
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Arvind Gavali College of Pharmacy, Jaitapur Satara India.
Among the most promising and flexible nanocarrier systems niosomes are highly biocompatible due to their excellent structural stability and ability to encapsulate hydrophilic and lipophilic therapeutic agents. In this review, composition, techniques of preparation, parameters for characterization and various applications are mentioned. The stability of the bilayer, the effectiveness of drug loading, and the shape of the vesicles depend on the non-ionic surfactants, cholesterol, and charge-inducing molecules. Recent advances in nanotechnology, such as ball milling and microfluidics, which increase their preparation and encapsulation efficiency, enable targeted drug delivery. Their potential for thermo-responsive variants and smart-stimuli systems highlights their innovative role in contemporary medicine. The ideal properties of niosomes like high stability, along with controlled release and reduced systemic toxicity, are used for anticancer therapy, gene delivery, and cosmetics. However, this has some restrictions: vesicle aggregation, drug leakage and in-vitro performance. According to this review, the niosome platform is an active and developing platform that has great potential to play a role in next-generation drug delivery systems due to its ongoing development.
Niosomes have attracted renewed interest due to recent advances in pharmaceutical nanotechnology, especially the last two decades, which have described the preparation techniques, merits, demerits, applications and comparison of niosomes with other nanocarriers. Niosomes are special nanocarrier, in which the active ingredients are entrapped in a vesicle made up of non-ionic surface-active agents. The study of niosomes is gaining much importance in advanced drug delivery, as they are nanoscopic in size. Recent developments like novel ball milling and microfludization have improved control the particle size and encapsulation efficiency.1iosomes are non-ionic spherical surfactants, which are safer and relatively less toxic than liposomes, and have sizes ranging from 20 to 1000 nm, making them nanoparticles and/or nanostructures. The polar and non-polar drugs are entrapped by the niosomes with equal efficiency.2 Niosomes are made from biodegradable and biocompatible materials and are used to load various drugs and allow targeted delivery by improving the chemical and physical stability for the pharmaceutical products.3 The additives for niosome include non-ionic surfactants to form a film, cholesterol to stabilise and rigidize the membrane, and the charge inducers, which helps in inducing a charge on the niosomes and stabilises the formulation.4 The niosomes represent a flexible stage with the ability to revolutionize drug delivery in multiple therapeutic areas, with potential to alter the drug release, enhance their firmness, and delivery to targeted locations. Niosomes have advantages over conventional drug carriers, like enhanced bioavailability and more accurate targeting. Green synthesis of niosomes by using biodegradable surfactant enhances the safety and suitability.5 Various drugs can be encapsulated in a niosome system, which can make them more biocompatible and effective, as the bilayer structure of these niosomes can accommodate polar and non-polar drugs. The major limitations that led to this change in focus was the instability of liposomes.6Advancement in niosomes will result in the development of a smart-stimuli niosomal system that inhibits ovarian cancer cells, MCF10-A cells and MCF10-A cells by biofunctionalizing curcumin, with potential for more potent tumour-inhibitory effects, and also the formation of the thermo-responsive niosomes using Pluronic® L64 and L64ox and the development of a nano silica substrate for HER2-positive breast cancer treatment.7
Composition of niosomes
Niosomes achieve thermodynamic stability through a bilayered structure formed by non-ionic surfactants, which can be produced by combining non-ionic surfactants with cholesterol at temperatures exceeding the gel-liquid transition point.
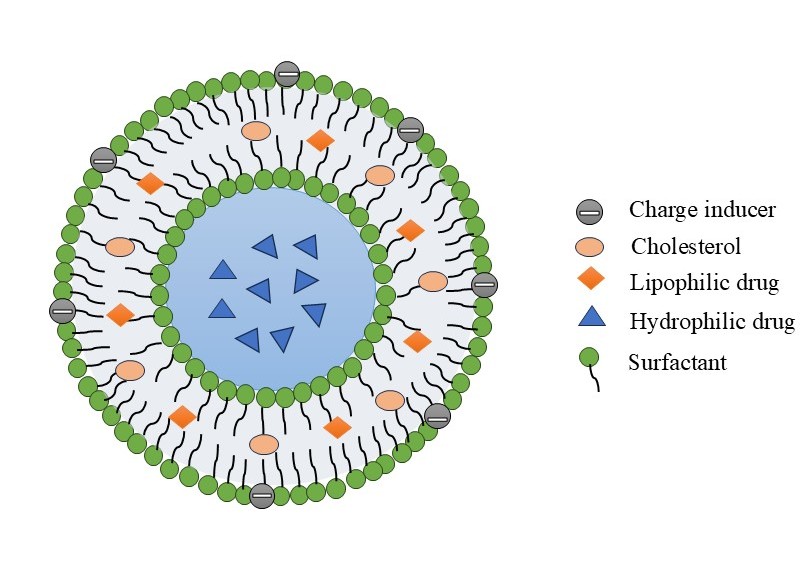
Figure 1: Composition of Niosomes
They have been frequently involved in niosome synthesis. Non-ionic surfactants are those that lack charged groups in their hydrophilic heads. They are less toxic, more steady and more bioabsorbable than anionic, amphoteric, or positive equivalents. Alkyl ethers, alkyl esters, alkyl amides, and fatty acids are the main non-ionic surfactant classes used in niosome production.8,27
Steroidal precursor cholesterol is used to strengthen and synthesize niosomal preparations. The non-ionic surfactants frequently contain a cholesterol, which provides them stiffness and spatial order. It can be absorbed at high molar ratios and has no effect on the bilayer. Additionally, it has been established that cholesterol inhibits leaks by preventing the conversion of gel to liquid.22
Introducing the charged molecules to the bilayer membrane makes the niosomes more stable. They reduce vesicle aggregation by raising their surface charge density. The stearyl amine and stearyl pyridinium chloride are positively charged substances; dicetyl phosphate is the negatively charged substance. Typically, 2.5–5% of the charged molecule are present in the niosomal formulation.8
Advantages of niosomes:
Disadvantages of Niosomes:
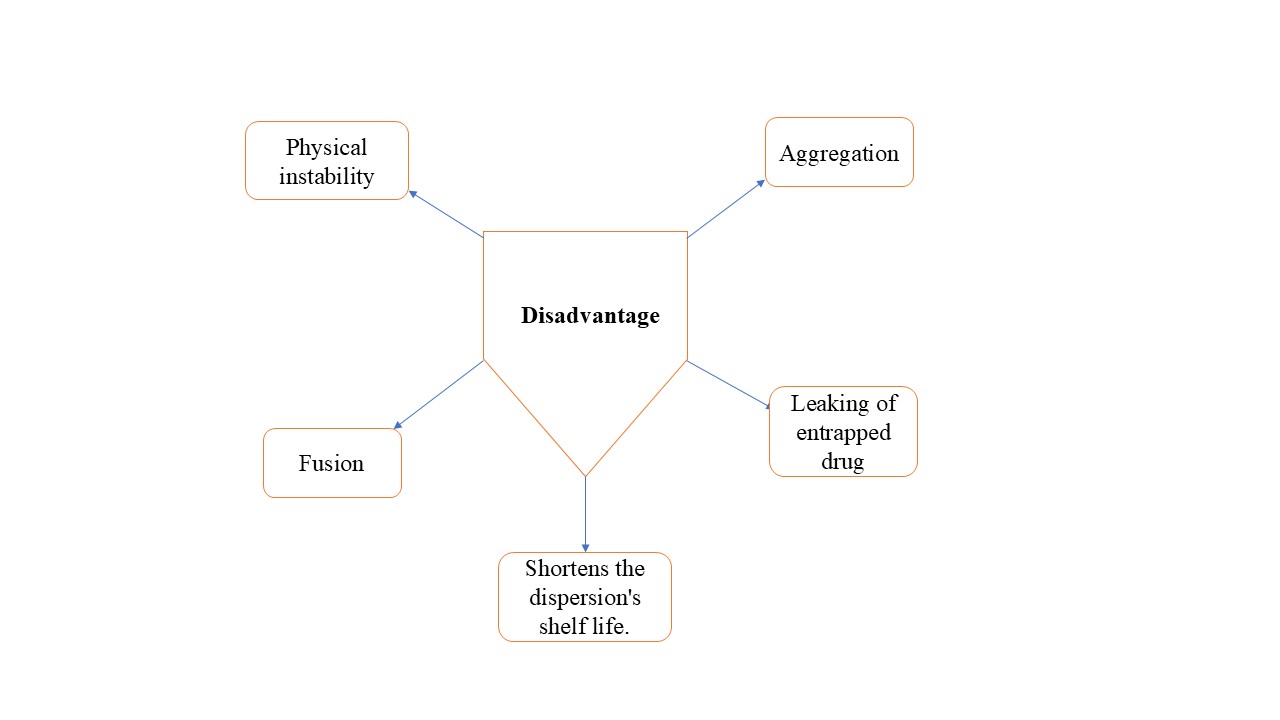
Figure 3: Disadvantages of niosomes 11
Niosomes Preparation Methods:
Niosomes are typically made by solvent evaporation to form a lipid film, followed by hydration. Variants of this technique, however affect sizes, number of bilayers, and drug entrapment effectiveness. Intended niosome application determines which preparation technique should be used.12
The methods of preparation are:
|
|
Dissolve phospholipids in the organic solvent like (ethanol/methanol/CHCl3). This solvent helps to dissolve lipids completely. |
Evaporation of solvent (rotary evaporation)
|
|
The solvent evaporates once the phospholipids dissolve in it. The lipid then forms a thin, dry coating on the inner wall of the round-bottom flask when the solvent is removed.
|
Hydration with Aqueous/Phosphate Buffer + Drug
|
|
Water or buffer are used to hydrate the lipid film. Multilamellar vesicles are formed and trap hydrophilic drugs in aqueous core. |
|
|
Large multilamellar vesicles are broken down into small unilamellar vesicle.12 |


Types of Niosomes
Table 1: Types of specialized niosomes
|
SR NO. |
TYPES |
INFORMATION |
|
|
Proniosomes
|
These are formed by coating of non-ionic surfactant to a carrier; the carrier must be nontoxic, safe, free flowing, and easily hydrated, and substances such as sorbitol, mannitol etc are used to synthesize them. This niosomes are usually in the form of dry mill which have certain merits over other types, including lower tendency for aggregation, and reduced drug leakage.19
|
|
|
Aspasomes
|
Ascorbyl palmitate, cholesterol along with charged lipid are mixed to form this niosome, which are then added into aqueous medium but niosomes are obtained after sonication. They can be utilized for enhancement of topical formulations.20 |
|
|
Elastic Niosomes.
|
They maintain their structural integrity while being deformable resulting, elastic niosomes to exit the pores that are smaller than them. Their capacity to penetrate the skin is enhanced by their structural flexibility.21
|
|
|
Niosomes in Carbopol Gel
|
The medication, non-ionic surfactant, and cholesterol were used to create niosomes in this system, which were subsequently mixed with a Carbopol gel along with glycerol.22 |
|
|
PEGylated Niosomes
|
These are tissue compatible, biocompatible niosomes with a polyethylene glycol modification. This prolongs drug's circulation time and increases its accumulation. They are typically employed when a medication has low bioavailability and poor solubility.15
|
B) Specialized niosomes
These nanoparticles range from 1 to 100 nanometers in size and are made of elements such as nickel, iron, or cobalt, etc. The tiny size, magnetic reactivity, and adaptability of MNPs make them particularly precious. MNPs are used therapeutically to target cancer cells more precisely by delivering medications to cells and causing localized warmth for cancer treatment. MNPs are an effective tool for biomedical exploration because of their numerous uses.24
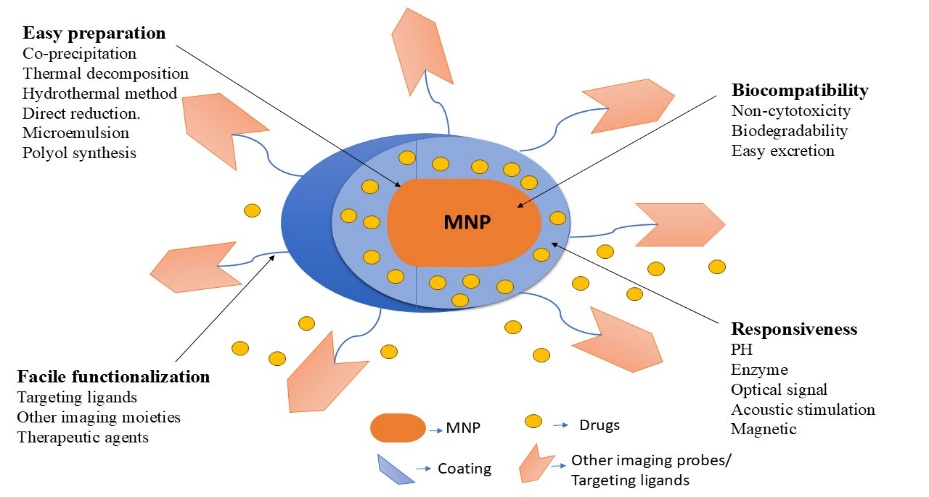
Figure 5: Key characteristics of a magnetic niosomes.
Super magnetic NiCoFe2O4 nanoparticles can be used to produce a magnetic niosome nanocarrier, followed by the development of a niosomal structure. This novel method makes it possible to load drugs onto the produced nanocarrier and combat breast cancer. The generated nanocarriers exhibited substantial cell killing for investigated MDA-MB-231 and SK-BR-3, but they were largely biocompatible with non-tumorigenic cell lines.25
Thermosensitive niosomes are advanced nanocarriers which release active material in reply to mild hyperthermia (generally 40–42 °C) in a controlled manner by using the high-temperature medium of tumors or externally induced hyperthermia.26Temperature-controlled release can be achieved using various compounds, including non-ionic surfactants, various polymers, and phase transition material. Since PCMs melt and release medications quickly at transition temperatures, they are potential tools for building advanced delivery. Their natural availability, low toxicity, and biodegradability enable to regulate a release of pharmaceuticals in desired location, enhancing medical ability, and minimizing negative effects.28 In various class of PCMs, a focus is on fatty acids, high availability, and lower price.27Because of the limited variety in natural fatty acids, it is challenging to manufacture PCMs at melting points close to body temperature using a one component. To overcome the problems, researchers suggested using binary types of fatty acids. So, the eutectic mixture can be created for intelligent thermoresponsive niosomes.28
Immuno-niosomes were created by conjugating niosomes with antibodies, and cyanuric chloride derivatization of Tween was utilized to bind IgG antibody to carrier periphery. It has been found that PEGylation of immuno-niosomes improves binding efficiency and protects the proteins that trigger the immunogenic response.29Kolahdooz H et al. created curcumin-niosomes (CUR-NIO) by film hydration method and mixed with prepared formulation. Comparing the curcumin niosome treated group with placebo-treated group, there was a noticeable improvement and reduction in redness and scaling. So, results of gene expression analysis showed that in lesions treated with CUR-NIO, IL17, IL23, IL22, TNF, S100A7, S100A12, and Ki67 were dramatically downregulated. Therefore, by inhibiting the IL17/IL23 immunopathogenic axis, CUR-NIO may offer treatment strategies for patients with mild-to-moderate PS.30
pH-sensitive niosomes are stimulant-responsive nanocarriers that can adjust drug release in reaction to pH changes in the surrounding environment. pH-responsive lipids or polymers like chitosan, cholesteryl hemisuccinate, or Eudragit derivatives are added to these vesicles.31,32The most popular cytobiocompatible polymers for pH sensitive niosome peripheral modification is polyethylene glycol (PEG), a hydrophilic, non-ionic polymer. PEG-modified niosomes improve the in vivo t1/2 of drug delivery vesicles because they are identical to the reticuloendothelial system.33 Even though vesicles have significant potential as medication carriers, their short blood circulation time and lack of stability in blood continue to be significant disadvantages for their use in vivo. It has recently been demonstrated that adding polyethylene glycol (PEG)-derived lipids to vesicular bilayers can prevent the reticuloendothelial system from quickly absorbing vesicles, leading to a significantly prolonged in-vivo circulation half-life. The negative impact of PEGylation on pH-sensitivity is a major worry for pH-sensitive vesicles and other polymer derivatives. The partial loss of pH responsiveness of polymer-coated vesicles during incubation in biological fluids is another problem.31
Factors affecting niosome preparation:
The drug interacts with the head of surfactant when trapped inside the niosome causing the repulsion between layers of surfactant, making the vesicles bigger. Some drugs are held inside PEG-coated niosomes, which helps prevent the vesicles from growing too much. 34
2. Amount of surfactant:
The mean amount of niosomes rises when the HLB value of surfactants such as Span 85 (HLB 1.8) and Span 20 (HLB 8.6) increases. This is since as the hydrophobic nature of the surfactant increases, the surface free energy drops. The vesicles' bilayers can be liquid or gel depending on the temperature, type of lipid or surfactant, and presence of additional components such as cholesterol.35
The presence of cholesterol affects the hydrodynamic diameter and trapping effectiveness of niosomes. Cholesterol causes the chain order of gel-state bilayers to drop and that of liquid-state bilayers to increase.37
A diameter of niosome suspension reduces as hypertonic salt fluid is mixed. In hypotonic salt solution, vesicles first release slowly and swell slightly, most likely as a result of the resistance of liquid eluting out of the nanocarrier.38
Characterization:
The morphology of niosomes can be identified by using various methods like, light microscopy, SEM (scanning electron microscope), and TEM (transmission electron microscope).39
Drug loading (%DL) was calculated as sum of total of entrapped drug by the total mass of nanoparticles and multiplying by 100.40
Vesicle dispersions are examined with a light microscope to see the formation and shape of the vesicles. Niosomes were studied by optical microscope under magnification up to 100 X.41
This can be indirectly identified by using size exclusion chromatography and dialysis methods, which both allowed separation of the unloaded drug from niosomes.42
In vitro discharge. Dialysis tubing is one way. The distilled liquid is utilized to wash and immerse a dialysis bag. The vesicular dispersion is pipetted in a tube sack and then closed. The vesicle-filled bag is placed in a 250-milliliter beaker containing 200 ml of the buffer mixture and agitated continuously until it reaches 25°C or 37°C. A appropriate test technique is employed to assess the buffer's medication content at different intervals.43
In vivo and in vitro, behaviour of niosome may be significantly influenced by vesicle surface charge. Compared to uncharged vesicles, charged niosomes are typically more stable against aggregation and fusion. Microelectrophoresis can be used to measure the zeta potential of individual naocarrier in order to guess the peripheral potential. The dynamic light scattering is used recently for zeta potential.44
The formation of a bilayer in niosomes can be seen using polarizing light microscopy. This is a characteristic of non-ionic surfactants assembling into bilayer structures.46
Application
Gene delivery is efficient system for treating genetic human illnesses by using non-vector carriers which enhance uptake of nuclear material by cells. Despite that niosomes have been around for nearly thirty years, very little study has been done to explore their eventuality as gene delivery vectors. Because niosomes contain non-ionic surfactants, which provides greater storage capacity than liposomes and help lower cost of niosome synthesis and their toxicity.
These properties motivate researcher to use niosomes in delivery of a gene. In reported studies, niosomes were used to deliver the pCMS-eGFP plasmid into retina.47
In recent times, the main goal of many cosmetic formulations has been to target oxidative stress. The use of flavonoids, which are naturally occurring antioxidants that scavenge free unstable radicle that cause oxidation of skin. When compared to a free resveratrol solution, Tavano et al.'s resveratrol-entrapped niosomes demonstrated regulated and improved percutaneous penetration. Additionally, they found that when drug is trapped with alpha-tocopherol and curcumin, niosomes capacity to scavenge free radicals is enhanced, as they show synergistic action.48
Drug-resistant tumor interactions, uncontrolled release, and untargeted delivery are only a few of the challenges associated with cancer therapy. With a high success rate, niosomes have been used to deliver cytotoxic medications selectively to particular tumor locations. Because they are more selective and have different pharmacokinetics and biodistribution, encapsulated anticancer medications enhance their therapeutic impact. Based on kind of tissue and the physical and chemical properties of the niosome, these medications can be administered by various methods. The term "passive delivery" describes the accumulating of nanocarrier inside tumor as a result of the malignant neoplasm's cellular features. Conversely, energy dependent delivery describes formative alteration of this nanocarrier with a compound/ligand so that nanocarrier are taken up by tumor cells.The characteristics of surrounding in which niosomes are injected, and substance are delivered via physical stimulant, determine physical delivery. Because of the features of the tumor surrounding, the passive delivery makes it easier for niosomes to work there by facilitating their uptake by cancer cells.15
Increasing selectivity towards the targeted tissue and lowering medicine toxicity are the main goals of a targeted drug delivery system. In 1909, Paul Ehrlich developed a way for delivering medication right to sick tissues .The medicine was delivered to the target cells and tissues using a variety of mechanisms, including immunoglobulins, liposomes, microspheres, and niosomes. Liposomes and niosomes have received special interest because to their unique properties and stability. Niosomes can trap water-soluble and lipophilic drugs. When non-ionic surfactants and hydrocarbons are present, niosomes perform better as drug carriers for tumors, the liver, and the nervous system. These days, they are frequently employed in medication targeting to more successfully treat viral, parasitic, cancerous, and other microbial disorders.49
The basis of the immune response elicited by pathogens was examined using niosomes. Niosomes can be used to deliver drugs to organs other than the reticuloendothelial system. Niosomes may be targeted to certain locations by connecting a delivery mechanism (such as antibodies) to them, as antibodies bind readily to the fatty coating on niosomes.50
After substantial study and inquiry into niosome preparations over the past few years, only a few studies have progressed to clinical studies using niosome nanocarriers. Topical administration was the main focus of a large portion of the clinical studies. Recent research over the past five years shows that adding medications to niosomes has improved therapeutic efficacy and decreased negative effects.51
CONCLUSION
The niosomes are the strong and adaptable nanocarriers that overcome many of the challenges that are seen in conventional drug delivery. The advantages of their structural stability, biocompatibility, and dual drug-loading capability are crucial for improving therapeutic efficacy and modifying pharmacokinetics. Advances in formulation science (microfluidic fabrication, ball-milling technology, PEGylation, and functionalization of ligands) have opened up new avenues for niosomal applications—in oncology, dermatology, gene therapy, and immunomodulation. Specialized niosomes like thermoresponsive and pH-responsive niosomes, are becoming more important in targeted delivery medicine. Recent studies show that niosomes are a promising and continuously evolving platform that may significantly influence next-generation drug delivery.
REFERENCES




Atharav Mane, Akash Bhise, Vishal Yadav, Vasant Lokhande, Niosomal Nanocarriers in Drug Delivery: Composition, Preparation, Stimuli-Responsive and Applications, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 4, 22036-2049, https://doi.org/10.5281/zenodo.19552968
 10.5281/zenodo.19552968
10.5281/zenodo.19552968