The goal of this idea is to create a new type of medicine that comes in the form of biodegradable polymer balls. These balls will be wrapped around liquid medicine inside a chewable, edible coating that can be colored and flavored. The balls are made using a reaction between calcium chloride and sodium alginate, forming calcium alginate. This method allows the use of polymers as alternatives to liquid medicines, making them more cost-effective and better for the environment. This new method of giving medicine is a fresh way to deliver drugs and improves on traditional approaches.It can give a new purpose to existing drugs, helping them work better in the body. This system can increase how much of the drug is absorbed, which is called bioavailability. It's also useful for long-term treatment of chronic conditions, keeping the right level of medicine in the blood over time, and reducing side effects because patients take fewer doses. This leads to better adherence to treatment and less medication being used, which means fewer unwanted effects. Targeted delivery also reduces harm to healthy tissues. Our main aim is to introduce and explain some of these newer drug delivery methods to understand the concept of this new type of medicine form.
Novel Pharmaceutical Dosage, Drug Release, Dosage Forms, Diseases, Sodium Alginate, Novel Drug Delivery System.
Sodium alginate and a soluble calcium salt are used in the spherification process to turn a liquid into small balls. When a liquid that contains sodium alginate is added into a solution that has calcium, like calcium chloride, tiny spheres can be made easily. A hard layer of calcium alginate forms on the outside of the liquid, trapping it inside like a flexible skin. These round balls are then washed with water to remove any leftover calcium or other substances from the solution. These formed balls can be used in food or drinks. In 2003, Bulli's work helped develop this method. When the liquid is dipped into a bath, it becomes gel-like and forms a sphere. These spheres can come in different sizes. The names of these spheres vary depending on their shape; for example, smaller ones are called caviar, while bigger ones are named after foods like ravioli, eggs, and gnocchi. These spheres are filled with flavored liquids that are sealed in a thin membrane. The flexibility of the spheres makes them easy to handle with care. They can also hold solid ingredients, allowing a single sphere to have different flavors and textures. This idea was first suggested in Britain in 1942 by William Peschardt. In 1999, Sergi Estragues and Joachim Vives, scientists at Unilever, showed the method to Ferran Adria. The restaurant El Bulli used the technique for more creative and surprising ways. The seaweed-tech company Skipping Rocks Lab is based in London. Yes, they used spherification to make edible water bottles.
Methods [3-5]
The Department of Pharmacy at S.G.S.I.T.S, NDDS, provided calcium lactate and sodium alginate for the laboratory. Molecular gastronomy kits make it easy to perform the spherification process at home. The Molecular Gastronomy kit includes all the ingredients and equipment needed to make any type of sphere. There are three different methods of spherification, each with its own advantages and disadvantages.
Technique 1 :
Spherification The liquid that is to be made into spheres contains sodium alginate at a concentration of 1%.
This liquid is then dripped into a dish containing a solution of any water-soluble calcium salt at room temperature.
Technique 2 :
Reverse spherification This method involves immersing the liquid, which contains sodium alginate dissolved in water, into a calcium salt solution.
It is the reverse of the first technique. This method is used for materials that contain calcium, like milk, or have a high acid or alcohol content. The process involves pouring the calcium lactate or calcium lactate gluconate into the bath water.
Technique 3 :
Frozen reverse spherification In this method, the spheres containing calcium lactate gluconate must be pre-frozen.
The frozen balls are then immersed in a sodium alginate solution.
The way a medicine is given can greatly affect how well it works. Some drugs work best when they reach a certain concentration level. If the concentration is too high or too low, it could be harmful or not work at all. This is why finding the right concentration is important. Treatment for serious illnesses has been moving slowly, so getting the right concentration is key. A team-based approach is often used to treat serious diseases. This has led to new ideas about how drugs move through the body, their effects, harmful side effects, immune responses, and how they nourish the body. These new methods, sometimes called drug delivery systems, involve many different fields. They combine molecular biology, polymer science, pharmacy, and bioconjugate chemistry. A drug delivery system is designed to deliver medicine to the right place in the body and keep the right amount of drug available. Research on new drug delivery systems is ongoing, focusing on technologies like hydrogels, nanoparticles, liposomes, implants, phytosomes, liquid crystals, and dendrimers.[6,7,8]
A new medication offers several advantages compared to traditional drug delivery :
- It allows for the right amount of drug in the blood or tissue to be maintained for a longer period.
- The release of the drug can be controlled over a longer time as planned.
- Drugs that normally break down quickly can have their lasting effect extended.
- By targeting specific areas of the body, unwanted side effects can be minimized.
- The need for large amounts of medication or frequent dosing can be reduced or avoided.
- Patients are more likely to follow their treatment plan consistently. [9]
Innovation In New Drug Delivery System :
1. Implants :
The categorization of implantable systems is quite broad.
Typically, they fall into two main types: "drug implants" and "implantable drug-loaded pumps." The first group includes items such as polymeric membranes and various polymers, and it is used to manage the release of the drug from the delivery system. The second type, known as implantable drug-loaded pumps, uses a mechanical pump to control the release of the medication. There is also a third, less common category that has emerged due to advancements in technology. These include different types of implant groups, each with its own delivery method. Examples include transurethral hydroxyapatite cement systems used for treating osteomyelitis, sustained-release intraocular systems for managing glaucoma, and others. For instance, injection systems are used to treat impotence.[10]
2. Liposomes :
Because of their unique properties, liposomes are commonly used in medication delivery.
They can enter areas of aqueous solution found within the water-replenishing membrane. Another way to look at this is that lipids do not easily pass through the hydrophobic membrane that dissolves hydrophilic substances. However, liposomes can carry both hydrophobic and hydrophilic molecules by dissolving chemicals in the membrane. When the lipid bilayer comes into contact with other surfaces, it melts. In the cell membrane, the molecules are then transported to the site of action.[11,12]
3. Nanoparticles :
Nanoparticles can be produced through chemical or physical methods.
Chemical synthesis methods often lead to harmful effects from the absorption of certain toxic compounds on the surface. Environmentally friendly alternatives include using biological pathways for nanoparticle synthesis, such as microorganisms, enzymes, fungi, and plant or plant extracts. These biological approaches combine with chemical and physical methods. Nanomedicine holds great promise for improving the diagnosis and treatment of diseases. The application of microorganisms in nanoparticle production is a sustainable process. Nanotechnology could transform various biotechnological applications, making them more personalized, portable, affordable, safe, and user-friendly.[13]
4. Hydrogels :
Hydrogels are three-dimensional, hydrophilic polymer networks that can absorb large amounts of water or biological fluids.
They are used to control drug release in ingestible and swelling-controlled systems, or as carriers in reservoir-based, controlled-release systems.[14]
5. Phytosomes :
Phytosomes are molecular complexes that are friendly to lipids.
These are referred to as "phyto," meaning plant, and "some," resembling cells. They are complex polyphenolic plant compounds. Phytosomes are a new method for delivering herbal medicine, initiated by choline. They are improved forms of herbal medicines that are more easily absorbed. Compared to traditional herbal extracts, phytosomes have better pharmacokinetic and therapeutic profiles, and are designed to give better results.
6. Dendrimers :
Dendrimers are nanometer-sized, highly branched, symmetric, non-dispersive macromolecules.
Dendrimers with polyethylene glycol chains (PEG) provide functionality that ensures their stability and protection from the mononuclear phagocyte system (MPS), while their structure is determined by their architecture.[15]
7. Liquid Crystals :
Liquid crystals combine properties of both liquid and solid states.
They can be made from a variety of materials. One of the geometries they can form is alternating polar and polar-polar layers, such as a lamellar phase, which can include aqueous drug solutions.[16]
New dosage forms are categorized.
New dosage forms, often called novel medication delivery systems, are new ways to give medicine into the body.
They offer better effectiveness, safety, and convenience than traditional forms like tablets and injections. These systems work in a few main ways: controlled release, targeted delivery, and using carriers.
Categorizing new dosage forms
1. Controlled drug delivery systems (CRDDS)
These systems slowly give medicine over time, keeping a steady drug level in the blood and target tissues.
They control the release of medicine using a polymer that manages the rate.
These are called diffusion-controlled systems.
Reservoir systems have a drug core wrapped in an insoluble polymer membrane.
The thickness and type of polymer control how fast the medicine moves through the membrane.
Examples: Nitro-Bid and Nico-400 are extended-release capsules that slowly release nitroglycerin.
The medicine is spread evenly in a polymer matrix that is either inert or expandable.
Examples : Glucotrol XL and Procardia XL are extended-release tablets.
- Dissolution-controlled systems :
The release of medicine depends on how quickly the drug or its polymer matrix dissolves.
- Drug dissolution in a matrix :
The medicine is in a matrix that dissolves over time. This often uses waxy materials.
Examples: Products with folic acid and ferrous sulfate.
- Dissolution of encapsulation :
Each medicine particle is wrapped in a material that dissolves slowly.
Examples: "Spansules," which are capsules filled with coated beads.
Medicine is released through a semipermeable membrane with a small hole.
This uses osmotic pressure to push the medicine out.
Examples: Some oral controlled-release medicines and the Alzet osmotic pump used in labs.
- Ion-exchange mechanisms :
Drug molecules are attached to a charged resin and are released when they exchange with ions in the digestive system.
Example: A liquid mix of hydrocodone and chlorpheniramine for long-term release.
2. Focused medication delivery systems (TDDS)
These systems deliver medicine to a specific part of the body, like an organ or cell type.
This helps achieve better treatment results and reduces side effects in non-target areas.
This relies on the natural buildup of medicine carriers in diseased areas.
A key process is the enhanced permeability and retention (EPR) effect. This happens when nanoparticles move through the leaky blood vessels of tumors and stay there because the lymphatic system is not working well.
Examples: Liposomes and polymer conjugates.
Doxil, a liposomal doxorubicin, builds up in cancer tissue.
This involves attaching specific targeting molecules, like antibodies or peptides, to the medicine carrier so it can bind to certain receptors on target cells.
Limits the medicine to a particular tissue or organ.
Delivers the medicine to a specific type of cell.
Releases the medicine into a specific part of a cell.
Example : Monoclonal antibodies conjugate that target cancer cells.
3. Drug delivery systems using nanoparticles :
These systems use materials that are very small, between 1 to 100 nanometers, to carry medicine and improve solubility, stability, and absorption.
These are round structures made from lipid layers that can hold both fat-soluble and water-soluble medicines.
Example : Doxil, used to treat cancer.
These form when certain molecules arrange in water, creating a center that holds medicines that are not very soluble.
Examples : Polymeric micelles used in cancer treatment in research settings.
- Microspheres and nanoparticles :
These are solid particles, either amorphous or crystalline, that either hold or spread the medicine.
Example : Biodegradable nanoparticles made from polylactide-co-glycolide (PLGA) for drug delivery.
- Niosomes and Transfersomes :
These are like liposomes but are made from nonionic surfactants, which makes them more stable or better at getting through biological barriers.
These are highly branched, tree-like polymers that can be modified to deliver or target medicine.
4. Transdermal drug delivery systems (TDDS)
These are patches applied to the skin to deliver medicine into the bloodstream in a controlled way.
Examples : Nicotine patches for quitting smoking and hormone patches for hormone replacement.
5. Implantable drug delivery systems (IDDS) :
These are devices surgically placed in the body to slowly release medicine over a long period.
Example : Contraceptive implants like Norplant.
6. Orally disintegrating tablets (ODTs)
These are tablets that dissolve quickly on the tongue, making them easier to use for children and older adults.
3D-printed dose forms :
These are customized tablets with specific dosages and tailored release patterns.
Benefits of a New Dosage Form :
1. Better effectiveness and safety :
New systems can make a drug work better while making side effects less likely. Targeted delivery, which sends medicine exactly where it's needed, and regulated release, which controls how much drug is given at a time, are two common ways to do this.
2. Increased bioavailability :
Many new drugs don't dissolve well in water, so they don’t get absorbed properly by the body. New formulations, especially those that use nanotechnology, can improve how well these drugs dissolve, which makes them more available and effective in the body.
3. Targeted drug delivery :
Unlike traditional drugs that spread throughout the body, new dosage forms can send medicine directly to the affected area. This is especially useful in treating diseases like cancer, where it's important to deliver chemo drugs only to the tumor and limit exposure to healthy cells.
4. Improved patient compliance :
New dosage forms can reduce the number of times a patient needs to take medicine, which helps them follow their treatment plan better. For example, a continuous release form may only need to be taken once a day instead of multiple times.
5. Controlled and continuous drug release :
These systems can release a drug at a steady rate over time. This avoids the ups and downs in drug levels that happen with traditional doses, keeping the right amount of medicine in the body consistently.
6. Drug protection :
Using methods like liposomes or nanoparticles, active drugs can be protected from being broken down by enzymes and other substances in the body. This helps keep the drug stable and effective, especially in harsh environments like the gastrointestinal tract.
7. Treatment of chronic diseases :
Because they can provide medicine steadily over a long time, new dosage forms are especially good for managing chronic conditions. For example, implantable devices can release drugs slowly over months or even years.
Drawbacks
1. High Cost :
Creating, researching, and making new drug delivery systems can be a difficult and expensive process. These costs are passed on to the final price, which can make some treatments less accessible.
2. Manufacturing Challenges :
It can be tough and costly to make the technologies used in new dosage forms, especially nanomedicines, in large amounts consistently. This can lead to problems with product quality and stability.
3. Regulatory Hurdles :
New dosage forms often need to go through a more strict and complex approval process. This can take more time and money to get a product to the market, as both the medicine and the delivery method must be carefully tested for safety and effectiveness.
4. Biocompatibility and Toxicity Concerns :
The materials used in new carriers, like nanoparticles or polymers, might be harmful or not compatible with the body. This could lead to unwanted immune responses or other harmful effects. There is often not enough long-term safety information available.
5. Risk of "Dose Dumping" :
If the delivery system fails, a large amount of the medicine could be released all at once, which might result in dangerously high levels of the drug and possible toxicity.
6. Limited Dose Adjustment :
Some implantable or controlled release devices have a fixed dose. This makes it harder to adjust the dose according to each patient's specific needs, which is an important part of traditional treatment.
7. Need for Specialized Administration
Inserting or removing certain advanced delivery systems may require surgery or skilled professionals, which can add to the cost and make the process uncomfortable for the patient.
New Gentle Dosage Forms For Pediatric Uses :
Since millions of children are treated by it every day, it holds international importance.
Because of the risk of life-threatening diseases, healthcare workers must ensure that patients follow their treatment plan [17] By providing age-appropriate drug delivery products and acceptable medications, healthcare professionals and caregivers often face daily challenges due to the availability of many medications that are not suitable for children [18] This often leads to the modification of drugs originally made for adults [19] The use of medications off-label, which is outside the terms of their marketing authorization, may increase the chance of [20]. Many studies have pointed out the difficulty of accurately dosing through tablet manipulation [21] These methods cannot be considered as manufactured in accordance with Good Manufacturing Practice [22], which is similar to commercially available pharmaceuticals. However, it still involves the dose adjustment by
Most of the time, when there is a shortage, the majority of cases are still handled by pharmacists, nurses, or parents when it comes to the treatment for children [23]
These practices have the potential to be harmful and carry a high level of risk.
The children population, those under the age of 18, is now 2,415,319,658 [25], and they are among those who suffer from toxicity and therapeutic ineffectiveness [24]
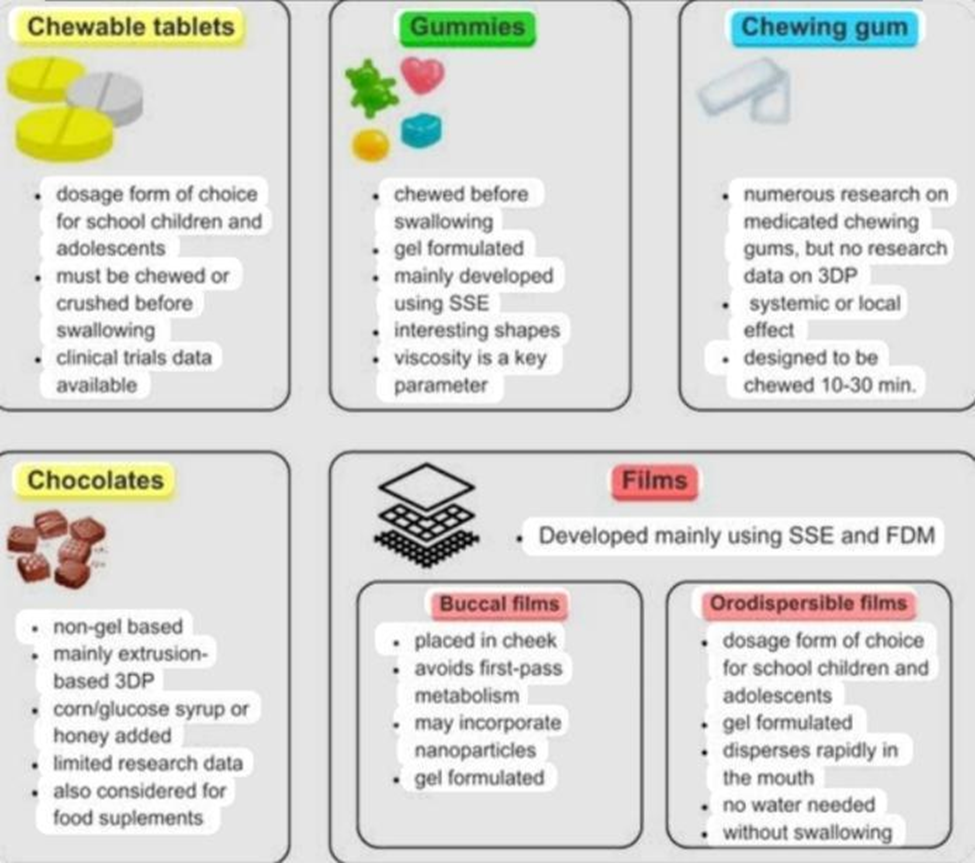
About 654,028,000 children under the age of five [26] Within this pediatric community, we can identify numerous groups based on their age in completed days [27], months, or years. The pharmaceutical industry must, with a Paediatric Investigation Plan (EU), involve youngsters in the drug development process.Or Paediatric Study Plan (USA). Additionally, numerous regulations have been put into place, including Title V of the Food and Drug Administration, which is part of the United States of America, FDASIA [28], which reauthorizes and modifies aspects of the Best Pharmaceuticals for Children Act (BPCA) and the Paediatric Research Equity Act (PREA). In a similar vein, the European Union established the EU Paediatric Regulation: The World Health Organization, along with (EC) Nos. 1901/2006 and (EC) No. 1902/2006 [29], launched the Make Medicines Child Size campaign [30] At the same time, a number of projects, such as the European Paediatric Formulation Initiative (EuPFI; 2007) and the Global Paediatric Formulations Accelerator (GAP-f) [31], are examples of this. These prioritize creating age-appropriate drug formulations. Additionally, several groups held seminars, including the workshop on the challenges of pediatric formulation development. The 6th APV Winter Conference on Patient-Centric Drug Development and Today and Tomorrow's Strategies in Product Development [32], which addresses the challenge of creating formulations specifically designed for children. Chewable pills and orally disintegrating dosage forms are thought to be the preferred dosage forms for school-age children (6-11 years) and teenagers. These include:
The use of gels is seen in novel drug delivery methods [33] Depending on the makeup and physicochemical characteristics of the medication, soft novel drug delivery systems (DDSs) might be divided into gel-formulated and non-gel-formulated systems. Three-dimensional printing (3DP) has the potential to be a valuable technique for creating tailored drug delivery systems for children [34]
New varieties of soft dose formulations :
1. Hydrogels and gels :
These are easy to take and come in different shapes and sizes.Because they look like candy, gel-based gummies are especially appealing to children.
2. Oral films :
These are thin, dissolvable strips that contain the active ingredient.
3. Orodispersible films (ODFs) :
These dissolve quickly in the mouth without needing water.
4. MBFs are mucoadhesive buccal films :
that stick to the inside of the mouth to help the medicine stay longer or be absorbed through the mucosa.
5. Chewable tablets and gummies :
This is a popular and commonly used form for older children (usually 2 years and older) who can chew safely.
To make it easier for children to take, gummies can be made in various shapes and flavors.
Mini-tablets that disintegrate in the mouth (ODMTs) :
These are small tablets (2–4 mm in diameter) that melt in the mouth.
The number of mini-tablets can be adjusted to give the right dose, and they are easier to swallow than regular tablets.
6. Sprinkles and granules :
These are small granules or pellets that are part of multi-particulate systems.
They can be added to soft foods or drinks. They are good for babies and young children because they allow for flexible dosing.
7. Lollipops :
Medicated lollipops and lozenges are flavored to cover up bitter tastes.They can be kept in the mouth for either local or systemic drug delivery through the buccal mucosa.


 Vedant Chimote*
Vedant Chimote*
 Vidya Karhale
Vidya Karhale

 10.5281/zenodo.18703471
10.5281/zenodo.18703471