
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmacology, Vidhyabharati College of Pharmacy, Amravati
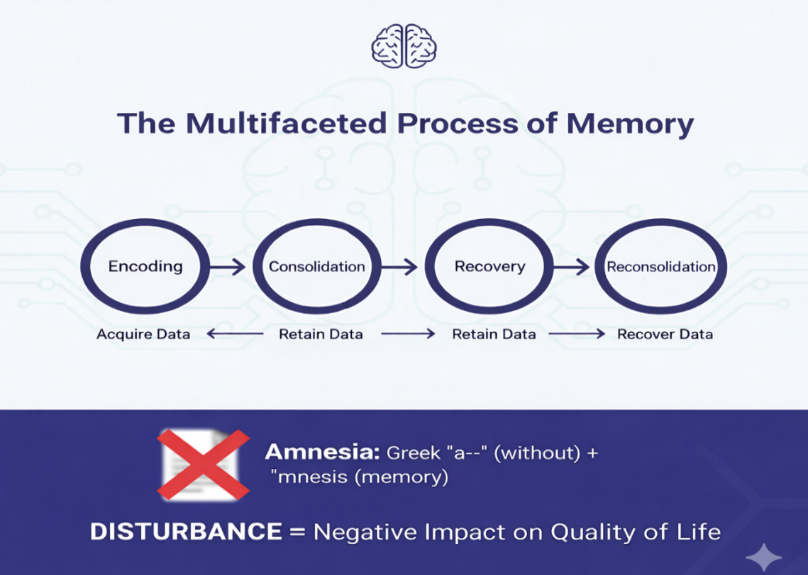
The word ‘amnesia’comes from the Greek roots a- (without) and mnesis (memory) or in simple words, amnesia means loss of memory. It usually happens because of brain injury, disease, or the effects of drugs or alcohol. The amnesia which is caused by drugs are drug-induced amnesia which is a significant clinical challenge caused by various medications that impair memory encoding, consolidation, or retrieval. This review explores classical drug classes such as benzodiazepines, anticholinergics, hypnotics-sedative, anticonvulsants and others like statins, immunosuppressant, proton pump inhibitors etc which are linked to memory deficits. It highlights recent advancements in understanding the neurobiological mechanisms, including neurotransmitter modulation, disrupted neural oscillations, synaptic plasticity impairments, neuroinflammation and neurogenesis. Additionally, emerging research on novel therapeutics, neurophysiological insights, pharmacovigilance data, and neurostimulation techniques offers promising strategies for prevention and reversal. The review underscores the importance of integrating mechanistic insights with clinical approaches to improve patient safety, optimize treatment, and explore future research directions in managing drug-induced cognitive side effects.
Memory is an essential cognitive function that permits individuals to acquire, retain, and recover data that defines a person’s identity. Memory is a multifaceted cognitive process that involves different stages: encoding, consolidation, recovery, and reconsolidation. Any disturbance in memory can negatively affect the quality of life [1]. The word ‘amnesia’comes from the Greek roots a- (without) and mnesis (memory). Broadly, it refers to a significant loss of memory, usually caused by brain injury, illness, or the effects of drugs and alcohol. Individuals with amnesia may struggle to remember past experiences, known information, and may also face difficulty in forming new memories or learning new skills [2].
|
Type |
Description |
Example |
|
Retrograde Amnesia |
Loss of previously stored memories |
Forgetting past events |
|
Anterograde Amnesia |
Inability to form new memories after trigger |
Common with benzodiazepines |
|
Transient Amnesia |
Short-term & reversible memory loss |
Alcohol intoxication |
|
Dissociative Amnesia |
Memory loss due to stress or trauma |
PTSD-related amnesia |
|
Drug-Induced Amnesia |
Memory impairment due to medications |
Sedatives, anesthetics |
There are various types of amnesia are as follows:-
Drug-induced amnesia refers to partial or complete impairment of memory resulting from pharmacological effects of medications.It may affect:-
Drug-induced amnesia refers specifically to the memory impairment produced by pharmacological agents, which can occur independently of sedation or other central depressant effects [3].Amnesia arising from drug exposure primarily affects the ability to form new memories (anterograde amnesia) but can also influence the retrieval of previously acquired memories (retrograde amnesia). Multiple drug classes including benzodiazepines, anticholinergics, hypnotics, anticonvulsants, and others are implicated [4].Among these, benzodiazepines are the most widely recognized agents causing anterograde amnesia. They potentiate the inhibitory effects of gamma-aminobutyric acid (GABA) through GABAA_AA? receptor modulation, thereby disrupting the formation of new memories without necessarily affecting retrieval of previously stored information. Similarly, intravenous anesthetics such as propofol and thiopental induce dose-dependent impairment in memory encoding and consolidation, even at sedative concentrations. Anticholinergic drugs like scopolamine, by blocking central muscarinic receptors, produce reproducible cognitive deficits that have made them valuable pharmacological tools for modelling amnesia in animals. Moreover, N-methyl-D-aspartate (NMDA) receptor antagonists, including ketamine and MK-801, interfere with glutamatergic neurotransmission, thereby impairing long-term potentiation (LTP) and learning processes.
Medications Associated With Amnesia:
Benzodiazepines:- Benzodiazepines enhance GABA_A receptor activity by acting as positive allosteric modulators, which increases inhibitory neurotransmission particularly in hippocampal circuits critical for memory encoding and consolidation. Drugs like midazolam induce transient anterograde amnesia by disrupting these processes, leading to impaired formation of new memories. This amnestic effect is linked predominantly to the α1 and α5 subunits of the GABA_A receptor, which are highly expressed in the hippocampus and amygdala—regions central to memory and learning. While α1 subunit modulation is associated with sedation and memory impairment, the role of the α5 subunit appears to affect hippocampus-dependent spatial memory, although its exact involvement in benzodiazepine-induced amnesia is complex and under active investigation. Behavioral and molecular studies have demonstrated that benzodiazepines impair recognition memory and contextual fear conditioning through these receptor subtypes. Importantly, antagonists like flumazenil can partially reverse these amnestic effects, supporting the specificity of benzodiazepine binding in mediating memory disturbances. Ongoing research also examines sex-specific hormonal influences and circuit-level neural ensemble changes to develop alternative therapeutic strategies with reduced cognitive side effects [5,6,7].
Anticholinergics:- Anticholinergics act by blocking muscarinic acetylcholine receptors, particularly the M1 subtype highly expressed in the hippocampus and prefrontal cortex, which are crucial for learning, episodic memory, and executive functions. This antagonism impairs cholinergic neurotransmission, leading to disruptions in sensory gating and information processing essential for memory formation and retrieval. Cognitive impairments depend on the cumulative anticholinergic load, baseline cognitive status, and individual pharmacokinetic variability and are exacerbated in populations with compromised blood-brain barrier permeability, such as diabetes patients or the elderly. Long-term use of anticholinergic drugs has been associated with brain structural changes including cortical atrophy and reduced cerebral metabolism, which resemble patterns seen in conditions like Alzheimer?s disease, suggesting potential acceleration of neurodegenerative processes[8,9]. Clinical studies reveal associations between prolonged anticholinergic exposure and increased risks of mild cognitive impairment and dementia, in a dose-dependent manner, although findings are not entirely uniform across populations[10,11,12]. Symptoms commonly reported include deficits in episodic memory, verbal fluency, visuospatial ability, and executive function, with some evidence indicating potential reversibility of cognitive decline upon reduction of anticholinergic burden [13].
|
Drug Class |
Examples of Medications |
Effect on Memory |
|
Benzodiazepines (Anti anxiety, Sedatives, Hypnotics) |
Alprazolam (Xanax), Diazepam (Valium), Lorazepam (Ativan), Midazolam (Versed), Temazepam (Restoril), Triazolam (Halcion) |
Cause anterograde amnesia (inability to form new memories after taking the drug) by dampening activity in memory-related brain areas. The risk increases with dosage and duration of use. |
|
Anticholinergic Drugs (Incontinence, Antihistamines, Antidepressants) |
Oxybutynin (Ditropan), Solifenacin (Vesicare), Diphenhydramine (Benadryl), Amitriptyline (Elavil) |
Block acetylcholine, a crucial neurotransmitter for learning and memory, which can lead to memory deficits and potentially increase dementia risk with long-term use. |
|
Non-benzodiazepine Hypnotics ("Z-drugs") |
Zolpidem (Ambien), Eszopiclone (Lunesta), Zaleplon (Sonata) |
Act on similar brain pathways as benzodiazepines and can cause amnesia, especially the inability to remember events that occurred while under the drug's influence. |
|
Anticonvulsants/Anti-seizure Drugs |
Gabapentin (Neurontin), Pregabalin (Lyrica), Topiramate (Topamax), Levetiracetam (Keppra), Valproic Acid (Depakote) |
Slow down signals in the central nervous system, affecting concentration, attention, and the ability to absorb new information. |
|
Opioid Analgesics (Narcotic Painkillers) |
Morphine, Hydrocodone (Vicodin), Oxycodone (Oxycontin), Fentanyl |
Interfere with how the hippocampus processes information and alter chemical messengers involved in cognition, leading to memory problems, especially with chronic use. |
|
Statins (Cholesterol-lowering) |
Atorvastatin (Lipitor), Simvastatin (Zocor) |
In some cases, statins have been reported to cause temporary memory loss, possibly by reducing brain cholesterol, which is essential for nerve cell connections. |
Hypnotics and Sedative:- Sedatives and hypnotics comprise a heterogeneous group of central nervous system depressants, including benzodiazepines, non-benzodiazepine “Z-drugs” (such as zolpidem), barbiturates, and intravenous anesthetics like propofol and thiopental. These agents primarily act by enhancing the effects of gamma-aminobutyric acid (GABA) through GABA_A receptor modulation, promoting neuronal inhibition within limbic and cortical structures critical for regulating arousal, sleep, and memory processing. Their amnestic effects predominantly manifest as anterograde amnesia, characterized by impaired encoding and consolidation of new memories, while largely sparing the retrieval of previously stored information . Notably, the impairment of memory by these drugs is distinct from their sedative properties—a phenomenon observed in experimental settings using electroencephalographic event-related potentials, which demonstrate that drugs like midazolam and propofol have specific, dose-dependent effects on memory circuits independent of sedation . The risk and severity of cognitive side effects are influenced by dose, duration, comorbid conditions, age, and concomitant use of other CNS depressants. Zolpidem, which selectively targets the α1 subunit of the GABA_A receptor, has been shown to produce potent anterograde amnesia with minimal residual psychomotor effects, while barbiturates and older hypnotics are associated with more pronounced residual sedation and impaired coordination. Clinical and epidemiological studies reveal an increased risk of memory impairment, falls, daytime drowsiness, and impaired attention in individuals using sedative-hypnotics, especially the elderly and those with underlying neurological vulnerability. Although sedative-hypnotics are therapeutically employed to prevent distressing recall during medical procedures and for the short-term management of insomnia and anxiety, repeated or long-term exposure elevates the risk of tolerance, dependence, and persistent cognitive adverse effects. Careful selection, titration, and periodic re-evaluation of these medications are therefore essential to optimize therapeutic benefit while minimizing the potential for neuropsychological harm [14,15,16].
Anti-convulsants:- Anti-convulsant are medications that prevent or treat seizures by controlling abnormal electrical activity in the brain. These medications commonly regarded as having a relatively high risk of cognitive impairment are Carbamazepine, valproate, clonazepam, ethosuximide, phenobarbital, phenytoin, primidone, topiramate, zonisamide, sulthiame. There are two types of cognitive impairement: Acute is for impaired attention and executive functions, delirium while chronic is for impaired attention, executive functions, processing speed, learning and also for impaired memory and speech. Anti- epileptic medications having the mechanism that it Reduced neuronal excitability through e.g. interaction with voltage-gated ion channels or amplification of GABA effects and also contain inhibition of carbonic anhydrase.Patients with neurologic or psychiatric comorbidity, pre-existing cognitive impairment, polytherapy, or high ASM serum concentration [17].
Other Medications:- Other Medications Associated with Amnesia include a range of drug classes that—though less directly implicated than sedatives or anticholinergics—have been linked to clinically meaningful memory impairment in certain contexts. Statins, commonly used for hyperlipidemia, have been associated in some observational studies with reports of cognitive slowing and short-term memory loss, though causality remains debated and effects appear reversible upon drug discontinuation[18] . Beta-blockers, prescribed for cardiovascular conditions, can cross the blood– brain barrier and may interfere with memory retrieval, particularly in stress-related or emotionally arousing contexts[19]. Immunosuppressants such as tacrolimus and cyclosporine, often used in organ transplantation, have demonstrated potential to induce reversible or persistent cognitive disturbances—including amnesia—via neurotoxic effects, particularly at high serum levels[20]. Additionally, proton pump inhibitors (PPIs), used in acid-related disorders, have recently be implicated in increased risks of cognitive impairment and incident dementia in large pharmacovigilance and epidemiological analyses, though mechanistic links remain under investigation. The expanding recognition of these “non-traditional” drug classes as possible contributors to drug-induced amnesia highlights the importance of clinician awareness and underscores the utility of ongoing pharmacovigilance to detect rare or delayed memory side effects in wider patient populations[21,22].
Mechanisms Of Drug-Induced Amnesia:
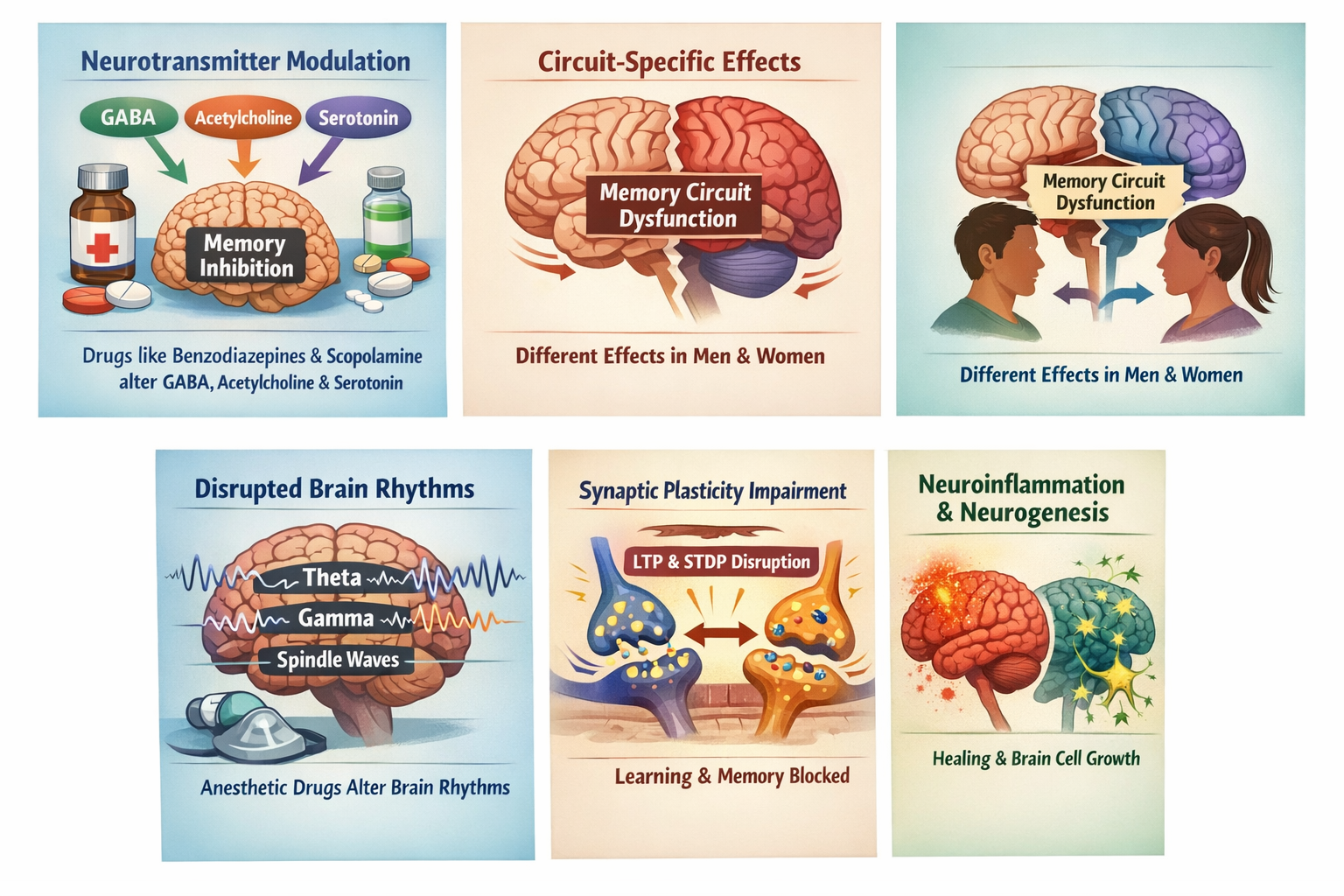
Drug-induced amnesia involves modulation of key neurotransmitters such as GABA, acetylcholine, and serotonin. Enhancement of GABAergic activity by drugs like benzodiazepines increases inhibitory neurotransmission, leading to reduced neural excitability in hippocampal and cortical circuits essential for memory formation. This is seen in GABAA receptor agonists such as propofol and benzodiazepines, which enhance inhibitory signaling and produce amnesia [23,24]. Cholinergic blockade, especially disruption of acetylcholine signaling by agents like scopolamine, impairs retention and memory consolidation, as acetylcholine is crucial for cognitive processes in the hippocampus and cortex [25,26]. Serotonergic modulation also plays a role, with some antipsychotic drugs like clozapine reversing drug-induced cognitive deficits by acting as serotonin and dopamine receptor antagonists[27].
Recent studies highlight sex-dependent effects of drugs like benzodiazepines on hippocampal ensembles, indicating that drug-induced amnesia may involve dysfunction of specific memory circuits with differences in male and female brain responses. This specificity in circuit disruption contributes to the complexity of drug-induced memory deficits [27].
Anesthetic drugs alter critical neural oscillations such as theta, gamma, and spindle rhythms that are vital for memory consolidation. For example, sevoflurane enhances hippocampal theta rhythms through TASK-3 channel modulation, which impairs memory processing by disrupting normal oscillatory patterns needed for encoding and consolidation of memories [28].
Drugs can impair synaptic plasticity mechanisms like spike-timing-dependent plasticity (STDP) and long-term potentiation (LTP), which underpin learning and memory. Enhanced GABAergic inhibition and cholinergic dysfunction interfere with these synaptic strengthening processes, leading to deficits in memory formation .
Some novel compounds have been found to mitigate drug-induced neuroinflammation and promote hippocampal neurogenesis, which may reverse or prevent amnesia. GABA receptor agonists and other neuroprotective agents can restore neuronal function and facilitate neurogenesis, which supports cognitive recovery after drug-induced impairment [29].
Recent Innovative Advances:
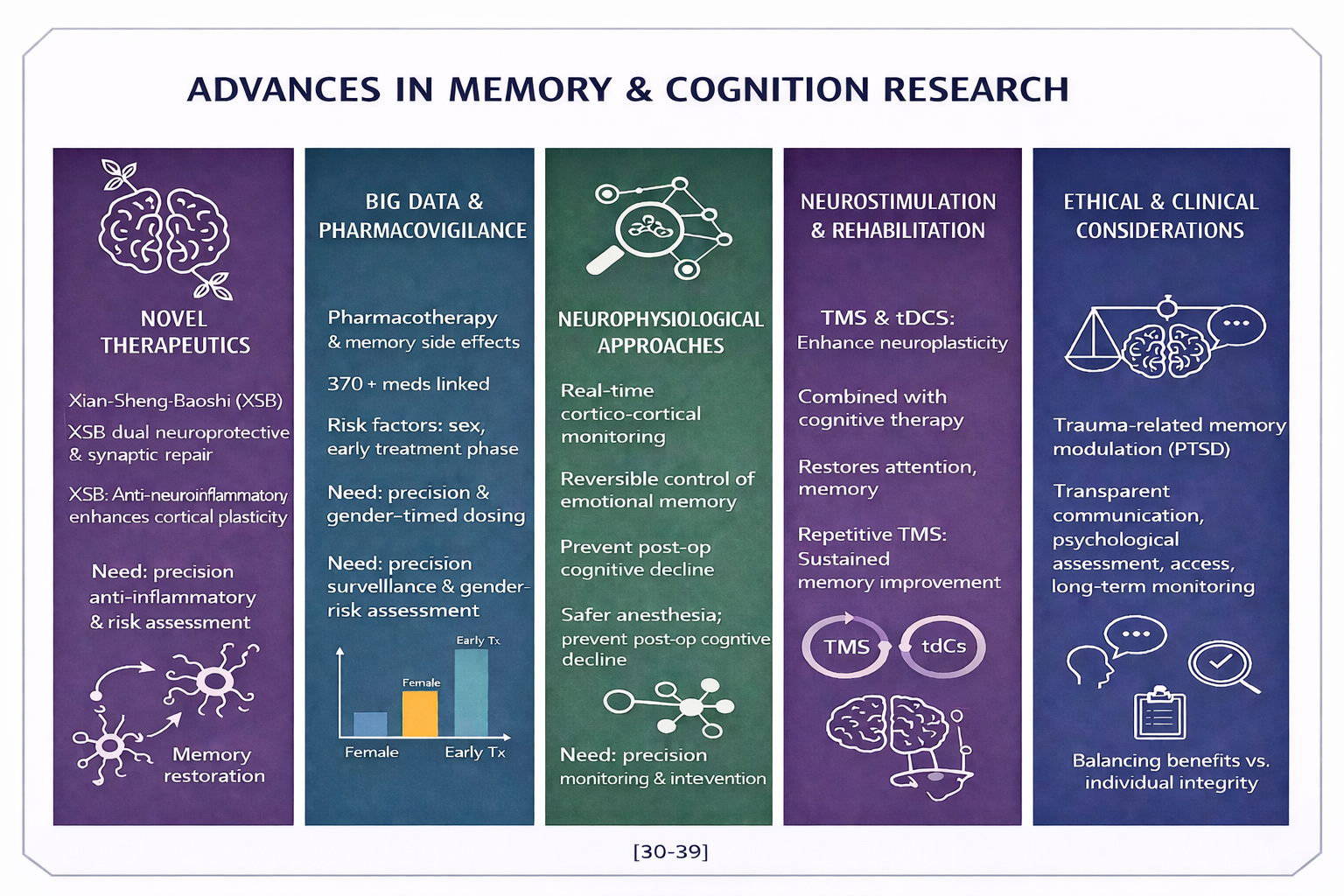
Xian-Sheng-Baoshi XSB) and GLII 73 represent emerging therapeutic compounds targeting cognitive impairment through dual neuroprotective and synaptic mechanisms. Preclinical data suggest that XSB reduces neuroinflammation by downregulating proinflammatory cytokines and oxidative stress pathways in hippocampal tissue, thereby supporting memory restoration in neurodegenerative models. GLII 73, a GABAA receptor subtype-selective modulator, enhances inhibitory-excitatory balance within cortical networks, improving plasticity and mnemonic performance. Together, these compounds illustrate the growing trend toward mechanism specific interventions aimed at reversing, rather than merely compensating for, memory dysfunction [30,31].
Large-scale pharmacovigilance analyses utilizing big data approaches have revealed extensive links between pharmacotherapy and memory-related adverse events. A study encompassing more than 370 commonly prescribed medications found that cognitive side effects are not limited to central nervous system drugs but also occur across cardiovascular, metabolic, and immunological agents. Statistical modeling identified female sex and early treatment phases as leading risk factors, potentially reflecting hormonal modulation of drug metabolism and neuroinflammatory sensitivity. These findings underscore the need for precision surveillance systems and gender-informed risk assessment in medication safety management[32,33].
Neurophysiological techniques such as EEG, optogenetics, and chemogenetics are transforming research into anesthesia-induced amnesia and broader mechanisms of memory alteration. EEG allows real-time monitoring of cortical oscillations linked to memory suppression and recovery, while optogenetic tools precisely modulate neuronal excitability to map amnestic circuits. Chemogenetic approaches, using designer receptors activated by custom ligands, enable reversible control of memory-related neuronal populations, shedding light on the role of hippocampal and thalamocortical pathways in anesthetic effects. Together, these methods provide mechanistic insights that could lead to safer anesthesia protocols and targeted prevention of postoperative cognitive dysfunction[34,35] .
Neurostimulation and rehabilitation strategies are emerging as effective interventions for reversing cognitive impairments induced by pharmacological agents. Techniques such as transcranial magnetic stimulation TMS) and transcranial direct current stimulation (tDCS) enhance neuroplasticity by modulating cortical excitability and synaptic strength. When combined with cognitive therapy, these approaches have shown additive benefits in restoring attention, working memory, and executive function. Clinical trials highlight that repetitive TMS, in particular, can induce sustained improvements in memory performance by reactivating dormant neural circuits, offering hope for patients with drug-related amnestic symptoms[36,37].
The therapeutic use of amnesia-inducing drugs in treating trauma-related conditions, such as PTSD, poses significant ethical challenges. While controlled memory modulation may alleviate distressing recollections and improve emotional regulation, it also raises concerns about patient autonomy, consent validity, and the alteration of personal identity. Ethical frameworks emphasize transparent communication, psychological assessment prior to intervention, and long-term monitoring of cognitive and emotional outcomes. The debate continues on balancing the potential benefits of memory suppression with respect for individual integrity and the moral limits of manipulating conscious experience [38,39].
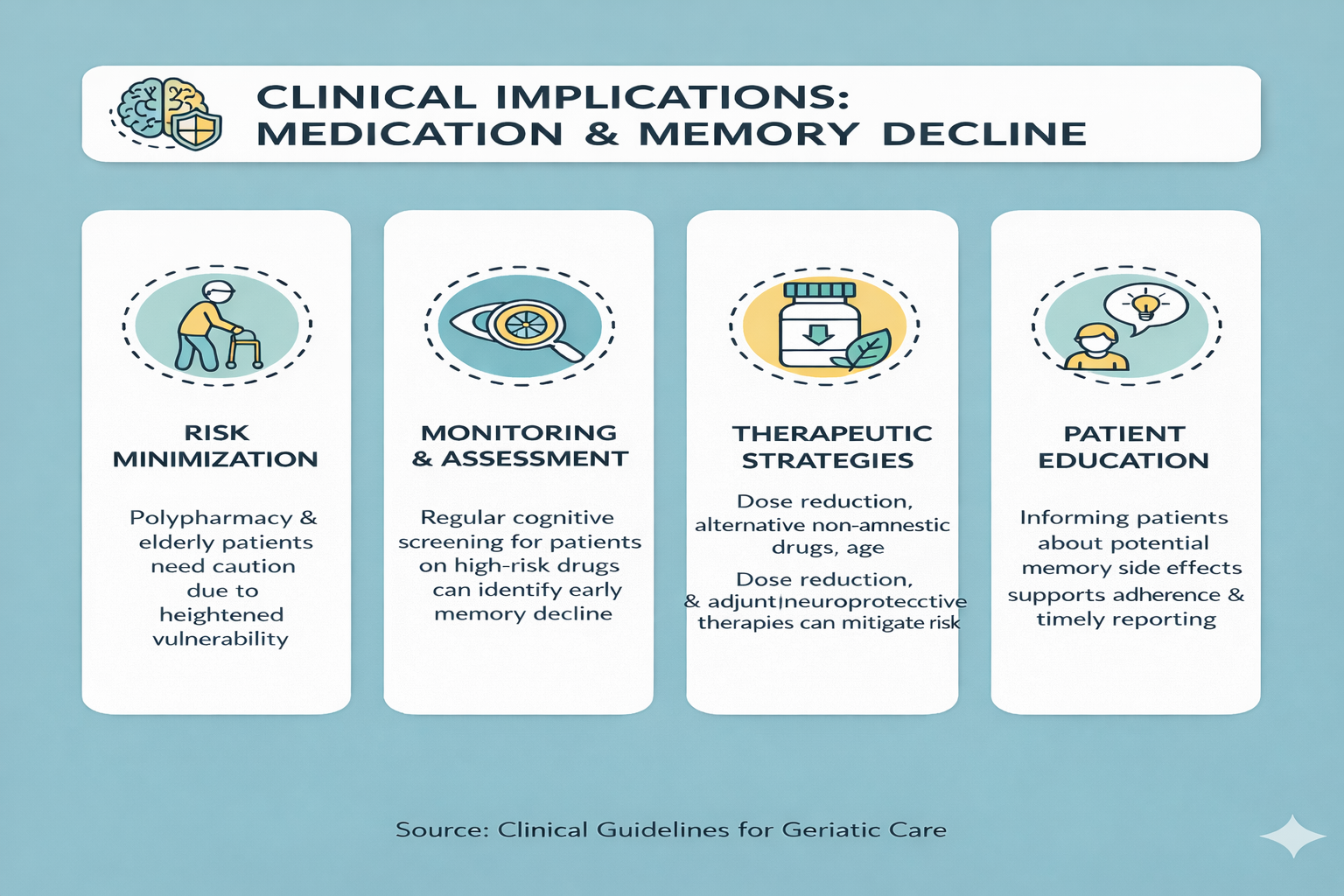
Clinical Implications:
CONCLUSION
Drug-induced amnesia encompasses a diverse spectrum of pharmacological agents that alter memory processes through multiple neural and biochemical mechanisms. These effects can arise from anesthetics, sedatives, antiepileptics, and other psychoactive substances that influence neurotransmitter systems, synaptic plasticity, and neural circuit function. By combining classical pharmacological understanding with emerging insights from molecular neuroscience, researchers can better delineate the pathways underlying drug-induced cognitive changes. Ongoing advancements in drug design, biomarker discovery, and neurotechnological tools such as functional imaging and neuromodulation provide novel opportunities to mitigate adverse memory effects. To achieve meaningful progress, continuous interdisciplinary research, vigilant clinical monitoring, and responsible therapeutic innovation will be crucial in preserving patient safety and sustaining cognitive well-being.
REFERENCES




Mitali Gajale, J.V. Vyas, Dr. Vivek Paithankar, Dr. Anjali Wankhade, Drug-Induced Amnesia: A Comprehensive Review of Medications Affecting Memory Function, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 822-832. https://doi.org/10.5281/zenodo.18923159
 10.5281/zenodo.18923159
10.5281/zenodo.18923159