
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Shri K. R Pandav Institute of Pharmacy
One of the most common cancers in women worldwide is cervical cancer, which is primarily caused by high-risk Human Papillomavirus (HPV) infection, especially types 16 and 18. By deactivating the tumor suppressor proteins p53 and retinoblastoma protein (pRb), respectively, the viral oncoproteins E6 and E7 are essential in the development of cervical cancer. E6 and E7 are very specific targets for therapeutic intervention because their continuous expression encourages unchecked cell proliferation, genomic instability, and resistance to apoptosis. The molecular mechanisms behind HPV-induced cervical cancer are highlighted in this review, along with cutting-edge tactics that target E6/E7 oncogenes. New molecular techniques like CRISPR/Cas9 gene editing, RNA interference (RNAi), and therapeutic vaccines show great promise in reestablishing normal cellular regulatory pathways and specifically inhibiting the expression of oncogenes. Furthermore, in preclinical studies, natural substances such as curcumin, resveratrol, epigallocatechin gallate (EGCG), and indole-3-carbinol have demonstrated encouraging inhibitory effects on E6/E7 activity. Current diagnostic and preventive methods, such as molecular HPV DNA testing, Pap smear screening, and HPV vaccination, are also covered in the review. All things considered, targeting E6/E7 is a precision-based therapeutic strategy that may enhance treatment results while reducing systemic toxicity in cervical cancer linked to HPV.
The broad family of non-enveloped, double-stranded circular DNA viruses known as papillomaviruses (PVs) infects the mucosa and skin. Despite the fact that there are over 200 different types of PV, the mechanisms of PV infection are essentially the same: Cellular binding, internalization, and virus trafficking are mediated by two viral capsid proteins, the major capsid protein (L1) and the minor capsid protein (L2). More than 150 different types of viruses make up the large family known as human papillomaviruses, or HPVs. Nearly all cases of cervical cancer and most cases of anal, head, and neck cancer are caused by HPVs with transforming potential. HPV16 is the most researched HPV type because it causes the majority of cervical cancer cases. The process of HPV16 infection involves several steps. The virus uses an epithelium injury to get to the basal keratinocytes, which are the only cells in the epithelium that undergo mitosis [1].
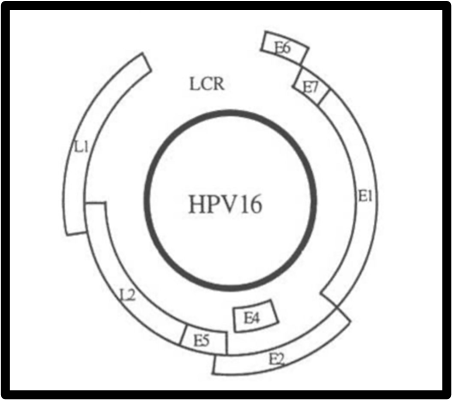
Fig1.1 Structure of HPV
Cervical cancer is the fourth most common cancer in women globally, causing hundreds of thousands of deaths annually, predominantly in low- and middle-income countries. Decades of research have unequivocally established that persistent infection with high-risk Human Papillomavirus (HR-HPV), particularly types 16 and 18, is the necessary causative agent for virtually all cases. The transition from transient infection to malignant neoplasia hinges entirely on the integration of the HR-HPV genome into the host cell's DNA and the subsequent, uncontrolled expression of the viral E6 and E7 oncogenes. These two proteins act as molecular master switches, dismantling the host cell’s critical tumor suppression pathways [2].
Given that E6 and E7 are solely responsible for initiating and maintaining the cancerous state, and are not expressed in normal, healthy cells, they present an exceptionally specific and high-value target for therapeutic intervention. Modern oncology is shifting towards precision medicine, making the targeted elimination or inactivation of E6/E7 an active and urgent area of research for developing curative strategies that minimize systemic toxicity [3].
Fig. 1.2 Carcinoma of Cervix Stock Vector
Human papillomaviruses (HPVs) are the cause of RRP. The Papillomaviridae family includes the non-enveloped, double-stranded circular DNA virus known as HPV. Together with the early and late harbor regions, which code for proteins involved in transcription, replication, and immunological resistance, the HPV genome also includes the non-coding upstream regulatory region (URR) . The structural proteins L1–L2 are encoded by the late (L) region, which also contributes to virus assembly. Conversely, the early proteins E5, E6, and E7 change the molecular mechanisms of HPV-infected cells and function as oncogenes. To date, over 200 distinct HPV types have been identified based on their genomes, and they are classified as low- and high-risk viruses. According to reports, at least 17 high-risk types—including HPVs 16, 18, 31, 33, 35, 39, 45, 51, 52, 55, 56, 58, 59, 68, 73, 82, and 83—co-occur with other oncogenes to contribute to the onset and progression of cancers [4]
The E6/E7 proteins of high-risk HPVs have been found to enhance DNA integration into the host genome, plausibly due to enhanced chromosome rearrangements in high-risk HPV-infected cells, leading to mutation and/or deletion of both viral and host genes . Furthermore, high-risk HPVs’ E6/E7 proteins have the ability to modify DNA repair pathways. Indeed, high-risk HPVs, especially HPV16, have been implicated in vulvar cancer [14], uterine cervical cancer and its precancerous (CIN) lesions [15], anal [16], and oropharyngeal cancers [17]. Low-risk HPV infections (6 and 11), however, are rarely associated with cancer and can result in benign anogenital papillomas and skin warts. Although several types of HPV may contribute to RRP, current research suggests that the low-risk HPV types (HPV6 and 11) are the most common causative factors for the condition. However, RRP has been linked to high-risk HPV infection. Low-risk HPV11 E6/E7 proteins are just as important for the stable maintenance of episomes as high-risk E6 and E7 proteins, per a study by Stephen et al. In RRP-infected individuals, HPV DNA is usually episomal (HPV infects the basal cells and develops a prolonged infection) rather than fused or integrated with host DNA. Numerous microbial agents, such as G. vaginalis and Sneathia spp., have been shown in studies to potentially aid HPV-induced infection, invasion, and multiplication [5].
1.1 HPV Classification and High-Risk Types
HPVs are non-enveloped, double-stranded DNA viruses with a circular genome. They are broadly classified based on their risk for oncogenesis.
Table no. 1
|
Classification |
HPV Types |
Associated Lesions |
Oncogenic Risk |
|
High-Risk (HR-HPV) |
16, 18, 31, 33, 45, 52, 58 |
Cervical, anal, vaginal, vulvar, penile, and oropharyngeal cancers. |
High |
|
Low-Risk (LR-HPV) |
6, 11, 40, 42, 43, 44 |
Genital warts (condyloma acuminata), Recurrent Respiratory Papillomatosis (RRP). |
Low |
HR-HPV types, particularly HPV-16 and HPV-18, account for over 70% of all cervical cancer cases.
The main risk factor for adults is increased sexual activity with multiple partners. Approximately 0.7% of mothers who have HPV genital warts infect their babies. Up to 80% of newborns born to mothers with genital HPV have HPV DNA. There are three types of vertical HPV transmission: perinatal (during or immediately following birth), prenatal (during pregnancy), and periconceptional (around the time of conception). In adults, the main risk factor is increased sexual activity with multiple partners. About 0.7% of mothers with HPV genital warts pass the disease to their infants. HPV DNA is detected in up to 80% of the neonates born to mothers with genital HPV . The vertical transmission of HPV can be categorized into periconceptional (around the time of conception), prenatal (pregnancy), and perinatal (birth or immediately after that). It is known that juvenile-onset RRP is acquired during delivery by vertical transmission, either through the birth canal or through infected placenta and amniotic fluid, from the mother's anogenital site to the child's respiratory tract . Children born to mothers with active HPV infection had a significant risk (231 times higher) of juvenile RRP than children born to mothers without the virus, according to a study by Venkatesan and colleagues [6].
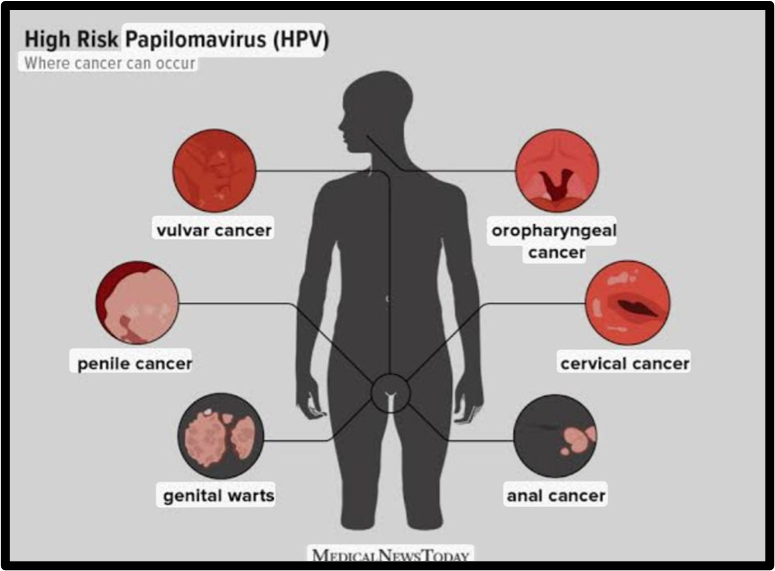
Fig. 1.3 Sexually transmitted infection -MAPEH G8
1.2 The E6/E7 Oncogenic Mechanism
The malignant transition occurs when the HR-HPV genome integrates into the host cell DNA, leading to constitutive, high-level transcription of the early genes E6 and E7.
The E6 oncoprotein primarily targets the host cell's major guardian of the genome, the p53 tumor suppressor protein.
The E7 oncoprotein disrupts the crucial G1-to-S phase cell cycle checkpoint maintained by the retinoblastoma protein (pRb).
The concurrent inactivation of both p53 and pRb by E6 and E7, respectively, provides the optimal environment for malignant transformation: the cell gains unlimited proliferative capacity (via E7) while simultaneously losing the ability to initiate cell death or repair DNA damage (via E6) [8] .
Fig. 1.4 HPV and its Effects
1.3 History:
The Foundational Science (1980s–1990s)
1.3.1 The Rationale: E6/E7 as the Achilles' Heel
The entire field of therapeutic targeting of cervical cancer began with a crucial discovery in the 1980s by Harald zur Hausen: High-risk Human Papillomavirus (HPV), specifically types HPV-16 and HPV-18, is the necessary cause of nearly all cervical cancers.
1.3.2. The Primary Molecular Mechanisms of E6/E7 Action
Targeting strategies are built upon understanding how E6 and E7 drive cancer:
Table no.2
|
Oncogene |
Primary Cellular Target |
Molecular Mechanism |
Oncogenic Outcome |
|
E6 |
p53 (Tumor Suppressor) |
E6 hijacks the cellular E3 ubiquitin ligase E6-AP to ubiquitinate and induce the proteasomal degradation of p53. |
Loss of DNA repair, failed apoptosis, genomic instability. |
|
E7 |
pRb (Retinoblastoma Protein) |
E7 binds to and inactivates the pRb pocket protein, releasing the E2F transcription factor. |
Constitutive cell cycle progression from G1 to S phase, loss of cell cycle control. |
1.3.3. First Generation Molecular Targeting: Gene Therapy
The initial attempts focused on genetically disrupting the E6/E7 mRNA or DNA, aiming to revert the malignant phenotype.
1.3.4. The Rise of Therapeutic Cancer Vaccines
The fact that E6 and E7 are viral proteins, foreign to the host, makes them perfect targets for the immune system. The goal of a therapeutic vaccine is to stimulate a robust, tumor-killing cytotoxic T-lymphocyte (CTL) response against E6 and E7.
1.3.5. The Rationale for Natural Strategies (Phytotherapy)
As an alternative to complex molecular and immunological approaches, significant research has focused on natural compounds (phytochemicals) that can disrupt the E6/E7 pathway.
1.3.6. Major Historical and Current Natural Agents
Table no.3
|
Strategy/ Compound |
Source |
Target Oncogene(s) |
Key Historical/ Preclinical Finding |
|
Curcumin |
Turmeric (Curcuma longa) |
E6/E7 (mRNA, Protein) |
Demonstrated the ability to reduce E6/E7 mRNA expression, leading to restoration of p53 and pRb function, and subsequent apoptosis in cervical cancer cell lines. |
|
Resveratrol |
Grapes, Red Wine (Polygonum cuspidatum) |
E6/E7 (Protein) |
Shown to interfere with the E6/p53 and E7/pRb interactions, potentially through structural changes or degradation of E6/E7 proteins themselves. |
|
Green Tea Polyphenols (EGCG) |
Green Tea (Camellia sinensis) |
E6 (Protein) |
Early studies demonstrated that epigallocatechin gallate (EGCG) could bind to E6, inhibiting E6's ability to degrade p53, thereby restoring p53 function. |
|
Indole-3-Carbinol (I3C) |
Cruciferous Vegetables |
E7 (Protein) |
I3C (and its metabolite DIM) has been shown to induce G1 cell cycle arrest, a phenotype consistent with pRb re-activation following E7 inhibition. |
Clinical Relevance: While in vitro data is strong, clinical translation is challenging due to low bioavailability, making these compounds better suited for high-grade pre-cancers (CIN 2/3) or as adjunct therapies [12] .
1.3.7 The Global Burden of Cervical Cancer and the HPV Link
Cervical cancer remains a significant global health challenge, particularly in developing countries. It is the fourth most common cancer in women worldwide, with an estimated 604,000 new cases and 342,000 deaths annually. The vast majority of cervical cancer cases (over 99%) are caused by persistent infection with high-risk human papillomaviruses (HPVs). Among these, HPV types 16 and
18 are responsible for approximately 70% of all cervical cancers.The journey from HPV infection to cervical cancer is a multi-step process. Initial HPV infection often clears spontaneously. However, in a subset of women, the infection persists, leading to precancerous lesions and, if left untreated, invasive cervical cancer. Understanding this progression has been crucial in developing preventive and therapeutic strategies.
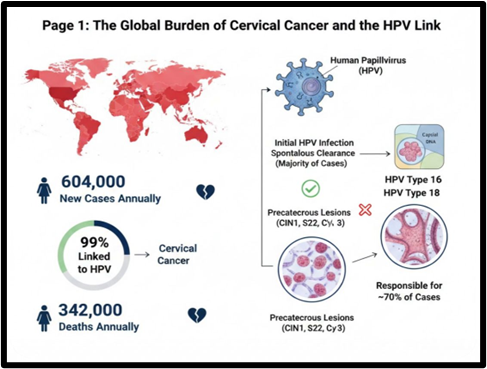
Fig.1.5 Global Burden of Cervical Cancer
1.3.8 The Role of HPV Oncogenes E6 and E7 in Carcinogenesis
The high-risk HPV types encode two key oncoproteins, E6 and E7, which are central to the development and progression of cervical cancer. These proteins manipulate cellular pathways to promote uncontrolled cell growth and evade immune surveillance.
The E6 oncoprotein primarily targets and degrades the tumor suppressor protein p53. p53 is a critical guardian of the genome, initiating cell cycle arrest or apoptosis in response to DNA damage. By degrading p53, E6 allows cells with damaged DNA to continue proliferating, accumulating mutations that can drive cancerous transformation.
The E7 oncoprotein primarily binds to and inactivates the retinoblastoma tumor suppressor protein (pRb). pRb is a crucial regulator of the cell cycle, preventing cells from entering the S-phase (DNA replication) until conditions are favorable. By inactivating pRb, E7 disrupts cell cycle control, leading to uncontrolled cell division. Additionally, E7 can interfere with other cellular pathways involved in DNA repair and immune responses.
The persistent expression of both E6 and E7 is essential for maintaining the malignant phenotype of HPV-positive cervical cancer cells. This makes these oncogenes attractive targets for therapeutic intervention [13].
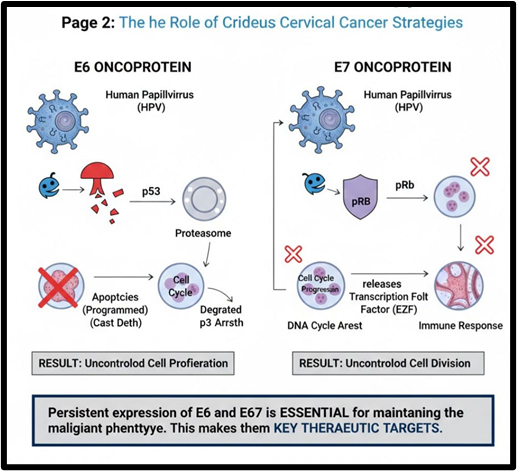
Fig.1.6 the Role of Crideus Cancer Strategies
1.3.8 Natural Strategies and Immunotherapy
Beyond direct molecular interventions, natural compounds and immunotherapeutic approaches are gaining traction in the fight against HPV-driven cervical cancer.
Natural Strategies: Several natural compounds have demonstrated anti-HPV activity by modulating E6/E7 expression or function. For example, compounds like curcumin (from turmeric), resveratrol (from grapes), and epigallocatechin gallate (EGCG) from green tea have been shown to downregulate E6/E7 expression, restore p53 and pRb function, and induce apoptosis in HPV-positive cancer cells. These compounds often work through various mechanisms, including epigenetic modulation and interference with signaling pathways critical for viral gene expression. While promising, further research and clinical trials are needed to establish their efficacy and safety as therapeutic agents.
Immunotherapy: The immune system plays a crucial role in controlling HPV infection and eliminating HPV-infected cells. However, E6 and E7 can also promote immune evasion. Immunotherapeutic strategies aim to re-engage the host immune system to recognize and destroy HPV-positive cancer cells. These include:
These natural and immunotherapeutic approaches offer diverse avenues for combating cervical cancer, often with fewer side effects than conventional treatments [14] .
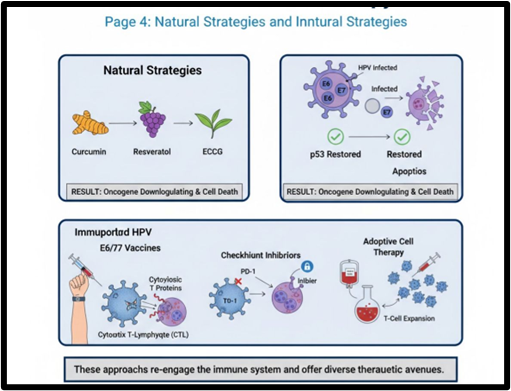
Fig.1.7 Natural Strategies and Inntural Strategies
1.4 Cervical Cancer
Cervical cancer (CC) is the fourth most common cancer in women globally, according to Bray et al. (2024). The International Agency for Research on Cancer (IARC) reports that the incidence of CC rose from 13.3 cases per 100,000 in 2020 to 14.1 cases per 100,000 in 2022. During that period, the mortality rate remained high at roughly 7 per 100,000 people (Sung et al., 2021; Bray et al., 2024). The outlook for CC is worrisome due to the increasing incidence rates and the consistently high mortality rates. Even more concerning is the disproportionate burden of CC, which is concentrated in low-income areas like Southeast Asia, South America, Melanesia, and sub-Saharan Africa (Bray et al., 2018, 2024; Sung et al., 2021). and vaccines. A thorough understanding of the natural history of HPV infection and the progression of precancerous lesions is essential for developing effective screening, prevention, and treatment strategies for CC. The underlying causes of this imbalance are probably disparities in screening accessibility, treatment uptake, health awareness, healthcare resource allocation (Qju et al., 2021). and vaccines. Creating efficient screening, prevention, and treatment plans for CC requires a deep comprehension of the natural history of HPV infection and the development of precancerous lesions. Particularly noteworthy is the link between cervical carcinogenesis and human papillomavirus (HPV) infection. The development of CC has been linked to persistent HPV infection (Arbyn et al., 2015; Wright et al., 2015), which calls for improvements in HPV testing methods and vaccinations. Creating efficient screening, prevention, and treatment plans for CC requires a deep comprehension of the natural history of HPV infection and the development of precancerous lesions .
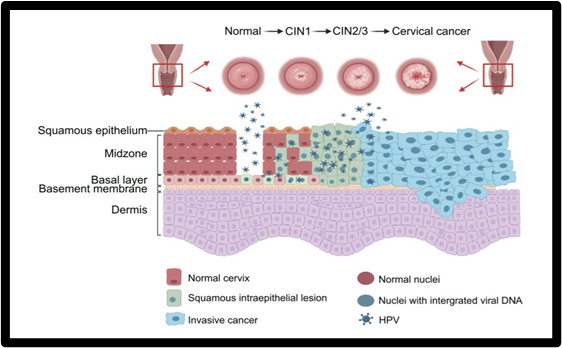
Fig. 1.8 HPV Lifecycle and CC Progression
CC progression and the HPV life cycle. The HPV life cycle starts when the virus infects basal epithelial cells, where it replicates at a low level. The viral oncogenes are progressively expressed as the cells differentiate, which causes the host cells to proliferate uncontrollably. Following this, the virus collects and emerges from differentiated epithelial cells. CIN, which can range from mild abnormalities (CIN1) to severe abnormalities (CIN3), can be caused by a chronic HR-HPV infection. This may eventually progress to invasive CC if treatment or reversal is delayed [15]
2. METHODOLOGY
2.1: Introduction and The Role of Cytology
Human Papillomavirus (HPV) infection is the primary cause of cervical cancer and is implicated in other anogenital and oropharyngeal cancers. The clinical utility of HPV detection methods lies in primary screening, triage of women with abnormal cytology, and monitoring after treatment. Given that persistent infection with high-risk (HR) HPV types (e.g., HPV 16, 18, 31, 33, 45, etc.) is the prerequisite for cancer development, most current assays are focused on detecting the nucleic acid (DNA or mRNA) of these carcinogenic types.
The Papanicolaou (Pap) smear, or cytology, is not a direct HPV detection method but is the traditional cornerstone of cervical cancer screening. It identifies the cytopathic effects of the virus on cervical epithelial cells.
Table no.4
|
Principle |
Procedure |
Key Findings (Morphology) |
|
Cellular Morphology |
Cells collected from the cervix are stained (Papanicolaou stain) and examined microscopically by a cytopathologist. |
Koilocytes: The morphological hallmark of HPV infection. These are squamous epithelial cells with enlarged, hyperchromatic, and often wrinkled nuclei, surrounded by a distinct perinuclear clear halo (vacuole). |
|
Purpose |
Detects abnormal cell changes (dysplasia or precancerous lesions, classified by the Bethesda System: ASCUS, LSIL, HSIL). |
Abnormal cells suggest the presence of a persistent, high-grade lesion, which is almost always driven by HR-HPV. |
2.1.2. Limitations of Cytology
2.2 Signal Amplification Assays (Hybrid Capture)
Nucleic acid hybridization methods were among the first molecular tests approved for HPV screening. These methods use probes to bind to target HPV DNA, which is then detected through a signal amplification system.
2.2.1 Hybrid Capture 2 (HC2) Technology
The HC2 test (QIAGEN) is a classic example of a signal-amplification assay and was historically the most widely used.
Table no.5
|
Step |
Principle |
Detail |
|
1. Target Hybridization |
Liquid-Phase Hybridization |
Target HPV DNA extracted from the clinical sample is denatured into single strands. These strands hybridize in solution with a cocktail of RNA probes specific for 13-14 high-risk HPV types (e.g., 16, 18, 31, 33, 45, etc.), forming stable RNA/DNA hybrids. |
|
2. Hybrid Capture |
Solid-Phase Capture |
The RNA/DNA hybrids are captured onto a solid phase (e.g., a plastic well) coated with universal capture antibodies that specifically bind to the hybrid structure, regardless of the HPV type. |
Providing a comprehensive, five-page, in-depth technical analysis requires a structured approach that details the principles, protocols, advantages, and limitations of the primary HPV detection methods.
Here is a detailed breakdown of HPV detection methods used in clinical and research settings [17]
2.2.2 Signal Amplification Assays (Hybrid Capture)
For HPV screening. These methods use probes to bind to target HPV DNA, which is then detected through a signal amplification system.
The HC2 test (QIAGEN) is a classic example of a signal-amplification assay and was historically the most widely used.
Table no.6
|
Step |
Principle |
Detail |
|
1. Target Hybridization |
Liquid-Phase Hybridization |
Target HPV DNA extracted from the clinical sample is denatured into single strands. These strands hybridize in solution with a cocktail of RNA probes specific for 13-14 high-risk HPV types (e.g., 16, 18, 31, 33, 45, etc.), forming stable RNA/DNA hybrids. |
|
2. Hybrid Capture |
Solid-Phase Capture |
The RNA/DNA hybrids are captured onto a solid phase (e.g., a plastic well) coated with universal capture antibodies that specifically bind to the hybrid structure, regardless of the HPV |
|
3. Signal Amplification |
Chemifluorescent Detection |
A second set of antibodies, conjugated to a reporter enzyme (e.g., Alkaline Phosphatase), binds to the captured hybrids. Multiple reporter molecules bind to each captured hybrid, resulting in a significantly amplified signal. |
|
4. Measurement |
Luminometry |
A chemiluminescent substrate is added. The reporter enzyme cleaves the substrate, producing light that is measured by a luminometer. The resulting light output is proportional to the concentration of target HPV DNA in the original sample. |
Advantages and Limitations of HC2
2.3 Target Amplification: Polymerase Chain Reaction (PCR)
2.3.2 Real-Time Quantitative PCR (qPCR)
2.4 Emerging and Future Technologies
Advancements in molecular biology are yielding even more sensitive and comprehensive HPV detection platforms, particularly for genotyping and cancer monitoring.
2.4.1 Next-Generation Sequencing (NGS)
NGS technology is moving into clinical research for HPV detection due to its ability to comprehensively type the virus and detect novel or rare variants [20].
Table no.7
|
Method |
Principle |
Clinical Relevance |
|
Targeted NGS |
Amplifies a broad set of text HPVgenes (e.g.L1 or E6/E7) using consensus primers, followed by deep sequencing. |
High-Resolution Genotyping: Can identify every HPV type present in a multiple infection and detect sublineages associated with higher cancer risk. |
|
Whole-Genome Sequencing (WGS) |
Sequences the entire viral and host genome. |
Integration Analysis: Provides definitive data on the precise integration sites of the HPV genome into the host cell DNA, a critical event in cancer progression. |
|
Liquid Biopsy (cfDNA NGS) |
Sequencing of circulating cell-free DNA (cfDNA) in plasma, especially for HPV associated cancers like oropharyngeal cancer. |
Non-invasive Monitoring: Highly sensitive for detecting residual disease or recurrence by tracking cfHPV-DNA levels after treatment. |
2.4.2 Comparison of Key Clinical HPV Assays
Table no.8
|
Assay Type |
Target |
End Point/Result |
Clinical Application |
|
Hybrid Capture 2 (HC2) |
HR-HPV DNA (13 types) |
Pooled HR status (Positive/Negative) |
Triage of ASCUS cytology, Co-testing |
|
Real-Time PCR (e.g., cobas) |
HR-HPV DNA (14 types) |
HPV 16/18 Genotyping + Pool HR status |
Primary Screening, Co-testing |
|
APTIMA (TMA) |
HR-HPV E6/E7 mRNA (14 types) |
E6/E7 mRNA status |
Triage and follow-up (high specificity for CIN2+) |
|
p16 IHC |
P16INK4a Protein |
Strong, diffuse nuclear and cytoplasmic staining |
Histological confirmation/triage of biopsy-proven lesions |
3. Prevention, Diagnosis and Treatment of Cervical Cancer
3.1 Prevention:
In recent years, HPV vaccines have emerged as a global re- search hotspot, with researchers worldwide dedicating efforts to develop efficient, attenuated, and cost-effective vaccines that cater to the diverse needs of different economic regions, thereby effectively preventing and treating HPV-induced CC. Vaccines are categorized into preventive and therapeutic based on their immunological functions (Oliver et al., 2017; Lei et al., 2020; Mix et al., 2021), and preventive HPV vaccines offer a broader range of applications compared to therapeutic vaccines.
Preventive vaccines are primarily administered to individuals to prevent HPV infection and its associated diseases . Currently, preventive vaccines mainly target the HPV cap- sid protein L1 antigen. Studies have shown that vaccines based on VLPs, constructed by expressing the virus capsid protein L1 in monkey kidney cells, induce specific immune responses in mice. Animal infection models with bovine papillomavirus and cotton tail rabbit papillomavirus have confirmed the effectiveness of L1 VLP-based preventive vaccines, driving the development of HPV preventive vaccines based on L1-VLPs. [21]
Six vaccines are currently on the market globally: HPV16/18 bivalent vaccines (GlaxoSmithKline Cervarix®, Xiamen Wantai Xincaning®, and Yuxi Zerun Wozehui®), HPV6/11/16/18 quadri- valent vaccines (Merck Gardasil® and Indian Serum Institute Cervavac®), and HPV6/11/16/18/31/33/45/52/58 nonavalent vaccines (Merck Gardasil®9), all utilizing HPV L1 VLPs as anti- gens. They effectively prevent infection by common oncogenic HPV types, estimated to prevent up to 90% of HPV-related cancers. In 2021, a new alum-adjuvanted bivalent vaccine protect- ing against HPV16 and HPV18 was licensed to further improve global supply and demand for HPV vaccines (Zou et al., 2020). Additionally, efforts are underway to develop next-generation vaccines based on the virus's minor structural protein L2, potentially preventing a broader range of HPV types.
HPV infection can be clinically diagnosed by the presence of warts in low risk types. Genital warts which are sexually transmitted can be present in the form of cauliflower like smooth popular, small elevated warts. Progressive cervical disease initiates as a benign non invasive squamous intraepithelial lesion or CIM. Differential diagnosis can be done with three procedures of molecular assays namely: signal amplification, target amplification, non amplified hybridization assays . Pap smears are the most commonly used lad diagnostic procedure for women. Cytological changes during HPV infection which are strongly associated with papaniclav (pap) smears screening, cell sampling of cervix is done and checked for signs of malignancy . In pap diagnosis atypical squamous cells are of undetermined significance (ASCUS) during cervical screening. Although this diagnosis shows cell abnormality and is not sufficient for definitive diagnosis of a squamous intraepithelial lesion. Colposcpic biopsy is referred to women with these types of abnormalities.[20]
3.2 Diagnosis of Cervical Cancer:
The diagnosis of cervical cancer is primarily associated with assessing the cervical epithelium through regular screening by Pap smear, colposcopy and viral DNA detection. By applying these methods, the onset and development of cervical cancer can be arrested as early as possible. Cervical cancer develops gradually through a series of processes from mild neoplasia (CIN1), through more severe degrees of neoplasia (CIN2 or CIN3), to invasive disease. These stages of development have been the basis for diagnosis and treatment. It has been observed that different HPV types are associated with varying degrees of neoplasia, with each neoplasia being a distinct process. CIN1 is usually observed as a self-limiting sexually transmitted HPV infection that is more likely to regress, while CIN2 and CIN3 are regarded as the true precursors of cervical cancer. The risk of progression from mild to severe dysplasia is only 1% per year, while the risk of progression from mild to severe dysplasia is 16% within 2 years, and 25% within 5 years Despite this, it is considered that early Infection and the treatment of HPV can prevent the progreson of precancerous lesions to cancer [21] .
Cervical cancer diagnosis is not a 'one size fits all' approach due to heterogeneity of the disease with respect to risk factors, immune status, lifestyle or socioeconomic status, race, and most importantly, the age of the patient. For asymptomatic infections, such as cervical neoplasia, and/or routine screening, Pap screening is recommended for women aged 21 years. HPV DNA testing and Pap smears are recommended for women aged 22-30 years, with a Pap screening every 3 years for those with negative results and at a low risk of developing cervical cancer. For those between 30-65 years of age, a Pap smear should be performed after every 5 years with HPV DNA testing. For women >65 years of age, and with continued negative screening, both cytology and HPV DNA testing should be discontinued [22] .
3.3 Treatment of Cervical Cancer:
The management of cervical cancer and other premalignant lesions is carried out according to the findings. Premalignant lesions are managed cautiously for women <25 years of age and in the majority of cases, the majority of findings are low-risk cervical dysplasia, which usually resolves spontaneously. However, lesions suspected to be greater than low-risk, are evaluated by colposcopy, low-risk legions are observed and re-evaluated more frequently, while high-risk lesions are treated based on staging, size and location. For premalignant lesions that are limited in size and depth, excision or cryo- therapy is done, while conization or the loop electrosurgical excision procedure is performed for lesions that have invaded the endocervical canal and beyond. For early-stage invasive cancer, the treatment is surgical resection either by conization or radical hysterectomy; however, women in this category who still require pregnancy are treated using a trachelectomy. Women with high-risk features post-resection may be subjected to adjuvant therapy with radiotherapy and/or chemotherapy [23] .
Surgery: Surgical resection is performed for patients with early-stage disease that is restricted to the cervix. This can range from non-invasive techniques, such as cervical conization to radical hysterectomy. Cervical conization is indicated for patients with carcinoma in situ or stage IA1 invasive cervical cancer, and it is performed with a cold knife cone (CKC) to remove the squamocolumnar junction and ~3 mm margin of the cervix. Pathological evaluation of the presence of lympho vascular invasion (LVI) and margins of the lesion is essential, and if present, further surgical intervention may be required. But if no LVI is present, nodal evaluation is not necessary. Patients that have undergone this procedure mostly have a 5-year survival rate >95%.
Radical hysterectomy is indicated for patients who do not desire to preserve their fertility. Four types of hysterectomy have been defined, with type A representing only a minimal parametrial resection, type B representing resection of the paracervix at the level of the ureter, type C representing resection of the paracervix at the level of the internal iliac vessels, and type D representing resection of the paracervix to the pelvic sidewall. This procedure however, is associated with complications, such as infection, hemorrhage, small bowel obstruction, vesicovaginal fistula, venous thromboembolism and pulmonary embolus.
Another form of surgery indicated for patients with stage IA1 who do not also desire to preserve their fertility is an extra fascial hysterectomy. This involves the removal of the entire cervix and uterus without removal of the parametria, while removal of the ovaries is optional. This technique may be done either by opening the abdominal cavity (laparotomy) or through the vagina. Complete parametrectomy or radiotherapy may be done post-operatively if adverse pathologic features are discovered[24] .
Radiotherapy: Radiotherapy is a treatment modality for cervical cancer as a definitive or adjuvant therapy with or without chemotherapy. For early-stage cervical cancer IA1-IIA1, radio- therapy may be used as the sole treatment modality. However, s are definitive concurrent platinum-based chemoradiotherapy with brachytherapy boost is essential for advanced cervical cancer. Post-operative radiotherapy with or without chemotherapy is gical essential when surgical pathologic conditions are observed. In aded post-radical hysterectomy patients, at least two of the following asive criteria must be met when using adjuvant radiotherapy without cation chemotherapy i.e., the tumor size must be >4 cm, there must egory be greater than one-third stromal invasion, or lymphovascular space invasion (LVSI) .
Radiotherapy is delivered either as brachytherapy or external beam radiotherapy (EBRT). The EBRT is composed of intensity-modulated radiotherapy (IMRT) and is directed at the primary and pelvic lymphatics, while Brachytherapy involves delivering radiation close to the tumor using a sealed radiation source either alone in early-stage lesions or as a boost following EBRT in more advanced lesions. A high dose rate of >12 Gy/h generated from a radioactive source (usually Iridium-192) is applied for a maximum of 8 weeks [25] .
Immunotherapy: It is currently considered an effective treatment strategy that guarantees successful recovery. Promising results have been obtained with this approach, with various classes of compounds being studied, such as tumor infiltrating lymphocytes, antibody-drug conjugate and vaccines (prophylactic and therapeutic). Among these, the most widely studied immunotherapy compounds are the ICI. Most importantly, ICIs with PD-1 and cytotoxic T-lymphocyte antigen 4 (CTLA4) as the most commonly targeted molecules are being explored in cervical cancer treatment . CTLA4, a homolog CD28 that have a higher affinity for B7, regulates effector G cells. T-cells are activated majorly by the binding of CD28 to 37 on APCs, and this is achieved only when there is CD28-B7 level less than that of CTLA4-B7. On the other hand, PD-1, a costimulatory receptor commonly found in exhausted T-cells due to prolonged stimulation caused by chronic inflammation or cancer, prevents the production of TCR signaling and decreases the production of IL-2, IFNy, and TNF-a when it binds to PD-L1 .
CONCLUSION
The molecular dependence of HPV-induced cervical cancer on the constitutive activity of the viral E6 and E7 oncoproteins sets it apart. Unlike most other solid tumors, this particular dependency offers a highly specific therapeutic window. Despite their effectiveness, conventional treatments are frequently morbid, underscoring the need for innovative, focused approaches.
A paradigm shift toward precision oncology is represented by current research on therapeutic vaccines, RNA interference, and CRISPR/Cas9 gene editing. Although delivery and safety issues must be resolved for these approaches to be successfully translated into clinical settings, their potential is revolutionary. Instead of only controlling the disease burden, these treatments offer the potential to functionally cure the cancer by restoring the native tumor surveillance systems (p53 and pRb).
FUTURE DIRECTION
Future research should focus on:
REFERENCES







Noorain Sayyad, Harshal Janbandhu, Nived Mukhi, Trupti Nimje, Diya Choudhari, Namrata Waghmare, Tanmay Khatarkar, Targeting HPV Oncogenes (E6/E7) For Cervical Cancer Therapy: Advances in Molecular and Natural Strategies, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 1496-1513. https://doi.org/10.5281/zenodo.19013154
 10.5281/zenodo.19013154
10.5281/zenodo.19013154