
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Shivlingeshwar College of Pharmacy, Almala, Latur
In pregnancy, changes are known to occur because of demand-related changes in hormones, the hcg stimulatory effect, and iodine requirements, resulting in thyroid enlargement with changes in TSH, T4, and T3 levels. Hypothyroidism is the most common endocrine cause of thyroid disease during pregnancy, seen in 2-3 percent as overt hypothyroidism and up to 10 percent as subclinical hypothyroidism, particularly in areas where iodine deficiency is a public health problem. This reduces the pregnancy outcomes to an increased risk of miscarriages, preeclampsia, growth restriction of the fetus, low birth weights, and neurodevelopmental problems for the offspring. Hyperthyroidism, resulting from either grave’s disease or thyrotoxicosis, increases the risk of premature births, low birth weights, and thyroid storms. Autoantibodies, such as TPO, are known to be the cause for miscarriages and preterm births. Diagnosis requires trimester-specific TSH/FT4 range, as current assays fail with TBG and estrogen elevation. Management of hypothyroidism includes an immediate rise in the dose of levothyroxine by 20-40%, while TSH is checked at regular intervals of 4 weeks. Management of hyperthyroidism includes the preferential use of PTU in the first trimester to avoid.Early treatment, as well as proper dose adjustments, are therefore important for the maintenance of maternal euthyroidism.
Thyroid hormones are crucial for metabolism, growth, and brain development, and even mild thyroid dysfunction during pregnancy can cause serious maternal and fetal morbidity. Pregnancy is a condition of physiological thyroid stimulation because of elevated estrogen levels, increased thyroid-binding globulin, and the stimulatory effect of Human chorionic gonadotropin (hCG). These effects cause an increase in thyroxine (T4) and triiodothyronine (T3) production and an increase in the size of the thyroid gland by 10% in iodine-sufficient areas and up to 20-40% in iodine-deficient areas [1].
The iodine requirement also increases by nearly 50% to fulfill the demands of the mother and the fetus. Although iodine-sufficient women can easily cope with the demands of pregnancy, iodine-deficient women are more susceptible to thyroid dysfunction. Overt thyroid disease affects 2-3% of pregnancies, whereas subclinical thyroid disease affects nearly 10% of pregnant women [1][3]. Hypothyroidism is the most prevalent thyroid disease in pregnancy, with a prevalence of 2.5% to 11%, especially in the Asian population compared to Western countries [3].
Untreated or undertreated hypothyroidism has been associated with miscarriage, preeclampsia, anemia, fetal growth restriction, placental abruption, preterm labor, low birth weight, and neuropsychological dysfunction in the child [1][2]. Subclinical hypothyroidism, defined as elevated serum levels of thyroid-stimulating hormone (TSH) and normal levels of free thyroxine (FT4), occurs in 3-5% of pregnancies, while overt hypothyroidism, defined as elevated TSH and low FT4, occurs in 0.3-0.5% of pregnancies [3].
In 2024, a cohort study was conducted on pregnant women at a tertiary facility in Gujarat, India. The study evaluated 900 women who were pregnant prior to 20 weeks of gestation, and because the comorbidities of pregnant women in the past had been significant barriers to their participation in studies, the originator of this study excluded many comorbidities from the evaluation to establish the causes of outcomes attributed to thyroid dysfunction. The results of this study support previous findings that indicate pregnant women with thyroid dysfunction present at a significantly greater frequency than women without thyroid dysfunction with complications at delivery (i.e., premature delivery and low APGAR scores) and after delivery (i.e., preeclampsia and postpartum complications) [2].
Epidemiology of Thyroid Disorders in Pregnancy
The prevalence of thyroid dysfunction is variable worldwide and is known to be strongly affected by iodine intake. Both iodine deficiency and excess are known to increase the risk of hypothyroidism and autoimmune thyroiditis. A large population-based study has shown increased rates of subclinical hypothyroidism and autoimmune thyroid disease in areas of iodine excess compared with mildly iodine-deficient regions [4]. These observations emphasize the need to achieve optimal iodine intake.
Autoimmune thyroid disease (AITD) is known to be more prevalent in infertile women, especially in those with polycystic ovary syndrome or endometriosis. While AITD is not known to affect implantation, it is known to substantially increase the risk of miscarriage in the first trimester, even in euthyroid women.
The baseline level of thyroid-stimulating hormone (TSH) can predict the development of thyroid dysfunction in patients with thyroid autoantibodies who are exposed to high iodine intake [4].
Assisted reproductive technology (ART) and ovarian hyper stimulation may also affect thyroid function, especially in patients with pre-existing AITD. Hence, thyroid evaluation before and during fertility treatment is advised to minimize pregnancy complications [5].
Physiology of Thyroid function in pregnancy
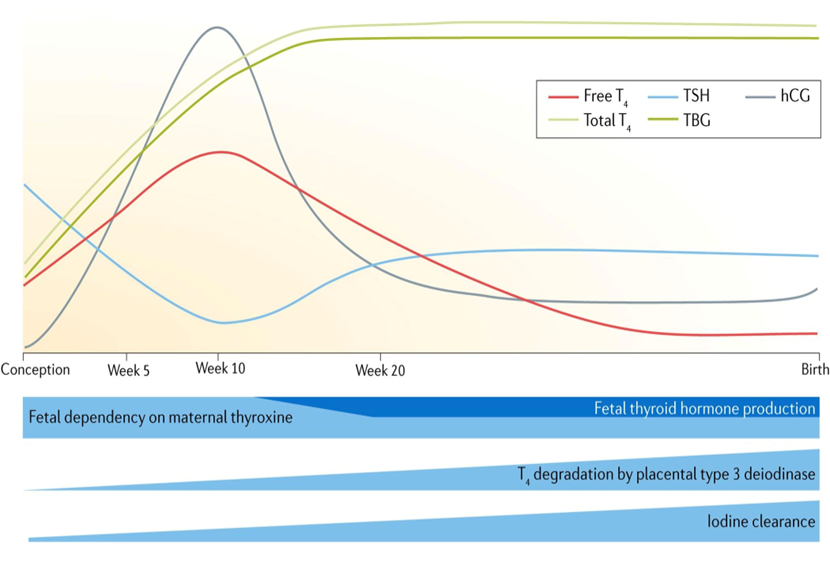
Pregnancy is a state of active endocrine adaptation, wherein the thyroid gland of the pregnant woman undergoes pronounced but reversible changes. These changes are caused by the elevated levels of estrogen, which in turn stimulate the liver to produce TBG, resulting in an increase in the total levels of thyroxine and triiodothyronine.
Estrogen and Thyroid-Binding Globulin (TBG) :-
The elevated levels of estrogen in pregnancy stimulate the liver to produce TBG, resulting in an increase in the total levels of thyroxine and triiodothyronine. However, the levels of free hormones remain relatively constant due to compensatory feedback mechanisms. This explains why the total levels of thyroid hormones increase, but it does not necessarily mean that the patient is hyperthyroid [6].
Increased Iodine Requirements:-
Pregnancy increases the demand for iodine by around half due to the increased clearance of iodine from the bloodstream and the difference in levels of fetal thyroid hormones. The World Health Organization suggests that pregnant women have a daily intake of 250 µg of iodine. Insufficient iodine intake can cause problems for the mother, including maternal hypothyroxinemia, and can also impact the developing baby’s brain development, especially in areas where there are very low levels of iodine in the diet [7].
Trimester-Specific Variations:-
Thyroid function varies significantly from trimester to trimester. In the first trimester, levels of TSH fall, and the amount of free T4 may transiently increase because of hCG. In the second and third trimesters, most thyroid tests stabilize in women with sufficient iodine. The level of neonatal TSH (3-4 days post-delivery) is an accurate indicator of maternal health.[8]
Role of Thyroid Hormones in Fetal Growth and Development
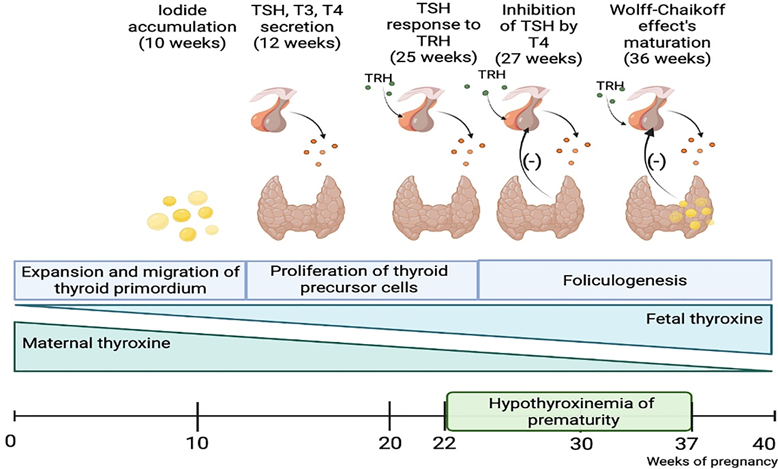
Fetal Thyroid Development and Maternal–Fetal Transfer: The mother remains the sole source of thyroxine for the developing fetal brain during early pregnancy, before midgestation. In spite of an existing uterine-placental barrier, it is not absolute and allows small amounts of maternal T4 and T3 to be measurable in the fetal compartment as early as 4 weeks after conception.
The sources emphasized a few key mechanisms of this transfer:
1. Steady increase: After the initial transfer at 4 weeks, T4 in the fetus increases linearly throughout the period.
2. Bioavailability: A large fraction of T4 in fetal fluids is unbound to proteins. This “free” T4 (FT4) is directly dependent on maternal serum T4 levels and achieves levels that are biologically relevant for tissue development.
3. First Trimester Surge: An increase in maternal FT4 during the first trimester is a biologically relevant event to make adequate substrate available for the developmental needs of the fetal cerebral cortex [9].
Effects on the Brain and Neurological Development:-
These hormones are required for the normal process of carcinogenesis, or the development of the cerebral cortex. Although low levels of T3 are normally found within the fetal fluids, the fetal cerebral cortex itself can produce T3 from maternal T4.
T3 synthesized in the fetal brain reaches adult concentrations by mid-gestation and acts on nuclear receptors to influence gene expression.
These sources imply that the presence of thyroid hormones is a necessity for these neurological activities very early in pregnancy, even before the time the fetus’s thyroid gland takes over as the central source of hormonal production.
Consequences of Thyroid Hormone Deficiency:-
The sources provided place significant emphasis on maternal hypothyroxinaemia, a clinical condition where the thyroid hormone FT4 is lower than normal in the pregnant woman’s blood, although Thyroid Stimulating Hormone levels fall within the normal reference range . The clinical condition is usually caused by a deficiency of iodine, ranging from mild to moderate in nature.
Developmental and Neurological Delays :-
Offspring developmental delays are independently predicted by maternal hypothyroxinemia in the early stages of pregnancy.
Mental and Motor Scores: On the Bayley Scales of Infant Development, children born to mothers with low FT4 at 12 weeks gestation have noticeably lower scores [10].
Diagnostic challenge in pregnancy
1.Trimester Specific Reference Range
One of the most critical requirements while diagnosing thyroid dysfunction among pregnant females is to make use of pregnancy-specific reference ranges. The sources clearly state that general reference ranges commonly obtained from any laboratory, which are applicable to general health, are not applicable to pregnant females . The thyroid function tests vary among the three trimesters of pregnancy as a result of endocrine changes, including a rise in human chorionic gonadotropin (hCG) and estrogen. Hence, it is strongly recommended that the obtained levels of TSH hormones must be assessed appropriately in relation to the gestational period[11].

2. Interpretation of TSH, FT4, and FT3
Interpretation of thyroid function tests (TFTs) changes profoundly in pregnancy:
Thyrotropin (TSH): While TSH will continue to be used as the initial investigation, it will be necessary to interpret it according to pregnancy-specific reference values.
Total T4 : Research suggests that Total T4 is increased substantially throughout all three trimesters when compared to non pregnant individuals .
Free T4 Index (FT4I): This index normally rises in the first trimester and then returns to the normal non-pregnant range in the second and third trimesters of pregnancy
TSH Relationship: TT4 and FT4I are considered better tests than certain direct tests because they have a good inverse relationship with TSH throughout the entire pregnancy.
3. Limitations of Laboratory Assays
A major diagnostic challenge identified is the fact that conventional Free T4 (FT4) immunoassays are not reliable during pregnancy.
Failure to Reflect Physiology: In contrast to FT4I tests, routine FT4 immunoassays usually fail to take account of established physiological changes.
Underestimation of Levels: While TT4 and FT4I levels have been found to be elevated or stable, FT4 levels, as measured by immunoassay, were found to be significantly reduced to 65% of non-pregnant control levels in the second and third trimesters.
Clinical Implications: Since these immunoassays are considered to be defective with respect to pregnancy, the sources suggest that the results of TT4 and FT4I give a much more reliable indication of the actual state of the patient’s free T4.age, they averaged 8 points lower on motor scales and 10 points lower on mental scales .Two-Year Results: Early childhood is affected by the delay; at age 2, these kids scored 10 points lower on motor scales and 8 points lower on mental scales [12].
Etiology of hypothyroidism in pregnancy
Hypothyroidism in pregnancy is most commonly linked to autoimmune disorders, especially Hashimoto's thyroiditis, which is determined by the presence of antithyroid peroxidase (TPO-Ab) and antithyroglobulin (Tg-Ab) antibodies in the blood [13]. Environmental factors, especially iodine deficiency, have been another important cause of hypothyroidism in pregnant women[11] Hypothyroidism has also been attributed to other iatrogenic , that is, to the consequences of medical treatment, for example, post-thyroidectomy or post-radioiodine ablation, especially in a thyroid patients who have to undergo large hormonal adjustments during subsequent pregnancies .
Clinical manifestations of hypothyroidism in pregnancy
The thyroid disorders during pregnancy is believed to be mediated through a complex interaction of maternal symptoms and normal biological changes, which need to be interpreted with respect to diagnosis . The implications of thyroid dysfunction on the fetus are significant because maternal hypothyroidism, especially decreased levels of free thyroxine in early pregnancy, can affect the development of psychomotor activity in the child and fetal growth, thereby altering the functioning of the thyroid in the newborn [14] .
Maternal and Fetal Complications:-
Regarding maternal and fetal complications, overt hypothyroidism is associated with a statistically significant risk of preterm birth. Although some research suggests that maternal thyroid hypofunction does not always result in a consistent pattern of adverse outcomes, some specific risks are evident; for example, first-trimester hypothyroxinemia is associated with preterm labor and macrosomin [15].Additionally, pregnant women who test positive for antithyroid antibodies in either the first or second trimester have an increased risk of preterm premature rupture of membranes. General pregnancy complications are one of the focal points of established management guidelines to ensure optimal care paradigms [16].

Management of hypothyroidism in pregnancy
Hypothyroidism in pregnant women has therefore been that the mother should not be made hypothyroid. The mechanism that is used in the management of hypothyroidism in pregnant individuals is the increasing demand for thyroid hormone by 20-40% during the course of the pregnancy. The strategy that is used to fulfill the increased demand for thyroid hormone that is required by the body during the course of the pregnancy in a pregnant woman who is hypothyroid and who is on replaced therapy with thyroid hormone for the management of hypothyroidism that is indicated by an imbalance in the thyroid hormone levels prior to conception is the administration of two more tablets of Levothyroxine (l-T4) immediately after conception. The effectiveness of the strategy has been very effective in that the TSH levels are not higher than 5.0 mIU/liter during the course of the first trimester of the pregnancy. The strategy is close to the physiological changes that are brought about by the pregnancy. The side effects of the strategy are that the TSH will fall below 0.5 mIU/liter. As indicated in the case scenario, despite the fact that even if the trimester-specific lower cut off is 0.1.
Specific patient populations are at greater risk for TSH suppression with this aggressive early adjustment strategy and include athyreotic patients, those with a pre-pregnancy TSH below 1.5 mIU/liter, and those receiving pre-pregnancy l-T4 doses of 100 µg/d or more[15]. Vigilant monitoring remains a hallmark of treatment, with TSH testing every 4 weeks recommended through mid-gestation to ensure that levels remain in the target range . This monitoring interval represents an adequate frequency, as testing every 4 weeks can detect 92% of abnormal thyroid values. The 2017 American Thyroid Association guidelines indicate that while all care must be individualized, adherence to these established evidence-based paradigms is the most optimal approach for managing pregnant and postpartum women . Several weeks after delivery, postpartum dose adjustment is required to transition the patient back to her pre-pregnancy levothyroxine needs, appropriately transitioning her to a state of safe baseline thyroid status [11].
Etiology of hyperthyroidism in pregnancy
Graves' disease, an autoimmune disease, is the most common cause of hyperthyroidism in pregnant women. However, other etiological factors for the syndrome include toxic goiter and solitary autonomously functioning nodules. Another cause of hyperthyroidism is Gestational transient thyrotoxicosis (GTT), described as hyperemesis gravidarum in women with the syndrome [16].The pathogenesis of the syndrome is due to elevated levels of human chorionic gonadotropin (hCG), which possesses in vitro thyroid-stimulating activity. Studies have established that the degree of biochemical hyperthyroidism in patients with the syndrome is directly proportional to the concentrations of hCG and the severity of vomiting[18].
Clinical manifestations of hyperthyroidism
It is very crucial to distinguish pathological hyperthyroidism from physiological changes in thyroid hormone economy during pregnancy, especially during the first trimester . In addition, it is critical to identify Maternal Symptoms as failure to manage hyperthyroidism may result in negative outcomes for both mother and child . To start with, fetal and neonatal effects may be severe since hyperthyroidism in the fetus may be life-threatening and needs to be recognized to enable timely administration of antithyroid drugs to the mother . In addition, Low Birth Weight may result from hyperthyroidism among pregnant women due to uncontrolled hyperthyroidism among these women .
Maternal and Fetal Complications :-
Thyroid storm: This is a severe and potentially life-threatening complication of thyrotoxicosis which can present in pregnancy . Heart failure: Though the sources are mainly centered on the general adverse effects on the woman, it is emphasized that the control of thyrotoxicosis is very important in the prevention of severe life-threatening complications.
Intrauterine growth restriction (IUGR) / Low birth weight: In the event of uncontrolled hyperthyroidism, there is a highly increased risk for low birth weight infants. It was seen that the risk for low birth weight was nine times more in the uncontrolled hyperthyroid group compared to the euthyroid group.
Fetal thyrotoxicosis is one condition that presents a life-threatening condition and therefore needs early detection so that the fetus can be treated through the mother.
Preeclampsia: Severe preeclampsia occurs due to uncontrolled hyperthyroidism fourfold [16] .
Management of hyperthyroidism in pregnancy
1. Pharmacological Management:
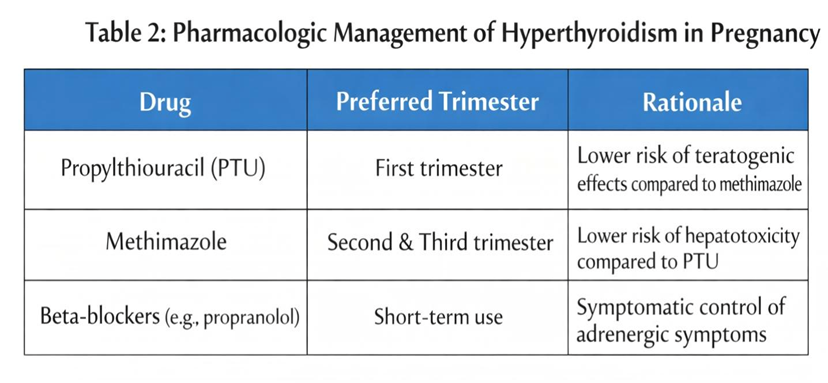
Antithyroid Drugs (The use of thioamides is still the primary treatment; however, this has been considered a controversial issue due to drug-related risks.
Drug Selection: The two primary choices for drugs for managing thyrotoxic crises are methimazole (MMI) (or its prodrug carbimazole)
Methimazole/Carbimazole: These have been associated with occasional teratogenic effects, that is, birth defects, when administered during early pregnancy.
Propylthiouracil (PTU): PTU is often used in the first trimester to prevent methimazole embryopathy, but the risk of hepatotoxicity to the mother is high.
Neonatal Impact: Various studies have examined the issue and established that, when managed properly, maternal thioamide therapy has no adverse effect on neonatal consequences [18]
2. Clinical goals and monitoring
In fact, the general approach to manage should always be the achievement of euthyroidism with a view to avert significant risk to mothers and babies.
Monitoring: Regular monitoring of thyroid function tests is required, by virtue of which the medication dosage can be readjusted to prevent the patient from continuing in a hyperthyroid condition.
Risk Reduction: Hyperthyroidism’s effective control will lead to a substantial reduction in severe preeclampsia’s risk, which is 4.74 times increased in uncontrolled thyroid diseases, as well as low birth weight’s risk.
Magnitude of Risk: The risk of having a low birth weight baby is 9.24 times increased among pregnant women whose hyperthyroidism is uncontrolled at delivery as opposed to non-hyperthyroid pregnant women.
3. Fetal and Neonatal Management
Management is not only limited to the mother; the fetus should also be closely monitored for signs of fetal thyrotoxicosis .
In-utero treatment: Fetal hyperthyroidism is a life-threatening condition. If detected, treatment is done by giving antithyroid drugs to the mother, which crosses the placenta to reach the fetus 4.
Postpartum Care: The management also goes up to the postpartum period when 5-10% of the women may develop postpartum thyroiditis leading to permanent hypothyroidism [18].
Thyroid Autoantibodies and Pregnancy Risks
The positive presence of maternal thyroid autoantibodies, such as thyroid peroxidase antibodies (TPOAb), is strongly linked to an increased risk of miscarriages and preterm birth, even among women who carry normal thyroid function status .
Miscarriage: A meta-analysis of cohort studies revealed that pregnant women who are positive for thyroid autoantibodies are more than three times as likely to experience miscarriages compared to their antibody-negative counterparts [19].
Preterm Birth: A substantial doubling of the odds of being premature is evident, and this is linked to the presence of those autoantibodies
Thyrotropin Receptor Antibodies (TRAb): Although the provided sources discuss the management of thyrotoxicosis and fetal considerations as essential aspects in the management of the patient during the course of her pregnancy, the statistical relationship in the management of the patient with regards to the presence of Thyrotropin Receptor Antibodies (TRAb) is not discussed in detail except as part of the overall guidelines from the American Thyroid Association (ATA) recommendations regarding fetal and neonatal care [11,20]
Screening for Thyroid Disorders During Pregnancy
Current International Guidelines:- International clinical practice guidelines, including those published by the Endocrine Society, offer evidence-based guidelines for the management and screening of thyroid dysfunction in pregnancy and the postpartum period. The international guidelines prepared through the application of the GRADE system and the United States Preventive Service Task Force criteria were reviewed by various international standards, including the Asia and Oceania Thyroid Association and the Latin American Thyroid Society .
Cost-effectiveness :- A study comparing different screening approaches, such as universal screening, risk-based or high-risk screening, as well as no screening, suggests that these two approaches are cost-effective compared to no screening at all [21].
Screening in Iodine-Deficient:- The sources illustrate that practice guidelines involve considerations regarding iodine nutrition, among other factors, in the medical management of thyroid issues that occur during pregnancy . Iodine deficiency has been identified as a causative factor of thyroid dysfunction among pregnant women and their fetuses, and there are evidence-based approaches that deal with the iodine nutritional needs of pregnant women who have thyroid problems and other thyroid-related medical issues.
Postpartum Thyroid Disorder
Postpartum thyroiditis (PPT) is defined by the occurrence of de novo autoimmune thyroid disease within the first year after childbirth, excluding the presence of Graves’ disease . It occurs in 5.4% of the general population, whereas women who already have other forms of autoimmune disease, like type 1 diabetes mellitus, have a decidedly greater chance of developing PPT than the general population. PPT in pregnant women is a completely predictable process, wherein 50% of pregnant women who are anti-thyroid peroxidase/anti-thyroglobulin antibody-positive during the first trimester will develop PPT . Even though PPT is a quite common phenomenon, the existence of a potential link between PPT or antibodies and postpartum depression is still debatable, with no conclusions being established yet .The clinical picture of PPT differs, with 22% showing the classical three-phase pattern of hyperthyroidism followed by hypothyroidism . Most affected women, however, experience an isolated hypothyroid phase, with 48% experiencing this while 30% experience an isolated thyrotoxicosis [22].
Special Situation and Clinical Challenges
The management of the diseases of thyroid disorders and hypertension is considered to be quite challenging in the field of obstetrics at present. “The term Subclinical Hypothyroidism has been defined as the presence of abnormal thyroid functions indicated by a TSH value outside the normal range for the period of gestation, in compliance with normal thyroxine values.” “Hypothyroxinaemia is defined by the presence of low levels of thyroxine in the blood of the mother, in spite of normal blood levels of TSH.” “Autoimmune thyroiditis is the major cause of Subclinical Hypothyroidism; however, both diseases have also been known to be caused by the presence of ‘Iodine Deficiency’ in mothers.” “These diseases have been known to cause adverse outcomes for the baby as well as neuro-intellectual impairment in the child.” “There has been no clinical basis for the role of levothyroxine and its ability to enhance the neuro-psycho-development of the child.” “However, the greatest barrier of the study is that it is extremely difficult in clinical practice to undertake appropriate antenatal screening of all those who may be suffering from thyroid disorders in the proportions indicated.”[23,24]
CURRENT GULIDELINE AND RECOMMENDATION
Guidelines for Thyroid Dysfunction in Pregnancy
Endocrine Society Recommendations (2012) :-
Clinical practice guidelines have been developed by Endocrine Society to update the recommendations made by them in the year 2007 on the treatment of thyroid dysfunction during and after pregnancy. The Endocrine Society used the GRADE system and the grading system proposed by the U.S. Preventive Service Task Force (USPSTF) to develop the recommendations.
Scope of Management: It includes management of hypothyroidism, hyperthyroidism, gestational hyperthyroidism, and thyroid autoimmunity [25].
Special Considerations: They provide evidence-based strategies to address thyroid tumors, iodine, and thyroiditis in the postpartum period.
Consensus Development: The development of the guidelines was carried out by a task force and reviewed by international organizations such as the Asia and Oceania Thyroid Association and the Latin American Thyroid Society [25] .
FUTURE PERSPECTIVES
Future directions in maternal-fetal medicine should emphasize studying biomarkers for thyroids to screen for any impending thyroid dysfunction to pregnant mothers, as prospective research trials noted a median gestational age of 13 weeks and 3 days for initiation of thyroxine, noting that it may be a period past the most crucial windows for fetal brain development There is a significant research void in studying personalized approaches to thyroxine supplements, as a standard dose for all thyroxine-positive pregnant mothers, as identified by a study, did not show any improvement in cognitive function compared to mothers who did not undergo thyroxine supplements . Long-term neurodevelopmental outcomes to assess if these treatment effects become more evident later on during a child's development . However, there remains a need for randomized clinical trials to assess its efficacy much earlier during the first trimester to settle the debate on the need for universal antenatal thyroid screening [26].
CONCLUSION
Pregnancy causes changes in the body of the expecting mother, leading to disruption in normal thyroid hormone levels. The onset of such changes in the body creates conditions for severe complications to occur during pregnancy for both the mother and her developing child. To diagnose thyroid dysfunction in an expectant mother, examinations must be conducted with individual reference ranges (expected normal values) during each trimester of pregnancy. When an expecting mother is misdiagnosed with thyroid dysfunction, both she and her child have an increased risk of experiencing complications, including the complications of pre-eclampsia (a hypertensive disorder during pregnancy), miscarriage, premature birth, low birth weight (less than 5.5 pounds), impaired neurodevelopmental outcomes in her infant, as well as experiencing complications herself.
The research study (IRB approved) evaluated the pregnancy outcomes of two groups of expectant mothers receiving care at a tertiary-level hospital in the state of Gujarat, India: Group 1 (Hypothyroid/Hyperthyroid) and Group 2 (Euthyroid). The study supports the notion that women who have thyroid dysfunctions (either hypothyroid or hyperthyroid) experience more premature births at earlier points in the gestational period, have lower APGAR scores, and experience more complications during pregnancy and delivery than do women who do not have thyroid dysfunctions.
These findings indicate that pregnant women with thyroid disorders must be screened as early as possible, and once identified as having a thyroid disorder, their treatment must be modified (i.e., an increase in levothyroxine) in accordance with the type of thyroid disorder and the trimester of pregnancy.
The identification of thyroid disease prior to, or early on in, pregnancy is critical. Properly managing the thyroid in the mother to allow for normal thyroid function (euthyroid) during the pregnancy creates the best possible environment for the baby to be born in a healthy way (i.e., having good perinatal outcomes). In order to help decrease preventable risks for negative perinatal outcomes and to improve maternal-fetal health outcomes (both short- and long-term), we need better antenatal screening processes and evidence-based mana gement protocols in place.
REFERENCES



Khond Arya, Mundhe BI Pushkar Pralhad, Giri Ashok, Pregnancy and Thyroid Disorder: Challenges in Management, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 1-14. https://doi.org/10.5281/zenodo.18834179
 10.5281/zenodo.18834179
10.5281/zenodo.18834179