
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmacology, Al-Ameen College of Pharmacy, opp. Lalbagh Main Gate, Bengaluru, Karnataka, 560027.
Hibiscus rosa-sinensis Linn., a globally distributed medicinal plant of the Malvaceae family, has gained prominence in ethnopharmacology and modern phytotherapy due to its extensive therapeutic applications and diverse phytochemical profile. This critical review synthesizes multidisciplinary research on the pharmacognostic characteristics, traditional usage, phytoconstituents, and pharmacological activities of H. rosa-sinensis, with a particular focus on flowers, leaves, and roots. The plant demonstrates significant antioxidant, antimicrobial, anti-inflammatory, antidiabetic, anticancer, cardioprotective, and antifertility properties, largely attributed to flavonoids, anthocyanins, phenolic acids, and triterpenoids. Additionally, emerging data reveal its neuropharmacological and antiviral potentials, including in silico inhibition of SARS-CoV-2 protease. The review highlights standardized extraction methods, mechanistic insights, and safety profiles, while identifying gaps in clinical validation and genotoxic assessments. Collectively, H. rosa-sinensis presents a promising lead for phytopharmaceutical development, warranting further translational and clinical investigations.
Hibiscus rosa-sinensis Linn., an evergreen shrub of the Malvaceae family, is taxonomically classified under the order Malvales, class Magnoliopsida, phylum Spermatophyta, and kingdom Plantae. Among over 160 species within the genus Hibiscus, H. rosa-sinensis is notable for its global distribution and pharmacological relevance [1,2,4].. Widely cultivated for ornamental and medicinal purposes, it holds a significant role in ethnomedicine and phytotherapy. The plant is known by various regional name “China Rose” and “Shoe Flower” in English; “Gurhal” (Hindi), “Japa” (Sanskrit), “Semparuthi” (Tamil), “Chembarathi” (Malayalam), and “Wadamal” (Sinhalese), among others—reflecting its widespread cultural integration[1–3]. Used extensively in traditional systems such as Ayurveda, Unani, and Siddha, its flowers and leaves are valued for emollient, anti-inflammatory, and antifertility effects. It is traditionally employed to treat cough, dysentery, epilepsy, venereal diseases, and menstrual disorders, as well as to promote hair growth, regulate menstruation, and act as a general tonic[1–3] . Native to China and tropical Asia, the plant is now naturalized across tropical and subtropical regions, especially in India, Pakistan, the Philippines, Malaysia, Iraq, the Caribbean, and the Pacific. It thrives in well-drained sandy or loamy soils with full sun exposure and is typically propagated by stem cuttings or grafting[1–4] .
2. Botanical Description
2.1 Macro Morphology
Hibiscus rosa-sinensis L. (Malvaceae) is a tropical, woody evergreen shrub cultivated for its ornamental and medicinal properties. Its simple, ovate to elliptical leaves possess dentate-serrate margins, acute apices, and palmate venation with basal actinodromous and semi-craspedodromous patterns. Leaf polymorphism is common among hybrids[5]. The plant features large, bisexual, actinomorphic flowers with five petals, a prominent central staminal column, and multiple epicalyx lobes. Crested peach variants show homeotic modifications in staminal and petaloid structures[7]. Stems are semi-terete, pubescent, and vascularized in an open arc; fibrous roots support propagation through vegetative means[5,6].
2.2 Microscopic Characteristics
Leaves exhibit a dorsiventral structure with a single-layered upper epidermis, well-defined palisade and spongy parenchyma, and amphistomatic surfaces dominated by anisocytic stomata, with paracytic and brachyparacytic types occasionally observed[5]. Trichomes include eglandular (conical, stellate, bifurcate) and glandular (capitate, sessile, stalked) form. Calcium oxalate druses and rosettes are frequent in the lamina and petiole. The midrib presents a biconvex shape with a clear vascular arc[5,6].
2.3 Chemotaxonomic Significance
GC–MS analysis identifies quercetin glycosides, cyanidin diglucoside, gentisic acid, malvalic acid, and fatty acids (margaric, lauric) as key chemotaxonomic markers. These compounds, along with morphological traits, help distinguish H. rosa-sinensis from related species, as confirmed by cluster analysis[5,6].
2.4 Powder Microscopy
Microscopic evaluation of powdered flowers reveals yellow, spiny pollen; stellate and hooked eglandular trichomes; oval to cordate glandular trichomes; and abundant druses, rosettes, and starch grains. The absence of oil globules and stone cells assists in differentiating it from adulterants such as Rhododendron arboreum[6].
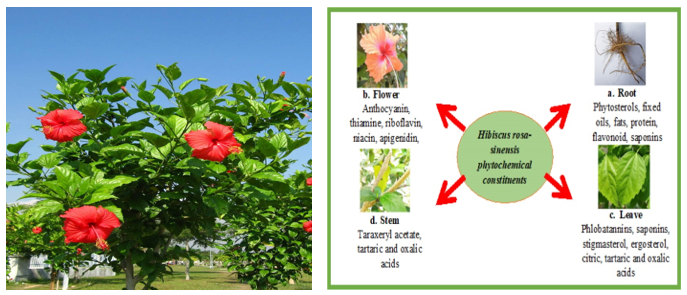
Fig- 1: Hibiscus Rosa-Sinensis Linn
Table- 1: Botanical and Taxonomical Classification of Hibiscus rosa-sinensis Linn
|
Kingdom |
Plantae |
|
Subkingdom |
Tracheobionta (Vascular plants) |
|
Division |
Magnoliophyta (Flowering plants) |
|
Class |
Magnoliopsida (Dicotyledons) |
|
Order |
Malvales |
|
Family |
Malvaceae (Mallow family) |
|
Genus |
Hibiscus |
|
Species |
Hibiscus rosa-sinensis Linn. |
3. History and Ethnobotanical Context
3.1 Use in Ayurveda, Chinese Medicine, and Other Traditional Systems
Hibiscus rosa-sinensis, known as “Japa Pushpa” in Ayurveda and “Gurhal” in Unani, has been traditionally employed across multiple medical systems. In Ayurveda, it is used to manage hair loss, premature graying, skin inflammation, and bleeding disorders, attributed to its cooling and emollient properties that pacify Pitta dosha[8]. Unani medicine describes it as “cold and dry,” utilized as a refrigerant, laxative, and aphrodisiac in treating gastrointestinal, cardiovascular, and reproductive ailments[8]. Traditional Chinese Medicine applies it to clear heat and toxins, addressing sore throat, dysentery, and hypertension[12].
3.2 Regional Folk Uses
In Indian ethnomedicine, the plant is applied for joint pain, cough, fever, and menstrual irregularities. For instance, in Karnataka, leaf paste is used to alleviate sprains and muscular discomfort [9]. In African traditions, hibiscus oil aids in treating sunburn and wounds, while in Caribbean cultures, hibiscus tea is consumed to reduce body heat and promote cardiovascular function [12].
3.3 Parts Used
Various plant parts—flowers, leaves, and roots—are used therapeutically. Flowers are primarily applied in hair care, as emollients, and for managing hypertension?10,11?. Leaves exhibit antimicrobial and anti-inflammatory properties and are often used in poultices and decoctions[10,12]. Roots serve as expectorants and digestive aids, with both flowers and leaves incorporated in Unani formulations for circulatory and reproductive health[8].
3.4 Preparation Methods
Preparation techniques vary by tradition:
4. Phytochemical Composition
4.1 Major Classes of Compounds
Hibiscus rosa-sinensis is rich in secondary metabolites such as flavonoids, anthocyanins, alkaloids, tannins, saponins, terpenoids, phenols, and quinones, which are associated with its antioxidant, antimicrobial, and anti-inflammatory effects[13]. Petals are particularly abundant in flavonoids and anthocyanins, while phenols and quinones are concentrated in the flowers. Steroids and cardiac glycosides are occasionally detected in leaves and bark[15].
4.2 Key Bioactive Compounds
Notable constituents include flavonoids (quercetin, kaempferol, myricetin, luteolin-7-glucoside, apigenin-hexuronide, catechin, epicatechin) and anthocyanins (cyanidin-3-glucoside, cyanidin-sambubioside, cyanidin-sophoroside), primarily in glycosidic forms Additional potent antioxidants such as hibiscetin-3-glucoside, quercetin-trihexoside, taxifolin-dihexoside, and quercetin-sophoroside have also been identified . Flavonoid profiles vary with flower color, with red cultivars rich in anthocyanins and white/yellow types higher in flavonols[14].
4.3 Extraction Methods and Solvents
Extraction techniques depend on target compound polarity and desired yield. Ethanol-based Soxhlet extraction (60?°C, 48?h) is effective for isolating flavonoids[16]. while aqueous maceration is commonly used for bioactivity screening[15]. Hydroethanolic extraction coupled with UHPLC-ESI?-Orbitrap-MS has enabled identification of 20 flavonoids across 16 cultivars[14]. Modern methods such as ultrasound-assisted, microwave-assisted, and supercritical fluid extraction improve efficiency and reduce solvent use . Ethanol, methanol, and water are commonly used solvents; acidified solvents enhance anthocyanin recovery, and 50–80% aqueous ethanol is optimal for flavonoid extraction 17].
5. Pharmacological Activities:
5.1 Antioxidant Activity of Hibiscus rosa-sinensis
Hibiscus rosa-sinensis demonstrates strong antioxidant activity, primarily attributed to its flavonoids, phenolics, and proanthocyanidins, especially in flowers and leaves. Methanolic and ethanolic flower extracts exhibited high total phenolic (61.45?±?3.23 and 59.31?±?4.31?mg/100?g GAE) and flavonoid contents (53.28?±?1.93 and 32.25?±?1.21?mg/100?g CE), with DPPH scavenging activities of 75.46% and 64.98%, respectively[18].Garg et al. reported superior antioxidant efficacy of methanolic stem and leaf extracts using DPPH, NO, H?O? scavenging, and FRAP assays, correlating with higher phenolic content[19]. Verma et al. observed potent DPPH radical inhibition with an IC?? of 19.54?μg/mL in flower extracts, confirming strong free radical neutralization[20]. Sheth et al. compared four cultivars, noting the red variant had the highest antioxidant effect (55.11% DPPH inhibition at 100?μg/mL), closely approximating ascorbic acid (56.38% at 5?μg/mL). Differences were linked to phenolic and flavonoid variability among cultivars[21]. Overall, H. rosa-sinensis possesses robust antioxidant potential, modulated by plant part, cultivar, and extraction method, supporting its traditional applications and potential in oxidative stress-related disorders.
5.2 Antimicrobial Activity of Hibiscus rosa-sinensis
The antimicrobial properties of Hibiscus rosa-sinensis have been well-documented, supporting its traditional use in treating infections. Various studies have highlighted its efficacy against both Gram-positive and Gram-negative bacteria, as well as fungi, across different extracts. A methanolic extract combining H. rosa-sinensis flowers, Aloe vera, and Coleus blumei exhibited significant antibacterial activity, with the highest inhibition observed against Klebsiella pneumoniae (22.33 mm) and Escherichia coli (16 mm) at 500 mg/mL. Additionally, it showed antifungal activity against Candida albicans (13.66 mm)[22]. In a bioprospecting study, both aqueous and ethanol extracts of H. rosa-sinensis leaves displayed antimicrobial activity against various soil microbes, including Staphylococcus aureus, Bacillus subtilis, Pseudomonas, and fungal species like Aspergillus niger. Ethanol extracts were particularly effective due to better solubility of active compounds, such as flavonoids and tannins[23]. A study on the ethanol and ethyl acetate fractions of H. rosa-sinensis flowers demonstrated strong bacteriostatic activity against clinical and antibiotic-resistant strains of Helicobacter pylori. The ethyl acetate fraction showed MICs of 0.2–0.25 mg/mL and MBCs of 1.25–1.5 mg/mL, inhibiting biofilm formation and reducing urease activity without cytotoxicity to human cells[24]. Recent research on methanolic flower extracts reported large zones of inhibition against Staphylococcus aureus (36 mm), Micrococcus luteus (45 mm), E. coli (45 mm), and K. pneumoniae (40 mm), with MICs ranging from 0.5–1 mg/mL. The extract also significantly inhibited biofilm formation (up to 47.6% for M. luteus) and caused notable morphological changes in bacterial cells[25]. The antimicrobial effects of H. rosa-sinensis are attributed to its diverse phytochemical composition, including flavonoids, tannins, phenolic acids, anthocyanins, and volatile compounds. These compounds likely disrupt microbial membranes, inhibit enzymes, and interfere with quorum sensing, highlighting the plant's potential for natural antimicrobial drug development [23,25].
5.3 Anti-inflammatory Activity of Hibiscus rosa-sinensis
Hibiscus rosa-sinensis demonstrates notable anti-inflammatory effects, attributed to flavonoids, anthocyanins, and polyphenols. In vitro studies showed that ethanolic leaf extract protected red blood cells from hypotonic-induced hemolysis (91.09% protection) and protein denaturation (89.45% inhibition), comparable to diclofenac sodium[26]. The ethyl acetate flower extract also inhibited RBC lysis by 90% at 100 µg/mL[27]. In vivo studies confirmed the plant’s anti-inflammatory action in carrageenan-induced paw edema and formaldehyde-induced arthritis models, with doses of 250–1000 mg/kg reducing inflammation and normalizing biomarkers[28]. Additionally, the extract reduced granuloma mass and edema in xylene-induced ear and cotton pellet models[29]. The mechanisms involve COX and LOX pathway inhibition, reduced histamine and prostaglandin synthesis, and suppression of neutrophil migration, with flavonoids like quercetin and cyanidin playing key roles[27,28]. These findings support H. rosa-sinensis as a candidate for further investigation in anti-inflammatory drug development.
5.4 Anticancer Activity of Hibiscus rosa-sinensis
Hibiscus rosa-sinensis has demonstrated significant anticancer potential through cytotoxic, antioxidant, and pro-apoptotic effects in various human cancer cell lines. The plant’s pharmacological efficacy is attributed to its rich phytochemical profile, including flavonoids, phenolics, triterpenoids, and saponins, which influence redox balance, apoptosis, and mitochondrial integrity. Arullappan et al. reported that methanolic leaf extract exhibited the highest cytotoxicity against K-562 chronic myeloid leukemia cells (IC?? = 30.9 ± 1.1 µg/mL), with minimal toxicity to normal cells[30]. Nguyen et al. found that aqueous flower extract induced mitochondrial depolarization and elevated ROS levels in both estrogen receptor-positive (MCF-7) and triple-negative (MDA-MB-231) breast cancer cells, enhancing the effects of chemotherapeutic agents[34]. Harini et al. demonstrated that ethanolic and aqueous extracts showed dose-dependent anticancer activity, with ethanolic extracts showing superior inhibition of cell proliferation[33]. Alam et al. identified triterpenoids such as ursolic acid and β-sitosterol in H. rosa-sinensis, which exhibited cytotoxicity against HepG2 and MCF-7 cells[32]. Mandade et al. linked the plant’s antioxidant capacity to its anticancer potential, as it inhibited free radicals and lipid peroxidation[31]. Overall, H. rosa-sinensis exhibits anticancer activity via multiple mechanisms, including ROS-mediated apoptosis, mitochondrial dysfunction, and synergistic effects with chemotherapeutic agents. Its bioactive compounds offer promise for further development in cancer therapy.
5.5 Antifertility Activity of Hibiscus rosa-sinensis
The antifertility potential of Hibiscus rosa-sinensis has been extensively studied in male and female animal models. Various plant parts, such as roots, flowers, and leaves, have demonstrated reversible effects on reproductive functions, including spermatogenesis, sperm motility, hormonal regulation, and implantation.
Male Reproductive Effects
Gupta and Yadav evaluated the effects of aqueous, ethanol, and benzene leaf extracts of H. rosa-sinensis in male albino mice over 35 days. The benzene extract notably reduced testis weight, induced severe histopathological changes in seminiferous tubules, and decreased sperm count, motility, and viability, without affecting libido. Although fertility rates dropped significantly, no systemic toxicity was observed, as liver and kidney histology and haematological indices remained unchanged, indicating selective action on reproductive organs [35]. Carolin et al. investigated the impact of methanolic flower extract on male rat reproductive tissues over 30 days. At higher doses (400 and 500 mg/kg), the extract caused significant reductions in the weight of the epididymis, prostate, and seminal vesicles, alongside thinning of the cauda epididymal epithelium. These changes suggest that hibiscus flower extract exerts antifertility effects via androgen-dependent organ atrophy and disruption of sperm maturation [36]. Al-Saily et al. compared the antifertility effects of a phenolic extract of H. rosa-sinensis flowers with cyproterone acetate (CPA) in male albino rats. Hibiscus extract (300 mg/kg for 60 days) significantly reduced sperm concentration, motility, and viability, while increasing morphological abnormalities. Although less potent than CPA, it delayed successful mating, with pregnancies occurring five weeks after treatment cessation, suggesting a prolonged antifertility effect. Additionally, the extract altered gonadotropin (FSH, LH) and testosterone levels, indicating endocrine disruption as a potential mechanism [37]
Female Reproductive Effects
Vasudeva and Sharma studied the ethanolic root extract of H. rosa-sinensis for post-coital antifertility and estrogenic activity in female rats. Oral administration of 400 mg/kg extract resulted in 100% anti-implantation activity without toxicity. The extract also exhibited uterotropic effects, increasing uterine weight and endometrial thickness, comparable to ethinyl estradiol, highlighting its estrogenic and anti-nidatory actions [38]. These studies collectively demonstrate that H. rosa-sinensis possesses multi-mechanistic antifertility effects through both anti-spermatogenic and anti-implantation pathways. Its reversible, non-toxic profile positions it as a promising candidate for plant-based contraceptive development for both sexes.
5.6 Antidiabetic Activity of Hibiscus rosa-sinensis
Hibiscus rosa-sinensis (HRS) exhibits significant antidiabetic effects through mechanisms including pancreatic β-cell regeneration, blood glucose reduction, lipid profile improvement, and anti-inflammatory actions. Both aqueous and ethanolic extracts from its flowers and leaves have shown hypoglycemic potential in animal models.
Pancreatic β-Cell Regeneration
Chauhan and Rani reported that the ethanolic flower extract (125 mg/kg) in STZ-induced diabetic rats reduced fasting glucose and restored body weight, with histopathological evidence of pancreatic islet regeneration, indicating β-cell protection [42].
Hepatoprotective and Nephroprotective Effects
Zaki et al. found that the aqueous methanolic leaf extract (400 mg/kg) reduced glucose, cholesterol, liver enzymes, and improved antioxidant levels, offering hepatoprotective and nephroprotective effects by mitigating organ damage. Key phytochemicals, orientin and verbascoside, contributed to these benefits [44].
Effects in Diabetic Pregnancies
Afiune et al. demonstrated that the flower extract improved maternal glycemia and fetal weight in STZ-induced diabetic pregnant rats, but noted potential reproductive risks in non-diabetic animals, suggesting caution in its use during pregnancy [40].
5.7 Anti-Inflammatory and Hypoglycaemic Effects
Oluwamodupe et al. reported that the aqueous flower extract downregulated inflammatory cytokines (TNF-α, IL-1β, IL-6) and reduced fasting glucose, indicating immunomodulatory and hypoglycemic actions via the JAK/STAT pathway [39].
α-Amylase Inhibition
Harini et al. confirmed that H. rosa-sinensis extract inhibited α-amylase activity in vitro, with effects comparable to metformin, supported by flavonoids, tannins, and saponins [41].
Red Hibiscus Flower Tea
Sanadheera et al. reviewed red H. rosa-sinensis flower tea, suggesting it may offer antihyperglycemic benefits, though further human studies are needed for confirmation [43]. H. rosa-sinensis shows promise as a complementary antidiabetic agent, with antioxidant, anti-inflammatory, and β-cell restorative properties. Further clinical validation is required.
5.8 Cardioprotective Activity of Hibiscus rosa-sinensis
Hibiscus rosa-sinensis demonstrates cardioprotective effects, particularly against oxidative damage in myocardial ischemia-reperfusion injury, primarily through enhancing antioxidant defenses and modulating lipid peroxidation.
Antioxidant Effects in Myocardial Infarction
Gauthaman et al. studied the effect of H. rosa-sinensis flower powder in isoproterenol-induced myocardial infarction in rats. Chronic administration (125, 250, and 500 mg/kg for 4 weeks) significantly increased antioxidant enzymes (SOD, catalase, GSH) and reduced lipid peroxidation markers (TBARS), with the 250 mg/kg dose offering the most protection, preserving myocardial structure and mitigating reperfusion damage [45,46].
Angiogenic Effects
Sharma et al. observed that H. rosa-sinensis flower extract induced dose-dependent neovascularization in the chick chorioallantoic membrane model, suggesting its potential in promoting vascular regeneration for myocardial recovery [47]. H. rosa-sinensis provides cardioprotection through antioxidant enhancement and angiogenesis, supporting its potential as a therapeutic agent in ischemic heart conditions.
6. Mechanisms of Action
6.1 Antimicrobial and Neurological Mechanisms
Hibiscus rosa-sinensis demonstrates antibacterial activity through urease inhibition, biofilm disruption, and morphological interference in Helicobacter pylori, impeding its colonization and viability[48]. Methanolic root extracts exhibit CNS depressant, anxiolytic, analgesic, and antipsychotic effects, likely via modulation of GABAergic, serotonergic, and dopaminergic systems[50].
6.2 Pharmacological Pathways
The plant enhances GABAergic signalling, as shown by prolonged pentobarbital-induced sleep and reduced locomotion . Serotonin pathway involvement is indicated by suppression of lithium-induced head twitches, while dopaminergic antagonism is evidenced by increased catalepsy with haloperidol[50]. Phenolic compounds such as naringenin, luteolin, and myricetin contribute to anti microbial effects by binding and inhibiting urease[48].
6.3 Molecular Targets
In silico analyses identified cyanidin-3-sophoroside-5-glucoside, rutin, and hibiscetin-3-glucoside as potent inhibitors of SARS-CoV-2 3CLpro, binding key residues (His-41, Cys-145) within the catalytic site[49]. Urease enzymes serve as primary bacterial targets, supporting non-antibiotic antimicrobial strategies[48].
6.4 Dose-Response Relationships
Methanolic root extract administered intraperitoneally (50–200?mg/kg) elicited dose-dependent CNS effects, including sedation, anxiolysis, and antipsychotic behaviors in animal models[50]. Ethyl acetate fractions demonstrated minimum inhibitory concentrations (MICs) of 0.2–0.25?mg/mL and minimum bactericidal concentrations (MBCs) of 1.25–1.5?mg/mL against H. pylori, achieving bactericidal activity within 36 hours at 1.5?mg/mL[48].
7. Toxicology and Safety Profile
7.1 Acute and Chronic Toxicity
Hibiscus rosa-sinensis demonstrates a wide safety margin in acute toxicity studies. Methanolic extracts of leaves and stem bark caused no mortality in rodents at oral doses up to 2000 mg/kg and 5000 mg/kg, respectively, with LD?? values beyond these limits[52,53]. Similarly, ethanolic leaf extracts administered at 2000 mg/kg over 14 days showed no toxic effect[51]Sub-acute evaluations at 400 mg/kg (methanolic extract) revealed no adverse haematological or organ-specific changes over 14 days. However, hepatotoxicity and nephrotoxicity were observed at 800 mg/kg, indicated by elevated AST, ALT, bilirubin, and renal function markers, alongside histopathological damage [53]. Repeated dosing of ethanolic extract at 200 mg/kg remained non-toxic.
7.2 Genotoxicity and Mutagenicity
Current literature lacks comprehensive genotoxicity or mutagenicity evaluations, such as Ames or micronucleus assays, underscoring the need for further investigation in this area.
7.3 Reported Side Effects and Contraindications
Extracts up to 400 mg/kg are generally well tolerated without observable side effects[51] However, doses at or above 800 mg/kg pose risks of hepatic and renal toxicity. While no formal contraindications are reported, the plant’s antifertility effects suggest caution in individuals of reproductive age.
8. Future Perspectives
8.1 Potential for Drug Discovery
The diverse phytoconstituents of Hibiscus rosa-sinensis, particularly flavonoids, anthocyanins, and triterpenoids, present promising leads for drug development. Computational studies have highlighted compounds like cyanidin-3-sophoroside-5-glucoside and rutin as potent SARS-CoV-2 3CLpro inhibitors, exhibiting superior binding affinity compared to remdesivir [55]. Its known antioxidant, anti-inflammatory, and lipid-lowering activities further position it as a candidate for managing metabolic and infectious disorders [54,56].
8.2 Areas Requiring Further Investigation
Despite extensive preclinical data, clinical validation remains limited. There is a need for standardized extract formulations and comprehensive toxicological assessments, including chronic, reproductive, and genotoxicity studies. Mechanistic exploration beyond known targets such as ACE inhibition is also warranted to better define therapeutic pathways [54,56].
8.3 Suggestions for Clinical Trials
Preliminary evidence suggests that daily intake of 2?g flower powder improves glycemic and lipid parameters in type 2 diabetes, indicating both efficacy and safety [54]. Future clinical studies should adopt standardized dosing, define metabolic and inflammatory endpoints, and incorporate safety assessments (hepatic, renal) to support translational application [55-57].
CONCLUSION
Hibiscus rosa-sinensis exhibits a broad spectrum of pharmacological activities, including antioxidant, antimicrobial, anti-inflammatory, antidiabetic, antifertility, cardioprotective, neuroprotective, and anticancer effects. These bioactivities are largely attributed to its rich phytochemical profile, encompassing flavonoids, anthocyanins, phenolic acids, and triterpenoids. Preclinical studies have substantiated its therapeutic potential through multiple mechanisms—ranging from modulation of oxidative stress and inflammatory pathways to hormone regulation and microbial inhibition. Despite promising results, clinical validation remains limited. Standardized extract development, mechanistic elucidation, and well-designed clinical trials are essential to translate this ethnomedicinal plant into evidence-based phytotherapeutics. Its safety profile appears favorable within certain dosage ranges, though long-term and reproductive toxicity require further investigation. Collectively, H. rosa-sinensis stands as a promising candidate for integrated phytomedicine and future drug discovery.
REFERENCES


Mohd Ashraf, Dr. Rupesh Kumar M.*, Hibiscus Rosa-Sinensis in The Spotlight: A Critical Review on The Bioactivity and Drug Development Potential of Hibiscus Rosa-Sinensis, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 6, 4431-4442. https://doi.org/10.5281/zenodo.15745324
 10.5281/zenodo.15745324
10.5281/zenodo.15745324