
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
SNJB’s Shriman Sureshdada Jain College of Pharmacy, Chandwad, Nashik, 423101.
Regulatory procedures for generic medication approval are crucial to guaranteeing the availability of safe, effective, and inexpensive medicines while preserving strong public health safeguards. This research compares the regulatory requirements for generic drugs in Myanmar, Malaysia, Madagascar, and India, with an emphasis on approval procedures, dossier forms, and compliance expectations. The key regulatory parameters analyzed include governing authorities, legal frameworks, dossier formats, submission systems, Good Manufacturing Practice (GMP) certification, bioequivalence (BE) requirements, stability data, local representation, review timelines, and post-marketing surveillance obligations. Myanmar and Malaysia generally follow the ASEAN Common Technical Dossier (ACTD), which reflects regional regulatory harmonization attempts, whereas India uses the CDSCO SUGAM portal to submit an Abbreviated New Drug Application (ANDA) based on the Common Technical Document. Madagascar uses a national regulatory approach that is consistent with internationally recognized quality, safety, and efficacy requirements. Regardless of procedural differences across jurisdictions, all four regulatory systems priorities proof of pharmaceutical quality, therapeutic equivalency, and manufacturing conformity. The assessment reveals both convergence and variation among national regulatory regimes, as well as potential obstacles in dossier preparation and approval timescales for pharmaceutical businesses. Regulatory affairs experts must have a thorough awareness of these regulatory variances in order to optimize submission strategies, assure compliance, and allow fast market entry for generic pharmaceuticals in varied regulatory settings.
Generic drugs are medications that are the same as brand-name drugs in dosage form, safety, strength, quality, and intended use, but are typically less expensive. They become available after the original brand-name drug's patent expires and must meet the same strict standards as the brand-name product for approval. Generic drugs contain the same active ingredients, have the same bioequivalence, and provide the same clinical benefit, though they may differ in inactive ingredients, colour, shape, and flavour.Generic drug product is comparable to an innovator drug product in following cases:
1. Dosage form
2. Strength
3. Route of administration
4. Quality
5. Intended Use
MYANMAR
In Myanmar, the regulation of pharmaceuticals, including generic drugs, is overseen by the Ministry of Health (MoH) through the Food and Drug Administration (FDA-Myanmar). Over recent years, Myanmar has strengthened its regulatory infrastructure to align more closely with global regulatory best practices, the ASEAN Common Technical Dossier (ACTD) format, and harmonized guidelines under the ASEAN Pharmaceutical Product Working Group (PPWG). The registration process for generic drugs in Myanmar requires submission of comprehensive administrative, quality, non-clinical, and clinical evidence, including proof of bioequivalence, manufacturing compliance with Good Manufacturing Practices (GMP), and product safety data. [1]
MALAYSIA
In Malaysia, the system for regulating medicines is managed by the National Pharmaceutical Regulatory Agency (NPRA) under the Ministry of Health (MOH). NPRA is responsible for checking the safety, quality, and effectiveness of generic medicines. Malaysia follows the Control of Drugs and Cosmetics Regulations 1984 and uses the ASEAN Common Technical Dossier (ACTD) format for submissions. It also requires GMP certification and bioequivalence studies to ensure generics are equal to the original products. [2]
MADAGASCAR
In Madagascar, the regulation of pharmaceuticals, including generic medicines, is under the authority of the Ministry of Public Health through the Madagascar Drug’s Agency (Agence du Medicament de Madagascar). The country has established a structured regulatory system to ensure the medical and technical evaluation of registration dossiers as an essential step in guaranteeing the quality of medicines placed on the market. The national registration procedures are designed to comply with current regulations and provide a common registration process for pharmaceutical sector operators. Applicants are required to submit comprehensive administrative, pharmaceutical, analytical, biological, and clinical documentation, along with evidence of Good Manufacturing Practices (GMP), product quality, safety, and efficacy, to support the granting of Marketing Authorization (MA). [3]
INDIA
Generic drug approval in India is handled by the Central Drugs Standard Control Organization (CDSCO), which requires an abbreviated new drug application (ANDA) for a generic drug to prove it is bioequivalent to the original product. While the applicant doesn't need to resubmit preclinical and clinical trial data for safety and efficacy, the generic drug must meet quality standards, and the application must be approved by the CDSCO before it can be marketed in India. [4]
MYANMAR
Generic Drug Registration Process In Myanmar [5]
Myanmar's drug regulatory authority is the Food and Drug Administration (FDA), operating under the Ministry of Health, responsible for ensuring the safety, quality, and efficacy of food, drugs, cosmetics, and medical devices, using the ASEAN Common Technical Dossier (ACTD) format for drug registration, requiring local representation and an online portal for submissions, with registrations typically valid for five years.
Application dossier requirements
The application dossier for registration has to comply with the ASEAN Common Technical Dossier format. Generic drugs dossier contains Part I (Administrative documents and product information) and Part II documentation (Quality documents for drug substance and drug product).
Application must be submitted in person by:
Local company representatives must be technically competent employees.
Registration Dossier Structure
The Dossier consist of Four parts, each covering a specific set of data. The dossier required for the generic drug
|
Part |
Section |
|
Part I |
Administrative and Product information |
|
Part II |
Quality Drug Substance, Drug Product, stability, Validation |
|
Prat III |
Non-Clinical Pharmacology, Toxicology summaries, non-clinical studied |
|
Part IV |
Clinical Clinical trial data, Bioequivalence report |
To register generic drugs in Myanmar documents require
Registration Process [5]

The process to register for the generic product can be summarized into the following steps
Prerequisites for submission of application
Submission Of Application:
The applicant has to start a new application and needs to obtain an application number automatically. The applicant needs to consider the following points to make an application.
Evaluation And Approval:
Form For Registration:
Approval Timeline:
In Myanmar, the generic drug registration process usually requires approximately 6-12 months. Validity of registration is 5 years. [6]
Fees Required For Registration:
MALAYSIA
Malaysia's drug regulatory authority is the National Pharmaceutical Regulatory Agency (NPRA), operating under the Ministry of Health (MOH), responsible for ensuring the quality, safety, and efficacy of medicines, health supplements, traditional products, and cosmetics through registration, licensing, and post-market surveillance (pharmacovigilance) to protect public health
Generic Drug Registration Process In Malaysia
Types of generic drugs in Malaysia
Generics are classified into two groups
Documents Require:
Registration Process: [8, 9, 10]
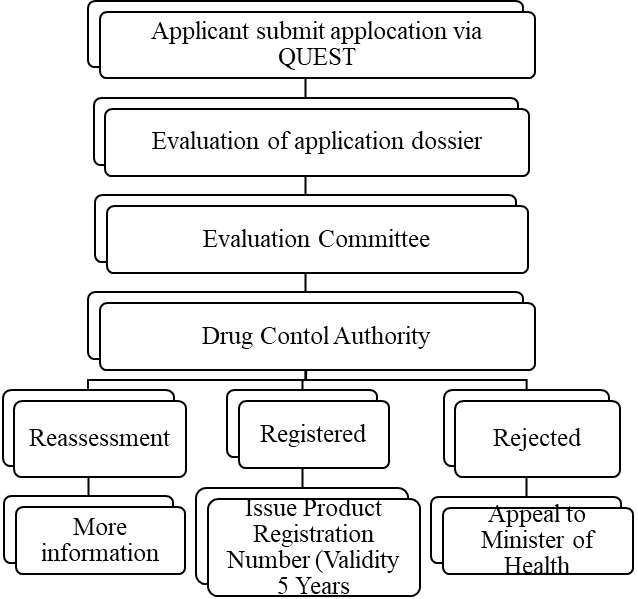
Fees Required For Registration: RM 2200-RM3000 (51,371-70,052 INR)
MADAGASCAR [3, 11]
Generic Drug Registration Process In Madagascar
For Imported Drugs 200 € (18000 INR)
Locally Manufactured Drugs 164,000 MGA (3372 INR)
Documents Required For Registration Process:
1. Administrative Documents
2. Pharmaceutical (Quality) Documents
3. Analytical Documents
4. Stability Data
5. Clinical / Bioequivalence / Safety Data
6. Sample Requirements
Samples must be:
(Quantities depend on dosage form such as tablets, capsules, injections, creams, suspensions, etc.)
7. Language & Format Requirements
INDIA
Registration Process In India
India's national regulatory body under the Ministry of Health, ensuring drug, cosmetic, and medical device quality, safety, and efficacy by regulating imports, new drug approvals, clinical trials, and licensing, working with state regulators for uniform enforcement nationwide from its New Delhi HQ, with zonal/port offices and labs across India.The generic drug registration process in India involves submitting an Abbreviated New Drug Application (ANDA) via the CDSCO SUGAM portal, providing a Common Technical Document (CTD) with CMC data, and crucially, conducting Bioequivalence (BE) studies to prove sameness to the reference drug, followed by technical review, potential site inspection, and final marketing approval, aiming for a 180-day timeline for faster approvals.
Documents Require For Registration Of Generic Drug In India:
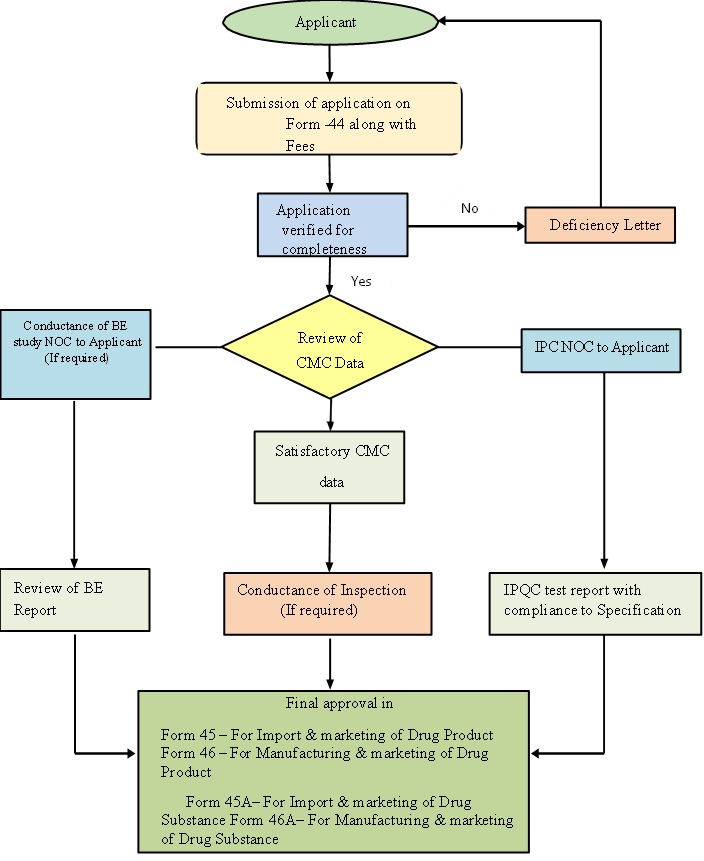
Approval Process For Drug Substance /Drug Product Approved By DCGI Within A Period Of Four Years From The Date Of Its First Approval.
Fees And Time: [12]
Generic drug registration costs INR 15,000.
6 to 12 months from submission.
COMPARATIVE TABLE OF MYANMAR, MALAYSIA, MADAGASCAR AND INDIA:
|
Parameter |
Myanmar |
Malaysia |
Madagascar |
India |
|
Flag |

|

|

|

|
|
Regulatory Authority |
FDA Myanmar (MoH) |
National Pharmaceutical Regulatory Agency (NPRA), MoH |
Madagascar Drug’s Agency (Ministry of Public Health) |
CDSCO (DCGI), MoHFW |
|
Regulatory Basis |
National Drug Law + ASEAN Guidelines |
Control of Drugs & Cosmetics Regulations 1984 |
National Medicines Regulations |
Drugs & Cosmetics Act, 1940 |
|
Dossier Format |
ACTD |
ACTD |
National format (similar to CTD) |
CTD (ANDA-based) |
|
Language |
English |
English |
English / French |
English |
|
Submission Portal |
FDA Myanmar online system |
QUEST 3+ system |
Physical submission |
SUGAM portal |
|
Local Authorized Representative |
Mandatory (resident in Myanmar) |
Mandatory |
Mandatory |
Mandatory (for imports) |
|
GMP Requirement |
Required |
Required |
Required |
Required |
|
CPP Requirement |
Required |
Required (for imports) |
Required (WHO format) |
Required (for imports) |
|
Bioequivalence (BE) |
Mandatory |
Mandatory |
Required (case-based) |
Mandatory |
|
Stability Data |
Required |
Zone IVB required |
Real-time + accelerated (3 batches) |
Required (as per Schedule Y) |
|
Clinical / Non-clinical Data |
BE + summaries |
BE only |
BE / safety justification |
BE (no full clinical data) |
|
Sample Submission |
Mandatory |
Not routine |
Mandatory |
Case-based |
|
Registration Timeline |
6–12 months |
12–18 months |
~4 months (new MA) |
6–12 months |
|
Registration Validity |
5 years |
5 years |
5 years |
Per approval (renewal applicable) |
|
Official Fees |
~800,000 Kyats (~35,040 INR) |
RM 2,200–3,000 (51,371-70,052 INR) |
Manufactured Drugs 164,000 MGA (3372 INR) |
?15,000–?25,000 |
|
Post-Market Surveillance |
Yes |
Yes (Pharmacovigilance) |
Yes |
Yes (PvPI) |
CONCLUSION
This research indicates that, while Myanmar, Malaysia, Madagascar, and India use distinct regulatory routes for generic drug approval, all four countries are committed to guaranteeing generic pharmaceutical quality, safety, and therapeutic equivalency. Myanmar and Malaysia closely follow ASEAN regulatory harmonization through the ASEAN Common Technical Dossier (ACTD), whereas India uses an Abbreviated New Drug Application (ANDA) pathway based on the Common Technical Document (CTD). Madagascar adopts a nationally defined approach that is compliant with international regulatory requirements. Good Manufacturing Practice (GMP) compliance, bioequivalence studies, stability data, and post-market surveillance continue to be critical regulatory criteria in all nations. Recognizing both common principles and country-specific regulatory variances is critical for efficient regulatory planning and timely market access. Further regulatory convergence and capacity building may improve approval efficiency and expand access to inexpensive generic pharmaceuticals.
REFERENCE


Atharva Gandhi*, Dr. G. D. Basarkar, Study Of Regulations for Generic Drug Approval in Myanmar, Malaysia, Madagascar and India, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 650-660. https://doi.org/10.5281/zenodo.18491264
 10.5281/zenodo.18491264
10.5281/zenodo.18491264