
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
KMCH College Of Pharmacy, Coimbatore, India
Resveratrol, a naturally occurring polyphenolic molecule found in grapes, berries, and peanuts, holds tremendous medicinal potential because of its antioxidant, anti-inflammatory, neuroprotective, and cardioprotective effects. However, its therapeutic usage is hampered by poor water solubility, fast metabolism, and low oral bioavailability. distribution techniques based on nanotechnology have shown promise in overcoming these obstacles and improving resveratrol's stability, absorption, and targeted distribution. This study summarises current breakthroughs in nanocarrier platforms, including solid lipid nanoparticles, nanostructured lipid carriers, polymeric nanoparticles, liposomes, nanoemulsions, dendrimers, and nano-in-microparticle systems designed for resveratrol encapsulation. Key formulation characteristics including as particle size, zeta potential, encapsulation efficiency, and release behaviour are highlighted to demonstrate how nanoscale engineering improves pharmacokinetic and pharmacodynamic profiles.Studies reveal greater antioxidant capability, improved cellular absorption, prolonged release, and superior therapeutic effects against cardiovascular illnesses, diabetes, neurological disorders such as Alzheimer’s disease, and many malignancies when resveratrol is administered using nanocarriers. In conclusion, nanotechnology plays a critical role in overcoming the innate limits of resveratrol, considerably enhancing its therapeutic efficacy. Continued research on optimised nanocarrier design, scalability, biocompatibility, and clinical translation is required for establishing resveratrol-loaded nanosystems as effective and safe treatment modalities.
Resveratrol (RSV), scientifically referred to as trans-3,4’,5-trihydroxystilbene, is a naturally occurring polyphenol found in various foods such as grapes, nuts, certain fruits, and red wine. It has strong anti-inflammatory and antiproliferative effects and is a strong antioxidant [1].

Fig. 1: Natural sources of Resveratrol.
The terms cis and trans isomers refer to the two structural variations of resveratrol. More stability and biological activity are shown by the trans isomer [2].
Trans-Resveratrol Cis-Resveratrol
The potential health benefits of resveratrol, including its anti-inflammatory, antioxidant, hepatoprotective, cardioprotective, neuroprotective, anti-carcinogenic, anti-aging, and ability to prevent obesity and anti-diabetes properties, have received a lot of attention lately. Even though resveratrol is a natural molecule with significant health advantages, commercial pharmaceutical, supplement, and functional food products struggle to incorporate it because of its limited bioavailability, poor water solubility, and chemical instability [3]. Numerous plants, such as peanuts, cranberries, blueberries, mulberries, and grapes, spontaneously create resveratrol, a polyphenolic phytoalexin, in reaction to UV radiation, fungal invasion, and physical harm [4].Nanotechnology-based delivery methods have become a revolutionary approach to improve the solubility, stability, and tailored bioavailability of resveratrol in order to get beyond these pharmacokinetic and physicochemical constraints. These sophisticated nanoformulations can enhance therapeutic results in a variety of illness scenarios, provide regulated release, and shield resveratrol from quick breakdown.Resveratrol demonstrates anticancer properties by triggering apoptosis, halting the progression of the cell cycle, inhibiting cell growth, and suppressing enzymes such as ribonucleotide reductase, ornithine decarboxylase, and cyclooxygenase involved in prostaglandin production. Additionally, promyelocytic leukemia cells' development is promoted by it [1].Reactive oxygen species (ROS), which are mostly generated in the mitochondria during regular cellular metabolism, can seriously harm a cell if their concentrations exceed its antioxidant capacity. This oxidative stress may damage vital biological components such as proteins, lipids, and DNA. Overproduction of ROS in the skin is thought to play a role in a number of conditions, including several types of skin cancer [1].A safe and efficient substitute for lipophilic colloidal drug carriers are solid lipid nanoparticles (SLNs), which were developed in the early 1990s. In oil-in-water emulsions, solid lipids are substituted for liquid lipids at both physiological and ambient temperatures. These solid lipids combine with oils to create nanostructured lipid carriers (NLCs), which are regarded as the second generation of lipid nanoparticle technology. They can make close contact with the stratum corneum due to their small size, which enhances drug absorption [1].Many natural compounds are unable to advance past preclinical and Phase I clinical trials. Creating nanocarrier systems that can deliver these substances straight to the target location while reducing toxicity is one possible solution to this problem. Recent advancements in nanotechnology have shown promise in this area and are increasingly seen as the future of medication delivery. One well-known example is liposomal doxorubicin (Doxil®), which has demonstrated notable effectiveness in treating metastatic breast cancer. With encouraging outcomes, natural products have been delivered using a range of carriers, such as liposomes, nanoparticles, and micelles. However, because of their structural diversity, limited solubility, and vulnerability to rapid oxidation under physiological conditions, polyphenols continue to be difficult to encapsulate [5].Resveratrol because of its strong anti-inflammatory, antioxidant, anticancer, antiplatelet aggregation, antihypertensive and cardioprotective qualities, resveratrol (RES) is unique among these substances. These numerous benefits have led to a great deal of interest in using it to its full medical potential [5].
NANOFORMS OF RESVERATROL:The ability of several nanoparticle-based methods to distribute, encapsulate, and shield resveratrol has been studied. With a LogP = 3.1, resveratrol is mostly a hydrophobic molecule that is a component of several lipid-based nanoparticles, including liposomes, solid lipid nanoparticles and nano-emulsions. Gelatine, bovine serum albumin, silk protein, gliadin, and zein are among the proteins having non-polar domains that have been incorporated into a range of biopolymer nanoparticles. Since zein is a cheap and accessible byproduct of maize manufacturing, it is an especially alluring protein source [6].
Nano-in-microparticles strategy:
Trans-resveratrol nanoparticles encapsulated in alginate and hyaluronic acid microparticles for targeted gastric ulcer treatment. Similarly, nanoemulgels combining resveratrol and curcumin demonstrated burn wound healing [7].
Liposomes in buccal formulations:
A strategy to get around obstacles that restrict the use of the phytoactive compound for mouth cancer chemoprevention and treatment [8].Nanostructured lipid carrier formulations loaded with trans-resveratrol for use in medical nebulizers for pulmonary medication delivery [9]. Liposomes' synergistic administration of resveratrol and docetaxel increases the antitumor efficacy against prostate cancer [10].When compared to free resveratrol, a study revealed that transferosome-encapsulated resveratrol had a higher penetration power (27.5%). The features included zeta potential, viscosity, deformability, entrapment efficacy, kinetics, drug retention, in vitro drug release, and particle size and shape. The solvent evaporation approach was used to create the transferosome; in contrast, the liposomes and niosomes were produced using the reverse-phase evaporation technique [11].Microemulsions help the skin absorb resveratrol by postponing photodegradation and protecting the virus from UVB rays for up to an hour. Moreover, zinc-pectinate microparticles and calcium-pectinate beads containing resveratrol have been developed and thoroughly examined for controlled release and extensively studied for their potential in controlled and site-specific drug delivery. Additionally, biodegradable double-layered ultrafine fibers can offer prolonged release [12].There have also been positive results from complexing with β-cyclodextrins to increase resveratrol solubility. Resveratrol's low bioavailability can also be addressed by incorporating it into liposomes or micelles [13].The lipid core usually absorbs resveratrol and other hydrophobic drugs with ease. According to reports, solid lipid nanoparticles are more effective than liposomes at boosting resveratrol's chemical stability. It has been shown to increase resveratrol's bioavailability while providing protection against oxidation, hydrolysis, and photodegradation [14].
Table 1: Summary of Nanocarrier-Based Delivery Systems for Resveratrol
|
Nanocarrier System |
Composition/ Material |
Key Advantages |
Therapeutic Application / Outcome |
|
Solid Lipid Nanoparticles (SLNs) |
Solid lipids (glyceryl monostearate, stearic acid) stabilized with surfactants (Poloxamer 188, Tween 80) |
Improves chemical stability, protects against oxidation and photodegradation, enhances bioavailability |
Brain-targeted delivery; improved neuroprotective and antioxidant effects in Alzheimer’s models |
|
Polymeric Nanoparticles (PLGA, PCL, PEG-based) |
Poly(lactic-co-glycolic acid), polycaprolactone, PEG |
Biodegradable, sustained release, enhanced circulation time |
Improved cardioprotective, anti-inflammatory, and neuroprotective activity; increased brain bioavailability |
|
Protein-Based Nanoparticles |
Zein, gelatin, bovine serum albumin |
Natural, biocompatible carriers; suitable for hydrophobic molecules |
Enhanced oral delivery and stability of resveratrol; reduced oxidative stress |
|
Nanoemulsions / Nanoemulgels |
Oil phase (isopropyl myristate, MCT oil) + surfactant (Tween 80) + co-surfactant |
Improves solubility and stability, enhances dermal penetration |
Synergistic wound-healing effect when combined with curcumin; improved photoprotection |
|
Nanocrystals |
Pure drug nanoparticles stabilized by surfactants |
High drug loading (>98%), rapid dissolution, minimal excipients |
Enhanced anticancer efficacy and localized drug concentration in tumor tissue |
In the treatment of myocardial injury, resveratrol-loaded PLGA nanoparticles demonstrated superior anti-inflammatory, antioxidant, and cardioprotective effects compared to free resveratrol [15].
CARDIO-PROTECTIVE EFFECTS:
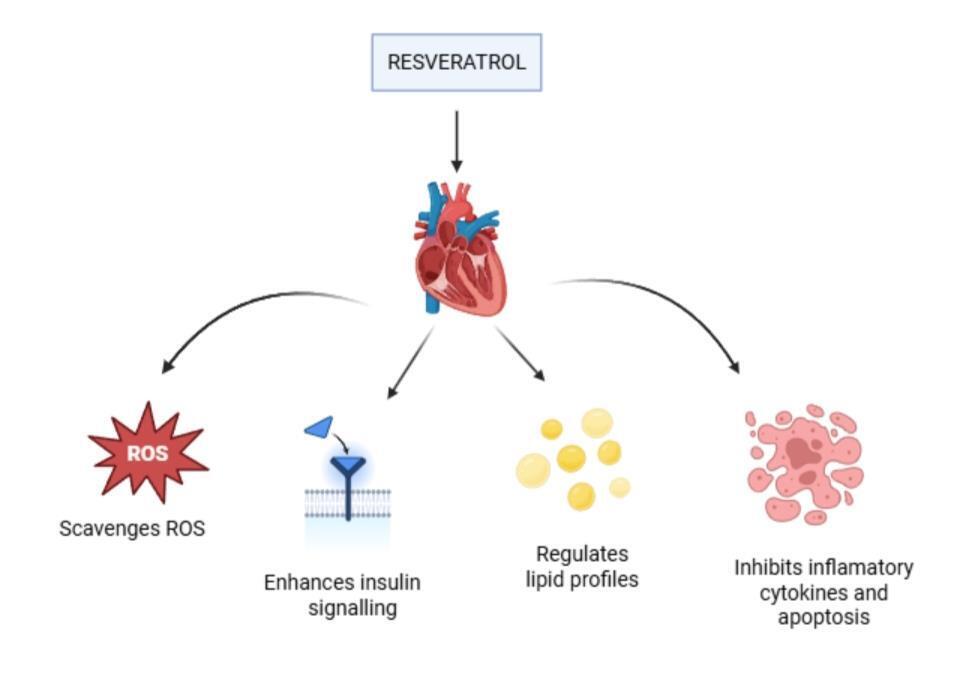
Red grape skin is the primary source of resveratrol, a bioactive polyphenolic stilbene that has received a lot of attention for its cardioprotective qualities. Its strong antioxidant activity and capacity to regulate cellular oxidative balance are major factors in its therapeutic importance in the prevention of coronary heart disease. By lowering oxidative stress, improving insulin sensitivity, and regulating lipid metabolism, resveratrol has a variety of cardioprotective benefits [16]. By efficiently scavenging reactive oxygen species, it shields cardiac tissue from thrombotic events, myocardial fibrosis, atherosclerotic development, ischemia-induced damage, and hypertensive cardiomyopathy [17]. Furthermore, resveratrol is effective in reducing ischemia-reperfusion injury and ventricular arrhythmias because of its special ability to block apoptosis even at very low concentrations [18]. Additionally, new research points to the possible significance of maternal resveratrol supplementation in prenatal metabolic programming and phenotypic adaptation by suggesting that it may reduce cardio-metabolic risks in children [19]. By controlling cellular survival signaling pathways, improving mitochondrial function, promoting adaptive myocardial remodeling, and having strong antioxidant and anti-inflammatory properties, resveratrol collectively supports cardiovascular health [20] (as shown in figure 2).
ALZHEIMER’S DISEASE:
Beyond Resveratrol’s cardiovascular benefits, it exhibits profound neuroprotective effects that are enhanced through nanoformulations. Neurocognitive diseases, like Alzheimer's disease, are non-psychiatric brain syndromes in which a patient's mental state is severely traumatized by a substantial decline in cognitive performance. The economy in particular suffers when neurocognitive problems are not adequately treated [21].In recent decades, scientists have had the opportunity to create novel treatments for neurocognitive disorders because of the discovery of resveratrol, a naturally occurring plant component. This is due to the fact that resveratrol successfully preserves the brains of those who suffer from neurocognitive disorders by targeting specific processes, such as inflammation and oxidative stress [21]. Neuroinflammation, the development of amyloid plaque and neurofibrillary tangles, hyper phosphorylation of tau proteins, and acetylcholine (Ach) depletion are some of the main pathogenic features of AD. The only treatment for AD is symptomatic, and there are now only six FDA-approved drugs available in the US [22]. The effects of oxidative stress can include mitochondrial dysfunction and cellular damage. According to research, Alzheimer’s patients have fewer intact mitochondria, and this reduced mitochondrial function is caused by the loss or failure of particular electron transport chain enzymes. The generation of reactive oxygen species is mostly attributed to mitochondria. The aberrant development of Aβ in the brain is the ultimate cause of cognitive impairment, which also causes synaptic and neuronal functional impairment and hyper phosphorylation of tau protein to create neurofibrillary tangles [23].In plants, the cis and trans versions are more prevalent and potent. The primary sources of this anti-inflammatory, antioxidant, cardioprotective, and neuroprotective compound are red wine, berries, and grapes. It helps prevent amyloid beta from aggregating, shields neurons from toxicity, oxidative damage, and apoptosis, and gets rid of plaque that has already formed inside the brain [22].In order to prevent AD, red wines and Concord grape juice can change several brain pathways, such as lowering Aβ oligomerization, lowering Aβ production, and encouraging neuroplasticity. Interestingly, several studies have demonstrated that resveratrol may enhance intracellular Aβ clearance, in part via inducing autophagy and AMPK signalling in vivo. Overall, these research results lend credence to the idea that autophagy and inflammation work together to produce resveratrol's anti-amyloidogenic effects. Polyphenols may also reduce hyperphosphorylation and abnormal tau aggregation, according to new studies [24].The low bioavailability of resveratrol limits its biological and medicinal efficacy. Moreover, it is chemically unstable and weakly soluble in water, degrading by isomerization in response to various enzymes, high temperatures, pH shifts, and UV light. Furthermore, resveratrol must cross the blood-brain barrier, a specialized barrier composed of brain endothelial cells that tightly regulate the flow of chemicals between the blood and the brain, in order to enter the brain, where it might demonstrate its neuroprotective properties [25].Resveratrol's ability to enter the brain is therefore restricted, and this work offers a novel way to get around these limitations by creating nanosystems that can shield resveratrol from the wide range of metabolism and radiation as well as improving the transport into a suitable therapeutic concentration in the brain. Considering the excellent solid-state encapsulation efficiency and high lipophilicity of resveratrol SLNs (lipid nanoparticles). In order to encapsulate and transport resveratrol to the brain, previously published SLNs were chosen. These nanoparticles combine the best features of most nanocarriers with the formulations' excellent stability and ease of manufacture, eliminating the need for organic solvents.SLNs are a visually appealing colloidal resveratrol delivery method that may target the brain, particularly when polysorbate 80 is utilized as the surfactant in their manufacturing. Consequently, the goal of this work was to create SLNs that were loaded with resveratrol in order to remedy the low solubility issues, shield it from degradation, and simultaneously target the brain. Resveratrol-loaded SLNs were functionalized using two distinct methods involving apolipoprotein E (ApoE) [26] (as shown in figure 3). High shear homogenization and ultrasonication were used to produce solid lipid nanoparticles encapsulated with Resveratrol, LissRhod-PE and 1,2-distearoyl-sn-glycero-3phosphoethanolamine-poly(ethylene glycol) (DSPE-PEG). SLN guaranteed its stability based on surface shape, size, Zeta Potential (ZP), and polydispersity index (PDI). Its encapsulation effectiveness was 94%. An antibody (OX-26) was added to SLN to boost its brain bioavailability [27]. To improve Resveratrol brain bioavailability, they developed Apo lipoprotein-functionalized Resveratrol-based SLN by employing the high shear homogenization technique. A polymer-based micelle called Resveratrol-loaded polymeric micelle was created to stop Aβ-induced oxidative damage. The formulation was made by lyophilizing and drying PEG as the hydrophilic shell and using polycaprolactone as the block copolymer for the hydrophobic core. It was reported that the encapsulation efficiency was an amazing 89% [27].
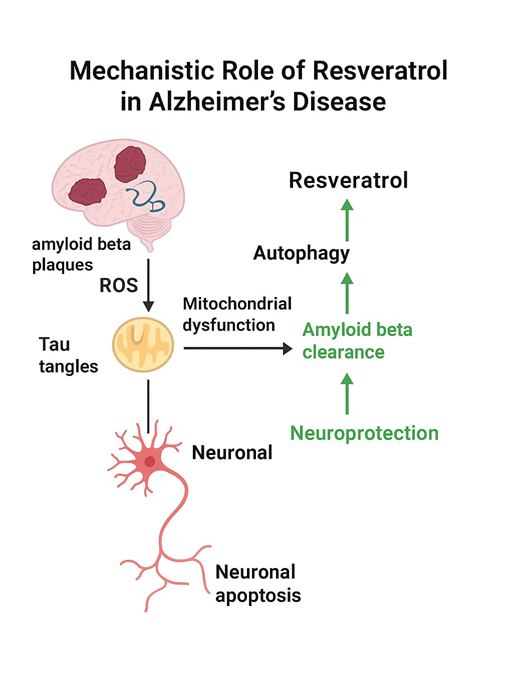
Fig 3: Schematic representation of the mechanistic role of resveratrol in mitigating Alzheimer’s Disease.
ANTI-CANCER EFFECTS:
A naturally occurring polyphenolic phytoalexin found in many different plant sources, resveratrol has garnered significant attention as a promising nanotherapeutic option for the treatment of lung cancer. Its addition to nano-formulations improves pharmacokinetic and pharmacodynamic characteristics, resulting in better intracellular bioavailability in malignant pulmonary tissues, controlled and prolonged drug release, selective tumor-site accumulation, and enhanced physicochemical stability. Mechanistically, resveratrol reduces carcinogenesis and cancer progression by modulating key carcinogenic signaling cascades, including the MAPK, mTOR, PI3K/Akt, and Wnt/β-catenin pathways [28].Resveratrol's capacity to cause programmed cell death and alter cell-cycle dynamics is primarily responsible for its anticancer potency. Its dual role in cancer interception and intervention is highlighted by the fact that its chemopreventive and therapeutic effects span the triphasic continuum of carcinogenesis—initiation, promotion, and malignant transformation [29]. Resveratrol has shown strong pro-apoptotic bioactivity in preclinical animals. By increasing caspase-3 and caspase-9 activity, inhibiting anti-apoptotic mediators like Bcl-2, Bcl-XL, and cyclin B, and upregulating pro-apoptotic and cell-cycle regulatory genes including BAX, p21, p27, and p53, it triggers intrinsic apoptotic pathways in HeLa cells. By stimulating Nrf2 transcription and encouraging the development of antioxidant enzymes like SOD, GPX, and CAT, it simultaneously strengthens cellular antioxidant defenses and successfully reduces oxidative macromolecular damage. By activating the Sirt1/AMPK and Nrf2 signaling pathways, resveratrol protects hepatocytes against cellular damage caused by oxidative stress.Furthermore, by blocking NF-κB's nuclear translocation and changing its post-translational changes, such as phosphorylation and acetylation, resveratrol reduces downstream inflammatory mediators in colorectal cancer cells. Activation of p53 and inhibition of the IGF-1R/Akt/Wnt oncogenic network further enhance its anticancer characteristics [30].In vivo studies utilizing the LNCaP-Luc prostate cancer xenograft model demonstrated that resveratrol analogues, including trimethoxy-resveratrol and piceatannol, exhibit improved systemic bioavailability and more pronounced chemopreventive activity, as evidenced by significant reductions in tumor burden and proliferative indices [31].Additionally, resveratrol modifies the AMPK/mTOR axis, which activates autophagy or apoptosis and inhibits the development of ovarian cancer cells. Notably, resveratrol's biological effects are dose-dependent and biphasic: at physiologically moderate concentrations, it primarily acts as an antioxidant, protecting genomic integrity and preventing cellular damage caused by reactive oxygen species; at supra-physiological doses, however, it may take on pro-oxidant characteristics, encouraging oxidative DNA damage in experimental systems [32].Because of their great drug-loading efficiency—typically containing >98% active medicinal ingredient with negligible excipient content—nanocrystals stand out among nano-delivery technologies. This reduces excipient-related toxicity while enabling exceptionally high localized medication concentrations [32]. Nanocrystal synthesis can be accomplished via bottom-up techniques like antisolvent precipitation or top-down techniques like media milling and high-pressure homogenization, which allow for exact control of particle size and crystallinity to maximize therapeutic performance [32].
ANTI-INFLAMMATORY EFFECTS:
Resveratrol exhibits strong anti-inflammatory potential by targeting multiple molecular mediators involved in the inflammatory cascade. It suppresses prostaglandin synthesis by dose-dependently modulating the enzymatic activity of cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2). Oral resveratrol considerably reduced leukocyte adhesion and vascular inflammation in an endotoxin-induced ocular inflammation model by downregulating the expression of monocyte chemoattractant protein-1 (MCP-1) and intercellular adhesion molecule-1 (ICAM-1). Additionally, it reduces the expression of pro-inflammatory cytokines in trabecular meshwork cells, including E-selectin, interleukin-6 (IL-6), and interleukin-8 (IL-8), indicating a widespread inhibitory effect on cytokine-driven signaling pathways. These results demonstrate the therapeutic potential of resveratrol as an efficient regulator of inflammatory responses in ocular and systemic diseases [33].Subsequent studies demonstrated that resveratrol downregulates NF-κB and AP-1 activation, decreases inducible nitric oxide synthase (iNOS) and nitrite production, and inhibits granulocyte–macrophage colony-stimulating factor (GM-CSF), IL-8, and cAMP response element-binding protein (CREB)-dependent transcription, showing greater inhibitory capacity than dexamethasone. In human mast cells (HMC-1) treated with phorbol 12-myristate 13-acetate (PMA) and calcium ionophore A23187, resveratrol suppressed IL-6, IL-8, TNF-α, and cyclooxygenase-2 (COX-2) expression through decreased intracellular Ca²? and inhibition of extracellular signal-regulated kinase (ERK1/2) activity [34].Collectively, these findings emphasize its strong antioxidant and anti-inflammatory potential, although its therapeutic impact is constrained by poor solubility and bioavailability. In cardiovascular diseases (CVDs), resveratrol exerts similar anti-inflammatory actions by preventing monocyte adhesion to endothelial cells, suppressing TNF-α, IL-1β, and IL-6 synthesis, and promoting anti-inflammatory cytokine release. It also modulates key signaling cascades, notably NF-κB and JAK/STAT pathways. Nevertheless, current experimental and clinical data remain variable, and its overall influence on systemic inflammatory biomarkers in CVD patients is yet to be fully clarified [35].Reductions in biomarkers like C-reactive protein (CRP) and tumor necrosis factor-alpha (TNF-α) have demonstrated that resveratrol greatly reduces systemic inflammation. Its effects on oxidative stress markers, such as total antioxidant capacity (TAC), glutathione peroxidase (GPx), and superoxide dismutase (SOD), are less consistent and probably depend on study population, duration, and dose. Overall, resveratrol has strong anti-inflammatory potential; however, further research is needed to determine how effective it is as an antioxidant, underscoring its significance as a dietary intervention for metabolic and chronic inflammatory diseases [36].
ANTIOXIDANT EFFECTS:
Resveratrol demonstrates potent antioxidant activity through both direct and indirect mechanisms of reactive oxygen species (ROS) regulation. Resveratrol significantly decreased ROS overproduction in trabecular meshwork cells subjected to oxidative or hyperoxic stress, maintaining mitochondrial integrity and cellular viability. It prevented oxidative damage and angiogenic activation in retinal pigment epithelial (ARPE-19) cells by suppressing oxidative stress-induced overexpression of vascular endothelial growth factor (VEGF), MCP-1, and IL-8. The maintenance of cellular redox equilibrium and the activation of endogenous antioxidant defenses complement these activities. All things considered, resveratrol's capacity to lessen oxidative damage supports its function as a multipurpose defense against oxidative stress-induced cellular dysfunction [33].
ADVERSE EFFECTS OF RESVERATROL:
Numerous studies have demonstrated that resveratrol (RSV) causes dose-dependent toxicities despite its potent anti-inflammatory, antioxidant, and anticancer activities. Shaito et al. (2020) state that while low concentrations (≤50 µM) activate protective pathways such SIRT1, AMPK, and Nrf2, higher concentrations (>100 µM) cause oxidative stress, mitochondrial dysfunction, DNA damage, and mortality by excessive reactive oxygen species generation. Organ-specific toxicities that have been observed in both clinical and experimental trials include hepatotoxicity, nephrotoxicity, and gastrointestinal problems. Additionally, RSV has endocrine-modulating action that impacts thyroid hormone metabolism and estrogen receptors, as well as immunosuppressive and genotoxic effects at supraphysiological levels. The biphasic or hormetic action of resveratrol, which is cytoprotective at low doses but pro-oxidant at large doses, makes determining suitable therapeutic limits difficult [37]. Resveratrol's health advantages are a "double-edged sword" due to its biphasic dose-dependent impact (Salehi et al., 2018). Cytotoxicity, pro-oxidant activity, mitochondrial dysfunction, endocrine disruption, and hepatic stress are among the negative consequences that may result from using it at larger concentrations or for longer periods of time, despite the fact that low dosages have been demonstrated to have antioxidant and protective properties. In order to preserve safety and therapeutic efficacy, these unfavorable results highlight the significance of regulated dosage and the application of targeted nanocarrier delivery systems [38].These findings highlight the necessity of targeted and dose-optimized delivery strategies, which can be successfully accomplished through nanocarrier-based systems that reduce off-target exposure and enable controlled administration. Incorporating resveratrol into nanotechnology platforms can minimize toxicity while preserving its therapeutic benefits.
RECENT TRENDS AND FUTURE PERSPECTIVES:
Recent advances in nanotechnology have opened up new ways to overcome the pharmacokinetic limitations of resveratrol. Li et al. (2023) describe numerous nanocarrier systems that enhance the stability, solubility, and bioavailability of resveratrol. These systems include liposomes, polymeric nanoparticles, and nanocrystals. When compared to free resveratrol, PEGylated and pH-responsive liposomes as well as PLGA-based nanoparticles can greatly increase the bioavailability and circulation half-life. Protein-based nanoparticles and cell membrane-coated structures are examples of biomimetic technologies that provide greater biocompatibility and targeted delivery, particularly for treating brain tumours. However, long-term safety, clinical translation, and large-scale production remain problems. Future studies should focus on improving the stability of formulations and their cost-effectiveness, while simultaneously enhancing pharmacokinetics and decreasing toxicity.To combat complicated diseases, we need to develop multifunctional imaging and targeted therapeutics. Applying these advancements requires clinical validation and compliance with regulations, which calls for further cooperation across nanomedicine fields [39]. Furthermore, we need to provide resveratrol with precision targeting in order to maximise efficacy and minimise off-target effects. Future study should leverage omics and systems biology to further understand tissue-specific mechanisms and develop response biomarkers. Integration into functional foods or combination therapies could broaden therapeutic and preventive uses, especially in aging and chronic diseases. Scalable and sustainable production is crucial, as are standardised clinical trials to evaluate the best dosage, methods of administration, and long-term safety. By fusing cutting-edge nanocarriers with a thorough comprehension ofWe could effectively convert resveratrol into safe, efficient, and widely available medicines with the help of mechanisms and regulatory backing [40]. Liu Peng et al. suggest that because resveratrol both stimulates ferroptosis in cancer and inhibits it in neurological and cardiovascular illnesses, its modulation of ferroptosis may have therapeutic effects. Future studies should focus on finding biomarkers, elucidating underlying mechanisms, and investigating nano-formulated resveratrol-based targeted therapeutic combinations. Linking genetic profiles to resveratrol sensitivity may improve precision medicine methods for a number of illnesses, and standardised clinical trials are required to evaluate safety and efficacy [41]. Standardised evaluation criteria and scalable production methods remain significant translational challenges despite preclinical progress.
Challenges and Gaps:
Future Prospects:
DISCUSSION
The low solubility, instability, and poor bioavailability of resveratrol have been successfully addressed by formulations based on nanotechnology, significantly increasing its therapeutic potential. Liposomes, solid lipid nanoparticles, and polymeric nanoparticles are a few examples of nanocarriers that provide enhanced stability, targeted delivery, and controlled release. When compared to free resveratrol, these nanoformulations exhibit better therapeutic results, such as increased antioxidant protection and decreased ischemic injury in cardiovascular illnesses, better brain targeting in Alzheimer's disease, and more potent tumor inhibition in cancer treatment. However, there are still issues like long-term safety, large-scale production, and regulatory approval that need for additional clinical and transdisciplinary study.
CONCLUSION
Numerous pharmacological characteristics of resveratrol, such as cardioprotective, neuroprotective, anticancer, anti-inflammatory, and antioxidant properties, demonstrate its potential for therapeutic advantages. However, physiochemical limitations such as limited systemic bioavailability, rapid metabolism, and poor water solubility restrict clinical translation. Nanotechnology-based medication delivery devices represent a feasible approach to the circumvention of these difficulties. Liposomes, polymeric nanoparticles, and nanoemulsions are examples of advanced nanocarriers that have been developed to improve resveratrol's physicochemical stability, cellular uptake, and bioavailability, thereby increasing its therapeutic efficacy across a wide range of disease contexts. Furthermore, by facilitating controlled release, targeted dispersion, and defence against degradation processes, these nanosystems optimise its pharmacodynamic potential. These novel formulations preserve the bioactivity of resveratrol while optimising its pharmacokinetic profile.Generally speaking, resveratrol delivery via nanotechnology appears to be very promising in terms of translation and offers a solid scientific approach for fully utilising its therapeutic potential in the management of complicated and long-term illnesses. Even if there are several encouraging preclinical results, more carefully planned clinical studies need to be carried out to verify the safety, effectiveness, and translational potential of nanoformulated resveratrol. The coupling of resveratrol with superior nanocarrier technologies heralds a new frontier in drug delivery research, opening new paths for its future application as a therapeutic agent.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
REFERENCES




Dharani Priya B., Athulya P. K., Kavya M., Kiruthika V., Advances in Nanotechnology-Based Delivery of Resveratrol: Challenges, Opportunities, and Therapeutic Applications with Mechanistic Insights., Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 1183-1194. https://doi.org/10.5281/zenodo.18976920
 10.5281/zenodo.18976920
10.5281/zenodo.18976920