
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Rathnam Institute of Pharmacy, Pidathapolur, Nellore, India
A simple, precise, accurate, and economical zero-order derivative UV spectrophotometric method was developed and validated for the quantitative estimation of Dapagliflozin in bulk drug and tablet dosage form. Methanol was selected as the solvent based on solubility studies. Background: Type 2 Diabetes Mellitus is a chronic metabolic disorder that requires effective therapeutic management and strict quality control of antidiabetic medications. Dapagliflozin, a sodium-glucose co-transporter-2 (SGLT2) inhibitor, is widely used in the treatment of this condition. Reliable analytical methods are essential for the quantitative estimation in pharmaceutical formulations. Methods: Methanol was selected as the solvent based on solubility studies. The maximum absorption wavelength (?max) was found to be 224 nm after scanning in the UV range of 200-400 nm. Beer-Lambert’s law was obeyed in the concentration range of 2-10 µg/mL. The method was validated for linearity, accuracy, precision, robustness, ruggedness, limit of detection (LOD), and limit of quantification (LOQ). Results: The calibration curve showed excellent linearity with a regression equation of Y = 0.085X + 0.002 and a correlation coefficient (r²) of 0.999. Accuracy studies showed recoveries between 99.2% and 100.1%, while precision studies demonstrated %RSD values below 2%. The LOD and LOQ indicated the good sensitivity. Assay of tablet formulations showed drug content within 99.5-100.3%. Conclusion: The developed method is simple, rapid, accurate and suitable for quality control analysis of dapagliflozin in pharmaceutical dosage forms.
Analytical chemistry plays a fundamental role in pharmaceutical sciences and serves as a critical component for ensuring that medications are safe, potent, and maintain superior quality standards. Each pharmaceutical product, spanning from initial development phases through final patient administration, requires thorough examination through analytical procedures [1]. The effectiveness of any medication relies heavily on the precision and dependability of these analytical evaluations [2]. Quality assurance within the pharmaceutical sector relies on analytical methodologies to guarantee that medications comply with established requirements [3]. These evaluations verify that pharmaceutical products are safe for consumption, possess appropriate concentrations of active components, and retain their potency throughout their designated storage period. In the absence of validated and reliable analytical procedures, maintaining uniformity and trustworthiness in pharmaceutical products becomes unattainable [4].
1.1 Role of Analytical Chemistry in the Pharmaceutical Industry:
Analytical chemistry is essential for connecting pharmaceutical research with clinical applications during drug discovery analytical tools are used to identify chemical compounds and study their physical properties during formulation [5]. It will make sure that the final product is stable and effective analytical testing checks that each batch of the pharmaceutical product meets predetermined quality standards throughout manufacturing & performing degradation studies, stability tests and impurity analyses to make sure that the product quality stays high [6].
1.2 UV-Visible Spectrophotometry in Pharmaceutical Analysis:
Due to its ease-of-use dependability and low cost uv-visible spectrophotometry is a common method for analyzing drugs this method works by having molecules absorb ultraviolet or visible light which causes electronic transitions within the compound many pharmaceutical compounds have chromophores that absorb ultraviolet radiation which makes this method good for determining how much of a drug is present [7,8,9].

Fig 1: UV-Visible Spectrophotometry
1.3 Beer-Lambert law:
The basis of quantitative analysis in UV spectrophotometry is the Beer-Lambert Law. This law explains that the absorbance of a solution is directly related to the concentration of the absorbing compound when the path length and molar absorptivity remain constant [10]. Under these conditions, an increase in concentration results in a proportional increase in absorbance [11]. However, this relationship is valid only within a certain concentration range. At higher concentrations, deviations may appear because of molecular interactions or instrumental limitations [12]. Therefore, it is essential to determine an appropriate concentration range where linearity is maintained [13,14].
1.3.1 Selection of Wavelength:
In UV spectrophotometric analysis, choosing the correct wavelength is very important for accurate measurements. The wavelength corresponding to the maximum absorbance is always known as λmax which provides higher sensitive and reliable results. To determine λmax, the analyte solution is scanned across the ultraviolet region, producing a spectrum with characteristic peaks. The highest peak in the spectrum is selected for analysis because it gives stronger absorbance signals and improves the precision of the measurement [15].
The relationship between absorbance and concentration follows the Beer-Lambert law.
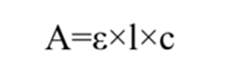
Where:
A = Absorbance
ε = Molar Absorptivity
l = Path length of the cuvette (cm)
c = Concentration of the solution (mol/L)
1.3.2 Zero-Order UV Spectrophotometric Method:
Zero-order UV spectrophotometry is a simple analytical technique in which the absorbance of a compound is measured directly at its wavelength of maximum absorption (λmax). Within a suitable concentration range, absorbance shows a linear relationship with concentration, allowing calibration curves to be constructed for quantitative determination. The method offers advantages such as simple operation, rapid analysis, minimal sample preparation, and in a low cost [16].
1.4 Drug Background:
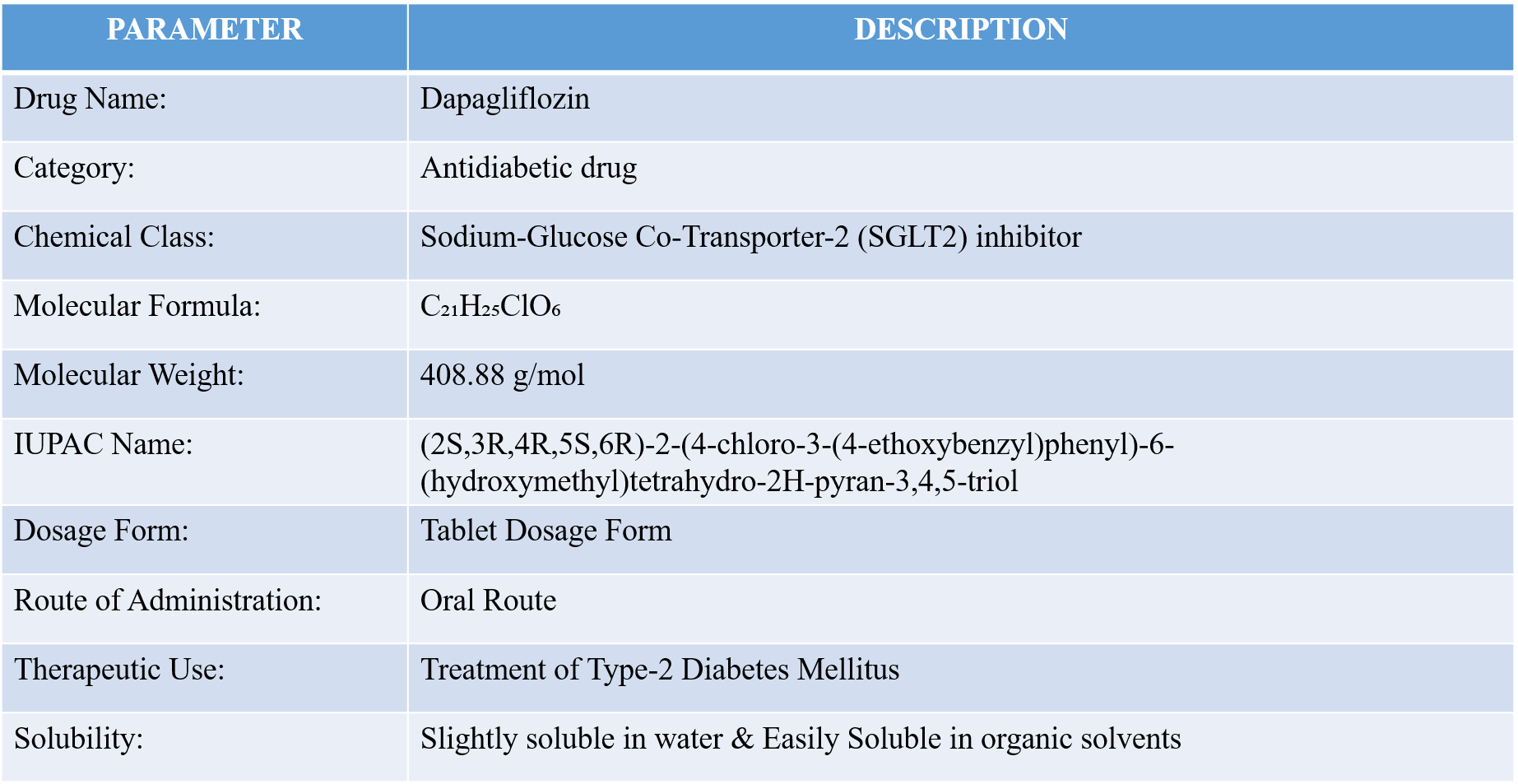
Dapagliflozin is an orally administered antidiabetic drug belonging to the sodium-glucose co-transporter-2 (SGLT2) inhibitor class. It is commonly used in the management of Type 2 Diabetes Mellitus [17]. The drug lowers blood glucose levels by inhibiting glucose reabsorption in the kidneys, thereby increasing glucose excretion through urine.
Fig 2: Tablet form of Dapagliflozin Drug
Several analytical methods, including HPLC, LC-MS, and spectrophotometric techniques, have been reported for the determination of dapagliflozin in pharmaceutical formulations [18]. Although chromatographic methods provide high sensitivity and selectivity, they require sophisticated instrumentation and higher operational costs [19]. Spectrophotometric methods provide a simpler and more economical approach for routine pharmaceutical analysis [20].
1.6 Objectives and Scientific Significance:
The primary goal of this study is to design and validate a simple, precise and dependable zero-order UV spectrophotometric method for the quantitative determination of dapagliflozin in both bulk form and tablet dosage forms. The specific objectives of the study include:
Table 1: Drug Profile of Dapagliflozin [21].
Dapagliflozin is an orally administered antidiabetic medication that belongs to the sodium-glucose co-transporter-2 (SGLT2) inhibitor class. It is widely prescribed for the treatment of Type 2 Diabetes Mellitus (T2DM) and offers an effective therapeutic option for regulating blood glucose levels through a mechanism that does not rely on insulin activity [22]. This drug produces its pharmacological effect by selectively inhibiting SGLT2 transporters present in the proximal renal tubules of the kidneys. Under normal physiological conditions, these transport proteins are responsible for reabsorbing nearly 90% of the glucose filtered by the glomerulus back into the systemic circulation. By blocking this transporter, dapagliflozin reduces glucose reabsorption and increases its elimination through urine, leading to a significant decrease in circulating plasma glucose levels [23].
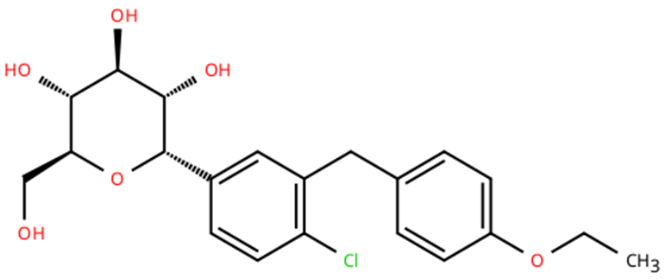
Fig 3: Chemical structure of Dapagliflozin
Beyond its primary Antihyperglycemic action, dapagliflozin has demonstrated additional therapeutic benefits. Clinical studies have shown that it will contribute to modest body-weight reduction, improvement in blood pressure, and protective effects on cardiovascular and renal functions. Because of these advantages dapagliflozin is also utilized in the management of heart failure and certain cases of chronic kidney disease. Chemically, dapagliflozin contains aromatic rings and conjugated functional groups that absorb ultraviolet radiation, making it suitable for UV spectrophotometric analysis [24].
3. MATERIALS AND METHODS:
3.1 Materials:
3.1.1 Drug:
The active pharmaceutical ingredient (API) of Dapagliflozin used in this study was provided as a gift sample by a reputed pharmaceutical company. The received sample was used directly for the experimental work without carrying out any additional purification procedures [25].
3.1.2 Chemicals and Reagents:
Analytical grade methanol, along with distilled water, was utilized during the entire experiment for preparing the required solutions and for performing necessary dilutions.
Fig 4: Methanol [Solvent used]
3.2 Instruments:
The experimental analysis was carried out using the following instruments:

Fig 5: UV-Visible spectrophotometer

Fig 6: Analytical balance

Fig 7: Sonicator

Fig 8: Standard laboratory glassware
4. METHOD DEVELOPMENT:
4.1 Solvent Selection:
Different solvents were evaluated to determine the solubility behavior of dapagliflozin. During the solubility assessment, the drug demonstrated high solubility in methanol compared with other tested solvents. Therefore, methanol was selected as the solvent for further analytical procedures [26].
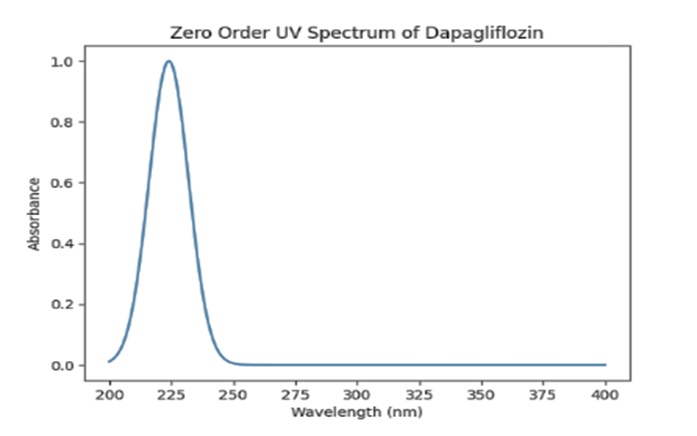
Graph 1: Zero-order UV Spectra of Dapagliflozin
4.2 Determination of Maximum Wavelength (λmax):
A standard stock solution of dapagliflozin with a concentration of 100 µg/mL was prepared using methanol. This solution was scanned in the UV spectrophotometer across the wavelength range of 200-400 nm, with methanol serving as the blank. The spectrum obtained showed the highest absorbance at 224 nm, which was considered the maximum wavelength (λmax) for dapagliflozin [27].
4.3 Preparation of Standard Stock Solution:
An accurately weighed 10 mg quantity of dapagliflozin was transferred into a 100 mL volumetric flask. Methanol was added to dissolve the drug completely [28]. The final volume was made up to the mark with the same solvent. This procedure produced a stock solution with a concentration of 100 µg/mL.

Fig 9: Preparation of Standard Stock Solution
4.4 Preparation of Working Standard Solutions:
Aliquots were withdrawn from the prepared stock solution and transferred into a series of 10 mL volumetric flasks. Each flask was then diluted with methanol to obtain working solutions with concentrations of 2, 4, 6, 8, 10, and 12 µg/mL. The absorbance values of these solutions were measured at 224 nm, using methanol as the blank solution.

Fig 10: Preparation of Working Standard Solutions
Table 2: Working Standard Solutions
|
Sr. No. |
Concentration |
Stock Solution |
Solvent added |
Final Dilution |
|
1. |
2 µg/mL |
0.2 mL |
9.8 mL |
10 mL |
|
2. |
4 µg/mL |
0.4 mL |
9.6 mL |
10 mL |
|
3. |
6 µg/mL |
0.6 mL |
9.4 mL |
10 mL |
|
4. |
8 µg/mL |
0.8 mL |
9.2 mL |
10 mL |
|
5. |
10 µg/mL |
1.0 mL |
9.0 mL |
10 mL |
Measurement:

Fig 11: UV-Visible spectrophotometer - Blank
4.5 Calibration Curve:
A calibration graph was constructed by plotting the measured absorbance values against their corresponding concentrations. The results showed that the method obeyed Beer - Lambert’s law within the concentration range of 2-12 µg/mL [31]. The regression equation derived from the calibration plot was:
Y = 0.085X + 0.002
The correlation coefficient (r²) was found to be approximately 0.999, indicating a strong linear relationship between concentration and absorbance, and confirming the reliability of the analytical method [32].
5. METHOD VALIDATION:
The developed UV spectrophotometric method was validated in accordance with ICH Q2 (R1) guidelines for analytical method validation. The validation parameters evaluated included linearity, accuracy, precision, robustness, ruggedness, limit of detection (LOD), limit of quantification (LOQ), and assay of tablet formulation [33].
5.1 Linearity:
Linearity was evaluated by preparing a series of standard solutions in the concentration range of 2-12 µg/mL. Aliquots were withdrawn from the standard stock solution and diluted with methanol to obtain concentrations of 2, 4, 6, 8, 10, and 12 µg/mL [34]. The absorbance of each solution was measured at 224 nm against methanol as a blank [35 v]. A calibration curve was constructed by plotting absorbance versus concentration. The regression equation and correlation coefficient (r²) were calculated to assess the linearity of the method [36].
5.2 Accuracy:
The accuracy of the method was determined by recovery studies using the standard addition method. Pre-analyzed tablet sample solutions were spiked with known quantities of standard drug at three levels: 80%, 100%, and 120%. Each level was prepared in triplicate and analyzed at 224 nm [37]. The percentage recovery was calculated using the following formula:
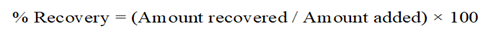
The mean percentage recovery and % relative standard deviation (%RSD) were calculated to evaluate the accuracy of the method [38].
5.3 Precision:
Precision of the method was evaluated in terms of intra-day precision (repeatability) and inter-day precision (intermediate precision) [39].
5.3.1 Intra-day Precision:
Three different concentrations (4, 6, and 8 µg/mL) were prepared and analyzed three times [40]. The absorbance was measured at 224 nm [41]. The mean, standard deviation (SD), and percentage relative standard deviation (%RSD) were calculated [42].
5.3.2 Inter-day Precision:
A selected concentration (6 µg/mL) was analyzed on three consecutive days under the same experimental conditions [43]. The absorbance values were recorded, and the mean, SD, and %RSD were calculated to assess inter-day precision [44].
5.4 Robustness:
Robustness of the method was evaluated by introducing small deliberate changes in analytical conditions [45]. The wavelength was varied by ±2 nm (222 nm and 226 nm) from the optimized wavelength of 224 nm [46, 47]. The absorbance of a standard solution (6 µg/mL) was measured under each condition, and %RSD was calculated to determine the effect of these variations [48].
5.5 Ruggedness:
Ruggedness was determined by performing the analysis under similar experimental conditions by two different analysts [49]. A standard solution (6 µg/mL) was prepared and analyzed independently by each analyst. The absorbance values were recorded, and %RSD was calculated to evaluate the ruggedness of the method [50].
5.6 Limit of Detection (LOD):
The limit of detection (LOD) was calculated using the following formula:

Where:
σ = Standard deviation of the response
S = Slope of the calibration curve [51]
5.7 Limit of Quantification (LOQ):
The limit of quantification (LOQ) was calculated using the following formula:
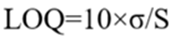
Where:
σ = Standard deviation of the response
S = Slope of the calibration curve [52]
5.8 Assay of Tablet Formulation:
Twenty tablets were weighed and powdered. A quantity of powder equivalent to 10 mg of dapagliflozin was accurately weighed and transferred into a 100 mL volumetric flask. The drug was dissolved in methanol, sonicated for complete extraction, filtered, and diluted appropriately [53]. The absorbance of the resulting solution was measured at 224 nm, and the percentage assay was calculated using the calibration curve [54].
6. RESULTS:
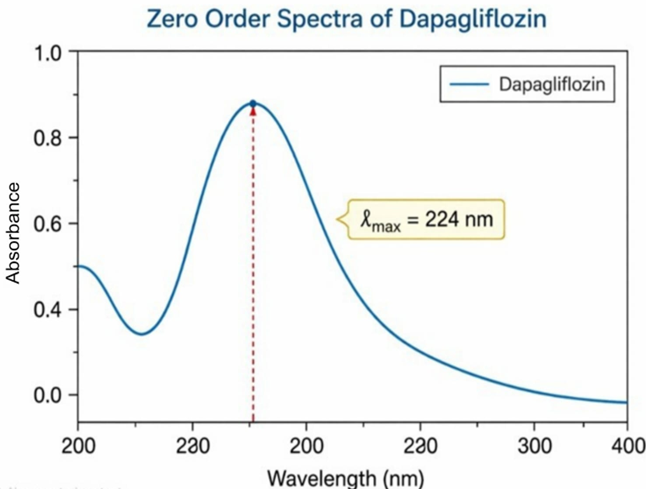
Graph 2: Zero-order UV Spectra of Dapagliflozin
6.1 Linearity:
Table 3: Linearity of Dapagliflozin
|
Sr. No. |
Concentration (µg/mL) |
Absorbance |
|
1. |
2 |
0.17 |
|
2. |
4 |
0.34 |
|
3. |
6 |
0.51 |
|
4. |
8 |
0.68 |
|
5. |
10 |
0.85 |
The calibration curve in the range of 2-10 µg/mL showed a strong linear relationship between concentration and absorbance with an r² value of 0.999. This confirms excellent absorbance of Beer-Lambert’s law. Since the value exceeds the general acceptance criterion (r² ≥ 0.995), the method demonstrates superior linear performance and is suitable for quantitative analysis.
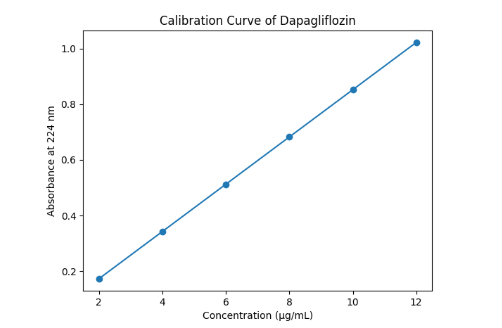
Graph 3: Calibration curve of Dapagliflozin
6.2 Accuracy:
Table 4: Recovery of Dapagliflozin
|
Sr. No. |
Level |
% Recovery |
|
1. |
80% |
99.2% |
|
2. |
100% |
100.1% |
|
3. |
120% |
99.8% |
Recovery studies at 80%, 100%, and 120% levels showed percentage recoveries between 99.2% and 100.1%. These values fall within the ICH acceptable range of 98-102%, indicating high accuracy and absence of interference from excipients. The results confirm the reliability of the method.
6.2.1 Intra-Day Precision:
Table 5: Intra-day Precision Data of Dapagliflozin
|
Sr. No: |
Concentration (µg/mL) |
Absorbance 1 |
Absorbance 2 |
Absorbance 3 |
Mean |
%RSD |
|
1. |
4 |
0.338 |
0.341 |
0.339 |
0.339 |
0.44 |
|
2. |
6 |
0.509 |
0.512 |
0.511 |
0.511 |
0.29 |
|
3. |
8 |
0.679 |
0.681 |
0.683 |
0.681 |
0.30 |
The intra-day precision study showed %RSD values of 0.44%, 0.29%, and 0.30% for 4, 6, and 8 µg/mL, respectively. All values are below 2%, demonstrating excellent repeatability and analytical consistency.
6.2.2 Inter-Day Precision:
Table 6: Inter-day Precision Data of Dapagliflozin
|
Sr. No. |
Day |
Absorbance (6 µg/mL) |
|
1. |
Day 1 |
0.510 |
|
2. |
Day 2 |
0.514 |
|
3. |
Day 3 |
0.508 |
Small deliberate changes in wavelength (±2 nm) produced %RSD values below 1%, indicating that minor variations in experimental conditions do not significantly affect the results. This confirms that the method is robust and reliable.
6.3 Robustness:
Table 7: Robustness Study of Dapagliflozin
|
Sr. No. |
Condition |
Absorbance |
%RSD |
|
1. |
224 nm (normal) |
0.511 |
– |
|
2. |
222 nm (-2 nm) |
0.507 |
0.78 |
|
3. |
226 nm (+2 nm) |
0.514 |
0.65 |
No significant variation was observed. Hence, this method is robust.
6.4 Ruggedness:
Table 8: Ruggedness Study of Dapagliflozin
|
Sr. No. |
Analyst |
Absorbance (6 µg/mL) |
|
1. |
Analyst I |
0.509 |
|
2. |
Analyst II |
0.513 |
Analysis performed by two different analysts showed a %RSD of 0.55%, demonstrating consistency irrespective of operator variation. The method is therefore rugged and suitable for routine laboratory use.
6.5 LOD and LOQ:
The LOD and LOQ were found to be approximately 0.25 µg/mL and 0.75 µg/mL, respectively, indicating good sensitivity. The method is capable of detecting and quantifying low concentrations effectively, making it appropriate for quality control applications.
6.6 Assay of Tablet Formulation:
Table 9: Assay of Dapagliflozin Tablets
|
Sr. No. |
Absorbance |
Amount Found (mg) |
% Assay |
|
1 |
0.846 |
9.96 |
99.6 |
|
2 |
0.851 |
10.02 |
100.2 |
|
3 |
0.849 |
9.99 |
99.9 |
The percentage assay of marketed tablets ranged from 99.5% to 100.3% with a mean value of 99.9 ± 0.30. These results fall within the Indian Pharmacopoeia limits, that is, (95-105%), which confirms accurate drug content and uniformity. The method is therefore suitable for the routine assay of Dapagliflozin tablet formulations.
7. SUMMARY & DISCUSSION:
The present study was carried out to develop and validate a simple zero-order UV-Visible spectrophotometric method for the estimation of Dapagliflozin in bulk and tablet dosage form [55]. Methanol was selected as the solvent based on solubility studies, and the drug showed maximum absorbance at 224 nm. Beer-Lambert’s law was obeyed in the concentration range of 2-10 µg/mL with a regression equation of Y = 0.085X + 0.002 and a correlation coefficient (r²) of 0.999, showing excellent linearity [56]. The method was validated as per ICH guidelines [57]. Accuracy studies showed percentage recovery between 99.2% and 100.1%. Intra-day precision revealed %RSD values of 0.44%, 0.29% and 0.30%, while inter-day precision showed a %RSD of 0.59%. Robustness tests under ±2 nm & wavelength variation produced %RSD values below 1%, and ruggedness evaluated between two analysts showed %RSD of 0.55%. The LOD and LOQ were found to be 0.25 µg/mL and 0.75 µg/mL, respectively [58]. The assay of marketed tablets showed a drug content ranging from 99.5% to 100.3%, with a mean value of 99.9 ± 0.30%, which is within Indian Pharmacopoeia limits [59].
The developed zero-order UV spectrophotometric method demonstrated excellent analytical performance across all validation parameters [60]. The high correlation coefficient (0.999) confirms a strong linear relationship between concentration and absorbance, indicating adherence to Beer-Lambert’s law. Recovery values close to 100% (99.2-100.1%) show the method is accurate and free from interference by tablet excipients [61]. The very low %RSD values observed in intra-day (≤0.44%) and inter-day precision (0.59%) studies confirm the method’s repeatability and reproducibility [62]. These values are well within the acceptable ICH limit of less than 2%, highlighting the method’s reliability. Robustness showed negligible variation even after slight wavelength changes, and ruggedness results confirmed consistency between analysts, proving the method’s practical applicability in routine laboratory conditions.
The low LOD (0.25 µg/mL) and LOQ (0.75 µg/mL) indicate good sensitivity, allowing detection and quantification of small drug concentrations [63]. The assay results (99.5-100.3%) further validate the accuracy and suitability of the method for tablet analysis. Overall, the findings demonstrate that the developed method performs comparably to more sophisticated analytical techniques while remaining simple and cost-effective [64].
8. CONCLUSION:
The developed zero-order UV spectrophotometric method for Dapagliflozin is accurate, precise, robust, and suitable for routine quality control analysis of bulk and tablet dosage forms. The present study successfully developed and validated a simple and economical UV spectrophotometric method for the estimation of Dapagliflozin in bulk and tablet dosage forms. The method showed excellent linearity (r² = 0.999), high accuracy (99.2-100.1% recovery), and strong precision (%RSD < 1%). The low LOD and LOQ values confirmed good sensitivity, and the assay results (99.9 ± 0.30%) were within pharmacopeial limits.
Therefore, the developed method is suitable for routine quality control analysis of Dapagliflozin in pharmaceutical formulations and can be effectively used in academic and industrial laboratories.
Mohammed Farheen played the main role in this research by conceptualizing the experimental study, designing the methodology, conducting the majority of the experimental work with team, analyzing and interpreting the data and drafting as well as critically revising the manuscript.
Prathapam Mokshitha, Surabhi Lalita Vyshnavi, Pinni Saikeerthi, and Reddy Praveena contributed to the experimental work under supervision, including assistance in processing tableted drug, Pure form from pharmaceutical company, performing laboratory procedures and supporting data collection and preliminary analysis.
Chejarla Shakina served as the project guide and provided overall supervision and guidance throughout the study.
All authors contributed to reviewing the manuscript and approved the final version for publication.
“This research received no external funding”.
The present study involved UV Spectrophotometer Method development & Validation of Dapagliflozin work using tableted drug and pure form that is API and did not involve human participants or animal subjects. Therefore, ethical approval from an Institutional Review Board (IRB) was not required for this research.
Not applicable. The study did not involve human participants or personal data requiring informed consent.
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
The authors express their sincere gratitude to the management and Principal of Rathnam Institute of Pharmacy, Pidathapolur, Nellore for providing laboratory facilities, equipment and Institutional support of Machinery to carry out this research work. The authors also acknowledge the guidance and supervision provided during the completion of this academic research project.
The authors declare no conflict of interest regarding the publication of this research work.
SGLT2 - Sodium-glucose co-transporter-2
LOD - Limit of Detection
LOQ - Limit of Quantification
RSD - Relative Standard Deviation
REFERENCES








Mohammed Farheen, Prathapam Mokshitha, Surabhi Lalitha Vyshnavi, Pinni Saikeerthi, Sakereddy Praveena, Ch. Sakhinamma, M Suchitra, Yadala Prapurna Chandra, Zero Order UV Spectrophotometric Method Development and Validation of Dapagliflozin, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 4, 2390-2409. https://doi.org/10.5281/zenodo.19594725
 10.5281/zenodo.19594725
10.5281/zenodo.19594725