
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Ashvin College of Pharmacy, Manchi Hill, Sangamner, Ahilyanagar, Maharashtra 413714
Adhatoda vasica, commonly known as Vasaka or Malabar nut, is an important medicinal plant widely used in traditional Ayurvedic medicine. It belongs to the family Acanthaceae and is well known for its therapeutic effects, especially in the treatment of respiratory disorders. The plant contains several bioactive compounds such as vasicine, vasicinone, and essential oils that contribute to its pharmacological properties. Traditionally, Adhatoda vasica has been used for the treatment of cough, asthma, bronchitis, and other respiratory diseases due to its bronchodilator, expectorant, and antitussive activities. In addition, the plant exhibits antimicrobial, anti-inflammatory, and antioxidant properties, making it valuable in herbal medicine. Various parts of the plant, including leaves, roots, and flowers, are used in herbal formulations and pharmaceutical preparations. This review highlights the phytochemical constituents, pharmacological activities, and therapeutic importance of Adhatoda vasica, emphasizing its significance as a natural medicinal resource.
For an estimated 75–80% of people worldwide, mostly in developing countries, herbal medicine continues to be their main source of healthcare [1]. According to the WHO, traditional medicine refers to methods that have been used for hundreds of years prior to modern biomedicine and are still in use today. Adhatoda vasica Nees is one of the most prominent plants in this global pharmacopoeia; in Hindu medical literature, it is often referred to as the "ruler of herbs" because of its unmatched therapeutic range [1].
The word's origin Note: The term "Adhatoda" comes from the Tamil words "Adha" (goat) and "toda" (won't touch). Because of its extremely bitter alkaloid content, goats and other animals steer clear of the plant.
In Sanskrit literature, the plant is called "Vasa," which reflects its main use as an expectorant. The Sanskrit word "vasaka" describes the same plant that is used in traditional Ayurvedic texts like the Charaka Samhita and Ashtanga Hridayam as a. first-line treatment for Tamaka shwasa (bronchial asthma) and Kasa (cough) [2, 3].
2. GLOBAL AND ETHNOBOTANICAL DISTRIBUTION
A. vasica is unquestionably one of the most culturally significant medicinal plants, according to ethnobotanical surveys conducted in a variety of geographical locations:
3. BOTANICAL CLASSIFICATION, MORPHOLOGY & MICROSCOPY
3.1 Taxonomic Classification
|
Taxonomic Rank |
Taxon / Notes |
|
Kingdom |
Plantae |
|
Division |
Angiosperms |
|
Class |
Eudicots (Liliopsida in some classifications) |
|
Order |
Lamiales |
|
Family |
Acanthaceae |
|
Genus |
Justicia |
|
Species |
J. adhatoda L. |
|
Synonyms |
Adhatoda vasica Nees; Adhatoda zeylanica Medic. |
3.2 Vernacular Names Across Languages
|
Language / Region |
Vernacular Name(s) |
|
Hindi |
Adosa, Adalsa, Vasaka |
|
Sanskrit |
Shwetavasa, Vasa, Vasaka, Vaidyamatasinghee |
|
Bengali |
Basak, Vasa |
|
Tamil |
Adatodai |
|
Kannada |
Adusoge |
|
Malayalam |
Ata-lotakam |
|
Telugu |
Adasaram |
|
Gujarati |
Aradusi, Adusa |
|
Punjabi |
Bansa, Basuti, Bhekkar |
|
English |
Malabar Nut |
|
Chinese |
Ya-Zui-Hua |
|
Arabic |
Adusha |
|
Nepali |
Asuro |
|
Assamese |
Boga |
|
Oriya |
Basango |
|
Manipuri |
Nongmangkha-agouba |
|
Konkani |
Adulasha |
3.3 Macroscopic Morphology
A. vasica is a dense, evergreen, perennial shrub that usually grows to a height of 1.2 to 2.5 meters. In the right circumstances, it can grow up to 6 meters in height. Important morphological characteristics consist of [1, 2, 3].
Stem & Bark: Younger sections are tomentose; the bark is smooth, round, and yellowish; there are numerous long, opposing, ascending branches.
Leaves: Acuminate, opposite, elliptic-lanceolate, 10-20 cm x 5-9 cm. Paler underneath, darker above. Glabrous when mature, slightly pubescent when young. 1-2.5 cm petioles and 10-13 pairs of secondary veins.
Inflorescence: Acuminate, opposite, elliptic-lanceolate, 10-20 cm x 5-9 cm. Paler below, dark green above. When young, slightly pubescent; when mature, glabrous. 1-2.5 cm petioles; 10-13 pairs of secondary veins.
Flowers: Subsessile, bisexual, zygomorphic, hypogynous. Calyx campanulate, 5-lobed (up to 1.2 cm). Corolla bilabiate, white with a pinkish-violet throat, 2.5–3 cm, pubescent outside; tube inflated above.
Stamens: 2, inserted at the corolla throat; filaments are hairy at the base; anthers are 2-celled.
Ovary: Bicarpellary, syncarpous, 2-celled, superior, tomentose; 2 ovules per cell. Style: filiform.
Fruit: Clavate capsule ~2 cm, contracted below into a solid stipe. Seeds: 1–2 per capsule, 5–6 mm, sub-orbicular, compressed, rugose.
Propagation: Hardwood cuttings (15-20 cm, 3-4 nodes, spring/early summer) or seeds (most common).thrives at elevations of up to 1,300 meters in wasteland, roadsides, and dry, stony soils [1].

Fig. 2: Different parts of Adhatoda vasica — plant habit, flower, seeds, leaf, and roots. Source: Hussain et al. (2025) [1]
3.4 Microscopic Characters
A highly ordered tissue architecture can be seen in the transverse section (T.S.) through the midrib of the leaf. [3]:
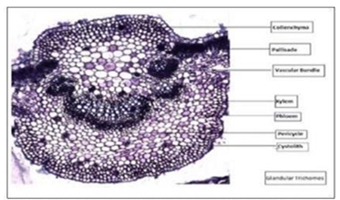
Fig. 3: Microscopic T.S. of Justicia adhatoda leaf through midrib showing collenchyma, palisade, vascular bundles, cystoliths,and glandular trichomes. Source: Varsha et al. (2025) [3]
4. PHYTOCHEMICAL CONSTITUENTS:
All of the main plant parts, including leaves, roots, bark, wood, flowers, fruit, and seeds, have undergone phytochemical analyses using HPLC, HPTLC, capillary electrophoresis, UV, mass spectrometry, and NMR spectroscopy [1, 2, 3]. Quinazoline alkaloids are the main class of bioactive substances. Flavonoids, terpenoids, steroids, fatty acids, and vitamins [2, 3]. Quinazoline alkaloids are the main class of bioactive substances. Flavonoids, terpenoids, steroids, fatty acids, vitamins, essential oils, and phenolics are some of the other classes. The most complete integrated phytochemical profile from all three source papers is shown in Table 1 below.
Table 1: Integrated phytochemical profile of Adhatoda vasica across all plant parts. Sources: [1,2,3]. FA = Fatty acid.
|
Plant Part |
Compound |
Mol. Formula |
M W (g/mol) |
Class |
Pharmacological Role |
|
Leaves |
Vasicine (Peganine) |
C11H12N2O |
188.23 |
Quinazoline alkaloid |
AChE inhibitor; Major alkaloid (0.85%); bronchodilator, antitussive, uterotonic, anti-inflammatory [1,2,3] |
|
Leaves |
Vasicinone |
C11H10N2O2 |
202.21 |
Quinazoline alkaloid |
Bronchodilator (1959); hepatoprotective; hypotensive; anticholinesterase [1,2,6] |
|
Leaves |
Adhatodine |
C20H21N3O2 |
355.41 |
Quinazoline alkaloid |
Immunomodulatory; antiinflammatory activity [1,2] |
|
Leaves |
Adhavasicinone |
C12H12N2O3 |
232.24 |
Quinazoline alkaloid |
Anti-inflammatory [1,2] |
|
Leaves |
Anisotine |
C20H19N3O3 |
349.39 |
Quinazoline alkaloid |
Anti-tuberculosis (docked against FabH); anti-inflammatory [1,14] |
|
Leaves |
Hydroxypeganine |
C11H12N2O2 |
196.21 |
Quinazoline alkaloid |
Anticholinesterase; CNS activity [1,2] |
|
Leaves |
Vasicinol |
C11H14N2O2 |
206.24 |
Quinazoline alkaloid |
Antioxidant; antimicrobial; AChE inhibition [2,3] |
|
Leaves |
Vasicol |
C11H12N2O2 |
204.23 |
Quinazoline alkaloid |
Anti-asthmatic derivative; bronchospasmolytic [2,3] |
|
Leaves |
Deoxyvasicine |
C11H12N2 |
172.23 |
Quinazoline alkaloid |
Anti-TB; molecular target: antigen 85C of M. tuberculosis [35] |
|
Leaves |
Vasicoline |
C11H14N2 |
174.24 |
Quinazoline alkaloid |
Anti-TB; docking studies confirm inhibitory potential [14] |
|
Flower |
Kaempferol |
C15H10O6 |
286.24 |
Flavonol |
Potent antioxidant; anti-inflammatory; anticarcinogenic potential [1,3] |
|
Flower |
Quercetin |
C15H10O7 |
302.24 |
Flavonol |
Anti-inflammatory; antiviral; cardioprotective; aldose reductase inhibitor [1,3] |
|
Flower |
Apigenin |
C15H10O5 |
270.24 |
Flavone |
Anxiolytic; anti-inflammatory; neuroprotective; anticancer [1,3] |
|
Flower |
Vitexin |
C21H20O10 |
432.38 |
C-glycosylflavone |
Antioxidant; antidiabetic; antiinflammatory; neuroprotective [1,3] |
|
Flower |
Astragalin |
C21H20O11 |
448.38 |
Kaempferol-3-glucoside |
Antioxidant; antiobesity; anti inflammatory [1] |
|
Flower |
4-dihydrochalcone-4'-glucoside |
C21H22O8 |
402.40 |
Chalcone glycoside |
Novel antioxidant flavonoid [1,2] |
|
Root |
Vascine (Vasicine) |
C11H12N2O |
188.23 |
Quinazoline alkaloid |
7.5% in roots — much higher than leaves; same broad activity [1] |
|
Root |
Vasicinal |
— |
— |
Quinazoline alkaloid |
Related alkaloid from root [1] |
|
Root |
Vasicinolone |
C11H12N2O2 |
204.23 |
Quinazoline alkaloid |
Anti-asthmatic [1] |
|
Root |
Daucosterol |
C34H59O6 |
563.84 |
Sterol glycoside |
Antitumor; antiinflammatory; immunomodulatory [1] |
|
Root |
beta-Sitosterol |
C29H50O |
414.72 |
Phytosterol |
Anti-inflammatory; lipid-lowering; anti-cancer [1,2] |
|
Root |
D-Galactose |
C6H12O6 |
180.16 |
Monosaccharide |
Carbohydrate component; structural role [1] |
|
Root |
Deoxyvasicinone |
C11H10N2O |
186.21 |
Quinazoline alkaloid |
Anti-TB molecular target [1,35] |
|
Root |
Vitamin C |
C6H8O6 |
176.12 |
Ascorbic acid |
Antioxidant; immune support [1] |
|
Seeds |
Oleic acid |
C18H34O2 |
282.47 |
Monounsaturated FA |
49.9% of seed oil; cardioprotective; antiinflammatory [1] |
|
Seeds |
Linoleic acid |
C18H32O2 |
280.45 |
Omega-6 PUFA |
12.3%; essential FA; immune function; skin health [1] |
|
Seeds |
Behenic acid |
C22H44O2 |
340.59 |
Saturated FA |
11.2%; hair conditioning; skin barrier [1] |
|
Seeds |
Lignoceric acid |
C24H48O2 |
368.65 |
Saturated FA |
10.7% of seed oil [1] |
|
Seeds |
Arachidic acid |
C20H40O2 |
312.54 |
Saturated FA |
3.1%; eicosanoid precursor [1] |
|
Seeds |
beta-Sitosterol |
C29H50O |
414.72 |
Phytosterol |
2.6%; anti-inflammatory [1] |
|
Whole plant |
Saponins, Tannins, Phenolics |
Various |
— |
Polyphenols |
Free-radical scavenging; antioxidant; antimicrobial; anti-typhoid [1,2,3] |
|
Aerial parts |
3-hydroxy-D-friedoolean-5-ene |
— |
— |
Triterpenoid |
Identified alongside epitaraxerol and peganidine; antiinflammatory [3] |
5. MAJOR ALKALOIDS — DEEP-DIVE MECHANISM ANALYSIS
5.1 Vasicine (Peganine) — The Master Alkaloid
Vasicine (1,2,3,9-tetrahydropyrrolo[2,1-b] quinazolin-3-ol; C??H??N O; MW 188.23) is the principal bioactive [11,12]. Compound of A. vasica, accounting for approximately 0.85% of the dry weight of the leaves and remarkably up to 7.5% of the roots [1]. It is a member of the pyrroloquinazoline class and is a levorotatory, optically active substance. It is one of the most adaptable plant alkaloids known due to its wide pharmacological profile.
5.2 Vasicinone — The Bronchodilator Pioneer
Vasicine's oxidized form is called vasicinone (C11H12N2O; MW 202.21). It was A. vasica's first alkaloid to [11,10, 22] have its bronchodilatory action officially recorded by Amin & Mehta in 1959 [6], signaling the start of the pharmaceutical history of A. vasica. It makes up around 0.027% of the dry weight of leaves.
Bronchodilator (1959 onward): Current and historical in vitro and in vivo data support the relaxation of bronchial smooth muscle [6].
Hepatoprotective synergy: In the CCl? model, vasicinone and silymarin (25 mg/kg/day) have a greater hepatoprotective synergy, lowering hepatic enzyme levels than silymarin alone [11].
Hypotensive: significant cardiovascular pharmacology that causes hypotension in cat models, contracts the intestine of an isolated guinea pig, and depresses the heart of an isolated frog [1, 2].
Anticholinesterase: inhibits AChE and, when combined with vasicine, enhances the anti-Alzheimer profile [12].
Vasicine — Confirmed Mechanisms of Action
Bronchodilation: Inhibits phosphodiesterase and activates the beta-adrenergic pathway to relax bronchial smooth muscle [6, 7].
Similar to codeine in mechanical, electrical, and irritant aerosol models, antitussive drugs work by suppressing the cough reflex by acting on the neuronal system [7, 19].
Uterotonic/ Oxytocic: Promotes uterine contraction by releasing prostaglandin (PG); similar to oxytocin and methyl geometries; strengthened by estrogen priming. [9]
Reversible and competitive, AChE inhibition (anti-alzheimer); IC50~294 µg/mL; binding profile similar to tacrine and galantamine [12,13].
Anti-tuberculosis: Molecular docking finds the antigen; vasicine acetate inhibits M.Tuberculosis (MDR strains). The main goal is 85C [16, 35].
Anti-inflammatory: at 20 mg/kg, carrageenan inhibits paw edema by 59.51%; this is equivalent to hydrocortisone in HET-CAM examination [15, 27]
In irradiated and CCl4-intoxicated models, the antioxidant DPPH radical scavenging and dosedependent FRAP lower LPO [20, 21].
Ethanolic extract exhibited 60-70% anti-implantation activity in female albino rats [2].
5.3 Landmark Clinical Translation — Bromhexine & Ambroxol
The semi-synthetic derivatives of vasicine from A. vasica are ambroxol and bromhexine, two of the most commonly prescribed mucolytic/expectorant medications in the world [8]. Translation mechanism: Both substances show pH dependent M. tuberculosis growth inhibition in Increase the amount of lysozyme in bronchial secretions, concentrate in alveolar macrophages, and facilitate removal of mucus containing bacilli from the lung cavities and bronchi [8, [8, 17]. This is among the best instances of ethnopharmacology being directly translated into mainstream international clinical medicine, demonstrating the efficacy of conventional Ayurvedic treatment.
5.4 Analytical Extraction — Traditional vs Modern Methods
A comparison of four extraction techniques by Soni et al. [29] demonstrated unequivocally that the traditional Putapak Vidhi (bolus) method produces the highest alkaloid content:
The following contemporary analytical techniques have been validated for the quantification of vasicine/ vasicinone: capillary electrophoresis (which separates in 11 minutes with high reproducibility), HPTLC [29], and HPLC. Alkaloid content has also been shown to vary seasonally; alkaloid accumulation peaks during specific seasons, which is a crucial factor to take into account when sourcing pharmaceutical raw materials [2].
6. PHARMACOLOGICAL AND THERAPEUTIC ACTIVITIES:-
6.1 Respiratory - Anti-asthmatic, Bronchodilator & Antitussive
The most well-established and historically recorded therapeutic area of A. vasica is respiratory pharmacology. Its effectiveness in a variety of respiratory conditions has been confirmed by 60 years of research involving controlled clinical studies, in vitro, and in vivo studies [1, 2, 3].
Anti-Asthmatic Evidence:
In guinea pigs with acetylcholine- and histamine-induced bronchospasm, ethanolic extract (250, 500, and 750 mg/kg) showed a spasmolytic effect comparable to ketotifen (1 mg/kg) [1].
In a mouse model of acute allergic asthma caused by an OVA allergen, aqueous extract (130 mg/kg) markedly reduced elevated airway resistance and inflammation [1].
Asthma symptoms were significantly reduced in both Vasa Avaleha (Swarasa vs. Kwath preparation) clinical trials; however, Swarasa (aqueous extract) was superior in terms of symptom relief and hematological parameters [2].
Rats' LPO caused by ovalbumin and aluminum hydroxide was decreased by the herbal compound "Pentapala04" (containing A. vasica), maintaining membrane integrity and averting lung damage [2].
RLX (6,7,8,9,10,12-hexahydro-azepino-[2,1-b]-quinazoline12one) was produced by chemically modifying vasicine. Similar to disodium cromoglycate + aminophylline with additional oral efficacy, oral RLX (10-20 mg/kg) inhibited antigen-induced mast cell degranulation and histamine secretion [2].
Antitussive Evidence — Comparable to Codeine [7,19]:
When rabbits and guinea pigs are made to cough mechanically or electrically, intravenous extract is 1/20-1/40 as effective as codeine [1].
In guinea pigs, oral extract exhibited antitussive activity similar to codeine against aerosols produced by irritants [1,7].
In a citric acid-induced cough model, arabinogalactan from A. vasica showed 67% cough suppression (compared to 62% for codeine) [2, 19].
Petroleum ether extract decreases secretions by 78.5%, 47%, and 36%, respectively; it increases the secretion of respiratory tract fluids more than ammonium chloride and eucalyptol [1].
Acute Upper Respiratory Tract (URT) Infection — Clinical RCT [18]:
The fixed herbal combination of A. vasica + Echinacea purpurea + Eleutherococcus senticosus was compared to the Echinacea mixture alone (standard control) and bromhexine in a randomized controlled trial called the Kan Jang trial. Compared to those receiving standard treatment, patients who received the A. vasica combination exhibited noticeably more improvement. The echinacea mixture's effectiveness was clearly increased by the addition of A. vasica extract, indicating synergistic action [18].
6.2 Anti-tubercular Activity — Molecular Mechanisms
A. vasica exhibits clinically significant anti-tuberculosis potential, especially when it comes to multi-drug resistant (MDR) strains of M. tuberculosis, which are a global health emergency. Evidence includes molecular docking simulations and in vitro research [14, 16, 17, 35].
Key Findings — In Vitro [17]:
Molecular Targets — Computational Evidence [14,35]:
FabH (beta-ketoacyl-ACP synthase III): uses type II fatty acid synthase (mtFabH) to catalyze the initial committed step of fatty acid biosynthesis in M. tuberculosis. Vasicoline, vasicinone, vasicine, and anisotine are alkaloids from A. vasica that exhibit inhibitory docking scores against this vital enzyme [14].
Antigen 85C of M. tuberculosis: determined to be the most effective biological target of vasicine and deoxyvasicine using comparative molecular docking [35].
Bromhexine and ambroxol, semi-synthetic derivatives, concentrate in macrophages, increase lysozyme in bronchial secretions, and aid in the removal of mucus containing bacilli -potentially helpful adjuvant therapy in tuberculosis [8].
6.3 Anti-inflammatory & Antioxidant — Pathways & Evidence
Targeting several biochemical pathways at once, A. vasica exhibits multi-modal anti-inflammatory and antioxidant properties, which is a significant benefit over synthetic medications that only target one [1, 2, 3].
Anti-inflammatory Mechanisms:
In the carrageenan-induced paw edema model, the most effective alkaloid tested, vasicine (20 mg/kg), achieves 59.51% inhibition at 6 hours postinjection [15].
Alkaloid fraction demonstrated activity comparable to hydrocortisone at 50 µg/pellet dose in the modified HETCAM test [15].
Aqueous fraction: Inhibits the arachidonic acid (AA) pathway, simultaneously affecting the enzymes cyclooxygenase (COX), thromboxane, and platelet-activating factor (PAF) [3].
Butanol fraction: Compared with metabolites of arachidonic acid to delineate mechanism [3].
In rat paw carrageenan and formalin models, ethanolic extract (200-400 mg/kg, oral) exhibited notable antiinflammatory activity [3].
Antioxidant Mechanisms:
Vasicine exhibits dose-dependent Ferric Reducing Antioxidant Power (FRAP) assay activity and significant DPPH radical scavenging [27].
Cytochrome P450, NADPH-cytochrome P450 reductase, cytochrome b5, glutathione S-transferase, DTdiaphorase, SOD, catalase, glutathione peroxidase, and glutathione reductase are significantly elevated in hepatic enzyme modulation [20].
Cadmium-induced stress model: LPO and xanthine oxidase are markedly inhibited; antimutagenic efficacy is achieved through restored antioxidant status [21].
Fe-NTA renal carcinogenesis model: chemopreventive and antioxidant effects against two-stage carcinogenesis and hyperproliferative response [2].
Hepatotoxicity from CCl4: Pretreatment raises liver tissue's levels of SOD, catalase, and GSH. [1]
Aflatoxin B1 detoxification: A. vasica's aqueous leaf extract specifically detoxifies aflatoxin B1, which has uses in hepatoprotection and food safety [2].
6.4 Hepatoprotective & Gastrointestinal Activity
A. vasica's traditional use in liver-related conditions is supported by its strong, multiply-validated hepatoprotective activity across a number of hepatotoxicity models [1, 2, 3].
Hepatoprotective Evidence:
CCl4 model: In Swiss albino rats, ethyl acetate extract (100-200 mg/kg) considerably preserves liver architecture and restores normal transaminase levels [1, 3].
D-galactosamine model: Below 100 mg/kg, aqueous leaf extract (50-100 mg/kg, oral) exhibits a notable hepatoprotective effect. Scientific evidence supports traditional use [1, 10].
Vasicinone + silymarin synergy: Hepatic enzyme elevations are lessened by pretreatment with vasicinone (25 mg/kg/day) + silymarin than by silymarin alone, indicating additive/synergistic hepatoprotection [11].
Perchloroethylene model: Highly elevated biochemical levels are returned to normal by ethanolic vasaka extract [1].
Whole plant powder: More proof of hepatoprotection from whole-plant preparations in various models of liver disorders [1].
Gastrointestinal Evidence:
Anti-ulcer (Ethanol model): In comparison to the pyloric ligation + aspirin model, leaf powder (500 mg/kg in
0.2% agar) exhibits the highest activity of any tested model, at 80% [1, 30].
Anti-Helicobacter pylori: Significant antiurease activity against H. pylori is demonstrated by A. vasica extract, which mechanistically supports anti-ulcer activity [2].
Non-ulcer dyspepsia (Amlapitta) — Clinical trial: A total positive clinical effect was confirmed when Vasa syrup (60 mL dry leaf extract, 6 divided doses, 6 weeks) significantly reduced pyrosis, burning sensation, free HCI levels, flatulence, and constipation [2].
Cholagogue effect (Dogs & Cats): Intravenous 5 mg/kg increased bilirubin excretion and bile excretion by 40100% [1].
6.5 Neuroprotective & Anti-Alzheimer Activity
One of A. vasica’s most intriguing new research topics is its neuroprotective profile. Both anti-Alzheimer potential and protection against diabetes-related cognitive decline are supported by a number of convergent mechanisms [12, 13].
Anti-Alzheimer Mechanisms [12,13]:
The main enzyme that authorized Alzheimer's medications target, AChE, is reversibly inhibited by an alcohol extract of A. vasica [12].
Active ingredients that contribute to ACHE inhibition include vasicine, vasicinone, vasicole, and anisotine [12].
Molecular docking has confirmed that vasicine's binding at the AChEACHE catalytic site is similar in profile to that of approved Alzheimer's medications tacrine and galantam [12]. • IC = 294 µg/mL (in vitro) for whole plant AChE inhibition [1]. 50
Diabetic Encephalopathy Model-STZ rats [13]:
For six weeks, AVEE (100, 300, and 400 mg/kg/day) resulted in statistically significant improvements:
AChE activity: Significantly normalized by AVEE after elevated 70% in the diabetic cortex [13].
LPO: Increased 94% (hippocampus) and 100% (cortex) → decreased toward normal [13].
Nitrite levels: significantly attenuated, elevated 170% in the cortex and 137% in the hippocampal region [13].
AVEE restores SOD, catalase, and GSH, which are reduced in diabetic brains [13].
TNF-alpha: markedly increased in diabetic rats markedly reduced by AVEE [13].
Behavioral assessments (passive avoidance, Y-maze): Notable gains in memory and learning [13].
6.6 Uterine & Reproductive System Effects
As an antifertility agent, A. vasica has a long history in both Ayurvedic and Unani traditions. The picture painted by scientific evidence is complicated, with some contradicting results that need to be carefully interpreted [1, 2, 9, 26].6.
Uterotonic action: Similar to oxytocin and methylergometrine, vasicine's uterotonic effect was investigated in vitro and in vivo across rat, hamster, guinea pig, and rabbit uteri under various hormonal influences [9].
Abortifacient action: More pronounced under oestrogen priming; mediated via prostaglandin (PG) release [9].
In vitro oxytocic: At concentrations greater than 1 mg/mL, synthetic vasicine and vasicinone derivatives exhibit oxytocic effects [1].
Abortive effect: Rats given leaf extract at a dose of 175 mg/kg 10 days after insemination exhibited a 100% abortive effect [2].
Anti-implantation: In female albino rats, ethanolic extract demonstrated 60-70% anti-implantation activity [2].
Survey evidence: According to a 1987 survey of women in Uttar Pradesh, A. vasica is one of the most often used herbs for anti-reproductive purposes [2].
6.7 Antimicrobial, Antiviral & Anti-Quorum Sensing
Antibacterial [1,2,3]:
Strong resistance to Bacillus subtilis, Proteus vulgaris, Pseudomonas aeruginosa, Staphylococcus aureus, S. epidermidis, Streptococcus faecalis, Micrococcus luteus, and E. coli.
Strong antibacterial activity against E. coli is demonstrated by vasicine (20 µg/mL) [2].
Vasicine acetate: Good zone of inhibition against P. aeruginosa, S. epidermidis, and Enterobacter aerogenes [2].
A. vasica leaf extract was used to create green-synthesised silver nanoparticles (average 20 nm, range 5-50 nm), which have strong antibacterial activity against P. aeruginosa (disc diffusion, agar cup, MIC methods) and are produced in an economical and environmentally responsible manner [28].
Antiviral — Influenza [24]:
Methanolic extract: 100% decrease in hemagglutination (HA) at 10 mg/mL in influenza virus tests conducted both concurrently and after treatment.
Aqueous extract: 16.67% (5 mg/mL); 33% ?? reduction (simultaneous assay, 10 mg/mL); dual mechanism of inhibiting viral attachment AND replication cycle [24].
Anti-Quorum Sensing [2]:
Anti-quorum sensing (AQS) activity against Chromobacterium violaceum-a novel antibacterial mechanism-is demonstrated by A. vasica leaf extract. Bacteria can coordinate antibiotic resistance and virulence through quorum sensing. By interfering with this communication without actually killing the bacteria, AQS agents lessen the selection pressure for the development of resistance, which is a tactical advantage in the fight against MDR pathogens [2].
6.8 Thrombolytic, Immunomodulatory & Radioprotective
Thrombolytic Activity [34]:
Methanolic fraction (MF): in vitro clot lysis model: 53.23% clot lysis vs. 80.65% (streptokinase positive control) vs. 4.08% (distilled water negative control) [34].
The clot lysis activity of root extract (5 mg/mL) was 19.63%, which is highly significant when compared to a normal saline control [2].
Mechanism: Fibrinolytic enzyme activation and platelet aggregation inhibition are probably involved [2, 34].
Immunomodulatory Activity [3]:
Leaf extracts containing methanol, chloroform, and diethyl ether (400 mg/kg oral, male Wistar rats):
→ Neutrophil adhesion to nylon fibers was significantly elevated (P<0.001) [3].
→ Sheep erythrocytes were used to induce a delayed type hypersensitivity (DTH) reaction (P<0.001) [3].
→ Positive immunomodulation of host immunity was verified, and results were significant compared to controls at all tested doses [3].
Radioprotective Activity [22,23]:
Mice given ethanolic leaf extract for 15 days in a row before being exposed to 8 Gy radiation:
→ In untreated mice, mortality was 100% by day 22, but it dropped to 70% at day 30 [22].
→ significantly avoided chromosomal abnormalities in bone marrow cells caused by radiation [22]. Testicular histology has been shown to be protected [22].
A. vasica leaf extract (800 mg/kg): Restored normal hematological parameters (GSH, LPO) in animals exposed to radiation [23].
6.9 Antidiabetic, Wound Healing & Anthelmintic
Antidiabetic / Anti-glucosidal [1,31]:
A. vasica methanolic leaf extract demonstrated the highest sucrose inhibitory activity among 40 traditional herbs screened for rat intestinal alpha-glucosidase inhibition, suggesting clinical potential for postprandial glucose control [31].
In STZ-diabetic rats, AVEE (ethanolic extract) also reverses diabetic encephalopathy markers (AChE, LPO, nitrite, TNF-alpha) [13].
Wound Healing [1]:
1% methanolic extract ointment: Excision model (Swiss albino mice) showed strong wound healing activity [1].
In calves with wounds caused by the vertebral column, alcoholic extract of A. vasica heals wounds better than chloroform extract [1].
Mechanism: Probably involves angiogenesis, re-epithelialization, and collagen synthesis [1].
Anthelmintic Activity [3,25]:
Significant ovicidal and larvicidal effects at 25-50 mg/mL (P<0.05); dose-dependent inhibition against gastrointestinal nematodes (sheep) in vitro [3].
Ethanolic extract (50 mg/mL): Best at preventing larval development and egg hatching [3].
Root powder: The most effective in vivo treatment for nematodes [25].
Anticestodal: Hymenolepis diminuta infections in rats can be effectively treated with A. vasica extract [2].
6.10 Antifungal, Cholagogue, Anti-cancer & Oral Health
Antifungal: Vasicine is effective against Candida albicans (>55 µg/mL), while alkaloid extract is effective against Aspergillus ruber and Trichophyton rubrum [2,3].
Aflatoxin B1 detoxification: Aflatoxin B1 is specifically neutralized by aqueous extract, which has important implications for food safety [2].
Cholagogue (dogs & cats): IV 5 mg/kg 40-100% increase in bile flow; increased excretion of bilirubin modulation of bile acid metabolism [1].
Anti-cancer / Cytotoxic: Cytotoxic studies (Duraipandiyan et al.) confirmed that vasicine acetate exhibits cytotoxic efficacy against the lung adenocarcinoma cell line through an antioxidant mechanism [2].
Antigingival / Oral Health (RCT [32]): Significant decreases in plaque index, bleeding on probing, pocket depth, and aerobic/anaerobic bacterial counts compared to a placebo were observed in a 12-week double-blind RCT of herbal toothpaste containing A. vasica [32].
Pyorrhea: Twenty-five patients had their inflamed gums massaged with leaf extract twice a day for three weeks, which resulted in a notable decrease in gum bleeding and inflammation [1].
Acaricidal: Tetranychus urticae, the red spider mite, is effectively repelled by A. vasica extract [2].
Larvicidal: Potential for controlling public health mosquitoes: Anopheles stephensi, Culex quinquefasciatus (the vector of filariasis), and Aedes aegypti (the vector of dengue) [2].
MASTER PHARMACOLOGICAL ACTIVITY TABLE :-
Table 2: Master summary of pharmacological activities with key compounds, evidence, models, and references. Sources: [1–35].
|
Activity |
Key Compound(s) |
Evidence / Findings |
Model / Dose |
Ref |
|
Anti-asthmatic |
Vasicine, Vasicinone |
Comparable to 1 mg/kg of ketotifen in cases of bronchospasm in guinea pigs; clinical RCT versus bromhexine |
Guinea pig, mice, Clinical RCT |
[1,2,18] |
|
Bronchodilator |
Vasicinone (1959), Vasicine |
Initially documented in 1959 (Amin & Mehta); verified both in vitro and in vivo; patent for an RLX derivative |
In vivo/in vitro |
[6,2] |
|
Antitussive |
Vasicine, Vasicinone, Arabinogalactan |
67% suppression (AV) versus 62% (codeine); 1/20–1/40 codeine potency in IV extract |
Guinea pig, rabbit |
[7,19,2] |
|
Anti-TB (MDR) |
Vasicine acetate, Alkaloids |
Vasicine acetate > 2-acetyl benzylamine at all concentrations; 86% inhibition of the JAL-1236 MDR strain |
In vitro L-J medium |
[16,17] |
|
Anti-TB (Molecular) |
Vasicine, Deoxyvasicine, Anisotine |
Antigen = strongest docking target; FabH enzyme inhibition |
In silico / docking |
[14,35] |
|
Anti-inflammatory |
Vasicine alkaloids, Aqueous fraction |
Carrageenan inhibits paw edema by 59.51%, which is comparable to hydrocortisone (HET-CAM) |
Rat models, In vitro |
[15,3] |
|
Antioxidant |
Vasicine, Flavonoids |
DPPH, FRAP, SOD, catalase, GSH elevation; LPO reduction |
Multiple models |
[20,21,27] |
|
Hepatoprotective |
Vasicinone, Leaf extracts |
Vasicinone + silymarin synergy, CCl4, D-galactosamine, and perchloroethylene models |
Rat, Mouse |
[10,11,1] |
|
Anti-ulcer |
Leaf powder |
80% activity (highest among those tested) in the ethanol-induced ulcer model |
Rat (500 mg/kg) |
[1,30] |
|
Anti-Alzheimer |
Vasicine, Vasicinone |
ACHE IC50: 294 µg/mL; comparable docking to Galantamine/tacrine |
In vitro, In silico |
[12,13] |
|
Diabetic encephalopathy |
AVEE (all alkaloids) |
ACHE, LPO, nitrite, TNF-alpha, and SOD levels in the STZ diabetic brain were normalized |
STZ rat (100–400 mg/kg) |
[13] |
|
Uterotonic |
Vasicine |
Comparable to oxytocin; mediated via PG release; 100% abortive at 175 mg/kg |
Multiple species |
[9,2] |
|
Antiviral (influenza) |
Methanolic extract |
100% HA reduction at 10 mg/mL; inhibits viral attachment and replication |
In vitro |
[24] |
|
Antibacterial |
Vasicine, Alkaloids, Ag-NPs |
Active vs P. aeruginosa, S. aureus, E. coli, MDR strains; Ag-NPs (20 nm) potent |
In vitro |
[2,28] |
|
Anti-quorum sensing |
Leaf extract |
Disrupts C. violaceum communication; novel anti-virulence mechanism |
In vitro |
[2] |
|
Thrombolytic |
Methanolic fraction |
53.23% clot lysis vs 80.65% streptokinase control |
In vitro clot model |
[34] |
|
Immunomodulatory |
Methanolic/ chloroform/ ether extracts |
↑ Neutrophil adhesion (P<0.001); DTH reaction confirmed |
Wistar rats 400 mg/kg |
[3] |
|
Radioprotective |
Ethanolic extract |
Mortality 70% at 30 d (vs 100% untreated at 22 d); chromosomal protection |
Swiss albino mice 8 Gy |
[22,23] |
|
Antidiabetic |
Methanolic leaf extract |
Highest alpha-glucosidase inhibition among 40 herbs tested |
Rat intestinal assay |
[31] |
|
Anthelmintic |
Aqueous & ethanolic extracts |
Significant ovicidal, larvicidal at 25–50 mg/mL (P<0.05) |
Sheep nematodes in vitro |
[3,25] |
|
Antifungal |
Alkaloid extract, Vasicine |
Active vs Aspergillus ruber, Trichophyton rubrum, C. albicans |
In vitro |
[2,3] |
|
Anti-cancer |
Vasicine acetate |
Cytotoxic vs lung adenocarcinoma cell line; antioxidant mechanism |
Cell line |
[2] |
|
Oral health |
Herbal toothpaste with AV |
↓ Plaque index, bleeding, pocket depth; ↓ bacterial counts (12-week RCT) |
Double-blind RCT |
[32] |
|
Wound healing |
1% methanolic ointment |
Strong wound healing in excision model; alcoholic extract superior in calves |
Mouse, Calf models |
[1] |
|
Larvicidal |
Leaf extract |
Effective vs Anopheles stephensi, Culex quinquefasciatus, Aedes aegypti |
In vitro |
[2] |
7. CHEMICAL STRUCTURES - VISUAL REFERENCE
The chemical structures of the main alkaloids and phytoconstituents of A. vasica are shown in the following figures, which were taken straight from the original publications. The pharmacological activities listed in Section 6 are directly caused by these structural characteristics.
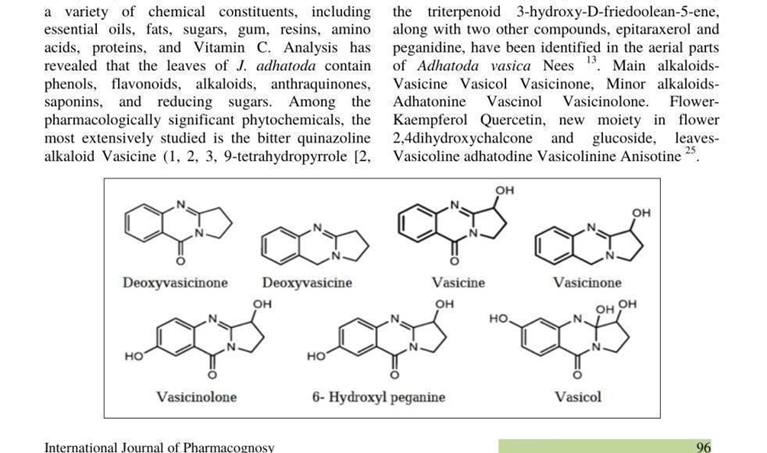
Fig. 4: Chemical structures of major alkaloids of Adhatoda vasica — Deoxyvasicinone, Deoxyvasicine, Vasicine, Vasicinone, Vasicinolone, 6- Hydroxyl Peganine, and Vasicol. Source: Varsha et al. (2025) [3]
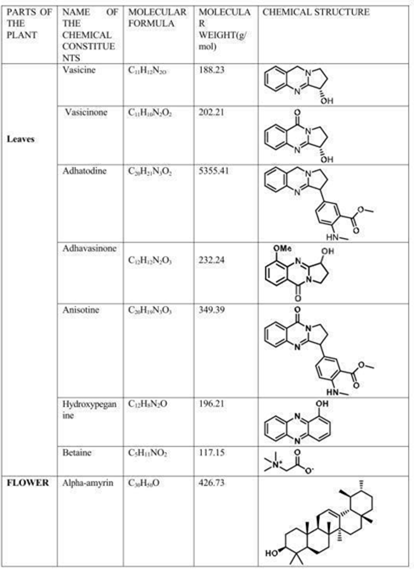
Fig. 5: Molecular structures and chemical data of leaf alkaloids (Vasicine, Vasicinone, Adhatodine, Adhavasicinone, Anisotine, Hydroxypeganine, Betaine) and flower triterpene (Alpha-amyrin). Source: Hussain et al. (2025) [1]
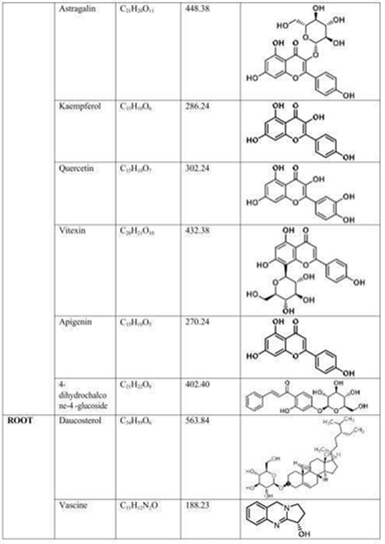
Fig. 6: Chemical structures of flower flavonoids (Astragalin, Kaempferol, Quercetin, Vitexin, Apigenin, 4dihydrochalcone4'glucoside) and root constituents (Daucosterol, Vascine). Source: Hussain et al. (2025) [1]
8. COMPARATIVE ANALYSIS — THREE SOURCE PAPERS:-
In order to find distinctive contributions, areas of agreement, and methodological variations, this section methodically compares and contrasts the three integrated review papers.
Table 3: Comparative analysis of three source review papers on Adhatoda vasica [1,2,3]
|
Aspect |
Hussain et al. 2025 [1] (IJES, India) |
Kapgate & Patil 2017 [2] (IJGP, Pune) |
Varsha et al. 2025 [3] (IJP, Karnataka) |
|
Primary focus |
phytochemical + pharmacological activities |
Therapeutic spectrum survey; phytochemical tables |
Critical ethnopharmacological + analytical review |
|
Alkaloid data depth |
Full table: molecular formulas |
weights, structures for all parts; analytical methods comparison |
(Putapak vs modern); seasonal variation |
|
Unique contribution |
Detailed chemical structure table; diabetic encephalopathy model; safety profile section |
analytical review |
— |
|
Clinical evidence |
Mostly experimental; some clinical RCTs cited |
Strongest: multiple clinical references |
clinical doses and duration specified |
|
Anti-TB coverage |
Basic alkaloid docking mention |
vasicine acetate, bromhexine/ambroxol |
molecular docking |
|
Safety profile |
Yes – uterotonic; minor GI at high doses |
Most detailed: hepatotoxicity, urticaria, exanthema |
Minimal safety discussion; contact dermatitis (Sweden) |
|
References |
47 citations |
85 citations (most comprehensive) |
31 citations |
|
WHO mention |
Primary Health Care Manual |
Both Primary Health Care + Fertility Research Programme |
Brief mention |
|
Ethnobotany |
Geographic distribution, vernacular names listed |
Extensive: 7 regions, index values, veterinary use |
Brief cultural information |
|
Formulations |
7 listed |
Putapak Vidhi traditional method detailed |
Not specifically listed |
9. TRADITIONAL FORMULATIONS — AYURVEDIC & UNANI:-
More than twenty Ayurvedic formulations contain A. vasica, mostly in the form of Vasa swarasa (leaf juice). Different preparation techniques that maximize various therapeutic qualities of the plant have been developed by both the Ayurvedic and Unani systems [1, 2].
|
Formulation Name |
Description, Uses & Evidence Reference |
|
Syrup Basakarista |
Ayurvedic liquid preparation with fermentation for bronchitis, asthma, and cough. requires a lengthy fermentation process with jaggery. |
|
Basadi Kwath |
A. vasica leaf decoction. Standard comparison preparation used in asthma clinical trials [2]. |
|
Vasa Avaleha |
Preparation of confections and decoction. Effectiveness for asthma symptoms was validated by a clinical RCT. The Kwath form is superseded by the Swarasa form [2]. |
|
Vasa Swarasa (Putapak Vidhi) |
The highest alkaloid content was found in traditional bolus extraction (5.93 mg/mL total alkaloids; 5.64 mg/mL vasicine). Gold standard preparation [2, 29]. |
|
Sarbat Ejaz |
Unani syrup; upper respiratory infections. |
|
Sarbat Tulsi |
Preparing a combination of herbs (Vasa and Tulsi); treating fever and cough. |
|
Sarbat Sadar |
Unani expectorant formulation. |
|
Sarbat Vasac |
Core Unani vasaka-based syrup; bronchitis and asthma. |
|
Basaboleho |
Ayurvedic bolus for respiratory conditions. |
|
Vasa Ghrita |
Ghee-based preparation for chronic respiratory conditions. |
|
Modern Herbal Toothpaste |
Significant decreases in plaque, bleeding, and bacterial counts were confirmed by a 12-week double-blind RCT for gingival health [32]. |
|
Vasicine Derivatives (Pharmaceutical) |
Global over-the-counter and prescription medications bromhexine and ambroxol are semi-synthesised from vasicine [8]. |
REFERENCES







Aniket Nirmal, Hindavi Adhav, Krishna Vishwasrao, Rutik Datir, Sanskar Khemnar, Tejashri Kudnar, Yuvraj Mhase, Phytochemical and Pharmacological Review of Adhatoda vasica (Vasaka) and its Therapeutic Applications, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 4, 1268-1287. https://doi.org/10.5281/zenodo.19475233
 10.5281/zenodo.19475233
10.5281/zenodo.19475233