
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Pravara Rural college of Pharmacy Loni.
Obesity is the multifactorial metabolic disease with worldwide interest, which leads to high non-communicable disease burden like type 2 diabetes, cardiovascular disease, and metabolic syndrome. Over the recent years, gut microbiota has been revealed as a master regulator of obesity pathogenesis. The imbalance of the composition of microbes in the gut or dysbiosis has been linked to higher energy harvest, lower energy expenditure, a change in lipid metabolism, and failing to control the appetite. Remarkably, a higher Firmicutes to Bacteroidetes ratio among the obese is a common behavior, which improves fermentation of the indigestible polysaccharides and leads to the production of short-chain fatty acids (SCFA) to facilitate adipogenesis. In addition, metabolites produced in the gut, including SCFAs, have context-specific effects on the metabolism of the host, and current evidence demonstrates that SCFAs exert both positive and negative effects on the rate of lipogenesis and inflammation. Neuroendocrine signaling by microbial metabolites regulating the gut-brain axis mediates the role of appetite and energy homeostasis. Moreover, Gram-negative bacterial endotoxin, lipopolysaccharides (LPS), causes metabolic endotoxemia and low-grade systemic inflammation to enhance insulin resistance and accumulation of lipids. Taken together, these mechanisms emphasise the imperative role of gut microbiota in development of obesity. Knowledge of the intricate host microbiota interactions may have therapeutic potentials, such as microbiota-specific treatments, namely probiotics, prebiotics, dietary manipulations, and fecal microbiota transplantation to treat the treatment of obesity.
Zoonotic diseases are the diseases caused by pathogens which are transmissible between animals and humans, represent a significant global public health challenge. These diseases account for a substantial proportion of emerging infectious diseases, with serious implications for both human and animal populations. India, with its diverse ecosystems, high population density, and close interactions between humans, livestock, and wildlife, faces a heightened risk of zoonotic outbreaks. The country’s socio-economic structure and agricultural practices further contribute to the vulnerability. Emerging infections such as the Nipah virus, Crimean-Congo haemorrhagic fever (CCHF), avian influenza, and zoonotic tuberculosis emphasize the critical importance of early detection and control. The COVID-19 pandemic has also underscored the interconnectedness of human, animal, and environmental health, propelling the adoption of the One Health approach in India. COVID-19 has demonstrated the critical importance of communication, coordination, and understanding within the healthcare sector to enhance public health outcomes. This interdisciplinary framework promotes collaboration across human health, veterinary, and environmental sectors to tackle zoonotic threats comprehensively. Recent advancements in zoonotic disease surveillance in India include the deployment of molecular diagnostic tools, geospatial mapping, and mobile health (mHealth) technologies, enabling real-time disease monitoring and reporting. However, the implementation of these innovations is often hampered by challenges such as fragmented healthcare systems, underfunded veterinary services, and limited public awareness. Strengthening zoonotic disease surveillance is pivotal for India to mitigate the impact of future outbreaks and safeguard public health.
This review explores the current status of zoonotic disease surveillance in India, focusing on the integration of innovative technologies, the role of One Health initiatives, and addressing persistent challenges to ensure robust preparedness and response systems
1. Overview of Zoonotic Diseases in India:
Zoonotic diseases account for a major proportion of infectious diseases in India, imposing a dual burden on public health and the economy.
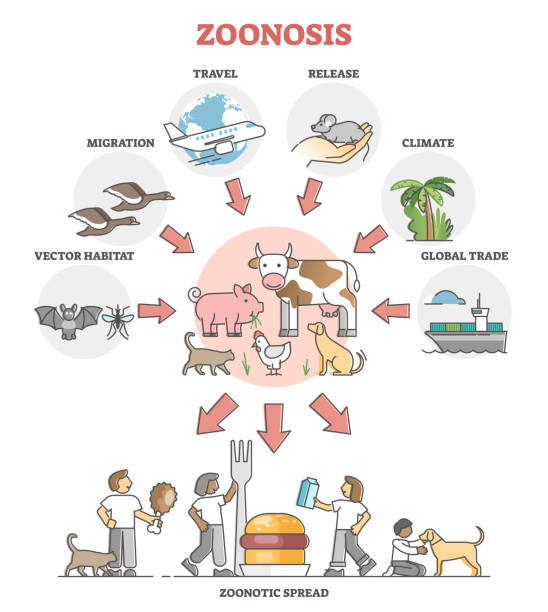
Key Examples:
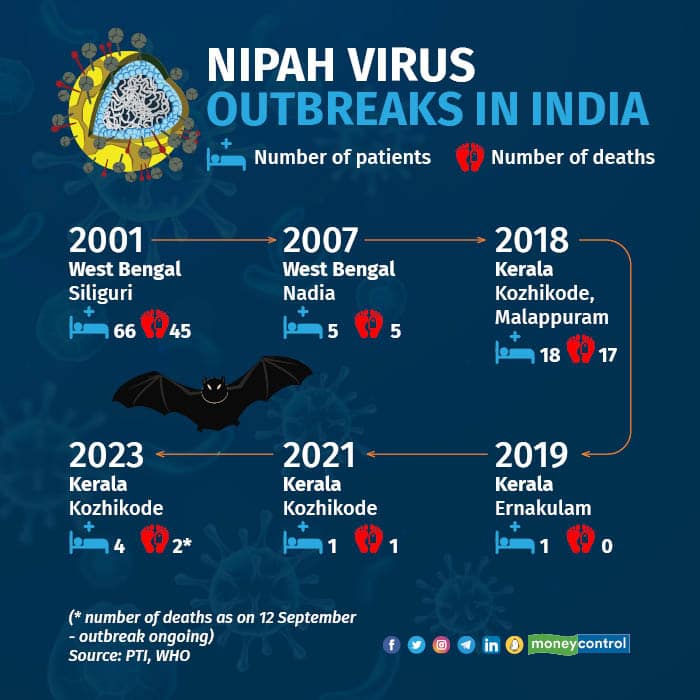
1. Nipah Virus:
Nipah virus (NiV) is a zoonotic pathogen that has emerged as a significant public health threat in India, particularly in the state of Kerala. It is a paramyxovirus first identified during an outbreak in Malaysia in 1998. In India, NiV outbreaks have been documented since 2001, with the state of Kerala experiencing recurrent episodes since 2018. The natural reservoir of the Nipah virus is fruit bats of the Pteropus genus. These bats are widely distributed across South and Southeast Asia and are often in close contact with human settlements. The virus is transmitted to humans through direct contact with bat secretions, consumption of fruits contaminated by bat saliva or urine, and, less commonly, through contact with infected animals such as pigs. Human-to-human transmission is also well-documented, making NiV a potential candidate for epidemics. The Kozhikode, Kerala had the most severe impact of Nipah virus outbreak in 2018. It resulted approximately in 18 confirmed cases, of which 17 were fatal, underscoring the virus’s high mortality rate. The government’s response included isolating patients, intensive contact tracing, and quarantining hundreds of individuals who had come into contact with infected persons. Subsequent outbreaks in Kerala in 2019, 2021, and 2023 further emphasized the virus’s persistent threat. For instance, the 2023 outbreak in Kozhikode resulted in six laboratory-confirmed cases and two fatalities, with extensive efforts made to contain the virus by identifying and monitoring over 350 contacts. Nipah virus infection presents with a range of symptoms, from fever, headaches, and respiratory distress to severe encephalitis. In many cases, patients progress to a coma within 24 to 48 hours of symptom onset. Currently, no specific treatments or vaccines exist for NiV, making supportive care the primary management strategy. Antiviral medications such as ribavirin have been used experimentally, but their efficacy remains unproven. Kerala’s proactive approach to managing Nipah outbreaks involves robust surveillance, rapid testing, and public awareness campaigns. However, the challenges persist, especially in managing the economic and psychological impacts of recurrent outbreaks. Efforts are ongoing to develop vaccines and improve diagnostic capabilities to address this critical public health issue. [1 ,2 ,3 ,4]
2. Crimean-Congo Haemorrhagic Fever (CCHF)
Crimean-Congo Haemorrhagic Fever (CCHF) is a tick-borne viral disease caused by the Nairovirus, primarily affecting humans through bites from infected Hyalomma ticks or contact with the blood or tissues of infected animals. The disease is characterized by severe haemorrhagic symptoms, fever, and multi-organ failure, with a case fatality rate ranging from 10% to 40%. In India, the first documented case of CCHF was reported in Gujarat in 2011. Since then, sporadic outbreaks have occurred primarily in Gujarat and Rajasthan, where the tick vector and susceptible livestock populations are prevalent. The disease’s geographic spread is influenced by the movement of livestock, as infected ticks are often transported along with animals. In recent years, several cases have been reported annually, with significant public health responses aimed at controlling the outbreaks. The transmission cycle of CCHF involves ticks, livestock, and humans. Hyalomma ticks acquire the virus by feeding on infected animals and serve as both reservoirs and vectors. Humans are typically infected through tick bites or contact with blood or tissues during the slaughter of infected animals. Human-to-human transmission occurs through exposure to infectious bodily fluids, posing a significant risk to healthcare workers and caregivers. The clinical presentation of CCHF ranges from mild febrile illness to severe haemorrhagic fever with complications such as disseminated intravascular coagulation (DIC), multi-organ failure, and death. Diagnosis is confirmed through laboratory tests, including reverse transcription-polymerase chain reaction (RT-PCR) and enzyme-linked immunosorbent assay (ELISA) for viral RNA and antibodies. Preventive measures for CCHF include controlling tick populations, avoiding direct contact with infected animals, and using personal protective equipment (PPE) for healthcare workers. Public health campaigns emphasize the importance of hygiene and safety in handling livestock. Although there is no specific antiviral treatment, ribavirin has been used with some success in managing CCHF cases. Vaccination efforts are under research to provide long-term solutions. [5 ,6 ,7 ,8 ]
3. Chandipura Virus:
The Chandipura virus (CHPV), a member of the Rhabdoviridae family, is an emerging zoonotic pathogen in India, associated with acute encephalitis syndrome (AES), particularly in children. First identified in 1965 in the village of Chandipura in Maharashtra, the virus has since caused multiple outbreaks in central India, including states like Madhya Pradesh, Maharashtra, and Andhra Pradesh. CHPV is transmitted primarily by sandflies (Phlebotomus spp.), which act as vectors. Humans are infected when bitten by an infected sandfly, and the virus primarily affects children under the age of 15. The disease’s rapid onset and progression to severe neurological symptoms make early diagnosis and intervention critical. Outbreaks of Chandipura virus have been sporadic but devastating. In 2003, an outbreak in Andhra Pradesh resulted in over 200 reported cases of AES, with a fatality rate exceeding 50%. Subsequent outbreaks in other regions highlighted the virus’s significant public health impact. The clinical course of CHPV infection involves high fever, seizures, altered mental status, and rapid progression to coma and death in severe cases. Laboratory confirmation is achieved through RT-PCR and serological assays. Efforts to control CHPV outbreaks focus on vector management, early diagnosis, and public health education. Since sandflies breed in humid and shaded areas, environmental management strategies such as clearing vegetation, draining stagnant water, and using insecticides are critical. Healthcare systems are also strengthening surveillance networks to detect and respond to outbreaks promptly. [9 , 10 , 11]
|
Disease |
Location |
Year |
Cases |
Deaths |
Reference |
|
Nipah Virus |
Kerala |
2023 |
2 |
2 |
The Sun |
|
Chandipura Virus |
Gujarat |
2024 |
133 |
56 |
Reuters |
|
Crimean-Congo Haemorrhagic Fever (CCHF) |
India (Gujrat) |
2023 |
1 |
1 |
National Centre for Disease Control |
Figure 1: Number of Reported Zoonotic Disease Outbreaks in India (2023-2024)[12 , 13 ,14]
Global and National Impact:
Zoonotic diseases impose significant economic burdens on affected countries, including India. The direct and indirect costs associated with outbreaks encompass healthcare expenditures, loss of livestock, trade restrictions, and long-term social impacts. These costs are particularly high for low- and middle-income countries (LMICs), where resources for outbreak prevention and control are often limited. One of the most visible economic impacts of zoonotic diseases is the loss of livestock. Outbreaks like foot-and-mouth disease, avian influenza, and CCHF necessitate mass culling of animals to prevent further spread. For instance, foot-and-mouth disease alone costs India an estimated $3.3 billion annually in terms of reduced productivity, loss of trade opportunities, and veterinary costs. Similarly, outbreaks of avian influenza have led to the culling of millions of poultry, affecting farmers’ livelihoods and the poultry industry’s overall stability. Trade restrictions are another significant economic consequence. Zoonotic disease outbreaks often lead to bans on the export of affected livestock and their products, disrupting international trade and reducing foreign exchange earnings. For instance, India’s status as a leading exporter of poultry products has been affected during avian influenza outbreaks. The healthcare sector bears a substantial burden during zoonotic disease outbreaks. The costs associated with diagnostics, treatment, and hospitalization are compounded by the need for enhanced surveillance and public health interventions. In addition, the long-term societal costs include reduced workforce productivity due to morbidity and mortality and the psychological impact of outbreaks on affected communities. [15 , 16 , 17]
India’s experiences with avian influenza and SARS have shaped its current strategies for managing zoonotic diseases. Avian influenza, caused by highly pathogenic avian influenza (HPAI) viruses such as H5N1 and H9N2, has been a recurring challenge since the first outbreak in Maharashtra in 2006. The country’s poultry industry, a significant contributor to the economy, has faced multiple setbacks due to culling operations and trade restrictions imposed during outbreaks. India has adopted stringent measures to combat avian influenza, including active surveillance, rapid diagnostics, and public awareness campaigns. The government’s Animal Husbandry Department collaborates with international agencies like the World Organisation for Animal Health (OIE) to monitor and respond to outbreaks. Vaccination of poultry, biosecurity measures, and training for farmers are integral components of the control strategy. The 2003 SARS outbreak, while not as severe in India as in other countries, highlighted the importance of global collaboration and robust surveillance systems. SARS-CoV, the causative agent, is believed to have originated from bats and spread to humans through civet cats in live animal markets. India’s response included strengthening the Integrated Disease Surveillance Programme (IDSP) and establishing rapid response teams to manage outbreaks. Lessons from SARS were instrumental in shaping the country’s response to subsequent zoonotic diseases, including COVID-19. India’s efforts to manage zoonotic diseases emphasize a One Health approach, recognizing the interconnectedness of human, animal, and environmental health. By integrating these aspects into a cohesive [18, 19, 20]
2. Key Components of Zoonotic Disease Surveillance Systems
Zoonotic disease surveillance is a cornerstone of global health security, essential for detecting, responding to, and mitigating the impact of zoonoses. These diseases, which can transmit between animals and humans, pose a persistent threat to public health, agriculture, and biodiversity. A robust surveillance system relies on three critical components: epidemiological surveillance, pathogen detection, and data integration. Together, these elements enable a comprehensive understanding of disease dynamics, bolster preparedness, and facilitate timely interventions.
Epidemiological Surveillance forms the backbone of zoonotic disease monitoring by tracking disease patterns and trends in both human and animal populations. Early detection of outbreaks is pivotal, as it provides the data necessary to guide effective public health responses. Human health surveillance, for example, plays a vital role in identifying unusual disease patterns through systems like India’s Integrated Disease Surveillance Programme (IDSP). The IDSP monitors diseases such as Nipah virus, Crimean-Congo Haemorrhagic Fever (CCHF), and Chandipura virus, using sentinel sites, hospitals, and diagnostic laboratories as hubs for reporting. Regular health screenings in high-risk areas further enhance detection efforts. [22]. Equally important is animal health surveillance, as many zoonotic diseases originate in animal reservoirs. India’s National Animal Disease Reporting System (NADRS) facilitates real-time data reporting from veterinary officers and field workers, enabling swift identification of unusual morbidity or mortality in livestock and wildlife populations [23]. Meanwhile, community-based monitoring engages local populations, training villagers and farmers to report unusual animal deaths or illnesses. This approach proved invaluable during avian influenza outbreaks in India, where early community reports triggered timely containment measures, including culling operations [24]. Despite these efforts, epidemiological surveillance faces significant challenges, including underreporting, inadequate infrastructure, and a lack of trained personnel. However, advancements in mobile applications, GIS tools, and automated reporting systems are addressing these limitations, making data collection and analysis more efficient[25].
3. Recent Advances in Zoonotic Disease Surveillance
3.1 One Health Approach
The One Health approach emphasizes the interconnectedness of human, animal, and environmental health. Given that over 60% of emerging infectious diseases originate from animals, a collaborative framework between these sectors is essential to control zoonotic outbreaks. In India, where rapid urbanization and environmental changes drive human-animal interactions, this approach is increasingly recognized as a cornerstone of public health strategies. The integration of veterinary sciences, human medicine, and environmental monitoring ensures early detection and timely response to zoonotic threats. However, implementing this strategy requires overcoming structural and policy challenges, including the fragmentation of responsibilities among various governmental agencies and a lack of standardized surveillance protocols [30] [31]. One notable success story is the PREDICT Project, launched under the Emerging Pandemic Threats (EPT) program. PREDICT monitors zoonotic pathogens at wildlife-human interfaces by conducting surveillance in high-risk regions, such as wet markets and areas of deforestation. By identifying novel viruses in animals before they spill over into human populations, PREDICT has played a crucial role in early detection and mitigation [32][33]. The project has provided vital insights into how viral spillovers occur and has helped train health professionals in zoonotic disease identification. However, challenges remain, including inadequate funding, lack of cross-sectoral coordination, and difficulties in data-sharing across governmental and research institutions [34]. The effectiveness of the One Health approach depends on sustained investment, capacity building, and a legislative framework that supports intersectoral collaboration. [35]
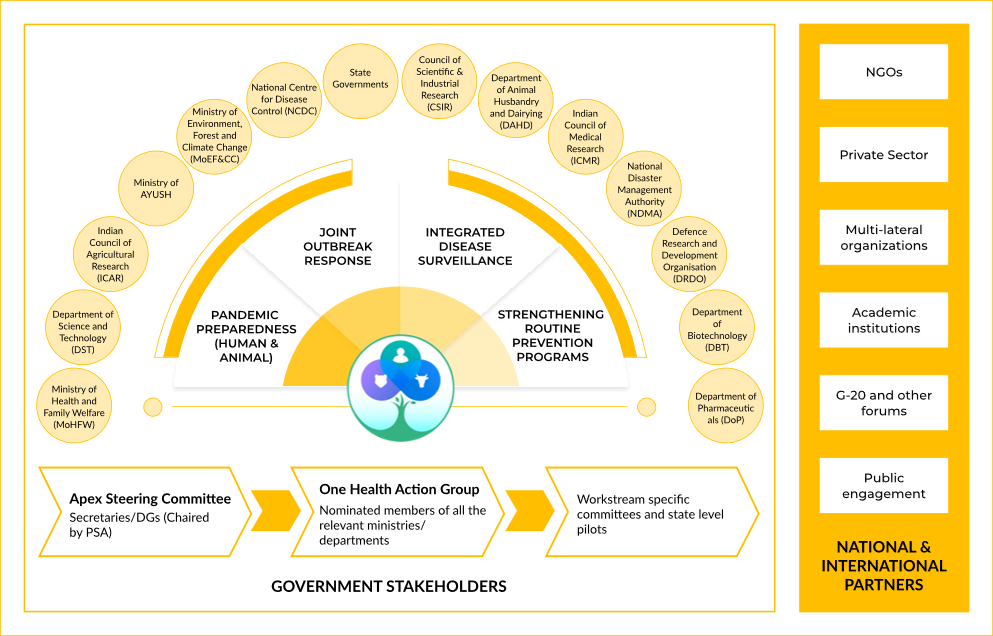
3.2 Technological Innovations
Technological advancements are transforming zoonotic disease surveillance in India. The integration of digital tools, artificial intelligence (AI), and genomic surveillance enables real-time monitoring, early warning systems, and precise pathogen identification. With the expansion of mobile technology and cloud computing, digital surveillance has become more accessible, allowing for the swift collection, analysis, and sharing of epidemiological data. Digital Surveillance Tools such as the Integrated Health Information Platform (IHIP) allow real-time disease reporting across states.

Launched by the Ministry of Health, IHIP provides an early warning system for outbreaks by linking data from human and veterinary health sectors. By integrating machine learning algorithms, IHIP enhances predictive analytics for outbreak trends, allowing authorities to take pre-emptive action [36]. Despite its advantages, implementation faces hurdles such as internet connectivity issues in rural areas and hesitancy among healthcare workers to adopt digital platforms [37]. Training programs and incentives for healthcare workers could enhance the platform’s adoption and effectiveness.
Artificial Intelligence (AI) and Machine Learning are revolutionizing outbreak prediction. By analysing climatic, migratory, and epidemiological data, AI models can identify potential hotspots for zoonotic spillovers. AI-driven tools have been deployed in India to track diseases like Nipah and avian influenza. For example, AI-based models using satellite and climate data have been instrumental in predicting outbreaks linked to monsoon-driven vector migration patterns [38]. However, the effectiveness of these models is constrained by the quality and availability of training datasets, raising concerns about biases and accuracy. Ethical concerns, such as the transparency of AI decision-making processes, also need to be addressed before AI-driven disease surveillance can be fully operationalized. [39] Genomic Surveillance plays a critical role in pathogen detection. Rapid sequencing technologies, including CRISPR-based diagnostics, have enabled the precise identification of zoonotic pathogens, aiding in outbreak containment. India’s Indian SARS-CoV-2 Genomics Consortium (INSACOG) has demonstrated the power of genomic surveillance by tracking COVID-19 variants [40]. Expanding similar genomic capabilities to zoonotic diseases could enhance preparedness, but challenges such as high costs and limited sequencing infrastructure must be addressed [41]. Additionally, integrating genomic data with epidemiological datasets could improve understanding of transmission pathways, but this requires significant investment in bioinformatics infrastructure and expertise.
3.3 Remote Sensing and GIS Technology
Remote sensing and Geographic Information System (GIS) technology provide crucial insights into environmental changes that influence zoonotic disease dynamics. Satellite imagery tracks deforestation, land-use changes, and climate variations, all of which affect disease transmission. These technologies facilitate risk assessments, helping policymakers devise targeted intervention strategies before an outbreak occurs. In India, GIS mapping has been instrumental in tracking vector-borne diseases such as malaria and dengue. The same principles are now being applied to zoonotic diseases. For instance, researchers use GIS tools to map bat populations in Kerala, helping predict Nipah virus spillover risks [42]. Additionally, remote sensing has been used to monitor rodent populations in agricultural zones, providing data that helps predict outbreaks of leptospirosis and other zoonotic diseases [43]. However, the effectiveness of these technologies depends on access to high-resolution satellite data and expertise in spatial epidemiology. Implementing GIS technology on a national scale requires extensive training programs for health officials and partnerships with satellite imaging organizations. [44]
3.4 Citizen Science and Crowdsourcing
Engaging the public in disease surveillance can significantly enhance early outbreak detection. Platforms like ProMED-Mail, an open-source system for reporting unusual health events, allow healthcare workers and citizens to share real-time updates on emerging threats. [45] These decentralized systems complement official reporting mechanisms by offering grassroots-level insights into disease spread. India has begun leveraging crowdsourcing initiatives to track zoonotic diseases. During the COVID-19 pandemic, community-driven apps helped monitor symptoms and exposure risks. Similar models can be adapted for zoonotic surveillance. Mobile-based participatory surveillance has also been deployed to track cases of rabies in rural areas, allowing for quicker response and containment efforts. However, challenges such as misinformation, data privacy concerns, and verifying reports from non-experts must be carefully managed to ensure accuracy and reliability [46].
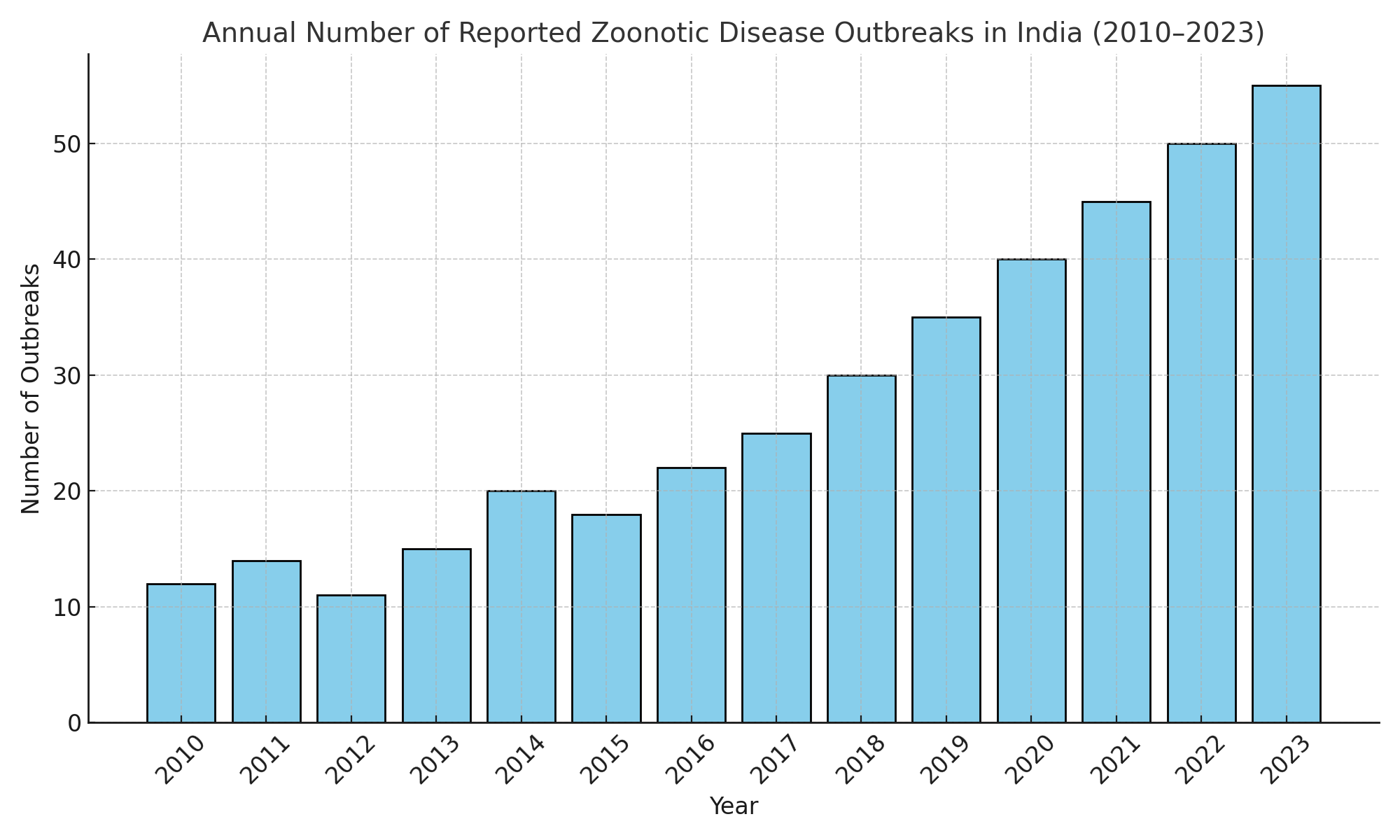
Figure 2: Bar chart image showing increasing trends in zoonotic disease outbreaks in India (2010–2023)
4. Integration of Advanced Data Analytics
Advanced data analytics is revolutionizing zoonotic disease surveillance by enabling efficient data collection, integration, and analysis across multiple disciplines. Technologies such as big data platforms, cloud-based collaboration, and blockchain technology have significantly improved information sharing, risk assessment, and early outbreak detection.
Cloud computing facilitates real-time data sharing, allowing researchers, government agencies, and health professionals to collaborate seamlessly across different regions and sectors. By leveraging cloud-based platforms, health authorities can access centralized databases, streamline disease reporting, and enhance coordination between national and international agencies [50]. One example of successful cloud-based collaboration is the Global Public Health Intelligence Network (GPHIN), which utilizes cloud technology to monitor infectious disease outbreaks worldwide. In India, platforms such as the Integrated Disease Surveillance Programme (IDSP) have integrated cloud computing to improve real-time reporting and resource allocation during outbreaks [51,52].
Despite its advantages, cloud-based collaboration faces challenges such as cybersecurity threats, high implementation costs, and disparities in digital infrastructure, particularly in low-resource settings. Ensuring data security through encryption protocols and regulatory compliance is critical for maintaining trust in cloud-based health surveillance systems [53].
Blockchain technology offers a secure and transparent framework for data sharing among stakeholders in zoonotic disease surveillance. By using decentralized ledger systems, blockchain ensures data integrity, traceability, and immutability, reducing the risks of data tampering and unauthorized access [54].
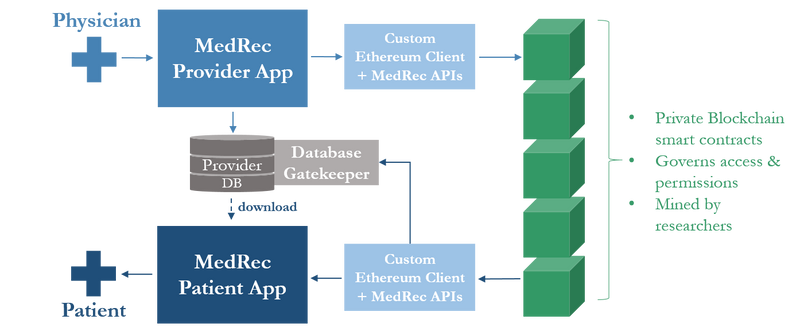
Fig. Example of Blockchain Mechanism (MEdRee) in healthcare.
In he context of zoonotic disease monitoring, blockchain has been explored for vaccine distribution tracking, livestock health records, and genomic data sharing. For example, blockchain-based systems have been implemented in veterinary health programs to track animal movement and disease status, ensuring transparency in disease control measures [55].
Despite its potential, blockchain adoption in public health faces technical barriers, including high computational requirements and the need for regulatory frameworks to govern data transactions. Integrating blockchain with existing digital health systems requires collaboration between policymakers, researchers, and technology developers to ensure scalability and interoperability [56].
5. Policy and Governance Enhancements:
Effective management of zoonotic diseases necessitates robust policy frameworks and governance structures. Strengthening these frameworks involves the implementation of international regulations, active participation of global organizations, and the development of national initiatives.
The International Health Regulations (IHR) of 2005 serve as a legally binding framework for 196 countries, including all WHO Member States, aiming to prevent, protect against, control, and provide a public health response to the international spread of diseases. The IHR require countries to develop and maintain core capacities for surveillance and response, ensuring prompt detection and reporting of public health events that could constitute a public health emergency of international concern (PHEIC) (57).
The World Health Organization (WHO), the Food and Agriculture Organization (FAO), and the World Organisation for Animal Health (OIE) play pivotal roles in global health security, particularly concerning zoonotic diseases. These organizations collaborate to provide guidance, build capacities, and mobilize resources to strengthen national and international responses to health threats.
India faces significant challenges with antimicrobial resistance (AMR) and zoonotic diseases due to its vast population, diverse ecosystems, and high burden of infectious diseases. In response, the Indian government has implemented several national initiatives to address these pressing issues. [62]
National Programme on AMR Containment: Launched during the 12th Five-Year Plan (2012-2017), this program aims to establish a comprehensive AMR surveillance system. Coordinated by the National Centre for Disease Control (NCDC), the program has expanded its network to include 35 state medical college laboratories across 26 states and union territories. These laboratories monitor AMR trends, providing critical data to inform treatment guidelines and policy decisions (63).
6. Challenges and Limitations
Zoonotic diseases, transmitted from animals to humans, represent a significant public health challenge in India. The country's diverse ecosystems, dense population, and intricate human-animal interactions provide fertile ground for zoonotic outbreaks. Effective surveillance systems are essential to mitigate the risks posed by these diseases; however, India faces numerous challenges in implementing robust and comprehensive mechanisms. Key challenges include inadequate infrastructure, data privacy concerns associated with AI-driven tools, and inconsistent intersectoral coordination. These issues are deeply interlinked, compounding the complexities of establishing a seamless surveillance framework.
A significant hurdle lies in the inadequate diagnostic infrastructure, particularly in rural areas. India's rural regions, often hotspots for zoonotic outbreaks due to close contact between humans and animals, are critically underserved in terms of healthcare facilities. Most rural health centres lack advanced diagnostic tools necessary for the early detection of zoonotic pathogens like the Nipah virus or Crimean-Congo Haemorrhagic Fever. Diagnostic facilities are often limited to basic symptomatic assessments, which can lead to misdiagnoses or delayed detection of outbreaks. Advanced diagnostic tools, such as genomic sequencing, rapid diagnostic kits, or molecular diagnostic technologies, are largely confined to urban centres and specialized research institutions, leaving rural regions vulnerable [67]. The logistical challenges in transporting biological samples from rural to urban laboratories further exacerbate the problem. Maintaining a cold chain for sample preservation is costly and often unfeasible in remote areas, leading to delays in testing and reporting results. Such delays can significantly hinder timely interventions and allow diseases to spread unchecked. Additionally, the financial constraints faced by rural healthcare systems limit investments in modern diagnostic equipment and infrastructure. Limited funding also results in a shortage of trained personnel capable of using advanced diagnostic technologies, further widening the gap between rural and urban healthcare capabilities [68]. Technological gaps remain a persistent issue. Although point-of-care diagnostic devices have the potential to revolutionize healthcare in rural areas, their availability and utilization are minimal. These devices, which could enable rapid and accurate testing at the site of outbreaks, are either unavailable or underused due to cost, lack of awareness, or inadequate training of healthcare workers. Consequently, the absence of timely diagnostic capabilities leads to significant delays in detecting and responding to zoonotic diseases in rural areas [67].
While technological advancements like artificial intelligence (AI) have greatly enhanced disease surveillance, they have also introduced challenges, particularly concerning data privacy and ethical considerations. AI-driven surveillance tools rely on vast amounts of data, including personal health records, environmental data, and livestock health information, to identify patterns and predict outbreaks. However, the collection and use of such data raise significant privacy concerns. Sensitive information, if not adequately safeguarded, is vulnerable to breaches and misuse. For instance, data about livestock health could be exploited by competitors in the agricultural sector, while breaches of personal health records could lead to stigmatization or discrimination [69]. The lack of a robust legal framework governing the ethical use of AI in health surveillance further exacerbates these concerns. India currently lacks comprehensive data protection laws that address the specific needs of AI-driven surveillance systems. This regulatory void creates ambiguities regarding data ownership, usage rights, and accountability in case of breaches. In many cases, communities and healthcare workers are hesitant to adopt AI tools due to fears of data misuse and surveillance overreach. The opacity of AI algorithms, which often function as "black boxes," adds to this scepticism, as users are unable to fully understand how their data is analysed and utilized [70]. Cybersecurity risks also loom large in AI-driven systems. These systems are susceptible to hacking, which can compromise critical health data during outbreaks and erode public trust in surveillance mechanisms. Ensuring the security of data collected through AI tools is paramount, but this requires substantial investment in cybersecurity measures, which is often overlooked in budget allocations. Without robust safeguards, the benefits of AI in zoonotic disease surveillance could be overshadowed by the risks associated with data breaches and privacy violations [71].
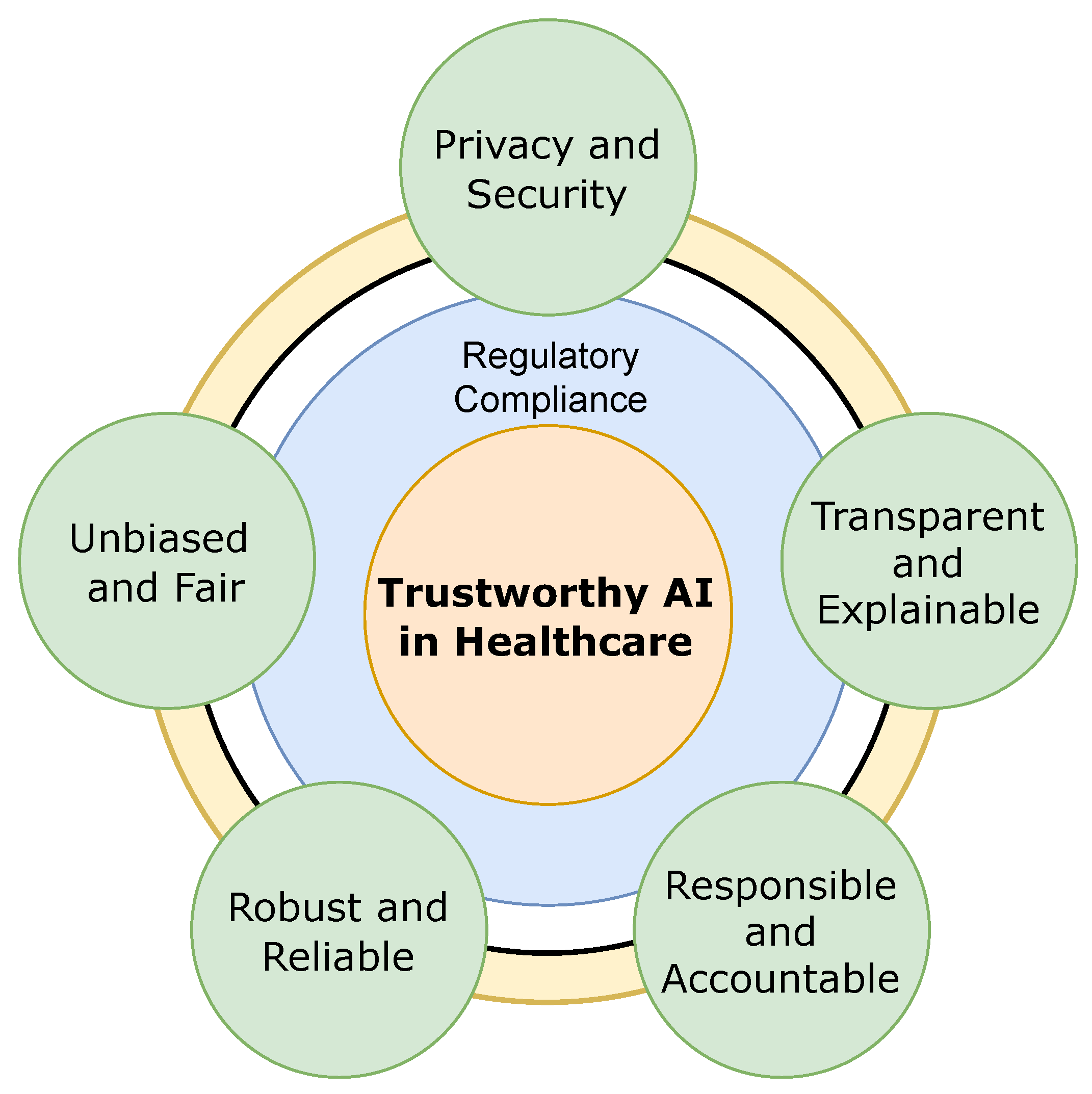
Fig. Framework of trustworthy AI in healthcare [5]
Another critical challenge lies in the inconsistent intersectoral coordination across human, animal, and environmental health sectors. Effective zoonotic disease surveillance requires seamless collaboration among these sectors to monitor disease emergence, identify potential threats, and implement timely interventions. However, in India, such collaboration is often fragmented and uneven. Different states exhibit varying levels of integration among health departments, veterinary services, and environmental agencies, leading to disjointed efforts in controlling outbreaks [68].
Administrative barriers further hinder effective coordination. Bureaucratic silos, overlapping responsibilities, and unclear lines of authority delay decision-making and resource allocation during crises. For instance, during a zoonotic outbreak, the lack of a clear chain of command can result in confusion about which agency should take the lead, leading to delays in implementing control measures. Additionally, communication gaps between state and central agencies exacerbate the problem. The timely sharing of critical information, such as disease outbreaks or pathogen identification, is often hampered by inefficient communication channels, resulting in delayed responses [72]. Resource disparities between states also pose a significant challenge. Wealthier states, with better-funded healthcare systems, are often better equipped to manage zoonotic disease surveillance and response. In contrast, poorer states struggle to align resources effectively, leaving them vulnerable to outbreaks. This disparity creates a patchwork of surveillance capabilities across the country, with some regions being well-prepared while others remain under-equipped [73]. Training and capacity-building efforts are also inadequate, limiting the effectiveness of intersectoral collaboration. Professionals in one sector often lack awareness of protocols and requirements in other sectors, reducing their ability to work together effectively. For example, veterinarians may not be familiar with human health reporting systems, while medical professionals may not fully understand the implications of zoonotic diseases in livestock. Cross-sectoral training programs are essential to bridge this gap, but they remain scarce and underfunded [72].
7. Case Studies of Successful Surveillance Systems in India:
7.1 H5N1 Avian Influenza: A Model for Effective Surveillance and Control in India:
H5N1 avian influenza, a highly pathogenic strain of the influenza virus, poses a significant zoonotic threat due to its potential to infect humans through direct or indirect contact with infected birds. India has experienced multiple outbreaks of H5N1 since its first recorded case in 2006, primarily affecting poultry farms across states such as Maharashtra, West Bengal, Assam, and Odisha. Given the severe consequences of the virus, the Indian government has implemented a series of surveillance and containment measures to prevent major human spillovers and mitigate economic losses in the poultry industry. [74] The foundation of India’s avian influenza surveillance program is the National Action Plan for Avian Influenza (NAPAI), which aligns with the broader One Health approach integrating human, animal, and environmental health sectors. This plan has facilitated the systematic monitoring of poultry farms, wetlands, and live bird markets, which are recognized as high-risk zones for virus transmission. A critical component of this strategy is the Avian Influenza Surveillance and Response System (AISRS), which is jointly operated by the Department of Animal Husbandry and Dairying (DAHD), the Indian Council of Agricultural Research (ICAR), and the National Centre for Disease Control (NCDC). This inter-agency collaboration has significantly enhanced India’s preparedness and response capacity.
7.2 Nipah Virus Monitoring: Kerala’s Model for Coordinated Surveillance and Response:
Nipah virus (NiV) is a zoonotic pathogen with a high mortality rate in humans, primarily transmitted through direct contact with infected fruit bats (Pteropus species), contaminated food, or human-to-human transmission. Kerala, a state in southern India, has experienced multiple outbreaks of Nipah virus, with the most significant incidents occurring in 2018, 2019, and 2023. The state’s ability to minimize large-scale outbreaks has been attributed to its robust disease surveillance system, rapid response protocols, and an integrated One Health approach.
During the 2023 outbreak in Kozhikode, authorities leveraged AI-driven contact tracing tools and GIS mapping to track case clusters efficiently. Hospitals were equipped with dedicated Nipah isolation wards, and medical teams were provided with personal protective equipment (PPE) to minimize nosocomial transmission. [77]
8. Future Directions: Strengthening Zoonotic Disease Surveillance in India and Beyond:
8.1 Scaling One Health Initiatives:
Expanding One Health frameworks to rural regions is essential for effective zoonotic disease control. Rural areas often serve as hotspots for zoonotic spillovers due to close interactions between humans, livestock, and wildlife. Implementing One Health approaches in these settings involves integrating human, animal, and environmental health sectors to facilitate early detection and response to emerging threats. This can be achieved by establishing multidisciplinary teams that conduct joint surveillance, share data, and coordinate interventions. Characterizing animal mobility across borders can enhance the prevention and control of zoonotic diseases in rural communities (80). Additionally, assessing One Health capacities for transboundary zoonotic diseases can help identify operational gaps and strengthen collaborative networks between neighbouring regions (81). In the Indian context, integrating veterinary and public health systems is crucial due to the country’s large livestock population, which increases the risk of zoonotic disease transmission. Strengthening coordination between veterinary health services and human healthcare providers can enhance disease detection and control. Programs like the National Animal Disease Control Program (NADCP) need to be better aligned with human disease surveillance efforts to ensure a more comprehensive response. Additionally, rural regions in India lack robust disease monitoring mechanisms. Establishing community-based disease monitoring networks and integrating digital platforms for early warning systems can help strengthen surveillance efforts. Since many zoonotic pathogens originate from wildlife, expanding India’s wildlife disease surveillance programs, particularly in biodiversity hotspots like the Western Ghats and Northeast India, can improve early detection and intervention.[82, 72, 83]
8.2 Improving Global Collaboration:
Strengthening cross-border data sharing is vital for managing zoonotic diseases that transcend national boundaries. Effective global collaboration enables timely exchange of information on emerging pathogens, facilitating coordinated responses and containment strategies. Establishing standardized protocols for data collection and sharing can enhance transparency and trust among nations. The World Health Organization's One Health Initiative emphasizes the importance of multisectoral collaboration to address health risks at the human-animal-environment interface (84). Implementing strategic frameworks for cross-border surveillance and information sharing can improve regional collaboration for emergency responses to public health events, particularly in disease-prone areas (85). India plays a pivotal role in disease surveillance across South Asia, as it shares borders with multiple countries that are zoonotic hotspots. Strengthening cooperation under frameworks like the South Asian Association for Regional Cooperation (SAARC) Disease Surveillance Network can improve cross-border information sharing and coordinated responses to emerging outbreaks. India has also been actively collaborating with global organizations such as the WHO and the Food and Agriculture Organization (FAO) to enhance zoonotic disease surveillance. Strengthening joint programs focused on research and response mechanisms can improve pandemic preparedness (86). Additionally, India’s advancements in artificial intelligence and biotechnology present an opportunity to develop AI-powered early warning systems for zoonotic diseases. Collaborating with global AI research initiatives to create predictive models for outbreak forecasting can significantly enhance preparedness and response efforts. [87, 88,89]
8.3 Promoting Equity:
Ensuring that low-income regions have access to advanced diagnostic tools and surveillance technologies is crucial for global health security. Resource-limited areas often face challenges in early detection and response to zoonotic outbreaks due to inadequate infrastructure and funding. Addressing these disparities involves providing financial support, capacity-building programs, and technology transfer to strengthen local health systems. Scaling up One Health approaches in regions like the Greater Mekong Subregion has proven effective in enhancing early warning and disease surveillance systems, ultimately improving preparedness and response capabilities (90). Additionally, a strong One Health approach to control zoonotic disease threats can help identify priority pathogens and combat antimicrobial resistance, benefiting underserved communities and enhancing global pandemic preparedness (91). In India, expanding low-cost diagnostic technologies is essential to bridge the gap in zoonotic disease detection between urban and rural areas. Many remote regions lack access to advanced diagnostic tools, which delays disease identification and containment efforts. Developing and deploying rapid, affordable, and point-of-care diagnostic kits for zoonotic diseases, such as those under India’s National Biopharma Mission, can significantly improve early detection rates. Furthermore, India’s major research and healthcare facilities are concentrated in urban centres, making it difficult for rural populations to receive timely medical intervention during outbreaks. Expanding telemedicine services and mobile health units in rural areas can help address this issue and ensure equitable healthcare access. Additionally, ensuring the equitable distribution of vaccines and therapeutics for zoonotic diseases is critical. Strengthening India’s vaccine distribution network, particularly through public-private partnerships, can enhance accessibility in underserved regions and improve overall disease control efforts. Lastly, integrating indigenous knowledge into disease surveillance and response strategies can provide region-specific solutions to zoonotic threats. Traditional knowledge systems, particularly those followed by tribal communities in India, often contain valuable insights into disease patterns and natural remedies. Incorporating these insights into formal surveillance frameworks can improve India’s overall preparedness against emerging zoonotic diseases. [92,93,94]. By focusing on these future directions, India and global stakeholders can collaboratively enhance zoonotic disease surveillance and control, ultimately safeguarding public health. Strengthening One Health initiatives, improving international collaboration, and promoting equitable access to diagnostic and treatment solutions will ensure a more resilient and prepared health system capable of responding effectively to emerging disease threats.
9.CONCLUSION:
Strengthening zoonotic disease surveillance in India requires a multi-sectoral approach that integrates human, animal, and environmental health. The One Health strategy has proven effective in outbreak management, particularly in Kerala’s Nipah virus containment efforts. Emerging technologies such as artificial intelligence, genomic surveillance, and blockchain-based tracking systems offer significant potential in improving disease prediction and response. However, challenges such as weak diagnostic infrastructure, data privacy concerns, and inadequate inter-agency coordination must be addressed. Moving forward, investments in digital surveillance, workforce training, and cross-border collaboration will be critical in enhancing India's preparedness against zoonotic threats. Policymakers must also ensure equitable access to advanced surveillance tools, particularly in rural and high-risk areas. By adopting a proactive and technology-driven approach, India can strengthen its resilience against future zoonotic outbreaks and contribute to global health security.
REFERENCES



Ketan Sonwane, Gauri Soni, Dr. Gaurao Damre*, Silent Invaders: In Masking the Hidden Epedemics of Zoonotic Disease in India Through Next Generation Surveillance, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 7, 1987-2018. https://doi.org/10.5281/zenodo.15912216
 10.5281/zenodo.15912216
10.5281/zenodo.15912216