
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmaceutics, Poona District Education Association’s Seth Govind Raghunath Sable College of Pharmacy, Saswad, Pune, Maharashtra, India
Autism Spectrum Disorder (ASD) is a complex neurodevelopmental condition characterized by persistent impairments in social communication, restricted behaviours, and sensory abnormalities. Despite extensive research, current therapeutic strategies remain largely symptomatic and fail to address the underlying neurobiological and immunological mechanisms involved in ASD pathogenesis. Emerging evidence highlights the critical role of synaptic dysfunction, excitatory–inhibitory imbalance, and chronic neuroimmune dysregulation in the development and progression of ASD. Recent advances in molecular medicine have introduced messenger RNA (mRNA)–based therapeutics as a promising tool for transient and targeted modulation of gene expression without permanent genomic alteration. In parallel, increasing attention has been directed toward regulatory T cells (T-regs), which play a crucial role in maintaining immune tolerance and regulating neuroinflammation. Independent studies suggest that both mRNA modulation and T-reg–mediated immune regulation may influence key pathological processes relevant to ASD.This review critically examines the existing literature on ASD pathophysiology, current therapeutic limitations, and the emerging potential of mRNA-based and T-regulatory approaches. Furthermore, it introduces a novel, integrated theoretical framework combining mRNA-mediated synaptic restoration with T-reg–driven immune tolerance. Although this dual approach has not yet been experimentally evaluated in ASD, it represents a forward-looking strategy that may guide future translational research aimed at developing disease-modifying therapies for ASD.
Autism Spectrum Disorder (ASD) is a heterogeneous neurodevelopmental condition that begins early in childhood and persists across the lifespan. It is clinically characterised by impairments in social communication, restricted and repetitive patterns of behaviour, and atypical sensory processing. In recent years, the reported prevalence of ASD has increased worldwide. This rise is largely attributed to improved diagnostic frameworks, greater clinical awareness and broader surveillance strategies, rather than to a true increase in disease occurrence. Owing to its long-term functional impact on affected individuals and their families, ASD represents a major public health challenge. [1,2] The aetiology of ASD is highly complex and reflects the interaction of genetic vulnerability, epigenetic regulation, environmental influences and immune-related mechanisms. Large-scale genomic investigations have identified a substantial number of susceptibility genes that regulate synapse formation, neuronal signalling pathways and chromatin organisation. These findings support the current view that ASD primarily represents a disorder of synaptic development and neural circuit organisation rather than a single-gene disease. In parallel, prenatal and perinatal factors, particularly maternal immune activation and inflammatory exposures, have been increasingly linked with altered neurodevelopmental trajectories in children later diagnosed with ASD. [3–5] At the biological level, accumulating evidence indicates that ASD is associated with disturbed synaptic plasticity, imbalance between excitatory and inhibitory neurotransmission, abnormal patterns of neural connectivity and persistent neuroimmune activation. Several clinical and experimental studies have demonstrated elevated pro-inflammatory cytokines, altered microglial activity and impairment of regulatory T-cell (T-reg) function in individuals with ASD. These immune abnormalities are now considered capable of influencing brain development and behaviour, rather than representing secondary consequences of the disorder. Consequently, ASD cannot be adequately explained or managed solely through behavioural interventions or symptom-directed pharmacotherapy. [6,7]
Currently available pharmacological treatments are largely directed toward associated behavioural symptoms such as irritability, aggression and hyperactivity and do not correct the underlying molecular, synaptic or immune disturbances. Therefore, there is a growing need for therapeutic strategies that directly address core disease mechanisms. In this context, messenger RNA (mRNA)-based therapeutic technologies provide a potential means for transient and targeted modulation of dysfunctional gene expression, while immune-regulatory approaches focused on T-reg cells offer a strategy to restore immune balance and attenuate neuroinflammation. The present review integrates existing evidence on ASD pathophysiology and therapeutic limitations and proposes a conceptual framework combining mRNA-mediated synaptic modulation with T-reg-driven immune tolerance as a future research direction. [8,22,23,26]
Novelty and scope of the present review
To the best of our knowledge, this is the first narrative review proposing an integrated therapeutic framework that combines mRNA-based synaptic modulation with regulatory T-cell-mediated immune tolerance restoration for autism spectrum disorder. While previous studies have independently explored synaptic dysfunction, immune dysregulation, mRNA therapeutics and regulatory T-cell biology in ASD and related neurodevelopmental disorders [3,5,7,23,26,36], no published review has conceptually integrated molecular and immunological strategies into a unified disease-modifying framework. The present review therefore introduces a novel translational perspective bridging molecular neuroscience and immune modulation as a future direction for pharmaceutical and therapeutic research in ASD.
REVIEW METHODOLOGY / LITERATURE SEARCH STRATEGY
This review was conducted using a narrative literature review approach to critically analyse existing evidence related to Autism Spectrum Disorder (ASD), with particular emphasis on synaptic dysfunction, neuroimmune dysregulation, messenger RNA (mRNA)–based therapeutics, and regulatory T-cell (T-reg)–mediated immune tolerance. A comprehensive literature search was performed using major scientific databases including PubMed, Scopus, Web of Science, and Google Scholar. Relevant peer-reviewed articles were identified using combinations of keywords such as “Autism Spectrum Disorder,” “synaptic dysfunction,” “neuroinflammation,” “mRNA therapeutics,” “regulatory T cells,” and “immune tolerance.” Foundational genetic and neurobiological studies that define ASD as a synaptic and neurodevelopmental disorder were considered essential for framing the review [3]. Original research articles, systematic reviews, and high-quality preclinical studies published in reputed international journals were included. Particular emphasis was placed on literature describing immune involvement and neuroinflammatory mechanisms in ASD, as immune dysregulation is increasingly recognized as a key contributor to disease pathophysiology [6,7]. Preference was given to studies published within the last 10–15 years, although landmark older studies were included where scientifically necessary. Non-English publications, conference abstracts without full texts, and studies lacking methodological clarity were excluded. The selected literature was critically evaluated and synthesized to identify established mechanisms, therapeutic gaps, and emerging molecular and immune-based strategies relevant to ASD. As this article is a theoretical review based solely on previously published literature, no experimental procedures involving human or animal subjects were conducted, and therefore ethical approval was not required.
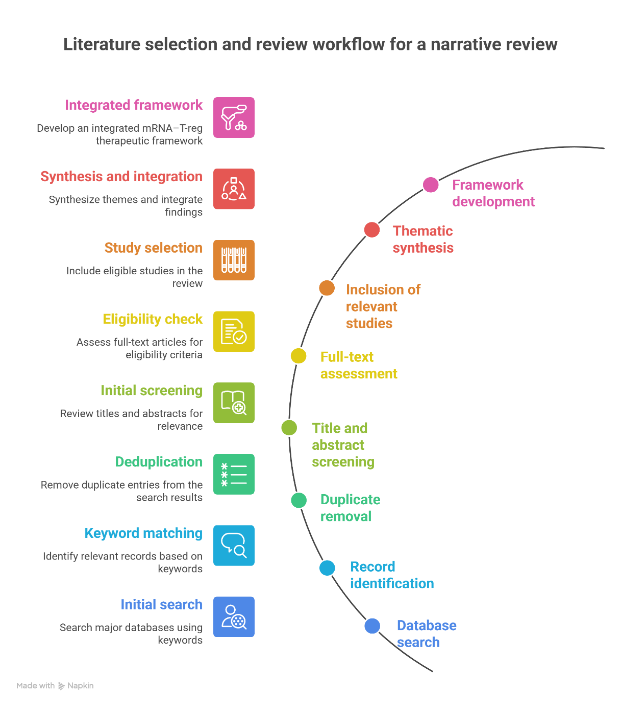
Figure 1. Literature selection and review workflow
EPIDEMIOLOGY OF AUTISM SPECTRUM DISORDER
Autism Spectrum Disorder (ASD) is currently recognised as one of the most prevalent neurodevelopmental disorders worldwide. Epidemiological investigations over the past two decades consistently demonstrate a progressive increase in reported prevalence. This rise is widely attributed to improved diagnostic definitions, increased professional and public awareness and enhanced surveillance practices, rather than to a true increase in disease incidence. [9,10] Recent global estimates indicate that approximately one in every 100 children is affected by ASD, although substantial geographical variation exists. Surveillance data from the United States generated by the Autism and Developmental Disabilities Monitoring (ADDM) Network report a prevalence of approximately one in 36 children. Similar upward trends have also been observed in several European and Asian regions, confirming the growing global burden of ASD. [9,10] A consistent sex-related difference is observed in ASD epidemiology. Most large-scale studies report that males are affected more frequently than females, with a male-to-female ratio of approximately 3–4:1. However, increasing evidence suggests that ASD in females may be under-recognised due to differences in clinical presentation and compensatory behavioural strategies, which has important implications for both diagnosis and research interpretation. [11] ASD is most commonly diagnosed during early childhood, typically between two and four years of age, although atypical developmental features may be identifiable earlier. Early identification is clinically important, as timely behavioural and educational interventions are associated with improved functional outcomes. Nevertheless, delayed diagnosis remains common in low- and middle-income settings because of limited access to specialised services and structured screening programmes. [10] Overall, the epidemiological profile of ASD highlights the urgent need for improved understanding of its biological underpinnings and for the development of therapeutic strategies that extend beyond symptomatic management. [9,10]
1. ETIOLOGY OF AUTISM SPECTRUM DISORDER
Autism Spectrum Disorder (ASD) is a multifactorial condition arising from a complex interplay of genetic, epigenetic, environmental, and immune-mediated factors. Rather than resulting from a single causative mechanism, ASD reflects cumulative disturbances in early brain development that alter neuronal connectivity, synaptic maturation, and neuroimmune regulation.
1.1 Genetic Factors
Genetic susceptibility plays a central role in ASD etiology, with heritability estimates ranging between 50–80% based on family and twin studies [12]. Large-scale genomic analyses have identified hundreds of ASD-associated risk genes involved in synaptic formation, neuronal signalling, transcriptional regulation, and chromatin remodelling. These findings support the concept of ASD as a disorder of synaptic and circuit-level dysfunction rather than a classical monogenic disease [3,12]. Both de novo mutations and copy number variations (CNVs) contribute significantly to ASD risk. De novo variants in genes such as SHANK3, SCN2A, CHD8, and MECP2 have been shown to disrupt synaptic scaffolding, neuronal excitability, and neurodevelopmental trajectories [3]. CNVs affecting chromosomal regions such as 16p11.2 and 15q11–13 further highlight the genetic heterogeneity underlying ASD.
1.2 Epigenetic Mechanisms
Epigenetic regulation serves as an important interface between genetic predisposition and environmental influences in ASD. Alterations in DNA methylation, histone modification, and chromatin remodelling can lead to long-lasting changes in gene expression without altering the DNA sequence itself [13]. Several ASD-associated genes are epigenetic regulators, suggesting that dysregulated transcriptional control may contribute to abnormal neuronal differentiation and synaptic plasticity. Epigenetic mechanisms are particularly relevant during prenatal and early postnatal development, when neural circuits are highly sensitive to external influences. Disruption of these processes may result in persistent neurodevelopmental abnormalities characteristic of ASD [13].
1.3 Environmental and Prenatal Factors
Environmental factors act in conjunction with genetic susceptibility to influence ASD risk. Prenatal exposures such as maternal infections, metabolic disorders, nutritional deficiencies, and exposure to environmental toxins have been associated with altered neurodevelopment [14]. Among these, maternal immune activation (MIA) has emerged as a critical etiological contributor. Experimental and epidemiological studies demonstrate that maternal inflammatory responses during pregnancy—particularly elevated cytokines such as interleukin-6 (IL-6) and interleukin-17A (IL-17A)—can disrupt fetal brain development and increase ASD-like behavioural outcomes in offspring [14,28]. These findings provide a mechanistic link between immune activation and neurodevelopmental alterations in ASD.
1.4 Immune-Related Etiological Contributions
Increasing evidence supports a role for immune dysregulation in ASD etiology. Individuals with ASD frequently exhibit altered immune profiles, including elevated pro-inflammatory cytokines, microglial activation, and reduced regulatory immune responses [6,7]. Notably, abnormalities in regulatory T cells (T-regs)—which are essential for maintaining immune tolerance—have been reported in both ASD individuals and their mothers [7,36]. Such immune disturbances during critical periods of brain development may influence synaptic pruning, neuronal connectivity, and circuit maturation, thereby contributing to ASD pathogenesis. These observations provide a strong etiological rationale for exploring immune-modulating therapeutic strategies alongside molecular interventions.
1.5 Gene–Environment Interaction
Current evidence suggests that ASD arises from gene–environment interactions, wherein genetic vulnerability amplifies sensitivity to environmental and immune-mediated insults. Epigenetic modifications may further mediate these interactions, leading to persistent alterations in neurodevelopmental pathways [12,13]. This integrative etiological model underscores the complexity of ASD and highlights the limitations of single-target therapeutic approaches.
2. PATHOPHYSIOLOGY OF AUTISM SPECTRUM DISORDER
Autism Spectrum Disorder (ASD) is characterized by complex and interrelated disturbances in neuronal development, synaptic organization, neural connectivity, and neuroimmune regulation. Current evidence suggests that pathological processes begin during prenatal or early postnatal brain development and persist throughout life, resulting in the core behavioural and cognitive features of ASD.
2.1 Synaptic Dysfunction and Altered Neural Development
One of the central pathological features of ASD is synaptic dysfunction, reflected by abnormalities in synapse formation, maturation, and pruning. Many ASD-associated genes encode synaptic scaffolding proteins, cell-adhesion molecules, and regulators of neurotransmission. Disruption of these proteins impairs synaptic stability and plasticity, leading to abnormal neuronal communication [3,12]. Postmortem and experimental studies demonstrate altered synaptic density and spine morphology in multiple brain regions implicated in social behaviour and cognition, including the prefrontal cortex, temporal cortex, and cerebellum [5]. These synaptic abnormalities interfere with the proper establishment of neural circuits required for learning, social interaction, and sensory integration.
2.2 Excitatory–Inhibitory (E/I) Imbalance
ASD pathophysiology is strongly associated with an imbalance between excitatory glutamatergic signalling and inhibitory γ-aminobutyric acid (GABAergic) signalling. Reduced expression of GABA-synthesizing enzymes and impaired inhibitory interneuron function contribute to cortical hyperexcitability and network instability [16]. This E/I imbalance has been linked to several ASD-related features, including sensory hypersensitivity, repetitive behaviours, seizures, and impaired information processing. Altered neurotransmission further disrupts synaptic plasticity mechanisms such as long-term potentiation and long-term depression, which are essential for cognitive development [5,16].
2.3 Abnormal Neural Connectivity
Neuroimaging studies consistently reveal atypical patterns of brain connectivity in individuals with ASD. These include increased short-range (local) connectivity and reduced long-range connectivity, particularly between frontal and temporal brain regions involved in social cognition and language processing [17]. Such connectivity alterations impair the integration of information across distributed neural networks, contributing to deficits in social communication, executive function, and emotional regulation. Abnormal network organization is now considered a key pathophysiological hallmark of ASD [17].
2.4 Neuroinflammation and Microglial Activation
Neuroinflammation plays a significant role in ASD pathophysiology. Microglia, the resident immune cells of the central nervous system, are essential for synaptic pruning and neuronal maturation. In ASD, microglial activation is frequently dysregulated, resulting in excessive inflammatory signalling and impaired synaptic refinement [6]. Elevated levels of pro-inflammatory cytokines such as IL-6, TNF-α, and IL-1β have been reported in both peripheral blood and brain tissue of individuals with ASD, indicating persistent immune activation [6,7]. Chronic neuroinflammation may exacerbate synaptic dysfunction and disrupt neural circuit development during critical developmental windows.
2.5 Immune Dysregulation and T-Regulatory Cell Involvement
Beyond innate immune activation, ASD is also associated with abnormalities in adaptive immune regulation. Several studies have reported reduced frequency and impaired function of regulatory T cells (T-regs) in individuals with ASD [7,36]. T-regs are essential for maintaining immune tolerance and controlling excessive inflammatory responses. Deficient T-reg activity may permit sustained neuroinflammatory states that negatively affect neuronal development and synaptic stability. The interaction between immune dysregulation and neural dysfunction underscores the importance of considering ASD as a neuroimmune disorder, rather than a purely neurological condition [6,26].
2.6 Integrated Pathophysiological Perspective
Taken together, ASD pathophysiology reflects the convergence of synaptic abnormalities, E/I imbalance, disrupted neural connectivity, and immune-mediated neuroinflammation. These mechanisms are not independent; rather, they interact dynamically to influence brain development and behaviour. This integrated understanding provides a strong biological foundation for exploring therapeutic strategies that simultaneously target neuronal and immune pathways, as proposed later in this review.
3. LIMITATIONS OF CURRENT THERAPEUTIC STRATEGIES IN AUTISM SPECTRUM DISORDER
Despite significant advances in the understanding of autism spectrum disorder (ASD), existing therapeutic strategies remain largely focused on symptom management rather than disease modification. Current interventions primarily aim to improve behavioural outcomes and associated psychiatric symptoms, while the underlying neurobiological and immunological abnormalities remain largely unaddressed.
3.1 Behavioural and Educational Interventions
Behavioural therapies, including applied behaviour analysis (ABA), speech therapy, and occupational therapy, constitute the cornerstone of ASD management. These interventions can improve adaptive behaviours, communication skills, and social functioning, particularly when initiated early in life [20]. However, such approaches are resource-intensive, require long-term commitment, and show variable effectiveness across individuals. Importantly, behavioural interventions do not target the molecular, synaptic, or immune-mediated mechanisms underlying ASD. As a result, while functional gains may be observed, these therapies do not alter the core pathophysiological processes driving the disorder [20].
3.2 Pharmacological Management
Pharmacological treatments approved for ASD are limited and primarily address associated behavioural symptoms such as irritability, aggression, and hyperactivity. Antipsychotic agents such as risperidone and aripiprazole are commonly prescribed but do not improve core deficits in social communication or cognitive function [21]. Moreover, long-term use of these medications is associated with adverse effects including weight gain, metabolic disturbances, sedation, and extrapyramidal symptoms, which further limit their utility [21]. The absence of pharmacological agents capable of correcting synaptic dysfunction, neural connectivity abnormalities, or immune dysregulation highlights a major therapeutic gap.
3.3 Lack of Disease-Modifying Therapies
A critical limitation of current ASD treatment paradigms is the absence of disease-modifying therapies. Existing interventions do not address key pathological mechanisms such as synaptic instability, excitatory–inhibitory imbalance, neuroinflammation, or impaired immune tolerance [5,6]. Consequently, treatment outcomes are often modest and inconsistent, particularly in individuals with severe or complex ASD phenotypes.
3.4 Limited Integration of Neuroimmune Targets
Although growing evidence implicates immune dysregulation and neuroinflammation in ASD pathogenesis, immune-modulating strategies are not part of standard clinical practice [6,7]. Regulatory T-cell dysfunction, chronic inflammatory signalling, and microglial abnormalities remain largely unexplored as therapeutic targets, despite their potential relevance to disease progression [36].
3.5 Implications for Future Therapeutic Development
The limitations of current therapeutic strategies underscore the need for novel approaches capable of targeting both neuronal and immune mechanisms. Therapies that solely focus on behavioural modification or symptomatic relief are unlikely to produce sustained improvements in neurodevelopmental outcomes. This therapeutic gap provides a strong rationale for exploring integrated molecular and immune-based strategies, such as the combined mRNA and T-regulatory approach proposed later in this review.
4. mRNA-BASED THERAPEUTIC MODULATION IN AUTISM SPECTRUM DISORDER
Recent advances in molecular medicine have positioned messenger RNA (mRNA)–based therapeutics as a promising platform for transient and targeted modulation of gene expression. Unlike traditional gene therapy approaches, mRNA therapeutics enable controlled protein production without permanent genomic integration, offering a favourable safety profile for neurodevelopmental disorders such as Autism Spectrum Disorder (ASD) [22,23].
Rationale for mRNA Therapeutics in ASD
ASD is strongly associated with dysregulated expression of genes involved in synaptic development, neurotransmission, and neuronal plasticity. Large-scale genetic studies have identified mutations and functional disruptions in genes encoding synaptic scaffolding proteins, ion channels, and regulators of neuronal connectivity [3,12]. These abnormalities result in impaired synaptic stability and circuit maturation, which are not addressed by current pharmacological treatments. mRNA-based modulation provides a potential strategy to temporarily restore deficient or dysfunctional proteins, thereby correcting downstream synaptic and cellular abnormalities without altering the host genome [23]. This reversibility is particularly relevant in pediatric and neurodevelopmental contexts, where long-term safety is a major concern.
4.1 Mechanistic Basis of mRNA Therapeutics
Therapeutic messenger RNA is synthetically designed to encode a specific protein of interest and is delivered into target cells, where it utilises the host translational machinery for protein synthesis. Following cellular uptake, the mRNA is translated by ribosomes to produce the encoded functional protein, after which the mRNA molecule is naturally degraded by intracellular processes. This transient and non-integrative expression profile allows precise control over both the dose and duration of protein production. [22,23] In the context of Autism Spectrum Disorder, mRNA-based constructs may theoretically be designed to express proteins that are directly involved in synaptic organisation, neuronal signalling and neuroplasticity. These may include proteins contributing to postsynaptic density structure, enzymes involved in neurotransmitter synthesis and regulation, and neurotrophic factors that support neuronal survival and synaptic maturation. Such targeted and temporary protein expression may assist in improving synaptic signalling and circuit stability during vulnerable developmental stages. [5,16,25] Importantly, the reversible nature of mRNA-based modulation is particularly relevant for neurodevelopmental conditions, where permanent genetic alteration raises significant safety concerns. The ability to titrate expression levels and repeat dosing if required offers a flexible therapeutic platform for exploratory interventions aimed at correcting functional protein deficits associated with ASD-related synaptic and cellular abnormalities. [22,23]
4.2 Potential Molecular Targets Relevant to ASD
Several molecular pathways implicated in ASD pathophysiology are conceptually amenable to mRNA-based intervention. These include synaptic scaffolding proteins associated with excitatory synapse stability, regulators of inhibitory neurotransmission, and neurotrophic factors involved in neuronal maturation [5,16,25]. Preclinical studies in non-ASD neurological models demonstrate that mRNA-mediated expression of neurotrophic or anti-inflammatory proteins can improve synaptic function and reduce neural injury, supporting the broader feasibility of this platform in central nervous system disorders [25,33].
4.3 Advantages of mRNA Over Conventional Gene-Based Approaches
Compared to viral gene therapy or genome-editing technologies, mRNA therapeutics offer several advantages:
No genomic integration, reducing mutagenesis risk
Transient expression, allowing dose titration and reversibility
Rapid design and modification, enabling target specificity
Reduced long-term safety concerns, particularly in developing brains These characteristics make mRNA-based approaches especially attractive for exploratory therapeutic strategies in ASD [22,23].
4.4 Current Evidence and Limitations
At present, no clinical studies have directly evaluated mRNA-based therapeutics in ASD. However, accumulating evidence from other neurological and inflammatory conditions supports the feasibility of mRNA delivery to neural tissues and its ability to modulate disease-relevant pathways [23,34]. The absence of ASD-specific mRNA studies highlights an important translational gap rather than a lack of conceptual validity. A major limitation for the application of mRNA therapeutics in ASD is efficient transport across the blood–brain barrier. The development of brain-targeted delivery systems capable of overcoming biological barriers while maintaining mRNA stability and controlled release remains a key pharmaceutical challenge and an important determinant of translational feasibility [34].
4.5 Positioning Within the Review Framework
Within the context of this review, mRNA-based modulation represents a theoretical yet biologically plausible strategy for addressing core molecular abnormalities in ASD. Its limitations, including delivery challenges and transient expression, emphasize the need for complementary approaches—such as immune regulation—which are discussed in subsequent sections.
5. PHARMACEUTICAL AND TRANSLATIONAL CONSIDERATIONS OF mRNA-BASED THERAPEUTICS FOR ASD
From a pharmaceutical development perspective, successful clinical translation of mRNA-based therapeutics for Autism Spectrum Disorder requires careful optimization of formulation and delivery systems. Therapeutic mRNA is intrinsically unstable and highly susceptible to enzymatic degradation, necessitating the use of protective delivery platforms such as lipid-based nanoparticles and polymeric carriers to improve stability, cellular uptake and translational efficiency [34,38]. Formulation parameters including particle size, surface characteristics and encapsulation efficiency critically influence biodistribution and tissue exposure [34].
For central nervous system applications, the presence of the blood–brain barrier represents a major pharmaceutical challenge, as it restricts the entry of large and negatively charged macromolecules such as mRNA. Advanced delivery strategies, including surface-modified lipid nanoparticles, receptor-mediated transport systems and intranasal administration approaches, are therefore of particular relevance for ASD-oriented mRNA therapeutics [34]. In addition, large-scale manufacturing, long-term storage stability, cold-chain requirements and batch-to-batch reproducibility remain important pharmaceutical considerations for regulatory approval and clinical translation of mRNA products [38]. These aspects indicate that formulation design and delivery optimization will be as critical as molecular target selection for the future development of mRNA-based interventions in ASD.
Table 1. Potential molecular targets and pathways suitable for mRNA-based modulation in autism spectrum disorder
|
Molecular target / pathway |
Role in ASD pathophysiology |
Rationale for mRNA-based modulation |
Expected therapeutic relevance |
References |
|
Synaptic scaffolding and postsynaptic density–associated pathways (e.g., SHANK3-related signalling) |
Disrupted synaptic organisation and impaired neuronal connectivity due to altered synaptic protein expression |
Transient restoration of deficient synaptic proteins using mRNA constructs may support synaptic maturation and stability. |
Improvement of synaptic architecture, synaptic stability and neural network integrity |
[3,5,12] |
|
Regulators of excitatory–inhibitory balance |
Cortical hyperexcitability and network instability caused by impaired inhibitory neurotransmission |
mRNA-mediated modulation of regulatory proteins may help normalise excitatory and inhibitory neurotransmission. |
Reduction of excitatory–inhibitory imbalance and improved network function |
[5,16] |
|
Neurotrophic signalling pathways |
Altered neuronal survival, differentiation and synaptic plasticity |
mRNA-based delivery of neurotrophic factors can transiently enhance neuronal support and synaptic remodelling. |
Improved neuronal maturation, synaptic plasticity and circuit refinement |
[25,33] |
|
Anti-inflammatory cytokine and signalling pathways (e.g., IL-10-related signalling) |
Persistent neuroinflammation and dysregulated microglial activation |
mRNA-encoded anti-inflammatory mediators may suppress excessive inflammatory signalling in neural tissues. |
Attenuation of neuroinflammation and protection of synaptic structures |
[6,7,32] |
|
Immune regulatory pathways supporting regulatory T-cell function |
Reduced regulatory immune control and impaired immune tolerance in ASD |
mRNA-mediated modulation of immune regulatory pathways may enhance tolerogenic immune responses. |
Restoration of immune balance and reduction of neuroimmune-mediated synaptic disruption |
[26,29,35] |
6. T-REGULATORY IMMUNE TOLERANCE IN AUTISM SPECTRUM DISORDER
Increasing evidence indicates that immune dysregulation plays a significant role in the pathophysiology of Autism Spectrum Disorder (ASD), extending beyond secondary inflammation to actively influence neurodevelopment. Among immune components, regulatory T cells (T-regs) have gained particular attention due to their central role in maintaining immune tolerance and controlling excessive inflammatory responses.
6.1 Immune Abnormalities in ASD
Individuals with ASD frequently exhibit altered immune profiles, including elevated pro-inflammatory cytokines, abnormal microglial activation, and imbalances in adaptive immune cell populations [6,7]. Persistent immune activation during critical periods of brain development may interfere with synaptic pruning, neuronal maturation, and circuit formation. Clinical and experimental studies suggest that immune abnormalities in ASD are not merely consequences of neural dysfunction but may actively contribute to disease progression by creating a chronic pro-inflammatory environment [6].
6.2 Role of Regulatory T Cells in Neurodevelopment
Regulatory T cells are a specialized subset of CD4? T lymphocytes characterized by the expression of FOXP3, a transcription factor essential for their development and suppressive function. T-regs play a key role in limiting excessive immune activation, maintaining peripheral tolerance, and regulating inflammatory responses that could otherwise damage host tissues [26]. Beyond their classical immunological role, T-regs are increasingly recognized as modulators of neuroimmune communication. They influence microglial activity, cytokine balance, and blood–brain barrier integrity, thereby indirectly affecting neuronal development and synaptic stability [26].
6.3 T-Regulatory Dysfunction in ASD
Multiple studies have reported reduced numbers and impaired function of T-regs in individuals with ASD, along with decreased FOXP3 expression and altered cytokine profiles [7,36]. These abnormalities may permit sustained inflammatory signalling, resulting in prolonged microglial activation and disruption of synaptic refinement. Maternal immune activation studies further support this association, demonstrating that immune imbalance during pregnancy can alter fetal brain development and increase ASD-like behavioural outcomes in offspring [14,28]. Reduced T-reg-mediated immune control during early development may therefore represent a critical etiological and pathophysiological factor in ASD.
6.4 Therapeutic Implications of T-Reg Modulation
Given their central role in immune tolerance, T-regs represent a biologically plausible therapeutic target in ASD. Strategies aimed at enhancing T-reg function or restoring immune balance may reduce chronic neuroinflammation and create a more favourable environment for synaptic development and neural circuit stabilization [26,30]. Importantly, T-reg-based approaches are not expected to directly correct genetic or synaptic defects. Instead, they may act as supportive modulators, limiting inflammatory damage and preserving neuronal integrity. This characteristic makes T-reg modulation particularly relevant as part of a broader, multi-target therapeutic framework.
6.5 Positioning Within the Review Framework
Within this review, T-regulatory immune tolerance represents the second core pillar of the proposed conceptual model. While immune modulation alone may be insufficient to reverse core ASD pathology, its ability to stabilize the neuroimmune environment provides a strong rationale for integration with molecular strategies such as mRNA-based modulation, discussed in subsequent sections.
7. INTEGRATED NOVEL CONCEPT: COMBINED mRNA AND T-REGULATORY IMMUNE TOLERANCE APPROACH IN ASD
Autism Spectrum Disorder (ASD) is increasingly recognized as a condition arising from the convergence of neurodevelopmental abnormalities and immune dysregulation. Evidence discussed in earlier sections highlights that synaptic dysfunction, excitatory–inhibitory imbalance, and chronic neuroinflammation coexist and interact throughout disease progression [3,5,6]. Therapeutic strategies that target only one of these domains have therefore shown limited efficacy. In this context, an integrated approach combining mRNA-based molecular modulation with T-regulatory (T-reg) immune tolerance restoration represents a biologically rational and potentially transformative concept.
7.1 Rationale for a Dual-Target Strategy
mRNA-based therapeutics offer a means to transiently modulate the expression of proteins involved in synaptic stability, neurotransmission, and neuronal plasticity without permanent genomic alteration [22,23]. However, molecular correction alone may be insufficient in an environment characterized by persistent immune activation and neuroinflammation. Chronic inflammatory signalling has been shown to impair synaptic pruning, alter neuronal connectivity, and exacerbate neural dysfunction in ASD [6,7]. Conversely, T-regulatory immune modulation can suppress excessive inflammatory responses, stabilize cytokine balance, and regulate microglial activation, but it does not directly correct synaptic or genetic abnormalities [26,36]. These limitations suggest that single-axis interventions are unlikely to fully address the multifactorial pathology of ASD.
Conceptual Framework of the Combined Approach
The proposed combined strategy is based on functional complementarity:
mRNA modulation targets intrinsic neuronal and synaptic deficits by restoring or supporting the expression of key molecular mediators of neural development and plasticity.
T-regulatory immune tolerance stabilizes the neuroimmune environment, reducing inflammatory stress and preventing immune-mediated disruption of neural circuits.
By acting simultaneously on neuronal function and immune regulation, the combined approach aims to create a permissive biological environment in which synaptic correction is more likely to be effective and sustained.
7.2 Mechanistic Synergy Between Neural and Immune Modulation
Neuroinflammation has been shown to interfere with synaptic maturation and plasticity through cytokine-mediated effects on neurons and glial cells [6]. Reduced T-reg function in ASD permits prolonged inflammatory signalling, which may counteract molecular interventions aimed at improving synaptic function [7,36]. In the proposed model, T-reg-mediated immune tolerance reduces inflammatory burden and microglial overactivation, thereby protecting developing synapses. Simultaneously, mRNA-mediated protein expression supports synaptic organization and neural circuit stabilization. This bidirectional interaction may enhance therapeutic efficacy beyond what either strategy could achieve alone.
8. NOVELTY AND THEORETICAL NATURE OF THE CONCEPT
Although mRNA-based therapeutic strategies and T-regulatory immune modulation have each been explored independently in neurological and immune-mediated disorders, their combined application in Autism Spectrum Disorder has not yet been investigated. The framework presented in this review therefore represents a conceptual integration derived from converging evidence across molecular neuroscience and immunology rather than from direct experimental validation in ASD. The intention of proposing this integrated model is not to suggest immediate clinical translation, but to identify a biologically plausible direction for future research. Current literature demonstrates that synaptic dysfunction and immune dysregulation coexist in ASD; however, therapeutic investigations have largely examined these domains in isolation. By combining transient molecular modulation through mRNA technology with restoration of immune tolerance via T-regulatory pathways, the present framework attempts to bridge this gap. It is important to emphasise that this proposal remains theoretical and requires systematic preclinical evaluation before any translational consideration. Presenting the concept within a clearly defined scientific context ensures methodological transparency and avoids overstating therapeutic applicability. Accordingly, the contribution of this review lies in outlining a research hypothesis grounded in existing evidence rather than proposing an established treatment strategy [22,23,26,36].
Implications for Disease-Modifying Therapy
If validated through preclinical and clinical research, the combined mRNA–T-reg approach has the potential to shift ASD treatment paradigms from symptom-oriented management toward disease-modifying strategies. By addressing both synaptic dysfunction and immune dysregulation, this framework may offer more durable improvements in neural development and functional outcomes.
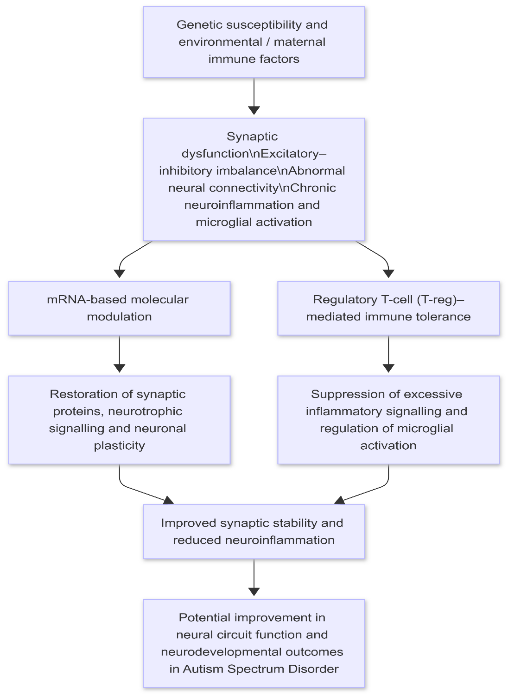
Figure 2. Conceptual framework of the combined mRNA-based synaptic modulation and regulatory T-cell-mediated immune tolerance strategy in autism spectrum disorder
9. EXISTING EVIDENCE SUPPORTING THE PROPOSED CONCEPT
Although no studies have directly evaluated a combined mRNA-based modulation and T-regulatory immune tolerance strategy in Autism Spectrum Disorder (ASD), multiple independent lines of evidence support the biological plausibility of this integrated approach. These findings arise from preclinical and clinical studies investigating synaptic modulation, neuroimmune regulation, and immune tolerance in ASD and related neurological conditions.
9.1 Evidence Supporting mRNA-Based Modulation of Neural Pathways
Emerging research in neurological and inflammatory disorders demonstrates that mRNA-based therapeutics can successfully induce controlled expression of biologically active proteins in neural and immune cells [22,23]. Preclinical studies have shown that mRNA-mediated delivery of neurotrophic and anti-inflammatory proteins can enhance synaptic plasticity, promote neuronal survival, and reduce neuroinflammatory damage [25,33]. Although these studies were not conducted in ASD models, the molecular targets involved—such as synaptic regulators and neurotrophic factors—overlap significantly with pathways disrupted in ASD [3,5]. This overlap provides indirect support for the feasibility of mRNA-based strategies in addressing ASD-related synaptic dysfunction.
9.2 Evidence Supporting Immune and T-Regulatory Modulation in ASD
Independent studies consistently report immune abnormalities in individuals with ASD, including elevated pro-inflammatory cytokines, activated microglia, and reduced regulatory T-cell populations [6,7,36]. These immune alterations have been associated with increased symptom severity and altered neurodevelopmental outcomes. Experimental models of maternal immune activation further demonstrate that immune imbalance during critical developmental periods can induce ASD-like behavioural phenotypes, highlighting the causal role of immune dysregulation in neurodevelopment [14,28]. Importantly, regulatory T cells are central to controlling such inflammatory responses and maintaining immune tolerance [26]. Clinical and preclinical evidence from autoimmune and neuroinflammatory disorders indicates that strategies enhancing T-reg function can suppress pathological inflammation and promote tissue homeostasis [30]. These findings support the rationale for exploring T-regulatory modulation as a therapeutic strategy in ASD, even though ASD-specific clinical trials remain limited.
9.3 Converging Neuroimmune Evidence
Synaptic development and immune regulation are increasingly recognized as interconnected biological processes. Neuroinflammation has been shown to interfere with synaptic pruning and plasticity, while immune tolerance mechanisms can indirectly influence neuronal maturation by regulating microglial activity and cytokine signalling [6,26]. Taken together, the available evidence suggests that molecular modulation of synaptic pathways and immune regulation through T-regs may act synergistically rather than independently. While current studies examine these mechanisms in isolation, their convergence provides a compelling scientific foundation for the integrated therapeutic concept proposed in this review.
9.4 Evidence Gap and Translational Opportunity
Despite strong supportive evidence from parallel research domains, the absence of studies evaluating combined mRNA and T-regulatory approaches in ASD represents a significant knowledge gap. Addressing this gap through systematic preclinical investigation may offer valuable insights into disease-modifying strategies for ASD. This review therefore positions the proposed integrated approach not as a proven therapy, but as a conceptual framework grounded in existing scientific evidence and deserving of future translational exploration.
10. EXPECTED OUTCOMES AND SIGNIFICANCE OF THE PROPOSED APPROACH
The integrated framework combining mRNA-based molecular modulation with T-regulatory immune tolerance is expected, at a theoretical level, to influence multiple biological processes implicated in autism spectrum disorder (ASD). Although direct experimental validation is not yet available, anticipated outcomes can be inferred from established evidence regarding synaptic biology, neuroimmune interactions and immune regulatory mechanisms.
10.1 Potential Neurobiological Impact
mRNA-mediated protein expression may support the restoration of synaptic components and signalling pathways that are frequently disrupted in ASD. By enabling controlled and transient production of proteins involved in synaptic stability, neuronal communication and plasticity, such an approach could contribute to improved neural circuit organisation during development. Experimental studies in related neurological contexts suggest that targeted molecular modulation can enhance synaptic resilience and neuronal survival, providing indirect support for this rationale. [3,5,16,25] Furthermore, modulation of pathways associated with excitatory–inhibitory balance may help reduce cortical network instability, which has been linked to sensory hypersensitivity and cognitive dysfunction in ASD. Although this remains hypothetical within ASD-specific models, the biological plausibility is supported by current understanding of synaptic regulation and neurotransmitter dynamics. [5,16]
10.2 Potential Immunological Impact
Enhancement of regulatory T-cell function is expected to attenuate chronic inflammatory signalling and restore immune tolerance. Regulatory T cells play a central role in limiting excessive cytokine production and maintaining immune homeostasis. In ASD, where elevated pro-inflammatory mediators and altered T-reg populations have been reported, restoring immune balance may create a more stable neurodevelopmental environment. [6,7,26,36] Reduction of sustained neuroinflammation may also indirectly protect synaptic structures from immune-mediated disruption, thereby complementing molecular interventions aimed at synaptic correction. Evidence from immune-modulatory research in other inflammatory disorders supports the concept that improved T-reg function can promote tissue stability and reduce pathological immune activation. [26,30]
10.3 Integrated and Synergistic Considerations
The principal significance of the proposed framework lies in its potential synergistic action. Molecular modulation alone may be insufficient in a persistently inflamed neural environment, while immune regulation without addressing synaptic dysfunction may not correct intrinsic neuronal deficits. By addressing both domains concurrently, the combined strategy conceptually targets interacting components of ASD pathophysiology. Such an integrated perspective aligns with the growing recognition of ASD as a neuroimmune disorder characterised by dynamic interaction between neuronal development and immune regulation. [6,23,26]
10.4 Translational and Scientific Relevance
From a translational standpoint, the reversible and non-genome-integrating nature of mRNA therapeutics, together with immune tolerance–based modulation, may offer safety advantages compared to permanent genetic interventions. However, rigorous preclinical investigation will be necessary to evaluate delivery strategies, dosing parameters and long-term safety, particularly in developing nervous systems. [22,23,30,34] Beyond ASD, this conceptual framework may stimulate broader research into integrated neuroimmune therapeutic strategies for other neurodevelopmental and neuroinflammatory conditions. The primary contribution of this section therefore lies in outlining scientifically plausible expectations rather than asserting established therapeutic efficacy.
11. LIMITATIONS OF THE PROPOSED mRNA–T-REGULATORY APPROACH
While the integrated mRNA-based modulation and T-regulatory immune tolerance framework presents a biologically plausible and innovative direction for autism spectrum disorder (ASD) therapy, several important limitations must be acknowledged. These limitations highlight the current gaps in evidence and the challenges associated with translating this conceptual model into clinical practice.
Theoretical and Preclinical Nature of the Concept
The foremost limitation of the proposed approach is its theoretical nature. At present, no experimental or clinical studies have evaluated the combined application of mRNA-based therapeutics and T-regulatory modulation in ASD. The rationale for this framework is derived from indirect evidence obtained from parallel studies in neurodevelopmental, neuroinflammatory, and immune-mediated disorders [23,26]. As such, the proposed model should be viewed as a conceptual hypothesis rather than an established therapeutic strategy.
11.1 Challenges in Targeted Delivery
Efficient and safe delivery of therapeutic mRNA to relevant neural cell populations remains a significant challenge. The central nervous system is protected by the blood–brain barrier, which limits the penetration of large or charged molecules [34]. Although advances in delivery platforms have shown promise, precise targeting of specific neuronal subtypes or brain regions relevant to ASD has not yet been achieved in a clinically scalable manner. Similarly, achieving controlled and selective modulation of T-regulatory cells without affecting broader immune function presents additional complexity [30].
11.2 Heterogeneity of Autism Spectrum Disorder
ASD is a highly heterogeneous condition, encompassing a wide range of genetic backgrounds, clinical phenotypes, and immune profiles. This heterogeneity may limit the universal applicability of a single therapeutic framework. It is likely that only specific ASD subgroups—such as those with pronounced immune dysregulation or identifiable synaptic deficits—would benefit from the proposed approach [12]. This variability underscores the need for biomarker-based patient stratification in future research.
11.3 Timing and Developmental Considerations
Neurodevelopmental timing represents another critical limitation. Synaptic development and immune maturation occur within tightly regulated developmental windows. Interventions applied too late may have limited impact on established neural circuitry, while early-life interventions raise ethical and safety concerns [5]. Determining the optimal therapeutic window for mRNA and T-regulatory interventions will therefore be essential.
11.4 Safety and Long-Term Effects
Although mRNA-based therapeutics are designed to be non-integrative and transient, long-term safety data in neurodevelopmental contexts remain limited [22,23]. Similarly, chronic immune modulation carries the potential risk of immune suppression or unintended alterations in host defence mechanisms [30]. Rigorous preclinical safety evaluation will be required before any clinical translation.
11.5 Regulatory and Translational Barriers
Finally, the combined nature of the proposed approach may pose regulatory challenges, as it integrates molecular and immune-based interventions. Establishing standardized manufacturing, dosing, and monitoring protocols for such a dual strategy may be complex and resource-intensive.
12. FUTURE SCOPE AND NOVEL DIRECTION
The integrated mRNA-based modulation and T-regulatory immune tolerance framework outlined in this review provides a foundation for several avenues of future investigation in Autism Spectrum Disorder (ASD). Given the heterogeneity and developmental complexity of ASD, any advancement toward clinical translation will require structured and phased evaluation. At present, both mRNA-mediated molecular strategies and T-regulatory immune modulation have been examined independently in neurological and immune-related disorders; however, their combined evaluation in ASD remains unexplored. The concept presented here should therefore be considered a research hypothesis that warrants systematic validation rather than an immediately applicable therapeutic model.
Preclinical and Experimental Validation
Initial efforts should focus on controlled preclinical studies using established ASD animal models and neurodevelopmental models involving immune dysregulation. Experimental designs may examine whether concurrent synaptic-targeted mRNA delivery and enhancement of regulatory T-cell function produce additive or synergistic effects on behavioural, synaptic and inflammatory endpoints. Such investigations would be essential for evaluating safety, optimal dosing intervals, route of administration and potential interactions between molecular and immune interventions. [3,6,26]
Biomarker-Based Stratification
Considering the clinical and biological heterogeneity of ASD, future research should incorporate biomarker-driven stratification strategies. Identification of immune signatures, inflammatory markers or synaptic pathway abnormalities may help determine which subgroups of individuals are most likely to benefit from integrated neuroimmune interventions. Precision-oriented approaches may increase translational relevance and reduce variability in therapeutic response. [12,36]
Developmental Timing and Therapeutic Windows
Another important aspect involves determining the developmental stage at which intervention may be most effective. Synaptic plasticity and immune maturation follow time-sensitive trajectories, suggesting that therapeutic impact may vary depending on age and neurodevelopmental stage. Carefully designed longitudinal studies will therefore be necessary to assess timing-dependent effects of both molecular and immune-modulatory strategies. [5]
Translational and Regulatory Considerations
Before clinical evaluation can be considered, comprehensive assessment of delivery systems, biodistribution, immunogenicity and long-term safety will be required. mRNA-based therapies intended for central nervous system applications must address challenges related to blood–brain barrier penetration and targeted cellular uptake. Similarly, immune-modulatory approaches must ensure preservation of systemic immune competence while enhancing regulatory pathways. Regulatory frameworks for advanced molecular and immune-based products will play a critical role in guiding responsible development and quality control. [23,29,34,38]
Broader Scientific Implications
Beyond ASD, the conceptual integration of synaptic modulation with immune tolerance may inform therapeutic exploration in other neurodevelopmental and neuroinflammatory conditions characterised by overlapping neuronal and immune disturbances. Continued interdisciplinary collaboration between neuroscience, immunology and pharmaceutical sciences will be essential to refine and evaluate such integrated strategies. Overall, the future direction proposed in this review emphasises systematic validation, safety assessment and translational feasibility, with the long-term objective of advancing disease-modifying research paradigms in ASD rather than solely symptom-oriented interventions.
CONCLUSION
Autism Spectrum Disorder (ASD) is a complex neurodevelopmental condition characterized by intertwined abnormalities in synaptic development, neural connectivity, and immune regulation. Despite substantial advances in understanding its biological basis, current therapeutic strategies remain largely symptomatic and do not address the core mechanisms underlying disease progression. This review critically synthesized existing evidence supporting the involvement of synaptic dysfunction and neuroimmune dysregulation in ASD and examined emerging molecular and immune-based therapeutic concepts. In this context, an integrated framework combining mRNA-based molecular modulation with T-regulatory immune tolerance restoration was proposed as a novel and biologically rational direction for future research. By targeting intrinsic neuronal deficits while simultaneously stabilizing the immune environment, this dual approach conceptually addresses multiple pathological dimensions of ASD. Importantly, the proposed mRNA–T-regulatory strategy is presented purely at a theoretical level. No experimental or clinical studies have yet evaluated this combined approach in ASD, and its feasibility, safety, and efficacy remain to be determined. Acknowledging this limitation is essential for maintaining scientific rigor and ethical transparency. The significance of this review lies not in proposing an immediate therapeutic solution, but in highlighting a new translational research direction grounded in current neurobiological and immunological knowledge. Systematic preclinical investigations, biomarker-driven stratification, and carefully designed translational studies will be required to assess the validity of this integrated model. In conclusion, ASD represents a disorder in which isolated therapeutic strategies are unlikely to yield sustained benefits. Integrated neuroimmune approaches, such as the conceptual framework presented here, may offer a pathway toward future disease-modifying interventions. This review aims to stimulate further interdisciplinary research bridging molecular neuroscience and immunology to advance the development of innovative therapeutic strategies for ASD.
REFERENCES




Mahak Shaikh, Pooja Petkar, Shruti Taware , Shravani Dhumal., Integrated Mrna-Based Synaptic Modulation and Regulatory T-Cell-Mediated Immune Tolerance as a Future Therapeutic Framework for Autism Spectrum Disorder: A Narrative Review, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 398-3215. https://doi.org/10.5281/zenodo.18710122
 10.5281/zenodo.18710122
10.5281/zenodo.18710122