
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmacology, Vidhyabharati College of Pharmacy, Amravati.
Kalanchoe tomentosa (Crassulaceae), commonly known as the Panda Plant, is a perennial succulent native to Madagascar and widely recognized for both its ornamental and medicinal value. The plant contains various biologically active secondary metabolites, including flavonoids, triterpenoids, bufadienolides, phenolic compounds, and sterols, which are responsible for its diverse pharmacological properties. Ethnobotanical and scientific studies have reported its antioxidant, anti-inflammatory, antimicrobial, antidiabetic, antileishmanial, wound-healing, and cytotoxic activities. Phytochemical analyses reveal compounds such as ?-amyrin acetate, ?-sitosterol, quercetin, and gallic acid, contributing to its bioactivity. Extraction methods using solvents of varying polarities (hexane, chloroform, methanol) have been optimized for isolating these metabolites. Despite promising biological effects, research on K. tomentosa remains largely preclinical, with limited data on its pharmacokinetics, molecular mechanisms, and safety profile. This review highlights the botanical features, traditional uses, phytochemistry, pharmacological activities, and future research directions of K. tomentosa, emphasizing its potential as a source of novel therapeutic agents for oxidative stress, inflammation, diabetes, and cancer management..
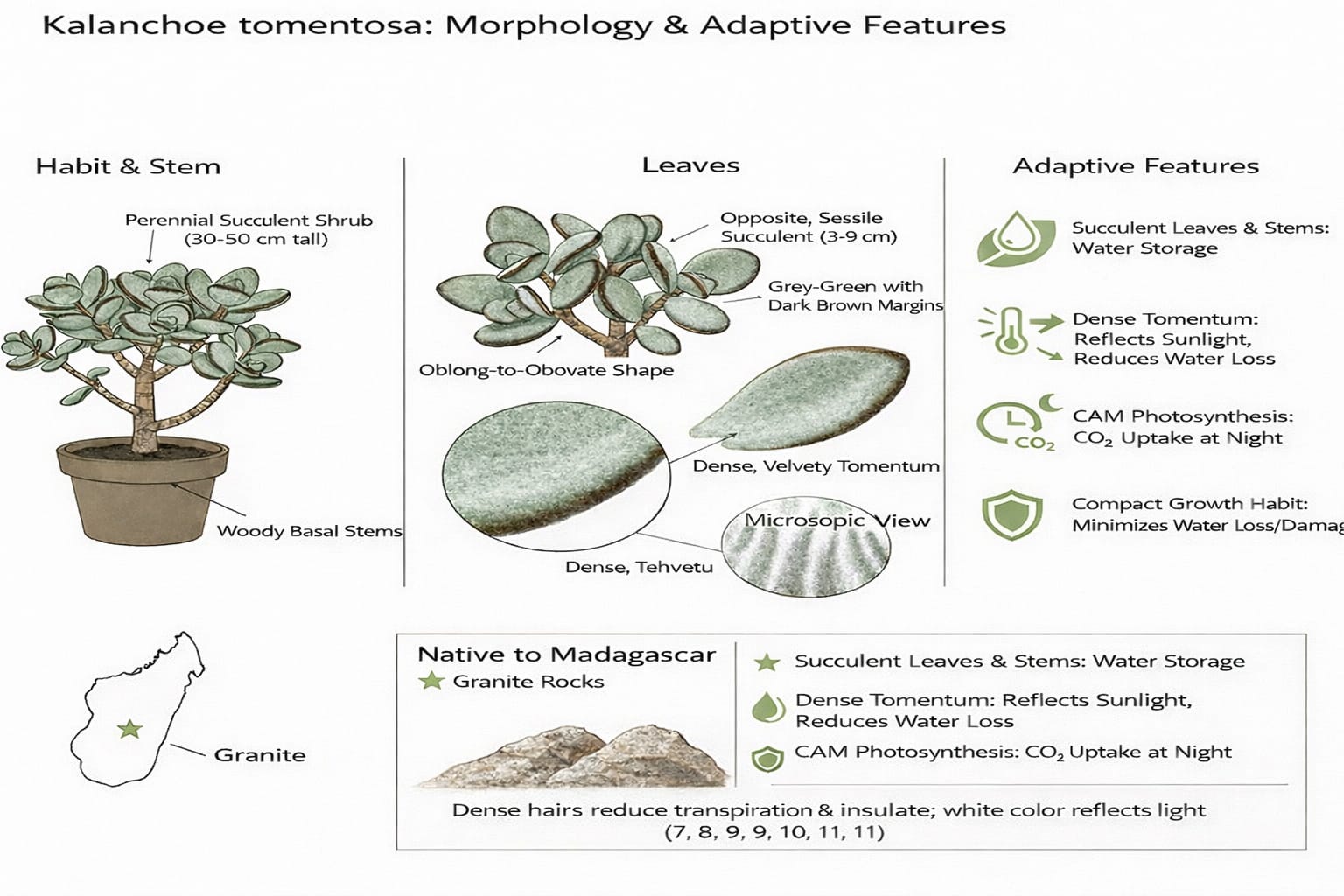
Kalanchoe tomentosa, belonging to the family Crassulaceae, is a perennial, succulent plant characterized by its dense covering of white, hair- like structures. Commonly known as the Panda Plant, it is native to Madagascar. The genus Kalanchoe comprises approximately 130 species of annual and perennial herbs, shrubs, climbers, and small trees. These species are typically found in semi-desert or shaded regions of Saudi Arabia, Yemen, Central Africa, Madagascar, Asia, Australia, and tropical America. The plant is often cultivated as an ornamental species in rock and sand gardens under moderate humidity conditions. Beyond its ornamental importance, Kalanchoe tomentosa is recognized for its medicinal properties, exhibiting antimicrobial, anti-inflammatory, antidiabetic, antileishmanial, and antitumor activities. (1)
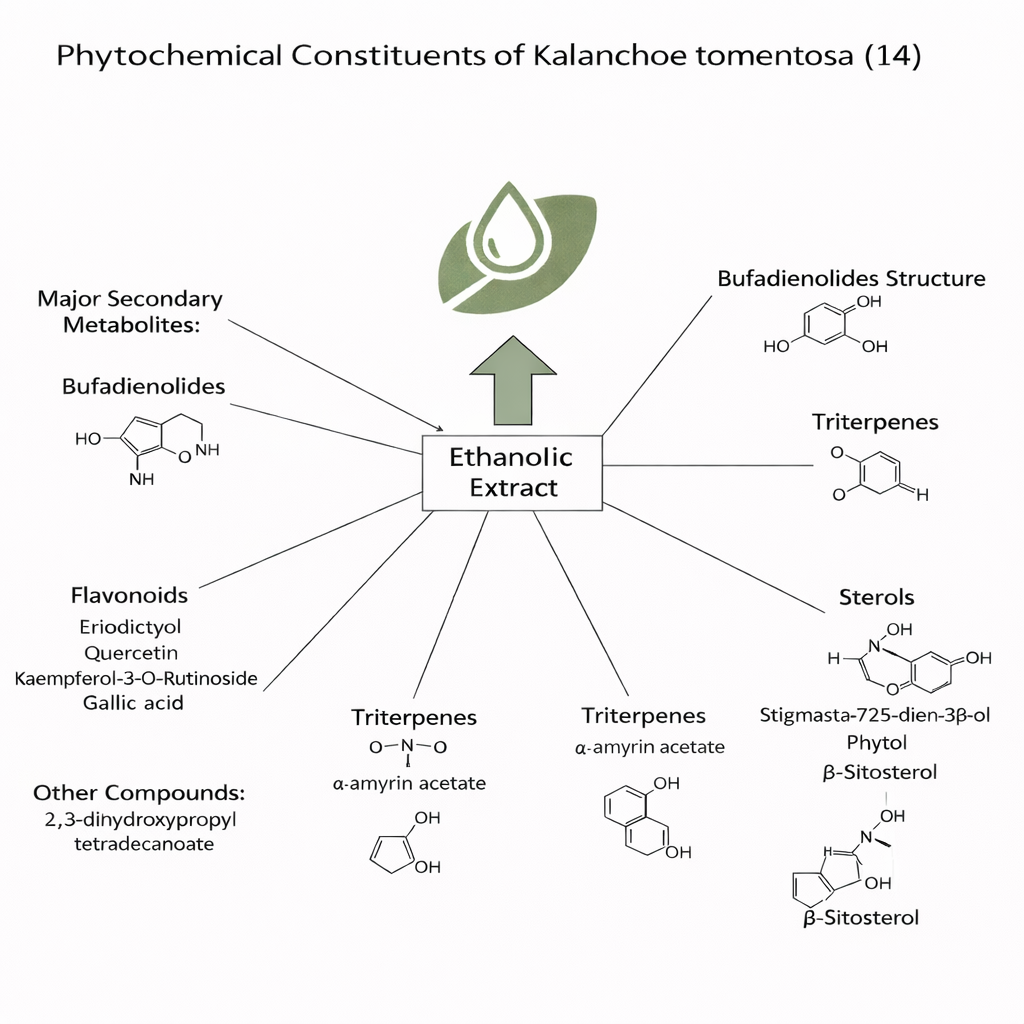
Apart from its ornamental value, Kalanchoe is also very well known as a medicinal plant in the folk medicine. In recent years, an increased interest in the phytochemistry of the genus Kalanchoe which reported to contain interesting biologically active constituents such as bufadienolides, flavonoids, triterpenes and sterols that constitute the major secondary metabolites and showing interesting spectrum of activities.(2)
Plants of the genus are widely used in traditional medicine in several countries, including India, China, South Africa and other African nations, and Brazil, as well as in alternative medicine systems. Indeed, in some of these countries, we can find reports of the use of Kalanchoe plants in Ayurveda, Chinese Traditional Medicine, and anthroposophic medicine. In particular, in Brazil, K. pinnata is part of a list of medicinal plants to be used in the national public health system (SUS-Sistema Único de Saúde). Among the ethnomedicinal uses, plants of the genus are reputed therapeutics in the treatment of inflammatory conditions, wounds, gastric ulcers, genito-urinary disorders, and other illnesses (3).

Fig. kalanchoe tomentosa
Kalanchoe tomentosa is a succulent sub-shrub native to Madagascar. It is found on granite rocks, growing up to 45cm (1.5 feet) tall. The dense covering of hairs performs a vital function for the plant as a water conservation adaptation. In the dry environment in which it lives, the plant must conserve what little water it can absorb from the soil. The dense mat of hairs growing from the leaf retards the movement of air directly across the leaf surface, thereby reducing water vapor loss due to the transpiration process. At the same time, this unventilated space created by the numerous trichomes insulates the leaf from its harsh external environment, too. In addition, the white-silver appearance of the leaves reflects light, lessening the chances of the leaves overheating.
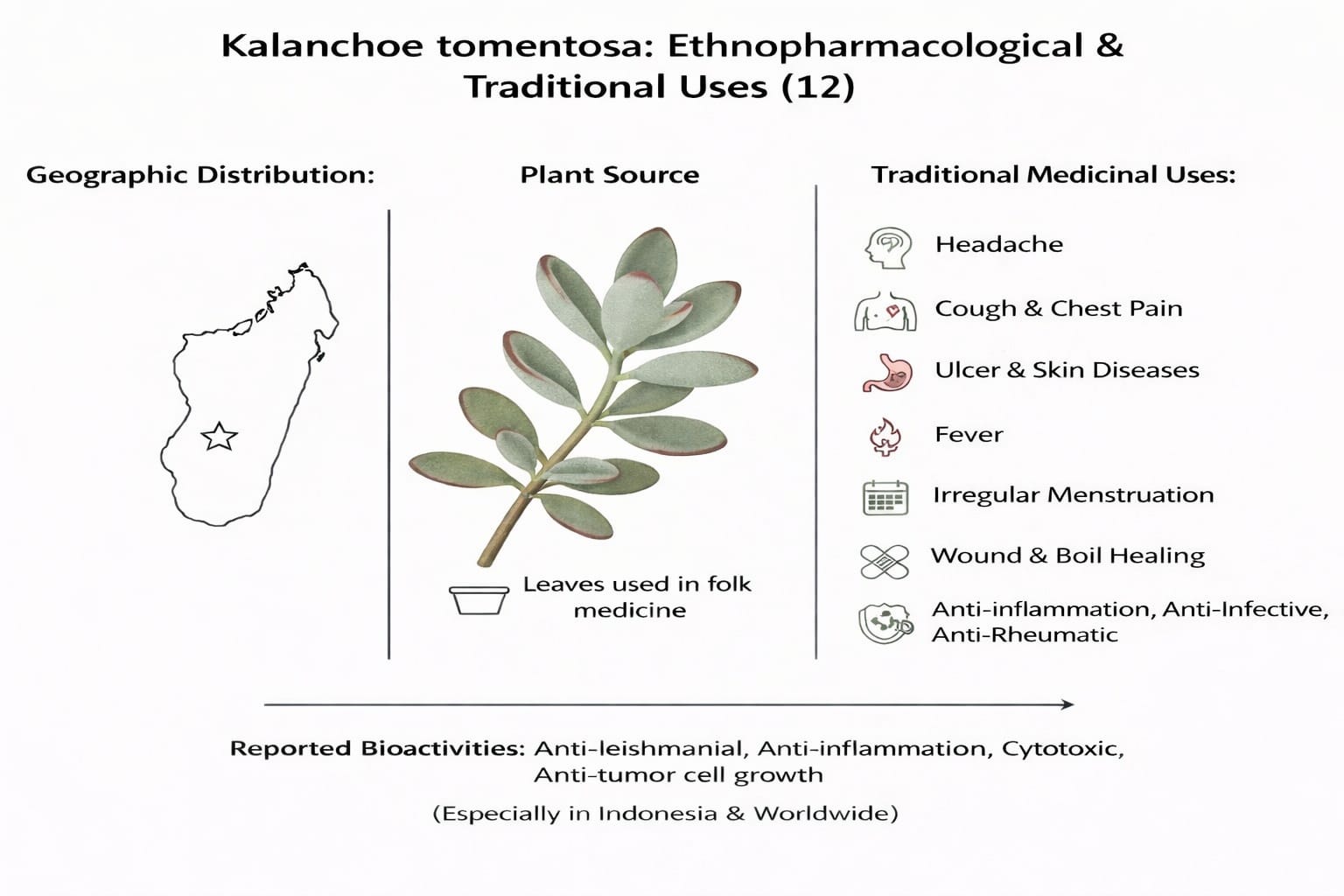
Kalanchoe plants are used as traditional medicines to cure headache, cough, chest pain. ulcer, and other skin diseases. They overcome fever, fix the irregular menstruation, heal wound and boil, not only in Indonesia but also almost everywhere in the world. Some researches reported that Kalachoe plants contain bufadienolide, triterpenoid, and flavonoid, and biological activities like antileismanial, antiinflammatory, cytotoxic, and inhibiting tumor cell growth One of unknown Kalanchoe plants ethnopharmacologically is Kalanchoe tomentosa, especially its anticancer activity. (12)
The leaves of K. tomentosa are used in Indonesian folk medicine for the treatment of fever, infections, rheumatism, and skin diseases (13).
In recent years, an increased interest in the phytochemistry of the genus Kalanchoe has been reported to contain interesting biologically active constituents such as bufadienolides, flavonoids, triterpenes, and sterols that constitute the major secondary metabolites and show an interesting spectrum of activities. Phytochemical study of the ethanolic extract of Kalanchoe tomentosa (Crassulaceae) resulted in the isolation of 14 compounds identified as: α-amyrin acetate, friedelin, glutinol, 1-dotriacontanol, phytol, Stigmasta-7,25-dien-3β-ol, β-sitosterol, Isorhamnetin, 2,3-dihydroxypropyl tetradecanoate, Eriodictyol, Gallic acid, quercetin, kampferol-3-O-Rutinoside, and isovitexin (14).
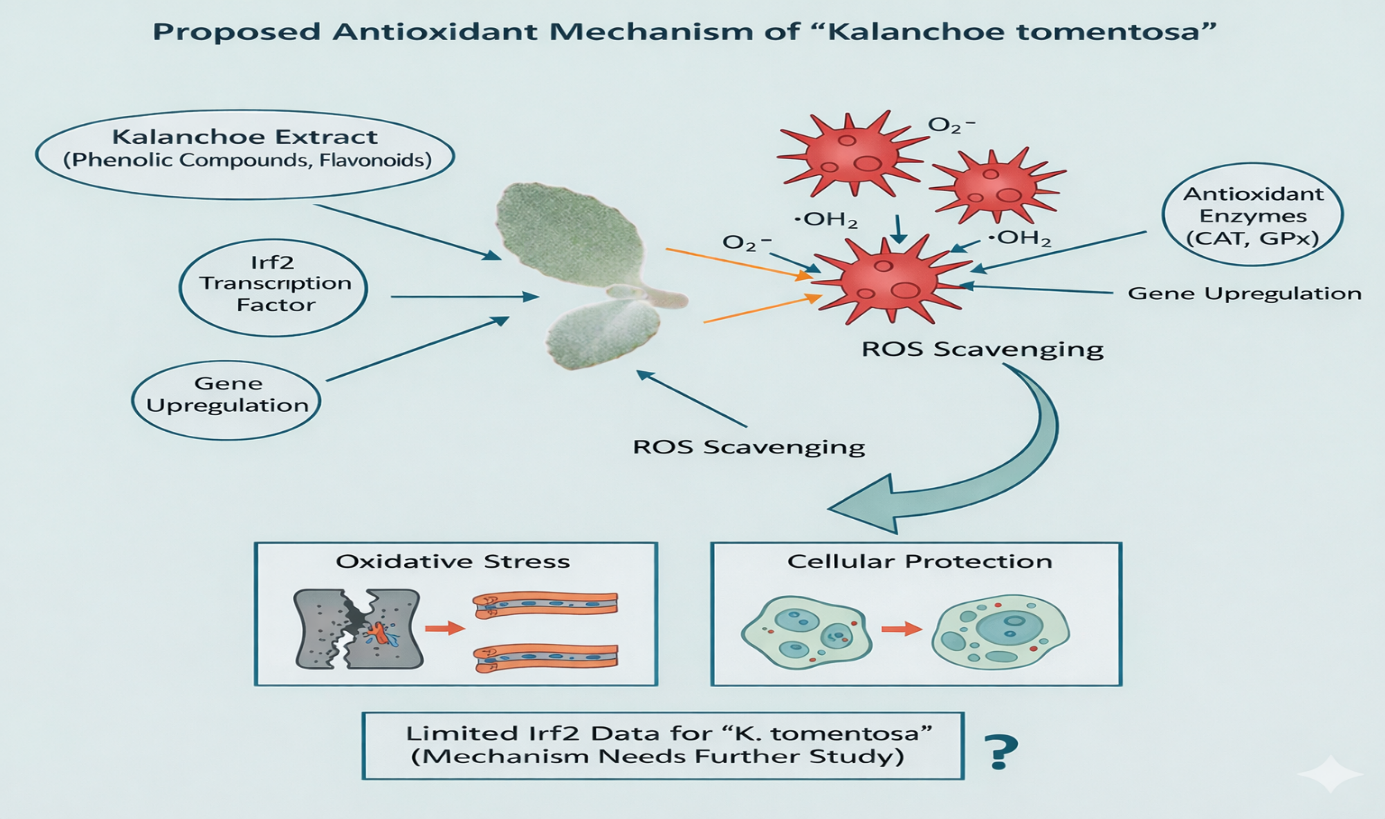
Direct ROS/RNS scavenging: flavonoids and phenolic acids donate electrons/hydrogen to neutralize reactive oxygen species (ROS) and reactive nitrogen species (RNS) in cell-free assays (DPPH, ABTS, FRAP), explaining the high in vitro antioxidant capacity reported for K. tomentosa extracts. (15) Upregulation / preservation of endogenous antioxidant defenses: several Kalanchoe spp. extracts increase levels or activity of antioxidant enzymes (SOD, catalase, glutathione peroxidase) or prevent GSH depletion in cellular models - this is consistent with cytoprotective effects against oxidative stress. Mechanistically, flavonoids can modulate Nrf2 signaling in other plant extracts (hypothesized for Kalanchoe but dire Irf2 data for K. tomentosa remains limited). (16)
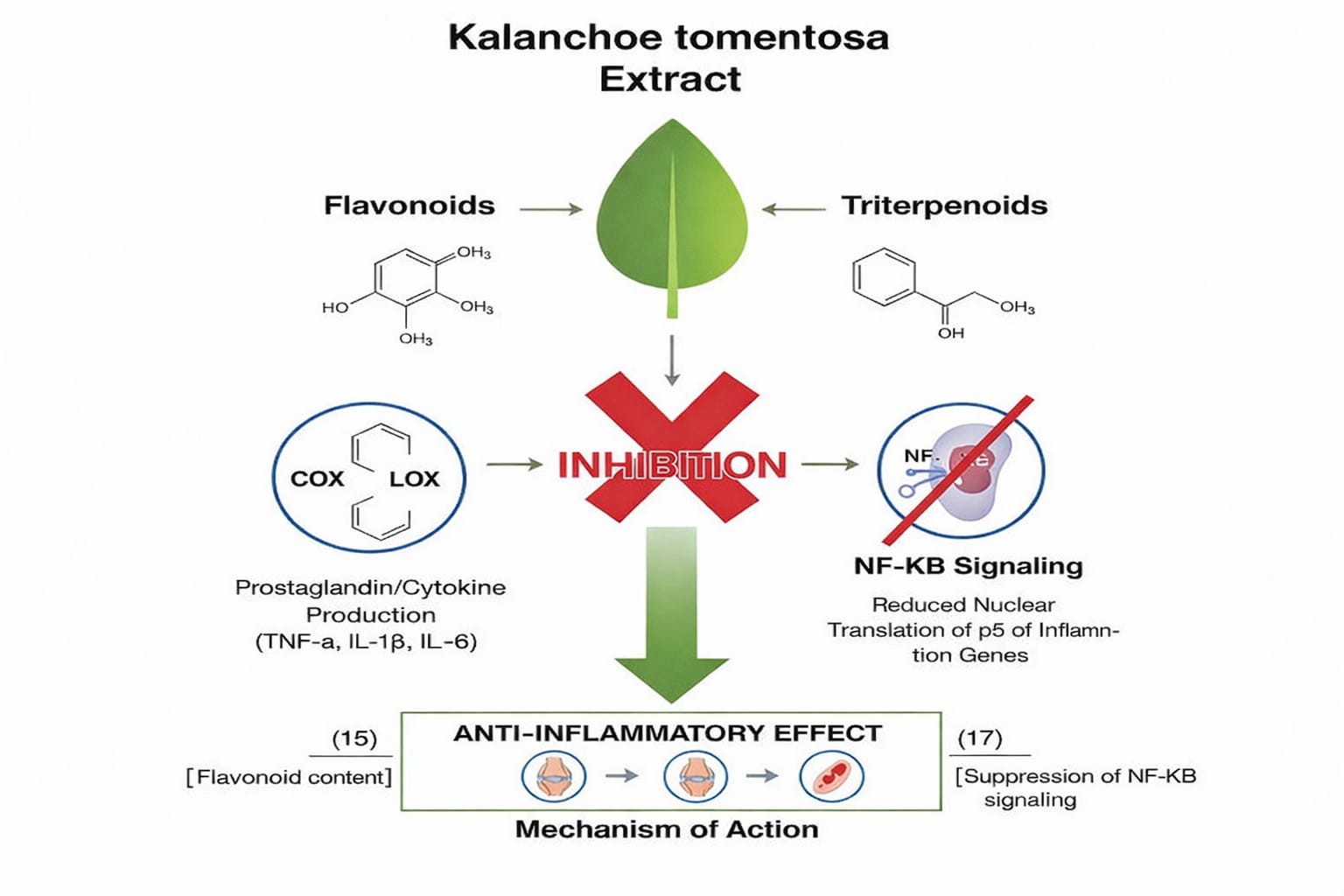
Traditional topical use for inflammation and wounds is supported by genus-level pharmacology and experimental reports showing inhibition of inflammatory markers by Kalanchoe extracts. Direct experimental data for K. tomentosa support anti-inflammatory potential via flavonoid content (15)
Inhibition of pro-inflammatory enzymes and mediators: flavonoids and triterpenoids in Kalanchoe extracts inhibit cyclooxygenase (COX) and lipoxygenase (LOX) activity in enzyme assays or lower prostaglandin/cytokine production (TNF-a, IL-1?, IL-6) in stimulated macrophage models. Suppression of NF-KB signaling: several Kalanchoe species show suppression of NF-KB activation (reduced nuclear translocation/phosphorylation of p65) resulting in decreased transcription of inflammatory genes; by analogy, K. tomentosa flavonoids are likely to act similarly, though direct mechanistic NF-KB studies on K. tomentosa remain limited (17)
Membrane perturbation and permeability changes: lipophilic constituents (fatty acids, sterols) and certain phenolics can disrupt bacterial/fungal membranes, increasing permeability and causing leakage of cytoplasmic contents.Enzyme and cell-wall biosynthesis inhibition: in silico docking on K. tomentosa flavonoids suggested interference with NAM/NAG peptide interactions important for gram-positive peptidoglycan formation - a plausible mechanism for reduced viability of Gram-positive bacteria in some assays. Bioactive single-compounds: ?-sitosterol isolated from K. tomentosa has shown activity against Staphylococcus aureus and Klebsiella pneumoniae in reported studies, indicating that isolated phytosterols contribute to the observed antimicrobial profile. (15)
1. Inhibition of Carbohydrate-Hydrolyzing Enzymes :
One of the primary mechanisms involves the inhibition of digestive enzymes α-amylase and α-glucosidase, responsible for the hydrolysis of complex carbohydrates into glucose. An in vitro study demonstrated that the ethyl acetate fraction of K. tomentosa leaves, containing a flavonol compound (3,7,4′-trihydroxyflavonol), exhibited significant α-amylase inhibitory activity (IC?? ≈ 346 µg/mL) (19). This enzyme inhibition delays glucose absorption in the intestine, thereby reducing postprandial hyperglycemia — a mechanism similar to that of synthetic antidiabetic drugs like acarbose (20).
2. Antioxidant-Mediated β-Cell Protection :
Oxidative stress plays a pivotal role in pancreatic β-cell dysfunction and insulin resistance in diabetes. K. tomentosa extracts exhibit potent antioxidant activities, as evidenced by DPPH and ABTS radical scavenging assays (15,17). These antioxidant compounds scavenge reactive oxygen species (ROS), protect β-cells from oxidative damage, and improve insulin secretion efficiency. By preserving β-cell viability, the plant indirectly enhances glucose homeostasis and insulin activity.
3. Improvement of Insulin Sensitivity :
Although direct in vivo studies on K. tomentosa are limited, related species such as Kalanchoe pinnata and Kalanchoe daigremontiana have been shown to enhance peripheral glucose uptake and improve insulin sensitivity via modulation of the PI3K/Akt signaling pathway and reduction of pro-inflammatory cytokines (e.g., TNF-α, IL-6) (16,21). Due to the structural and phytochemical similarities among Kalanchoe species, it is plausible that K. tomentosa exerts comparable effects on insulin signaling mechanisms.
A central mechanistic theme in Kalanchoe anticancer literature is the role of bufadienolides (cardenolide-like steroids) and other phenolics:
Na+/K+-ATPase inhibition (primary molecular target for bufadienolides):
bufadienolides bind to and inhibit the Na+/K+-ATPase (the same target as cardiac glycosides like digoxin). This inhibition disrupts ion homeostasis, causes intracellular Na+ accumulation → reduced Na+/Ca2+ exchange → increased intracellular Ca²+, which can trigger mitochondrial dysfunction, activation of caspases, and apoptosis in cancer cells. Several Kalanchoe-derived bufadienolides display cytotoxicity correlated with Na+/K+-ATPase inhibitory PMC +1 potency.
Induction of apoptosis via mitochondrial (intrinsic) pathway: treated cancer cells show loss of mitochondrial membrane potential, cytochrome-c release, caspase-9/3 activation and DNA fragmentation. Some flavonoids and triterpenes also contribute to ROS generation that potentiates apoptosis.(22)
CONCLUSION
Kalanchoe tomentosa (Crassulaceae) represents a highly valuable medicinal plant owing to its rich phytochemical diversity and wide spectrum of pharmacological activities. Numerous studies have demonstrated the presence of bioactive compounds such as flavonoids, triterpenoids, bufadienolides, phenolic acids, sterols, and glycosides, which collectively contribute to its therapeutic potential. These metabolites are largely responsible for its reported antioxidant, anti-inflammatory, antimicrobial, antidiabetic, wound-healing, and cytotoxic properties, thereby validating many of its traditional medicinal uses.
Despite substantial preclinical evidence, current research on K. tomentosa remains limited by a lack of standardized extraction protocols, quantitative marker compounds, and in-depth mechanistic studies. Most investigations focus on in vitro or in vivo models without comprehensive pharmacokinetic, toxicological, or clinical assessments, which restricts its translation into evidence-based therapeutics. Additionally, the potential cardiotoxicity associated with bufadienolide derivatives necessitates rigorous safety evaluations and structure–activity optimization. Future studies should emphasize the development of standardized extracts, advanced chromatographic and spectroscopic profiling, and bioactivity-guided isolation of lead compounds. Integration of molecular docking, omics-based target identification, and in vivo pharmacological validation can help elucidate the precise mechanisms underlying its therapeutic actions. Moreover, exploring novel delivery systems such as nanoformulations or biotechnological production of key metabolites could enhance bioavailability and therapeutic efficacy while ensuring sustainability. In conclusion, K. tomentosa is a promising reservoir of pharmacologically active molecules with potential applications in managing oxidative stress, inflammation, metabolic disorders, microbial infections, and cancer. Comprehensive, multidisciplinary research integrating phytochemistry, pharmacology, and formulation science is essential to transform this traditional medicinal plant into a modern, clinically relevant natural therapeutic.
REFERENCES




Prachi Gupta, J. V. Vyas, Dr. Vivek Paithankar, Dr. Anjali Wankhade, A Comprehensive Review on Kalanchoe tomentosa, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 802-812. https://doi.org/10.5281/zenodo.18920907
 10.5281/zenodo.18920907
10.5281/zenodo.18920907