
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Shree Sai College of Pharmacy, Khandala, Vaijapur, Chatrapati Sambhajinagar 431116
Microsphere-based topical drug delivery systems are gaining importance as an effective approach to improve the performance of non-steroidal anti-inflammatory drugs (NSAIDs).[5 ,7,20]These systems provide sustained drug release and localized action, which helps reduce systemic side effects. Lornoxicam is a potent NSAID with strong pain-relieving and anti-inflammatory properties; however, its conventional dosage forms are limited by short duration of action and gastrointestinal adverse effects.[9-21] Encapsulating lornoxicam in polymer-based microspheres offers a promising solution by enhancing drug stability, prolonging drug release, and improving local therapeutic efficacy.[15,16] This review discusses recent developments in microsphere-based topical drug delivery systems for prolonged anti-inflammatory action.[ 7,19]The use of biodegradable polymers such as PLGA, ethyl cellulose, chitosan, and natural polymers is reviewed along with commonly used preparation methods including solvent evaporation, emulsion diffusion, and spray drying. Important formulation parameters affecting particle size, drug entrapment efficiency, and release characteristics are highlighted. Evaluation techniques such as physicochemical characterization, skin permeation testing, anti-inflammatory studies, and stability assessment are also discussed. Overall, microsphere-based topical delivery of lornoxicam presents a safe and effective strategy for prolonged anti-inflammatory therapy with improved patient compliance.
Inflammation is a complex biological response of body tissues to harmful stimuli such as infection, injury, or chemical [30]irritation. It is commonly associated with pain, redness, swelling, and loss of function. Non-steroidal anti-inflammatory drugs (NSAIDs) are widely used for the management of inflammatory conditions; however, conventional oral therapy is often associated with systemic side effects such as gastrointestinal irritation, cardiovascular risks, and hepatic toxicity.[11,31] Topical drug delivery systems have therefore gained significant attention as they offer localized drug action at the site of inflammation while minimizing systemic exposure [9,21]and adverse effects.
Despite the advantages of topical administration, conventional topical formulations such as gels, creams, and ointments often suffer from poor skin penetration, rapid drug release, short duration of action, [23,24]and frequent dosing requirements. These limitations reduce patient compliance and therapeutic effectiveness. To overcome these challenges, advanced drug delivery systems have been developed, among which microsphere-based topical drug delivery systems have emerged as a promising approach for achieving prolonged and controlled anti-inflammatory action. [5,7,12]Microspheres are spherical, free flowing particulate systems typically ranging in size from 1 to 1000 µm, composed of biodegradable or non-biodegradable polymers.[9,19] When incorporated into topical formulations, microspheres can encapsulate anti-inflammatory drugs and provide sustained release by controlling drug diffusion and polymer degradation. This leads to prolonged drug residence time at the site of application, reduced dosing frequency, improved therapeutic efficacy, and enhanced patient compliance.
Additionally, microsphere-based systems can improve drug stability, protect the drug from degradation, and reduce skin irritation by preventing high initial drug concentrations. Polymers such as poly(lactic-co-glycolic acid) (PLGA), ethyl cellulose, chitosan, and alginate are commonly used for microsphere preparation due to their biocompatibility and controlled release properties. The integration of microspheres into topical dosage forms such as gels or creams further enhances their applicability for the treatment of chronic inflammatory conditions including arthritis, dermatitis, and musculoskeletal disorders. Thus, microsphere-based topical drug delivery systems represent an advanced and effective strategy for prolonging anti-inflammatory action, improving therapeutic outcomes, and reducing systemic side effects. This review focuses on the principles, Formulation strategies, evaluation parameters, and therapeutic potential of microsphere based topical drug delivery systems for sustained anti-inflammatory therapy.
Concept of Microspheres
Microspheres are spherical, free-flowing particulate drug delivery systems with particle sizes typically ranging from 1 to 1000 µm, composed of natural or synthetic polymers. They are designed to encapsulate, entrap, or adsorb active pharmaceutical ingredients (APIs) within a polymeric matrix or shell, enabling controlled, sustained, or targeted drug release.[5,7,16] Due to their small size and large surface area, microspheres offer improved drug stability, bioavailability, and therapeutic efficacy.]The fundamental concept behind microsphere-based drug delivery lies in modulating the release profile of drugs by controlling polymer composition, particle size, surface morphology, and preparation technique. Drugs can be uniformly dispersed throughout the polymer matrix (matrix-type microspheres) or enclosed within a polymeric membrane (reservoir-type microspheres), depending on the formulation strategy. In pharmaceutical applications, microspheres serve as multiparticulate carriers that minimize dose dumping, reduce systemic side effects, and improve patient compliance. Their versatility allows incorporation into various dosage forms such as topical gels, creams, transdermal systems, oral capsules, and injectable formulations. In topical drug delivery, microspheres are particularly advantageous as they enable localized drug action, prolonged residence time on the skin, and reduced percutaneous absorption into systemic circulation. Microspheres can be formulated using biodegradable polymers such as chitosan, gelatin, alginate, and polylactic-co-glycolic acid (PLGA), or non-biodegradable polymers[6,26,27] like ethyl cellulose and polymethacrylates. Biodegradable microspheres are preferred in most pharmaceutical applications due to their safety, biocompatibility, and controlled degradation into non-toxic by-products. The drug release from microspheres occurs through mechanisms such as diffusion, polymer erosion, swelling, or a combination of these processes. By manipulating formulation variables, microspheres can be engineered to deliver drugs over extended periods, making them highly suitable for the management of chronic conditions such as inflammation, arthritis, and dermatological disorders. Overall, the concept of microspheres represents an advanced and flexible drug delivery approach that bridges conventional dosage forms and modern controlled release systems. Their ability to provide sustained therapeutic action, enhanced stability, and targeted delivery makes microspheres an integral component of novel pharmaceutical formulations, particularly in topical sustained-release anti-inflammatory therapy.
Methods of Microsphere Preparation
Emulsification is a widely used method for microsphere fabrication Due to its simplicity and ability to control particle size precisely. The products of this procedure can be classified as single emulsions (oil-in-Water: O/W) and double emulsions (water-in-oil in-water: W/O/W and Oil-in-water-in-oil: O/W/O), with the choice depending on the drug’s Properties (Fig. 3). This technique can be used to encapsulate Both hydrophilic and hydrophobic drugs, making it versatile for a range Of therapeutic applications. Additionally, emulsification allows for Controlled drug release, improving the stability and bioavailability of The encapsulated drugs. However, the process can be time-consuming And may involve the use of organic solvents, raising concerns about toxicity and environmental impact . Furthermore, emulsification Can result in broad particle size distributions, leading to inconsistencies In drug release profiles. The efficiency of drug encapsulation is influenced by factors such as emulsifier concentration, stirring speed, and Temperature requiring optimization for specific applications. Despite these challenges, emulsification remains a popular and adaptable method for microsphere fabrication.[ 27,29,30]
The solvent evaporation/extraction technique is commonly used to encapsulate hydrophilic drugs in polymer-based microspheres, resulting in a W/O/W structure In this method, the aqueous phase containing the hydrophilic drug is first encapsulated within an organic phase and then dispersed into an aqueous solution. For example, a hydrophilic drug is dissolved in water and mixed with a polymer solution (e.g., PLGA) in an organic solvent to create the first emulsion. This W/O emulsion is then added to an aqueous solution .This method is widely applied to encapsulate hydrophilic drugs using a double emulsion system. The drug is dissolved in an aqueous phase and emulsified within a polymer solution in an organic solvent, forming a primary emulsion. This is then dispersed into an external aqueous phase to create a double emulsion. The organic solvent is removed by evaporation, resulting in hardened microspheres. This technique allows sustained drug release but requires precise control to prevent drug loss and ensure complete solvent removal.
Spray drying is a technique in which a liquid polymer solution or Suspension is sprayed into fine droplets, which are then rapidly dried to Form microspheres This process is widely used in the pharmaceutical and biomedical fields for producing drug delivery microspheres Spray drying typically involves four stages. First, the drug or Polymer issue dissolved or dispersed in a solvent to create a uniform solution. This solution Is then sprayed through a nozzle to form fine droplets The droplet size is influenced by factors such as nozzle design, Solution viscosity, and spray pressure. Next, the are exposed to Hot drying air (150–220 ?C), which causes rapid solvent evaporation and Solidifies the surface of the droplet. Drying continues inside the droplet, Resulting in the formation of complete microspheres. For heat sensitive Drugs, adjustments to the drying temperature or spray rate may be Necessary to prevent degradation. The dried microspheres are then Collected and can be further coated or surface-treated to enhance their Functionality. The advantages of spray drying include fast processing speed, high Scalability for industrial production, and the ability to control particle Size and shape by adjusting parameters such as spray speed, drying Temperature, and solution concentration . Additionally, it enables The production of uniform, highly dispersible microspheres. However, The process requires careful control to prevent thermal degradation, Particularly for heat-sensitive drugs Moreover, spray drying may Not be suitable for unstable drugs, and achieving precise microsphere Morphologies can be challenging. Despite these limitations, spray drying Remains a versatile and widely applied method in drug delivery. [28,29]
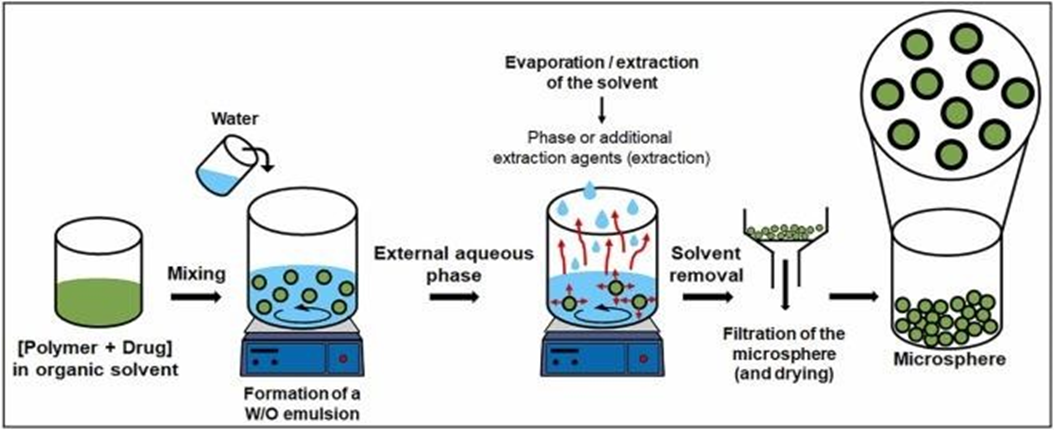
Fig:2:Solvent evaporation/extraction technique for encapsulating hydrophilic drugs in polymer-based microspheres, forming a W/O/W structure.
This method involves emulsifying a hydrophilic drug within an internal aqueous phase, which is then dispersed in an organic polymer solution before being emulsified in an external aqueous phase. Controlled solvent removal facilitates microsphere formation, enabling sustained drug release.
Phase separation/coacervation is a process where two or more Initially mixed phases separate into independent phases under specific Conditions such as temperature, concentration changes, or solvent alterations. This technique is commonly used for microsphere preparation And occurs in three stages. First, the polymer is dissolved In a solvent to form a uniform solution, into which the drug is either Mixed or dispersed . In the next stage, a coacervation inducer is Added, altering conditions such as temperature, pH, ion concentration, Or solvent ratio, causing the polymer to separate from the solution. The Polymer droplets formed at this stage represent the initial microsphere Form. During the second stage, conditions are adjusted to encourage the Aggregation of the droplets into more compact structures over time. Particle size and shape can be controlled by the environmental conditions . Finally, crosslinking agents are introduced to stabilize the Microspheres, enhancing their structural integrity and allowing for the Fine-tuning of drug release characteristics.This technique offers several advantages, including high drug Loading efficiency, ease of controlling particle size for tailored drug. [19,20]
The single emulsion technique, commonly used to create microspheres, involves forming an O/W emulsion by dissolving a hydrophobic polymer in an organic solvent and incorporating the drug into the solution. This solution is then added to an aqueous medium, where emulsification occurs . Afterward, the organic solvent is evaporated or extracted, causing the polymer to solidify and form microsphere. For example, PLGA and a hydrophobic drug are dissolved In an organic solvent and added to an aqueous solution containing polyvinyl alcohol (PVA), where emulsification is achieved through vigorous stirring. Once the organic solvent is removed, microspheres are formed and purified by centrifugation, washing, and drying. This method is simple, cost-effective, and suitable for large-scale production of microspheres, particularly for encapsulating hydrophobic drugs and enabling sustained drug release . However, challenges include difficulties in encapsulating hydrophilic drugs due to their poor solubility in organic solvents, resulting in low encapsulation efficiency. Additionally, the use of organic solvents raises toxicity concerns, and achieving a uniform particle size distribution can be
The double emulsion technique is frequently used for encapsulating Hydrophilic drugs in polymer-based microspheres, forming a W/O/W Structure. Unlike the single emulsion method, this technique involves Encapsulating an aqueous phase containing the hydrophilic drug within An organic phase, which is then dispersed into an aqueous solution . For example, a hydrophilic drug is dissolved in water and mixed With a polymer solution (e.g., PLGA) in an organic solvent to form the First emulsion. This W/O emulsion is then added to an aqueous solution Containing PVA to form the W/O/W emulsion [fig 3]. Following this, the Organic solvent is evaporated or extracted, solidifying the polymer and Forming microspheres, which are then purified through centrifugation, Washing, and drying. This method is particularly effective for encapsulating hydrophilic Drugs, such as proteins, peptides, and vaccines, which are difficult to Encapsulate using single emulsions . It also facilitates the development of controlled-release systems for sustained drug delivery and Can incorporate a variety of biocompatible polymers. However, the Process is more complex and time-consuming due to the need to form Two emulsions, which can reduce efficiency. Additionally, drug distribution within the microsphere may be uneven, impacting release pro-Files, and some drug loss may occur during emulsification, resulting in Lower encapsulation efficiency. Incomplete solvent removal could also Lead to toxicity concerns. Despite these challenges, the double Emulsion technique remains highly
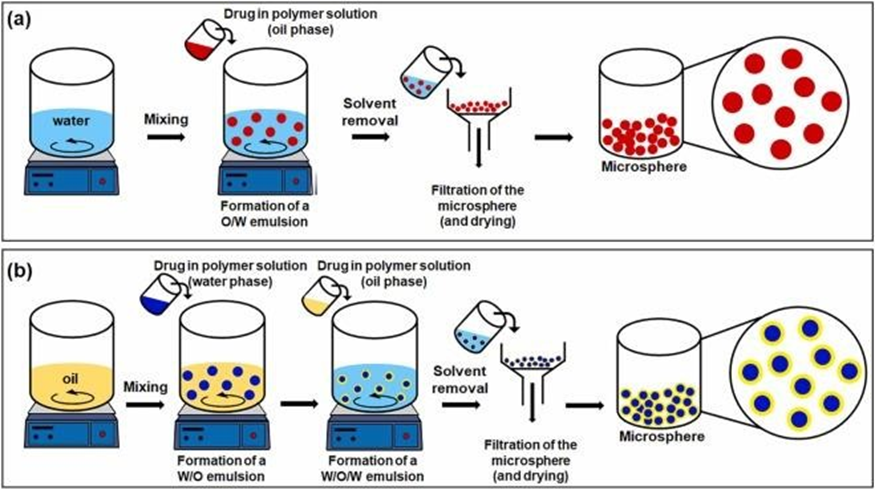
Fig 3: Microsphere fabrication via emulsification technique, using single (O/W) and double (W/O/W, O/W/O) emulsions.
This method enables precise control over microsphere size and morphology by adjusting formulation parameters, allowing for the customization of drug release profiles based on specific therapeutic needs.
Polymers Used in Microsphere Preparation
1. Natural Polymers
Advantages: Biocompatibility, biodegradability, non-toxicity
Limitations: Batch-to-batch variability, microbial contamination [26,27]
2. Synthetic Polymer
Advantages: Controlled release, reproducibility, mechanical strength [6,16,24]
Rationale for Microsphere-Based Topical Delivery
The incorporation of anti-inflammatory drugs into microspheres offers several therapeutic advantages:
Microspheres can be incorporated into conventional topical dosage forms such as gels, creams, or lotions for ease of application.
Evaluation of Microsphere-Based Topical Systems
Particle size plays a crucial role in topical drug delivery as it influences skin penetration, Spreadability, and drug release. Microsphere size is commonly determined using optical microscopy, laser diffraction, or dynamic light scattering techniques. A narrow size distribution is preferred for uniform drug release and better formulation stability.
Surface morphology provides information about the shape, texture, and integrity of microspheres. Scanning Electron Microscopy (SEM) is widely used to evaluate whether microspheres are spherical, smooth, porous, or aggregated. Surface characteristics significantly affect drug release and skin interaction.
The percentage yield indicates the efficiency of the microsphere preparation method. It is calculated by comparing the practical yield of microspheres obtained with the theoretical yield of polymer and drug used. Higher yield reflects a reproducible and economical formulation process.
Drug loading represents the amount of drug incorporated within the microspheres, while entrapment efficiency indicates the effectiveness of the encapsulation process. These parameters are determined by dissolving microspheres and analyzing the drug content using UV–Visible spectroscopy or HPLC.
FTIR analysis is performed to study drug–polymer compatibility. It helps identify any chemical interaction between the drug and excipients by comparing characteristic functional group peaks of pure drug and microsphere formulation.
DSC studies are carried out to evaluate the thermal behavior of microspheres. Changes in melting point or disappearance of drug peaks indicate drug entrapment and possible transformation from crystalline to amorphous form.
In-vitro release studies are conducted using diffusion cells or USP dissolution apparatus. These studies help evaluate the sustained or controlled release behavior of the drug from microspheres. Release data are often fitted to kinetic models such as zero-order, first-order, Higuchi, and Korsmeyer–Peppas models.
Ex-vivo permeation studies are performed using animal or human cadaver skin to assess drug penetration and retention in skin layers. Franz diffusion cells are commonly used to evaluate the topical performance of microsphere formulations.
Skin irritation studies are carried out to ensure the safety of topical microsphere formulations. These studies are usually performed on animal models to evaluate erythema, edema, or any adverse skin reactions.
Stability studies are an essential part of the formulation and development of microsphere-based topical drug delivery systems. These studies help to ensure that the formulation maintains its physical, chemical, microbiological, and therapeutic properties throughout its shelf life under specified storage conditions. Stability evaluation is particularly important for microsphere systems because changes in polymer structure, drug entrapment, or surface characteristics can significantly affect drug release and efficacy.
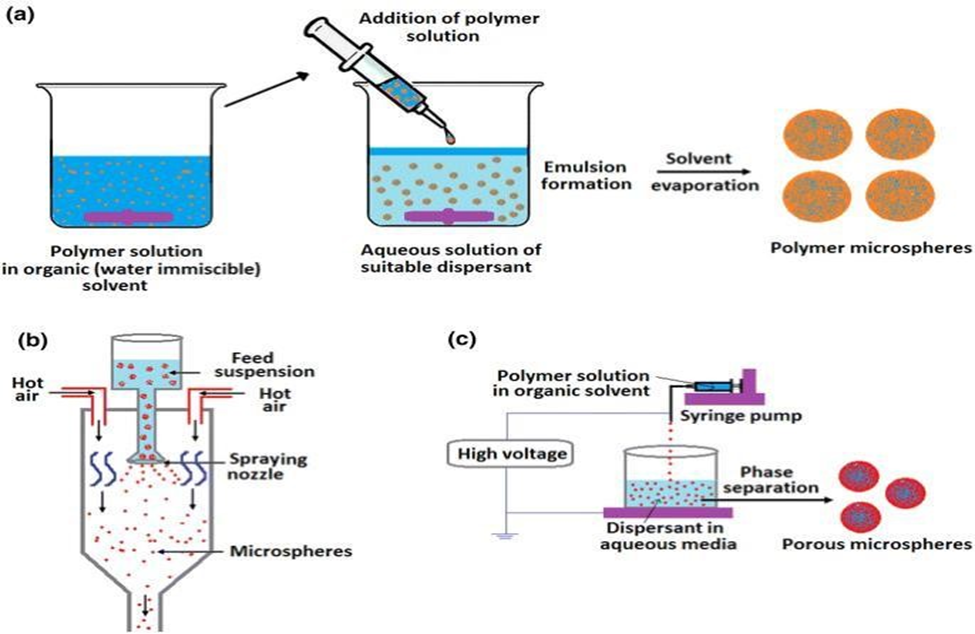
Fig:4: This image describe evaluation of microsphere in different steps.
In Vivo Anti-Inflammatory Effects
Studies demonstrate enhanced anti-inflammatory action with microsphere-based systems: Microsponges-laden gels of Lornoxicam show sustained anti-inflammatory response in carrageenan-induced rat paw edema models.
PMC
Topical microspheres with permeation enhancers significantly reduced edema and provided prolonged effect compared to pure drug formulations. Mechanisms for Prolonged Action Sustained drug release from microspheres extends drug residence at the application site. Slower release reduces peak-to-trough fluctuations. Microspheres act as depots delivering drug over extended periods (8–12+ hours). [32,33]
Tablet:1: Common Anti-inflammatory drugs and their therapeutic use
|
CLASS OF DRUG |
NAME OF DRUG |
DOSAGE FORM |
INDICATION (DISEASE) |
|
NSAID |
Ibuprofen |
Tablet, suspension gel |
Arthritis, pain, fever |
|
NSAID |
Diclofenac |
Tablet, injection gel |
Musculoskeletal, pain, Arthritis |
|
NSAID |
Lornoxicam |
Tablet, injections |
Acute inflammatory pain, Arthritis |
|
NSAID |
Naproxen |
Tablet |
Gout, Rheumatoid arthritis |
|
Cox-2 inhibitor |
Celecoxib |
Capsule |
Osteoarthritis, Rheumatoid arthritis |
|
Corticosteroid |
Prednisolone |
Tablet, Syrup |
Asthma, Autoimmune disorder |
|
Corticosteroid |
Hydrocortisone |
Cream, Injection |
Dermatitis, Skin inflammation |
|
DMARD |
Methotrexate |
Tablet, Injection |
Rheumatoid arthritis |
|
Biological agent |
Infliximab |
Injection |
Crohn’s diseases , Rheumatoid arthritis |
|
Tropical NSAID |
Piroxicam |
Gel, Cream |
Localized inflammation |
Advantages of Microsphere-Based Topical Drug Delivery Systems
Microsphere-based topical drug delivery systems have emerged as an advanced pharmaceutical approach for improving the therapeutic efficacy of drugs intended for local application. These systems involve the incorporation of active pharmaceutical ingredients into polymeric microspheres, which are subsequently dispersed in suitable topical dosage forms such as gels, creams, or ointments. The unique structural and functional properties of microspheres offer several advantages over conventional topical formulations. A major advantage of microsphere-based systems is their ability to provide controlled and prolonged drug release. The polymeric matrix regulates the diffusion of the drug, thereby maintaining a sustained drug concentration at the site of application. This controlled release profile minimizes fluctuations in drug levels and reduces the frequency of administration. Encapsulation of drugs within microspheres significantly enhances drug stability by protecting them from environmental and biological degradation. Factors such as light, moisture, oxidation, and enzymatic activity, which commonly affect topical formulations, are effectively minimized through microsphere entrapment. Microsphere based topical delivery systems also contribute to localized drug action with reduced systemic exposure. By retaining the drug at the application site, systemic absorption is limited, leading to a reduction in systemic side effects and toxicity. This characteristic is particularly advantageous for potent drugs and long-term topical therapies. Another important benefit is enhanced skin retention and penetration. Microspheres adhere to the skin surface and gradually release the drug into the epidermal and dermal layers, resulting in prolonged residence time and improved local bioavailability. This enhances therapeutic outcomes in the management of localized inflammatory and dermatological conditions.
The use of microspheres further helps in minimizing skin irritation and sensitization. Controlled drug release prevents high initial drug concentrations on the skin, which are often responsible for irritation and erythema associated with conventional topical formulations. In addition, microsphere-based systems ensure uniform drug distribution over the skin surface, leading to consistent dosing and predictable therapeutic response. These systems are also formulation-flexible and can be easily incorporated into various topical dosage forms without compromising their physicochemical properties. Improved patient compliance is another notable advantage, as microsphere-based formulations offer better cosmetic acceptability, smooth texture, non-greasy nature, and reduced application frequency. Such characteristics enhance patient adherence to topical therapy.
Future Trends and Directions
1. Advanced polymers and stimuli-responsive systems (e.g., pH or enzyme-triggered release).Integration with nanotechnology and microneedles to enhance skin permeation. Personalized therapy through tunable release profiles based on patient needs. Clinical translation and regulatory approval pathways remain areas for development. Future microsphere-based topical systems may move toward personalized drug delivery, where formulations are tailored based on[42,43]
2. Customized lornoxicam release profiles could significantly enhance treatment effectiveness in chronic inflammatory disorders such as osteoarthritis and localized musculoskeletal pain.
Future research will also emphasize:
Well-designed clinical studies demonstrating superiority over conventional topical formulations will be critical for successful commercialization.
CONCLUSION
Microsphere-based topical drug delivery systems provide a compelling platform to achieve prolonged anti-inflammatory action . These systems can enhance local therapeutic efficacy, reduce systemic exposure, and improve patient compliance. Selection of optimal polymers, preparation techniques, and topical vehicles — along with comprehensive physicochemical and pharmacodynamics characterization — are essential for successful product development. Continued research into skin permeation enhancement and controlled release kinetics will further optimize these formulations for clinical use. [5,7,15]Microsphere-based topical drug delivery systems represent a promising and advanced approach for the effective management of inflammatory conditions. By encapsulating anti-inflammatory drugs within biodegradable and biocompatible polymeric microspheres, these systems successfully overcome several limitations associated with conventional topical formulations, such as poor drug penetration, short duration of action, frequent dosing, and local irritation. The ability of microspheres to provide sustained and controlled drug release enhances therapeutic efficacy while minimizing systemic absorption and adverse effects. Their small particle size, high drug loading capacity, and adaptability to various topical bases (gels, creams, ointments, and patches) make them highly suitable for dermal and transdermal applications. Additionally, the selection of appropriate polymers and formulation techniques allows optimization of drug stability, skin retention, and release kinetics, leading to improved patient compliance.[ 9,21,24]
Overall, microsphere-based topical delivery systems offer a significant advancement in localized anti-inflammatory therapy by ensuring prolonged action, reduced dosing frequency, and improved safety profiles. Continued research focusing on polymer innovation, skin permeation enhancement, large-scale manufacturing, and clinical validation will further strengthen their potential for commercial development and clinical application in the treatment of inflammatory disorders.
REFERENCES



Pratiksha Raut, Prachi Udapurkar, Ravindra Dukare, Microsphere-Based Topical Drug Delivery System for Prolonged Anti- Inflammatory action, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 3321-3332. https://doi.org/10.5281/zenodo.18716001
 10.5281/zenodo.18716001
10.5281/zenodo.18716001