
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Poona District Education Association’s Seth Govind Raghunath Sable College of Pharmacy, Saswad, Pune, Maharashtra, India
The endolysosomal system is crucial for cellular homeostasis. Historically, the ESCRT machinery was considered the sole mechanism responsible for sorting cargo and generating multivesicular bodies (MVBs). However, the discovery of the hemifusome a novel organelle with a distinct 'snowman-like' architecture challenges this traditional view. Composed of two vesicles connected by a stable hemifusion diaphragm and organized by a proteolipid nanodroplet, the hemifusome functions as a dynamic platform for cargo sorting independent of the ESCRT pathway. This review synthesizes current knowledge regarding the hemifusome's structure and function, presenting it as a paradigm shift in intracellular trafficking. Furthermore, we propose a new pathological framework termed "hemifusome-opathy." We postulate that hemifusome dysfunction is a primary driver of major human diseases, specifically contributing to the toxic protein accumulation seen in neurodegenerative conditions like Alzheimer's and Parkinson's disease. By outlining a roadmap for future research, we identify the hemifusome as a critical new frontier in cell biology with significant potential for developing novel diagnostic and therapeutic strategies.
Eukaryotic cellular function relies on precise compartmentalization, coordinated by a complex network of membrane-bound organelles. The endolysosomal system is central to this organization, managing the sorting, processing, and degradation of intracellular and extracellular materials.[1] For decades, the biogenesis of multivesicular bodies (MVBs) a critical event within this system was defined by the Endosomal Sorting Complexes Required for Transport (ESCRT) machinery. This multi-component pathway creates intraluminal vesicles and packages cargo for lysosomal degradation, and it has long been considered the canonical mechanism for these processes.[2]
?However, the ESCRT-centric model remains incomplete. Evidence for ESCRT-independent MVB formation has accumulated for years, suggesting that alternative mechanisms exist. Furthermore, the classical ESCRT pathway depends heavily on cargo ubiquitination, which fails to explain how non-ubiquitinated proteins and bulk lipids are efficiently sorted for degradation.[3]
?The recent discovery of the hemifusome addresses these gaps and represents a significant advancement in cell biology. Identified via cryo-electron tomography, the hemifusome exhibits a unique 'snowman-like' architecture consisting of two vesicles connected by a stable hemifusion diaphragm and organized by a proteolipid nanodroplet. Contrary to traditional views of membrane fusion, this structure is not a transient intermediate but a stable, functional platform for vesicle biogenesis.[4]
?This review provides a comprehensive synthesis of current research on the hemifusome. We examine its unique architecture, which challenges the assumption that membrane hemifusion is inherently fleeting. We then explore its function as the core of an ESCRT-independent cargo sorting pathway, potentially resolving long-standing questions regarding cellular trafficking. Furthermore, we propose that hemifusome dysfunction acts as a pathological driver in human disease, particularly in neurodegenerative conditions. Finally, by outlining key unanswered questions, we provide a roadmap for future research, positioning the hemifusome as a fundamental new component of cellular homeostasis.
2. THE ARCHITECTURAL NOVELTY: A STABLE BRIDGE BETWEEN VESICLES
The discovery of the hemifusome, made possible by the near-native visualization of cryo-electron tomography, has unveiled a structure that is as novel as it is elegant. Its architecture challenges long-held assumptions about membrane dynamics and presents a fascinating new puzzle for cell biologists. The structure can be understood through its three defining, and revolutionary, features.
2.1 The Hemifusion Diaphragm: A Paradigm Shift in Membrane Biology
At the heart of the hemifusome lies its most paradigm-shifting feature: a stable hemifusion diaphragm. In classical membrane biophysics, the fusion of two vesicles is a high-energy process that proceeds through a series of transient intermediates. Hemifusion where the outer leaflets of the two lipid bilayers merge while the inner leaflets remain separate was considered a fleeting, energetically unfavorable state on the path to full fusion pore opening [5]. The hemifusome directly refutes this model, demonstrating that this intermediate state can be captured and stabilized as a persistent, functional structure. This stable bridge, which connects two vesicles of differing sizes to create the organelle's characteristic 'snowman-like' appearance, must be maintained by a specific molecular machinery that counteracts the thermodynamic drive towards complete fusion [6]. Understanding the precise lipid composition (e.g., the potential presence of conical lipids) and the specific proteins that clamp this diaphragm in a stable state is a critical goal for future research.
2.2 The Proteolipid Nanodroplet (PND): An Enigmatic Organizing Center
Consistently observed at the junction of the two vesicles is a 42-nanometer particle identified as a proteolipid nanodroplet (PND). This dense, non-membranous particle appears integral to the hemifusome's structure and is hypothesized to function as its organizing center [5]. Its "proteolipid" nature suggests it is a rich mixture of proteins and lipids, possibly existing as a phase-separated condensate, similar to other non-membranous cellular bodies like P-bodies or stress granules. Its strategic location at the rim of the hemifusion diaphragm suggests multiple potential roles:
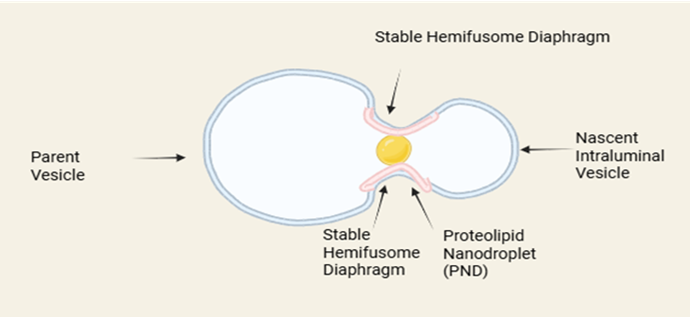
Fig 1: Architecture of Hemifusome
2.3 Structural Plasticity: A Hint at a Functional Cycle
Hemifusome have been observed in two distinct configurations: a "direct" form, where a smaller vesicle is attached to the exterior of a larger one, and a "flipped" form, where the smaller vesicle is located within the larger vesicle's lumen [5]. This structural plasticity is not trivial; it strongly implies that the hemifusome is a dynamic entity that undergoes a regulated, multi-stage functional cycle. The "direct" confirmation likely represents the initial, cargo-loading stage of the process. The "flipped" conformation, in contrast, may represent a later stage, post-scission, or even an alternative pathway where the hemifusome is involved in forming vesicles within another vesicle. These static snapshots provide tantalizing clues to the choreography of a dynamic biological machine that is yet to be seen in action.
3. A NEW FUNCTIONAL PARADIGM: THE ESCRT-INDEPENDENT PATHWAY
The hemifusome's unique structure is the foundation for a novel functional pathway in cellular cargo sorting, representing a significant departure from the canonical mechanism that has dominated the field for years. This new pathway suggests a previously unappreciated level of sophistication in the cell's ability to manage its internal environment.
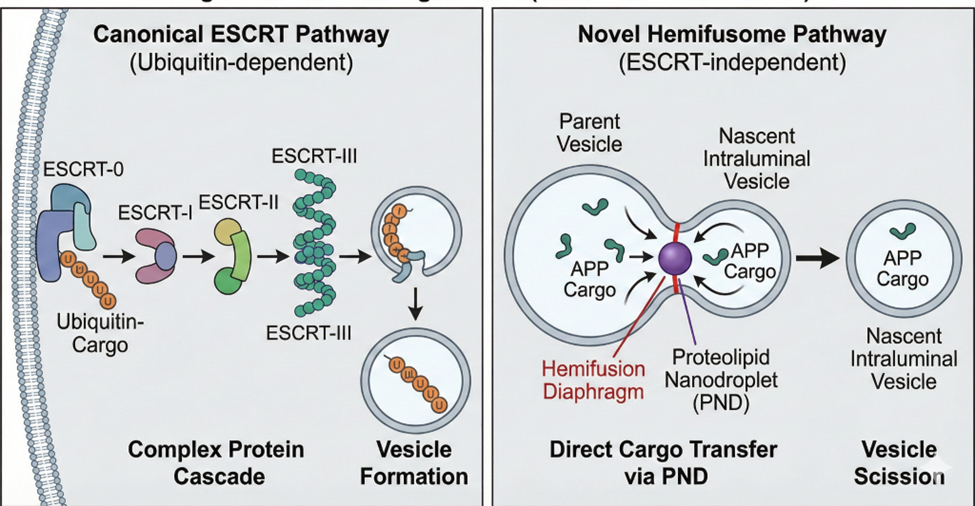
Fig 2:The “Paradigm Shift” (ESCRT vs Hemifusome)
3.1 The Classical Pathway: A Multi-Stage ESCRT-Dependent Cascade
The formation of intraluminal vesicles (ILVs) within MVBs has long been considered the exclusive domain of the ESCRT machinery [2,3]. This well-characterized pathway functions as a sequential, multi-stage cascade. It begins with the ESCRT-0 complex recognizing and clustering membrane proteins tagged with ubiquitin, a molecular signal for degradation. Subsequently, ESCRT-I and -II complexes are recruited to initiate the inward budding of the membrane. The critical step of membrane constriction and scission is then performed by the ESCRT-III complex, which forms spiral filaments that narrow the neck of the budding vesicle. Finally, the Vps4 ATPase is recruited to disassemble the ESCRT-III complex, an energy-dependent step that releases the machinery for another cycle [7]. This reliance on a complex, multi-component cascade underscores the intricacy of the established pathway.
3.2 A Streamlined Alternative: The Hemifusome's Proposed Mechanism
The hemifusome offers a striking, ESCRT-independent alternative. We propose a model where it functions as a self-contained sorting and budding platform. In this model, the stable hemifusion diaphragm acts as a unique conduit between the parent membrane (the larger vesicle) and the nascent ILV (the smaller vesicle). Cargo, such as transmembrane proteins or lipids, could be sorted via lateral diffusion across this bridge and become concentrated in the smaller vesicle.
The final, critical step is the scission mechanism that releases the ILV. While the precise mechanism is unknown, it likely differs from the ESCRT-III constriction method. One possibility is a lipid-driven scission, where the accumulation of specific lipids like lysobisophosphatidic acid (LBPA) in the neck of the bud creates high negative membrane curvature, leading to spontaneous pinching off [8]. Alternatively, a dedicated, yet-to-be-discovered protein "scissionase" might be recruited to this site to sever the connection. The elegance of this proposed mechanism lies in its apparent simplicity, potentially circumventing the need for the large, sequential protein cascade and the ATP-dependent disassembly step employed by ESCRT.
3.3 Implications of a Dual Pathway System: Specialization and Regulation
The existence of parallel pathways strongly suggests specialization. The hemifusome may have evolved to handle specific cellular tasks that are less efficiently managed by the ESCRT machinery. We propose several non-mutually exclusive possibilities:
4. HEALTH & DISEASE: THE CORE MEDICAL ARGUMENT (COMPREHENSIVE ELABORATION)
The integrity of intracellular trafficking pathways is paramount to cellular health. Their disruption is a known hallmark of numerous human diseases, from rare genetic disorders to common neurodegenerative conditions. The discovery of the hemifusome and its associated ESCRT-independent pathway provides a new and powerful lens through which to re-examine the pathophysiology of these conditions. We formally propose that defects in hemifusome formation or function a state we term a "hemifusome-opathy" may represent a fundamental, previously overlooked disease mechanism that contributes significantly to cellular pathology.
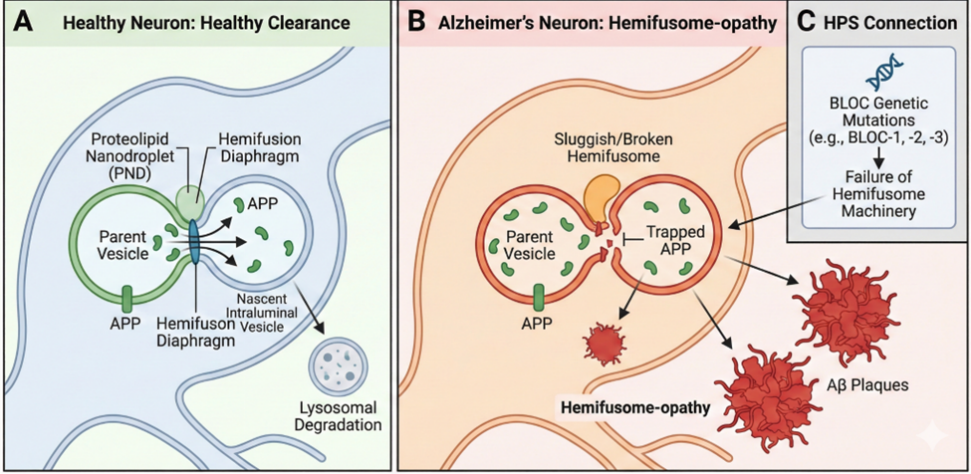
Fig 3: “Hemifusome-opathy” The Hypothesis
4.1 Re-examining Neurodegeneration: A "Hemifusome-opathy" Hypothesis
The well-established link between defective cellular clearance or a failure of "proteostasis" and neurodegeneration is a central concern [10]. Due to their post-mitotic nature and high polarization, neurons are particularly susceptible to the buildup of toxic substances. We propose that the hemifusome pathway serves as an essential, specialized mechanism for maintaining neuronal homeostasis, and that its deterioration with age significantly contributes to the initiation and advancement of sporadic neurodegenerative disorders.
The central pathology in AD is the accumulation of amyloid-beta (Aβ) plaques and neurofibrillary tangles of tau protein. Aβ peptides are generated from the amyloid precursor protein (APP) through sequential enzymatic cleavage. The critical step in the amyloidogenic pathway occurs in endosomes, where APP is cleaved by the β-secretase (BACE1) and subsequently by the γ-secretase complex [12]. We propose that the hemifusome is a primary sorting station for shunting APP towards a non-amyloidogenic fate in the lysosome. In a healthy neuron, hemifusomes would efficiently sort APP into intraluminal vesicles for degradation, limiting its availability to BACE1 and γ-secretase. An age-related decline in hemifusome efficiency would cause APP to become trapped in the endosomal membrane, increasing its residency time and promoting its cleavage into the toxic Aβ42 species [14]. This model provides a direct mechanistic link between a specific trafficking defect and the central molecular event in AD.
The defining features of PD are the buildup of α-synuclein in Lewy bodies and the failure to clear damaged mitochondria (mitophagy) [13]. While the PINK1/Parkin pathway ubiquitinates damaged mitochondria to tag them for disposal, the sheer size of a mitochondrion (often >1 μm) makes it a challenging cargo for the classical ESCRT machinery, which typically generates vesicles of <100 nm [15]. The hemifusome, with its ability to form a large vesicle and its unique engulfing architecture, is a far more plausible candidate for this large-scale clearance. We hypothesize that the hemifusome is a key effector in the final stages of mitophagy. A faulty hemifusome would directly lead to the accumulation of dysfunctional, reactive-oxygen-species-producing mitochondria and toxic α-synuclein aggregates, creating a vicious cycle of cellular stress that drives the progressive death of neurons in the substantia nigra [16].
4.2 A Molecular Basis for Rare Genetic Disorders
For certain rare diseases, the hemifusome may be the missing mechanistic link that unifies a constellation of seemingly disparate symptoms.
HPS is a group of autosomal recessive disorders caused by mutations in genes encoding proteins that form biogenesis of lysosome-related organelle complexes (BLOCs) [11]. These proteins are known to be involved in vesicle formation and transport, but the precise mechanism has been elusive. We hypothesize that several BLOC proteins (e.g., BLOC-1, BLOC-2) are, in fact, essential structural or regulatory components of the hemifusome itself, perhaps acting as adaptors or stabilizing factors for the PND. A mutation would therefore directly impair hemifusome formation, providing a direct molecular explanation for the widespread intracellular trafficking defects affecting melanosomes, platelet-dense granules, and lysosomes seen in HPS patients [17].
4.3 New Avenues for Diagnosis and Therapeutics
The discovery of this new pathway, the hemifusome, unlocks a wealth of potential. It provides fresh, untapped targets for advancements in both disease diagnostics and therapeutic drug development.
Measuring the activity of the hemifusome in patients can be achieved by targeting its core machinery. Specifically, key proteins associated with the hemifusome, particularly those within the PND complex, could be quantified in cerebrospinal fluid or exosomes isolated from plasma. Changes in the concentration or post-translational modification of these proteins may serve as an early, sensitive biomarker for neurodegenerative disease risk, appearing significantly before clinical symptoms become apparent.
The hemifusome presents an entirely new, untapped target for drug development, aligning with the growing interest in modulating intracellular trafficking for therapeutic benefits [17].
Small-molecule drugs, termed "hemifusome activators," could be developed to stabilize or enhance hemifusome formation and function. These activators could be discovered via high-throughput screening. For instance, identifying compounds that promote the phase separation of PND components or boost the activity of a crucial hemifusome-associated kinase would effectively strengthen the innate cellular cleaning mechanism within neurons.
A promising direction for future research involves the development of targeted therapies, such as novel brain-penetrant small molecules or nucleic acid-based treatments, to overcome the critical hurdle of ensuring cell-type specificity, especially for central nervous system (CNS) disorders [17]. The overarching ambition is to create a therapeutic intervention that can restore the proteostasis network in aging neurons. This, in turn, could effectively slow or even stop the progression of neurodegenerative diseases.
5. FUTURE PERSPECTIVES AND UNANSWERED QUESTIONS
Future Directions: The discovery of the hemifusome represents a significant advance in cell biology, but it has simultaneously generated a host of critical, unanswered questions. A comprehensive understanding of this pathway requires a collaborative, multidisciplinary approach spanning advanced imaging, proteomics, and genetic engineering. The following roadmap outlines key areas for future investigation:
I. Molecular Characterization
The immediate priority is to fully identify the protein and lipid components of the hemifusome, particularly the enigmatic PND.
II. Regulation and Dynamics
Investigate the cellular cues and signaling pathways that govern the dynamic lifecycle of the hemifusome.
III. Mechanistic Insights
Determine how cargo is selected for the hemifusome pathway and the physical mechanism of vesicle release.
IV. Functional and Evolutionary Scope
Determine the prevalence and functional breadth of the hemifusome pathway across biology.
V. Genetic and Clinical Relevance
Solidify the hemifusome's role in cellular homeostasis through targeted genetic manipulation.
Broaden the clinical significance of this pathway.
6. CONCLUSION
The discovery of the hemifusome signifies a major breakthrough in cell biology, demonstrating that even fundamental cellular processes still hold unknown elements. This is more than just a new organelle for textbooks; it is the cornerstone of a novel, ESCRT-independent pathway that radically redefines our knowledge of intracellular trafficking. This review consolidates the initial findings, presenting the hemifusome as a new framework for understanding membrane biology, cellular cargo sorting, and critically, its role in human disease.
We introduce the concept of "hemifusome-opathy" to describe diseases potentially stemming from the dysfunction of this pathway, carrying significant implications for currently challenging disorders like neurodegeneration. While the full mechanistic and clinical understanding is nascent, the research roadmap we've laid out offers vast opportunities for further discovery. Unlocking the mysteries of the hemifusome promises to drive innovative diagnostic and therapeutic strategies, providing renewed hope in the battle against some of medicine's most difficult illnesses.
REFERENCES



Dr. Jayashri Jagtap, Sumit Musale, Shruti Bembade, The Hemifusome: A Paradigm Shift in Cellular Cargo Sorting and a New Frontier in Disease Research, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 1, 1173-1181. https://doi.org/10.5281/zenodo.18224475
 10.5281/zenodo.18224475
10.5281/zenodo.18224475