLT 0.5 percent of flavone glycosides, which are flavonoids (Mr. 757) (drug that is dried) (Commission B. P., 2023). |
Flavonoids are expressed as flavone glycosides, which (Mr 756.7). The following percentages apply to dried extracts: 2.8 % - 3.4% for ginkgolides A, B and C; 2.6 % - 3.2% for bilobalides; -ginkgolic acids: NMT 5 parts per million (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Identification by TLC |
Test solution |
Mix two grams of powdered herbal product (710) with 10 milliliters of methanol R (2.9.12). Place in a bath of water and heat for 10 minutes at 65°C. Shake often. Let it come to room temperature and then filter (Commission B. P., 2023). |
Two milligrams of the extract to be analyzed should dissolve in ten milliliters of a solution that contains eight milliliters of methanol and two quantities of water (Commission B. P., 2023). |
|||||||||||||||||||||||||||||
|
Reference solution |
Mix 20 milliliters of methyl alcohol R with three milligrams of rutoside trihydrate R and one milligram of chlorogenic acid R (Commission B. P., 2023). |
Mix 20 milliliters of methyl alcohol R with three milligrams of rutoside trihydrate R and one milligram of chlorogenic acid R (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Plate |
Silica gel surface R of TLC (Commission B. P., 2023). |
Plate R of silica gel for TLC (5–40 micrometer) or [Plate R of silica gel for TLC (2–10 micrometer)] (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Elution solvent |
7.5 amounts of anhydrous ethanoic acid R, 7.5 amounts of methanoic acid, 17.5 amounts of ethyl ethanoate R and 67.5 amounts of water R in volumes (Commission B. P., 2023). |
7.5 amounts of methanoic acid, 7.5 amounts of anhydrous ethanoic acid R, 17.5 amounts of ethyl acetate R, and 67.5 amounts of water R in volumes (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Application |
20 µL as bands (Commission B. P., 2023). |
20 µL [or 5 µL], as bands (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Development |
Over a path of 17 cm (Commission B. P., 2023). |
Over a path of 17 cm (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Dry |
At 100-105°C (Commission B. P., 2023). |
At 100-105°C (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Detection |
Transfer to the heated plate a 10 grams per liter solution containing diphenyl aminoethyl ester R of boric acid in methyl alcohol R. Utilize the same amount of a macro gel 400 R in methyl alcohol R solution containing 50 g per liter. Examine the plate under 365 nm ultraviolet light after allowing it to dry in air conditioning for roughly 30 minutes (Commission B. P., 2023). |
While the plate is still warm, Add 10 grams per liter of diphenyl boric acid aminoethyl ester R to methyl alcohol R. In methanol R, add a 50-gram per liter macro gel 400 R solution after that. After letting it air-dry in the air for about 30 minutes, look at it with an ultraviolet light source at 365 nm (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Assay by HPLC-1 |
Test solution |
Using a reflux condenser, 2.500 grams of the powdered herbal medication (710) are heated for 30 minutes in 50 milliliters of a 60 % V/V acetone R solution. Gather the filtrate after it has been filtered. The residue was extracted again with 40 milliliter of a 60 percent V/V acetone R solution and a filter. Gather the filtrates and use a 60 percent V/V acetone R solution to dilute them to 100 milliliters. Transfer the mixture to a 50-milliliter vial and rinse with 30 milliliters of methanol R after the acetone has been removed from 50 milliliters of solution by evaporation. Centrifuge after adding 4.4 milliliters of hydrochloric acid, RI, and diluting with water, R, to make a volume of 50mL. Fill a 10-milliliter brow glass vial with 10 milliliters of the supernatant. After covering it with an aluminum metal cap and rubber seal, put it in a water bath to heat it for twenty-five minutes. Once it's room temperature, let it cool. (Commission B. P., 2023). |
To prepare it for analysis, melt 0.2 gram of the extract in 20 milliliter of methyl alcohol R. Combine 15 milliliters of diluted HCL R and 5 milliliters of water R to make 50 milliliters. With methanol R, dilute. Spoon 10 milliliter of this mixture into amber glass vial that holds 10 mL. Use an aluminum-bound cap to fix the rubber membrane stopper that was used to seal the vial securely. Place in a bath of water and heat for 25 minutes. Let it cool down to 20 °C (Commission B. P., 2023). |
|||||||||||||||||||||||||||||
|
Reference solution |
One milligram of quercetin dihydrate R should dissolve in twenty milliliters of methyl alcohol R. Combine 15 milliliters of diluted HCL R and 5 milliliters of water R to make 50 milliliters. With methanol R, dilute (Commission B. P., 2023). |
Combine 10 milligrams of quercetin dihydrate CRS with twenty milliliters of methanol R. 50 milliliters of methanol are added to 15 milliliters of diluted HCL and 5 milliliters of water to dilute the mixture (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Column |
Measurements: l = 0.125 m, ø = 4 mm; temperature: 25 °C; phase of stationary: octadecyl silyl gel of silica for the chromatography R (5 micrometer) (Commission B. P., 2023). |
Measurements: l = 0.125 m, ø = 4 mm; temperature: 25 °C; phase of stationary: octadecyl silyl gel of silica for the chromatography R (5 micrometer) (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Elution solvent |
Elution solvent A: methanol R; Elution solvent B: phosphoric acid R solution (0.3 g/L) adjusting to pH 2.0;
(Commission B. P., 2023). |
-Elution solvent A: methanol R; -Elution solvent B: phosphoric acid R solution (0.3 g/L, pH 2.0);
(Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Flow rate |
1.0 milliliters/min (Commission B. P., 2023). |
1.0 milliliters/min (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Detection |
Spectrophotometer at 370 nm (Commission B. P., 2023). |
Spectrophotometer at 370 nm (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Injection |
10 µL (Commission B. P., 2023). |
10 µL (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Assay by HPLC-2 |
Test solution |
- |
Pour 10 milliliters of pH 5.8 R phosphate buffer solution into a 25-milliliter beaker, then add 0.120 gram of the extract to be tested to dissolve it. Transfer the mixture into an analytical chromatography R column that has a 30 mm inner diameter and a 0.15 m length. The column can hold 15 grams of kieselguhr. The beaker was cleaned using two 5-milliliter volumes of a solution of phosphate buffer with a pH of 5.8 R. After that, the laundering were transferred to the column to be used in liquid chromatography. Give it a fifteen-minute rest. Ethyl acetate R (100 ml) is used to elute. At a pressure of not more than four kPa, it permeates the solution to the point of drying out in a bath of water that is set at 50 °C. An air current extracts the remaining solvent. Collect the leftovers in the mobile phase's 2.5 milliliters (Commission B. P., 2023). |
|||||||||||||||||||||||||||||
|
Reference solution |
- |
Reference solution (a): Dilute 30 milligrams of phenyl methanol CRS to 100 milliliters by dissolving it in the mobile phase (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Column |
- |
Specifications: Temperature: 25°C; l = 0.25 m, ae = 4 mm; phase of stationary: octyl silyl gel of silica for liquid chromatography R (five µm); (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Elution solvent |
- |
20 amounts of methyl alcohol R, 75 amounts of water R and 10 amounts of tetrahydrofuran R in volumes (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Flow rate |
- |
1.0 millimeters/min (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Detection |
- |
Refractometer maintained at 35°C (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Injection |
- |
100 µL (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Assay by HPLC-3 |
Test solution |
- |
0.500 grams of the powdered materials for testing should be dissolved in 8 milliliters of methyl alcohol R (sonicate if needed), the solvent should then be diluted to 10 milliliters. Centrifuge if necessary (Commission B. P., 2023). |
|||||||||||||||||||||||||||||
|
Reference solution |
|
If necessary, sonicate Eight milliliters of methanol R with 10 milligrams of ginkgolic acids CRS to aid in dissolving it. Then, dilute the solution to a total amount of 10 mL using the same solvent. Afterward, take 2 milliliter of the prepared mixture and dilute it further to 10 milliliters with methyl alcohol R. (Commission B. P., 2023) |
||||||||||||||||||||||||||||||
|
Column |
- |
Measurements: l = 0.25 m, ø = 4.6 mm; phase of stationary: liquid chromatography R (5 micrometer) octyl silyl silica gel; The temperature is 35 °C. (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Elution solvent |
- |
Trifluoroacetic acid (0.1 milliliter) diluted with water to prepare 1000 milliliters of the first elution solvent. For the second elution solvent, dilute the same volume of trifluoroacetic acid R with acetonitrile R. (Commission B. P., 2023).
(Commission B. P., 2023) |
||||||||||||||||||||||||||||||
|
Rate of flow |
- |
1 milliliter per minute (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
The detection |
- |
UV-Vis Spectrophotometer at 210 nanometers (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
|
Injection |
- |
50 µL (Commission B. P., 2023). |
||||||||||||||||||||||||||||||
Table 5 Monograph followed by CP (Chinese Pharmacopoeia)
|
TESTS |
Ginkgo Folium |
Ginkgo Semen |
|
|
Identification by TLC |
Test solution |
|
Ten grams of the powder, a tiny quantity Add 40 milliliters of methanol, let it reflux for an hour, strain it, and then dry the filtrate that comes out. Use 15 milliliters of water to dissolve any leftover residue, then pass the mixture through a few cotton swabs to filter it. Utilize the filtrate and then elute with 70 ml water, collect the eluates, and extract by shaking the two 40-ml components of ethyl acetate using a column (10–15 mm) packed with polyamide (80–100 mesh, 3 g). Mix the ethyl acetate solutions, let them evaporate until the residue is completely dry, and then dissolve them in the test solution—one milliliter of methanol (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
Reference solution |
Ginkgo Folium same as test solution (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
Mixture of bilobalide A CRS and bilobalide C CRS in methanol containing 0.5 mg each per ml (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
|
Mobile phase
|
Combination of water, formic acid, butanone, and ethyl acetate (1:1:3:5) (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020) |
Toluene, acetone, ethyl acetate, and methanol combined (10:5:5:0.6) (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
|
Application |
6 µl as bands (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
10 µl as bands (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
|
Development |
- |
- |
|
|
Drying |
At Room temperature (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
At 140-160°C (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
|
Detection |
Ultraviolet light at 365 nm (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
Ultraviolet light at 365 nm (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
|
Assay by HPLC 1 |
Reference solution |
Weigh exactly the same amount of dried quercetin CRS, kaempferol CRS, and isorhamnetin CRS over the phosphorous pentoxide the previous evening. The material need to be mixed with methanol to create a mixture containing 20 µg of isorhamnetin and thirty µg of every one of the two kaempferols and quercetin whenever ml are used as a reference solution (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
- |
|
Test solution |
One gram of the powder should be precisely weighed, added to a Soxhlet extractor along with the chloroform, and allowed to reflux for two hours. Methanol is added, After four hours reflux heating, chloroform solution is discarded, the remaining substances is dried, and the extraction is done. After transferring the residue to a 50- milliliter graduated flask and thoroughly mixing everything, the resulting blend gets heated under the reflux for thirty minutes. Over the residue, add 25 milliliters of a 4:1 methanol to 25% hydrochloric acid mixture, and stir to dissolve (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
- |
|
|
Column |
Use octadecylsilane bonded silica gel as the stationary phase (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
- |
|
|
Mobile phase |
Mixture of methanol and 0.4% phosphoric acid (50:50) (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
- |
|
|
Detection |
Spectrophotometer at 360 nm (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
- |
|
|
Injection |
10µl (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
- |
|
|
Assay by HPLC 2 |
Reference solution |
The amounts of ginkgolide A in the CRS, ginkgolide B in the in the CRS, ginkgolide C in the in the CRS, as well as bilobalide in the in the CRS that have been dried across phosphorous pentoxide require exact measurements. The reference solution is then created by adding 50% methanol to the mixture. The concentration of ginkgolide A, B, and C in this solution is 0.18 milligram, 0.08 milligram, 0.10 milligram, and 0.20 milligram of bilobalide per milliliter. (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
- |
|
Test solution |
After correctly weighing 1.5 grams of a powder and putting 30 to 60°C of petroleum ether, heat a bath of water to 70°C with reflux for an hour. Discard the petroleum ether solution (between 30 and 60°C), dry the residue and the filter paper in a 60°C oven, add methanol, warm the extract under reflux for six hours, and then let it dry. After dissolving the residue in methyl alcohol, precisely move the mixture to a 10-milliliter graduated flask, cool it with ultrasonication for 30 minutes, add methyl alcohol to the volume, stir thoroughly, and let it stand until. Take exact measurements. After 30 minutes of ultrasonically agitation, 4.5 ml of water are added to a 10-milliliter volumetric flask, which is then allowed to cool. Methanol is then added to the flask to the full amount, thoroughly mixed, and filtered. An acidic aluminum oxide column is filled with five milliliters of the supernatant, which is then eluted with twenty-five ml of methanol, collected as the eluate, vaporized until it is completely dry, and the remaining substance dissolves in 5 milliliters of methanol. (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
|
|
Column |
Use octadecylsilane bonded gel of silica as the stationary phase (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
- |
|
|
Elution solvent |
Tetrahydrofuran, methyl alcohol, and water combined in a 25:10:65 ratio was used as the mobile phase (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020) |
- |
|
|
Detection |
Evaporative light scattering detector (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
- |
|
|
Injection |
10 – 20 µl (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
- |
|
Table 6 Physicochemical Test
|
Specification Tests |
IP |
USP |
EUP |
BP |
CP |
|
Foreign organic matter |
NMT 2% ((IPC), 2022). |
- |
NMT 2% of extra foreign matter and 5% of stems (Commission E. P., European Pharmacopoeia, 2023). |
NMT 2% of extra foreign matter and 5% of stems (Commission B. P., 2023). |
NMT 2% (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
ESE |
NLT 11% ((IPC), 2022). |
- |
- |
- |
- |
|
WSE |
NLT 17% ((IPC), 2022). |
- |
- |
- |
NMT 12% (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
TA |
NMT 4% ((IPC), 2022). |
NMT 11% ((USP), 2023). |
NMT 11% (Commission E. P., European Pharmacopoeia, 2023). |
NMT 11% (Commission B. P., 2023). |
NMT 10% (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
AIA |
- |
- |
- |
- |
NMT 2.0% (Commission C. P., Chinese Pharmacopoeia (Zhonghua Renmin Ghongheguo Yaodian), 2020). |
|
Heavy Metals |
The limit test is met with 1 g ((IPC), 2022). |
- |
- |
- |
- |
|
LOD |
For Ginkgo Leaf- NMT 11%. For Ginkgo Dry Extract- NMT 5%. For Ginkgo Tablet- NMT 5% ((IPC), 2022). |
For Ginkgo- NMT 11% For Powder Ginkgo Extract- NMT 5% ((USP), 2023). |
NMT 11% (Commission E. P., European Pharmacopoeia, 2023). |
NMT 11% (Commission B. P., 2023). |
- |
|
Microbial Contamination |
Complies with the microbial contamination tests ((IPC), 2022). |
- |
- |
- |
- |
Table 7 Phytochemical composition of Ginkgo biloba- (Beek, 2002)
|
Compounds category |
% |
|
The glycosides of flavonols |
24 |
|
Trilactones of terpenes |
6 |
|
The proanthocyanidins |
7 |
|
Acids carboxylic |
13 |
|
The catechins |
2 |
|
Non flavanol glycosides |
20 |
|
Inorganic components |
5 |
|
Water, solution |
3 |
|
Compounds with high molecular mass |
4 |
Table 8 Phytoconstituent of Ginkgo biloba [1-29]
|
Compounds Name |
Plant Part |
Class |
Structure |
References |
|
Cymene |
Leaf |
Monoterpenes |
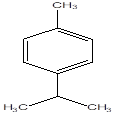
|
[1] |
|
Isopropyl-phenol |
Leaf |
Monoterpenes |
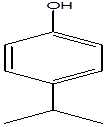
|
[2] |
|
Thymol |
Leaf |
Monoterpenes |
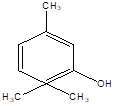
|
[3] |
|
Linalool oxide |
Leaf |
Monoterpenes |
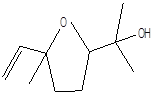
|
[4] |
|
Lonone |
Leaf |
Monoterpenes |
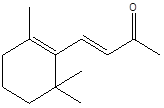
|
[5] |
|
Ginkgolide A |
Leaf |
Diterpenes |
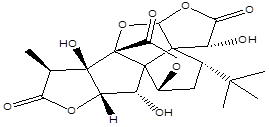
|
[6] |
|
Ginkgolide B |
Leaf |
Diterpenes |
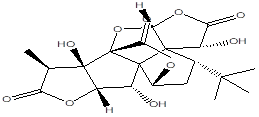
|
[7] |
|
Ginkgolide C |
Leaf |
Diterpenes |
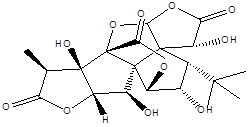
|
[8] |
|
Ginkgolide J |
Leaf |
Diterpenes |
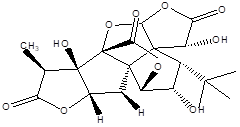
|
[9] |
|
Bilobalide |
Leaf |
Sesquiterpenes |
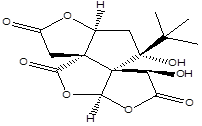
|
[10] |
|
Bilobanone |
Leaf |
Sesquiterpenes |
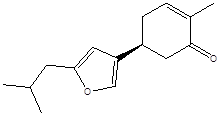
|
[11] |
|
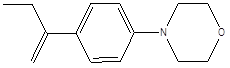
Elemol |
Leaf |
Sesquiterpenes |
|
[12] |
|
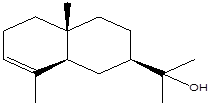
Eudesmol |
Leaf |
Sesquiterpenes |
|
[13] |
|
β -Sitosterol |
Leaf |
Steroids, phytosterols |
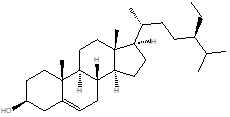
|
[14] |
|
Stigmasterol |
Leaf |
Steroids, phytosterols |

|
[15] |
|
Kaempferol |
Leaf |
Flavonoid Glycoside |
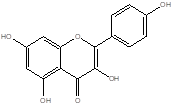
|
[16] |
|
Quercetin |
Leaf |
Flavonoid Glycoside |
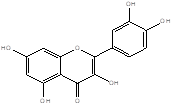
|
[17] |
|
Myricetin |
Leaf |
Flavonoid Glycoside |

|
[18] |
|
Isorhamnetin |
Leaf |
Flavonoid Glycoside |
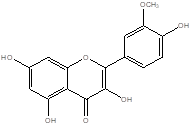
|
[19] |
|
Luteolin |
Leaf |
Flavonoid Glycoside |

|
[20] |
|
Tamarixetin |
Leaf |
Aglycones |
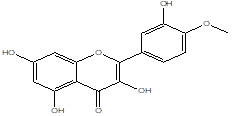
|
[21] |
|
Catechin |
Leaf |
Aglycones |
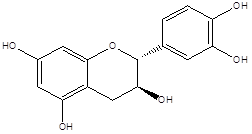
|
[22] |
|
Procyanidin |
Leaf |
Anthocyanidin |
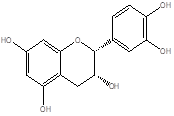
|
[23] |
|
Bilobetin |
Leaf |
Biflavones |

|
[24] |
|
Ginkgetin |
Leaf |
Biflavones |
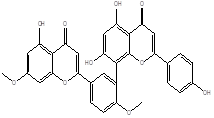
|
[25] |
|
Isoginkgetin |
Leaf |
Biflavones |

|
[26] |
|
Sciadopitysin |
Leaf |
Biflavones |

|
[27] |
|
Ginkgol |
Leaf |
Alkyl phenols Cardanols |

|
[28] |
|
Bilobolol |
Leaf |
Cardols |
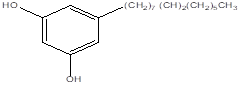
|
[29] |
Table 9 Therapeutic activity of Ginkgo biloba [1-16]
|
Therapeutic effect |
Bioactive compound |
Mechanism of action |
References |
|
Anticancer |
Ginkgol, Bilobalol |
The process through which fresh blood vessels form is inhibited by ginkgolides. Restricting angiogenesis can stop tumor growth because angiogenesis is necessary for the development and spread of tumors. VEGF (vascular endothelial growth factor) and other angiogenic factors are inhibited in order to initiate this process. |
[1] |
|
Anti-Alzheimer |
Ginkgolides, Bilobalide |
Strong antioxidant properties of ginkgolides and bilobalides aid in scavenging free radicals that accumulate in the brain. By doing this, oxidative stress is lessened, which is important in the etiology of neurodegenerative diseases like Alzheimer's. |
[2] |
|
Antidiabetic |
Ginkgolides, Bilobalide |
By modulating signaling pathways and influencing glucose uptake and metabolism, ginkgolides and bilobalides enhance insulin sensitivity. Improved glucose regulation is facilitated by ginkgolides and bilobalides. |
[3] |
|
Antidepressant |
Ginkgolides, Bilobalide |
Platelet-activating factor (PAF) is inhibited by ginkgolide B, helping to prevent oxidative stress, inflammation, and neuronal damage—all of which are linked to depression. Bilobalide improves neuronal survival from excitotoxicity-induced damage, a process that may be involved in depression. These substances have the capacity to alter important neurotransmitter systems, including GABA, dopamine, and serotonin. These substances can lessen the signs of depression by enhancing neural health and lowering brain stress. Nevertheless, additional clinical research is required to completely understand their mechanism and potential for human therapeutics. |
[4] |
|
Antihypertensive |
Ginkgolide B, Quercetin |
A strong antagonist of platelet-activating factor (PAF) is ginkgolide B. PAF is involved in platelet aggregation and vascular inflammation, both of which can lead to the onset of hypertension. Ginkgolide B can lower inflammation and aggregation by blocking PAF, which lowers blood pressure. Because it shields the inner endothelium, quercetin aids in the maintenance of regular vascular function. |
[5] |
|
Antioxidant |
Ginkgolides, Bilobalide, Quercetin, Kaempferol, Isorhamnetin |
Scavenging free radicals and blocking signaling pathways induced by oxidative stress, all of these compounds function as antioxidants. Ginkgolides and bilobalides function by preventing oxidative stress-related inflammatory pathways and safeguarding mitochondria. Strong flavonoid antioxidants include isorhamnetin, kaempferol, and quercetin. When combined, they can lessen inflammation, shield cells against oxidative damage, and promote general cellular growth. |
[6] |
|
Antilipidemic |
Ginkgolide B |
Ginkgobiloba B is a strong PAF antagonist. PAF is involved in both vascular inflammation and lipid metabolism. Ginkgolide B may lower the probability of atherosclerosis and enhance lipid regulation by blocking PAF. Lipid-modulating qualities are present. To completely comprehend the precise molecular mechanism in humans, more research is necessary. |
[7] |
|
Antiplatelet |
Ginkgolide B |
Ginkgobiloba B is a strong PAF antagonist. PAF is involved in both vascular inflammation and lipid metabolism. Ginkgolide B may lower the probability of atherosclerosis and enhance lipid regulation by blocking PAF. Lipid-modulating qualities are present. To completely comprehend the precise molecular mechanism in humans, more research is necessary. |
[8] |
|
Antimicrobial |
Ginkgolides, Bilobalide |
Certain kinds of microorganisms (fungi, bacteria, etc.) may have their cell membranes damaged by ginkgolides and bilobalides. Compounds interact with the lipid bilayer of microbial membranes, increasing the membrane's permeability. This compromises the authenticity of the membrane, leading to essential intracellular components to leak out and, eventually, cell death. |
[9] |
|
Anti-Glaucoma |
Ginkgolides, Bilobalide, Quercetin |
The potential antiglaucoma effects of compounds found in ginkgolides, bilobalides, and quercetin have been studied. A strong inhibitor of the platelet-activating factor (PAF) is ginkgolide B. Patients who suffer from normal-tension glaucoma gain from ginkgolide B's reduction of inflammation and improved blood flow to the optic nerve caused by blocking PAF. Quercetin increases the evacuation of aqueous humor, which may help lower IOP. |
[10] |
|
Anti-inflammatory |
Ginkgetin, Isoginkgetin, Bilobetin |
These substances work against inflammation by blocking NF-kB signaling, reducing the production of cytokines that promote inflammation and the reduction of inflammatory-processing enzymes like COX-2 and iNOS. They also help to lessen inflammation because of their antioxidant qualities. |
[11] |
|
Cardioprotective |
Ginkgolide B, Quercetin, Kaempferol, Isorhamnetin |
By decreasing oxidative stress, lowering inflammation, maintaining mitochondrial function, improving the functioning of endothelial cells, and stopping thrombosis or atherosclerosis—all of which are crucial in preventing heart disease—these substances aid in cardio protection. |
[12] |
|
Neuroprotective |
Ginkgolide B, Bilobalide, Quercetin, Kaempferol |
Ginkgolide B reduces inflammatory processes and neuronal damage brought on by excessive PAF signaling by acting as an inhibitor of the PAF receptor. Bilobalide's neuroprotective and anti-excitotoxic impacts are partly attributed to its enhancement of GABAergic transmission. Brain-derived neurotrophic factor, or BDNF, and other growth factors are upregulated by quercetin, promoting the survival and regeneration of neurons. Kaempferol improves cognitive performance and neuronal survival by encouraging neurogenesis and synaptic plasticity. |
[13] |
|
Immunomodulatory |
Ginkgolide B, Bilobalide |
Bilobalide acts via antioxidant mechanisms, defense against mitochondrial damage, and the modulation of microglia as well as cytokine action. Ginkgolide B functions by inhibiting the PAF receptor and reducing the release of pro-inflammatory cytokines. Both substances help to lessen overreactions from the immune system, which may have therapeutic advantages for autoimmune and inflammatory diseases. |
[14] |
|
Dietary supplement |
Quercetin, Kaempferol, Isorhamnetin, Bilobalide |
When taken as dietary supplements, these substances work in concert to offer a variety of health advantages. Strong antioxidant activity, decreased inflammation, neuroprotection, and possible cancer-prevention effects are some of their mechanisms. |
[15] |
|
Wound healing |
Ginkgolides, Bilobalide |
These substances promote angiogenesis, collagen synthesis, and cell division, which aid in the development of tissue granulation and re-epithelialization. By regulating inflammation, lowering oxidative stress, boosting fibroblast activity, and boosting cellular migration and proliferation all of which are essential for tissue repair and regeneration ginkgolides and bilobalide facilitate wound healing. |
[16] |
DISCUSSION
Ginkgo biloba, one of the oldest surviving tree species, holds a significant position in traditional and modern medicinal systems. The extensive research cited in this study demonstrates the plant's pharmacological adaptability, which includes its cardiovascular, anti-inflammatory, anti-cancer, and neuroprotective properties. It has been demonstrated that standardized Ginkgo biloba extracts (GbE), in particular EGb761, are effective in treating diseases like mood disorders, dementia, and Alzheimer's disease. Flavonoids, terpenoids, and ginkgolides are among the distinctive phytochemicals that are responsible for these qualities. G. biloba is well recognized as a valuable phytopharmaceutical agent, as evidenced by its regular inclusion in international pharmacopoeias such as IP, USP, BP, and CP. Although ginkgo is widely used for therapeutic purposes, including cognitive enhancement, its popularity as a herbal supplement contributes to its commercial success. Ginkgo-based products are guaranteed to be safe and of high quality thanks to analytical methods like TLC and HPLC that are standardized in different pharmacopeias. The monographs that are reviewed highlight the rigorous quality control procedures that are in place for its extracts, leaves, and tablets. These procedures include preparation techniques and assay methodologies. But even with its wide range of therapeutic uses, there are some major concerns about possible side effects, including bleeding risks, allergic responses, and interactions with anticoagulants. Therefore, even if ginkgo biloba has a lot of promise, its usage needs to be governed by clinical research and legal requirements in order to mitigate risks.
CONCLUSION
The comprehensive review of Ginkgo biloba underscores its status as one of the oldest and most therapeutically significant plants in traditional and modern medicine. Pharmacopoeias like IP, USP, BP, CP, and others have standardized analytical techniques, such as TLC and HPLC, which guarantee consistency and quality in goods that originate from G. biloba. Its phytochemical profile is validated by these techniques, especially that of flavonoids and terpenoids, which are important active ingredients that give it its medicinal properties. The assay, detection, and identification standards offer a strong foundation for its quality control. Ginkgo biloba is a remarkable example of how current pharmacological research and traditional medical knowledge may coexist. Its importance as a worldwide medicinal agent is highlighted by its inclusion in several pharmacopeias and its proven effectiveness in treating neurological and cardiovascular conditions. This review consolidates essential information, paving the way for future research to address existing knowledge gaps and enhance the clinical utility of Ginkgo biloba. Top of FormBottom of Form
ACKNOWLEDGMENT
I would like to express my gratitude to Dr. Meenakshi Kanwar Chauhan, Department of Pharmaceutics, Delhi Institute of Pharmaceutical Science and Research, Mehrauli-Badarpur Rd, Sector 3, New Delhi, Delhi 110025, for her unwavering support and guidance throughout the development of this review article. Her insightful suggestions and expert advice have been instrumental in shaping this work and deepening my understanding of the subject. I am sincerely thankful for her mentorship. Thank you for your continuous support and guidance.
REFERENCES




Uvaish Aalam, Ritu Tiwari, Meenakshi Kanwar Chauhan, Meenakshi Dahiya, Ginkgo biloba Monograph: A Comprehensive Review of its Representation Across Pharmacopoeias, Physicochemical Properties, Identification, and Therapeutic Applications, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 7, 3285-3319. https://doi.org/10.5281/zenodo.16411195
 10.5281/zenodo.16411195
10.5281/zenodo.16411195