
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmacy, Mahatma Jyotiba Phule Rohilkhand University, Bareilly, Uttar Pradesh, India-243006.
Helminthiasis is a significant public health problem affecting millions of people worldwide. This review article provides a comprehensive overview of helminthiasis, including its introduction, history, epidemiology, etiology, causes, transmission routes, specific parasites, life cycle of helminths, pathogenesis, pathophysiology, clinical manifestations, precautions from helminthiasis, diagnosis and treatment. Additionally, it discusses anthelmintic drugs and traditional drugs, highlighting their differences, advantages and disadvantages. The article also explores the mechanism of action, causes, symptoms, signs and side effects of anthelmintic drugs in the therapy of helminthiasis and highlights herbal remedies for worm infections, focusing on medicinal plants families, properties, bioactive compound, bioactive compound responsible for anthelmintic activity and plants parts used for determining anthelmintic activity and they exhibiting anthelmintic activity. The major problem with helminthiasis is its widespread prevalence and the limitations of current treatment options. Anthelmintic resistance and side effects are significant concerns, emphazing the need for new and effective treatments. Traditional plants have shown promise in treating helminthiasis, but further research is needed to validate their efficacy and safety. Helminthiasis control and treatment require a comprehensive approach combining anthelmintic drugs, traditional plants and public health educations. Future directions include developing new anthelmintic drugs, validating traditional plants, vaccine development and public health education. In conclusion, helminthiasis is a complex and multifaceted problem requiring a comprehensive approach to control and prevent. By combining anthelmintic drugs, traditional plants and public health education, we can effectively reduce the burden of disease and improve the lives of those affected. This review provides a valuable resource for researcher’s; healthcare professionals and policy makers seeking to understand and address helminthiasis.
The word "helminths" derived from the Greek meaning worm (1). The parasites that infect humans are classified as heirlooms or souvenirs. Parasites are inherited from African ancestors are termed heirlooms, whereas those acquired from the animals through our evolution, migrations, and agricultural practices are called souvenirs (2). In developing nations, helminthic infections are a major cause of human infections. Approximately 2 billion people worldwide, or more than a quarter of the global population, suffer from helminthic infections, which disproportionately affected children in developing countries. (3). Helminths are primarily classified into two major phyla: nematodes and platyhelminths. The nematode phylum, also known as roundworms, includes soil-transmitted helminths and filarial worms responsible for lymphatic filariasis and onchocerciasis (4). Other phyla platyhelminths also known as flatworms, which include flukes schistosomes and tapeworms such as the pork tapeworm that causes cysticercosis (5). Trematodes are commonly known as flukes, while cestodes are known as tapeworms (6). Soil-transmitted helminthiasis is caused by three main types of worms i.e. roundworm (Ascaris lumbricoides), whipworm (Trichuris trichiura), and hookworm (Ancylostoma duodenale and Necator americanus) (7). The soil-transmitted helminths infect humans through contact with contaminated soil that contains eggs of A. lumbricoides and T. trichiura. Some helminths such as hookworm larvae penetrate the skin directly (8). In tropical areas, helminthic infections tend to be neglected because of their insidious effects on human growth and development (9). Furthermore, the study of these diseases is severely underfunded, receiving less than 1% of the global research budget (10). STH infections are prevalent in impoverished areas marked by inadequate sanitation and overcrowding, similar to other parasitic infections. Generally, it's observed that Ascaris lumbricoides and Trichuris trichiura are more frequent in urban areas, whereas hookworms are more frequent in rural areas. Available data indicates a complex situation, with Ascaris lumbricoides and Trichuris trichiura prevalence showing variability between urban and rural communities, but lacking a clear pattern. Hookworms are equally common in both urban and rural areas. The reasons for these differences are unclear, but may be related to sanitation, population density and socio-economic factors. As the global population becomes increasingly urbanized, with over half of people living in cities by 2007, the transmissions of soil transmitted helminths infections may increase particularly in urban slums with inadequate water and sanitation (11).
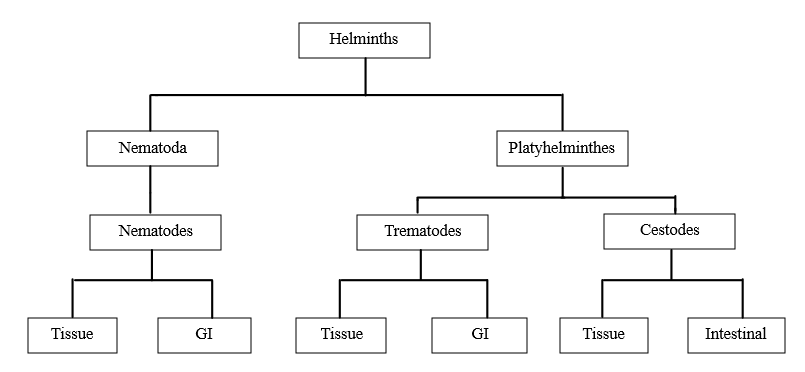
Figure no. 1 Classification of Helminths
Helminthiasis is a condition caused by infections from worms like tapeworms, pinworms, and round worms (12). These worms usually live in the gastrointestinal tract, but they can also enter in the liver and different organs of the body through burrowing (13). For example: Fasciolosis – It affects the liver, Paragonimiasis: It affects the lungs, Cysticercosis: It affects the muscles, Strongyloidiasis: It affects the skin, Filariasis: It affects the lymphatic system, River blindness: It affects the eyes (14). It is estimated that over a billion people are infected by helminths that are spread through soil, mostly in developing nations (15). Helminths infection is danger for global food security because they affect the health of animals. When animals are infected with these worms then they become sick due to which their productivity decreases such as milk production or growth rates. This can lead to a shortage of food supply for people who depend or rely on livestock for meat, milk and other products (16). 80 % of the world’s population in impoverished or poor nations uses plant materials as their major source of heath care according to traditional medicine. Traditional medicines involve the use of natural products such as plants, herbs and minerals to prevent and treat diseases (17). Traditional methods are used for removing parasitic worms from the body of human or animal include stunning or killing those using anthelmintic herbs (18). Helminthiasis is transmitted through the intake of contaminated food and water as well as mosquitoes and flies. Helminthiasis is more common in tropical and sub-tropical areas including sub-Saharan Africa, central and East Asia and the. Helminths infections are more common in men than female (19). Helminthiasis causes malnutrition, iron deficiency anemia, cognitive changes and increased susceptibility to tuberculosis, HIV and malaria (14). Schistosomiasis is the second most common parasitic disease of humans after malaria due to poor management practices. Anthelmintic works as vermifuges (stunning) or vermicides (killing). Common side effects of anthelmintic drugs include gastrointestinal issues stomach pain, nausea and vomiting, dizziness, spinning sensation, headache and temporary hair loss (14). Worms are classified into three main categories i.e. Nematoda, Cestoda and Trematoda. Antinematodal, Anticestodal and Antitrematodal medications are used for these three categories (17). Antinematoda drugs such as piperazine, mebendazole, thiabendazole, pyrantel, ivermectin and diethyl carbamazine are effective against nematoda worms like pinworm, hookworms, ascaris and strongyloides. Antitrematodal drugs such as praziquantel, bithional sulfoxide, oxamniquine, albendazole and metrifonate are used to treat trematoda worms. Anticestoda drugs like niclosamide, albendazole and praziquantel are effective against cestoda worms also known as tapeworms (20). Resistance development occurs when synthetic medicines are used repeatedly. Synthetic anthelmintics drugs are benzimidazoles, piperazine, diethylcarbamazine citrate, ivermectin, levamisole etc. (21). Some of the examples of benzimidazoles drugs are albendazole, mebendazole, fenbendazole, oxibendazole, flubendazole, thiabendazole, netobimin, febantel, thiophanate, albendazole sulfoxide etc. (22). Allopathic drugs are not sufficient for the treatment of helminthiasis of large population because many of these drugs developed resistance and are ineffective (23). It is essential to have scientific evidence to back up the uses of plant-based products in ayurveda for treating helminthiasis (24). The side effects are diarrhea, cough, difficulty breathing, fatigue, dizziness, weight loss, abdominal tenderness, rash or itching, fever, malaise (25). Worms lack the ability to store energy which means they need a constant intake of food to meet their metabolic needs. Anthelmintic medicines work by either making them starve by stopping them from getting food or by making them immobilizing which means unable to move (26). If there is any interruption in the feeding of helminths its leads to energy loss. Helminths can be eliminated by inhibiting their ability to feed for a period of 24 hours or even less. Helminths will die or perish if they are paralyzed for some time because they cannot maintain their position in the gut. To control the helminthic infections anthelminthic medications are used (27). Resistance development occurs when synthetic medicines are used repeatedly. Synthetic anthelmintics drugs are Benzimidazoles, Piperazine, Diethylcarbamazine citrate, Ivermectin, Levamisole etc. (28).
Benzimidazoles- Benzimidazoles involves disrupting the metabolism and microtubules of worms. Microtubules are essential for the structure and function of cells. When benzimidazoles inhibit these microtubules it affects the worm’s energy, production and nutrient absorption leading to their weakening and eventual death. It is used against roundworms, hookworms and tapeworms. Some of the examples of benzimidazoles are albendazole, mebendazole, fenbendazole, oxibendazole, flubendazole, thiabendazole, netobimin, febantel, thiophanate, albendazole sulfoxide etc.(29).
Piperazine- Piperazine works on the neuromuscular system of worms. It disrupts the muscles contractions of the worms leading to paralysis. It acts as neuromuscular blocking agent which prevents the worms from attaching to their host and allowing them to be expelled out from the body. Piperazine drug is effective against roundworms such as Ascaris lumbicoides and Enterobius vermicularis (pinworms) (29).
Diethylcarbamazine citrate - It is used to treat parasitic infections such as filariasis. It works by affecting the nervous system of the parasites. It immobilizes microfilariae (larval stage of filarial worms) which helps to separate them from the host tissues and allowing the body to eliminate them. It also stimulates body defense system response boosting the body's natural defenses against parasites. It is effective against filarial worms such as Wuchereia bancrofti, Brugia malayi. It is useful in treating lymphatic filariasis (28).
Ivermectin – It is used to treat parasitic infections. Ivermectin works by affecting the nervous system of parasites. It targets the muscle and nerve cells of parasitics organisms leading to paralysis and death. It binds to chloride channels which helps in killing the parasites. Ivermectin is used for several types of infections including – Strongyloidiasis (a type of intestinal infection) Onchocerciasis (river blindness) Scabies (a skin infection) Lice (parasites found in hair) (30).
Levamisole – It is used to treat parasitic infections. Levamisole works by affecting the nervous system of the parasites. It alters muscle contraction, leading to paralysis and death of the parasites. Its action targets the muscle cells of the parasites, inhibiting their movement. Levamisole is mainly used to treat roundworm infections such as ascariasis and hookworm infections. It is used to boost the immune system. The common side effects of anthelmintic drugs are - anorexia, nausea, vomiting, disorientation, diarrhea, weight loss, sporadic fever and rashes (31).
History of Helminthiasis
Helminthiasis has been described in ancient texts such as the Ebers Papyrus (1550 B.C.) and the Hippocratic Corpus (400 B.C.) The Ebers Papyrus describes various intestinal parasites including tapeworms and roundworms. Hippocrates and Galen described various parasitic diseases including helminthiasis. Avicenna’s (980-1037 A.D.) described various parasitic diseases including helminthiasis in his book “The Cannon of Medicine”. Aristotle, a renowned Greek philosopher and scientist (384-322 BC) who made significant contributions to the field of biology. He described various types of parasites including tapeworms and roundworms. Rhazes (865-923 A.D.) described various parasitic diseases including helminthiasis in his book “Kitab al-Hawi fi al-Tibbs”. Francesco Redi (1626-1697 A.D.) discovered the lifecycle of parasites including helminths and demonstrated that they are transmitted through the contaminated food and water. Antonie Van Leeuwenhoek discovered microorganism is including bacteria and protozoa and described their role in diseases (32).
Overview of Helminthiasis
These worm-like parasites are known as helminths or parasitic worms. They are classified based on the morphological features of their life stages, including eggs, larvae, and adults, examining both external and internal structures. Some helminths, like tapeworms and most flukes (except blood flukes), are hermaphroditic. Some helminths, like roundworms, are bisexual species, and nematodes are one of the most prevalent groups globally (33). There are three major types of helminths (parasitic worms): trematodes (flukes), cestodes (tapeworms) and nematodes (roundworms). Understanding the epidemiology of helminths is crucial because they are widespread in many regions including: Tropical and sub-tropical regions including Sub- Saharan Africa, South America, Central America, Middle East, Caribbean and Asia. In 1998, World Health Organization (WHO) launched the Global Atlas of Helminth Infectious (GAHI) to collect and analyze the data on helminth infections worldwide. The GAHI website (link unavailable) provides a database and graphical tool for researchers and policymakers (34). The GAHI collected data between 1998 and 2010 on the prevalence of helminth infections. However, some countries were excluded due to: limited socioeconomic data, comprehensive control measures (e.g. Mauritius, Maldives and Mayotte) (35). For 7 countries in Oceania with no available data, the GAHI assigned mean prevalence rates based on regional data. Similarly, eight countries with no data were assigned mean prevalence rates based on data from neighboring countries with similar ecological and epidemiological conditions. Examples of these neighboring countries include: Iran ( for Georgia, Iraq and Turkmenistan), Morocco (for Algeria), Turkey (for Syria and Tunisia), China and Indonesia (36).
Table no.1 shows the global distribution and prevalence of various helminths
|
Helminthiasis |
Region |
|
Ascariasis Lumbricoides (roundworm) |
Asia, Africa and Latin America |
|
Trichuris trichiura (whipworm) |
Asia, Africa and Latin America |
|
Hookworm (Necator americanus, Ancylostoma duodenale) |
Asia, Africa and Latin America |
|
Strongyloidiasis stercoralis (threadworm) |
Asia, Africa and Latin America |
|
LF Wuchereia bancrofti, Brugia malayi |
India, Southeast Asia and Sub- Saharan Africa |
|
Onchocerciasis (River blindness) Onchocerca volvulus |
Sub- Saharan Africa |
|
Loiasis Loa |
Sub- Saharan Africa |
|
Dracunculiasis (guinea worm) Dracunculus medinensis |
Sub- Saharan Africa |
|
Schistosomiasis (Schistosoma haematobium) |
Sub- Saharan Africa |
|
Schistosoma mansoni |
Sub- Saharan Africa and Eastern Brazil |
|
Schistosoma japonicum (blood flukes) |
China and Southeast Asia |
|
Clonorchis sinensis (liver fluke) |
Developing region of East Asia |
|
Opisthorchis viverrine (liver fluke) |
Developing region of East Asia |
|
Paragonimus supp. (lung flukes) |
Developing region of East Asia |
|
Fasciolopsis buski (intestinal fluke) |
Developing region of East Asia |
|
Fasciola hepatica (intestinal fluke) |
Developing region of East Asia |
|
Cysticercosis Taenia sodium (pork tapeworm) |
Sub- Saharan Africa |
Methods for Studying Anthelmintic Activity
In vitro studies using earthworms: Most studies assess anthelmintic properties of plants, oils or extracts based on their toxicity to earthworms. Compounds toxic to earthworms often causes initial irritability or agitation, leading to worm death. Anthelmintics may sometimes eject parasites due to this effect if their concentration is not sufficient to kill the worm. Researchers have also used hookworms (H. contortus), tapeworms, and A. lumbricoides to assess in-vitro anthelmintic activity of plant materials (37). Modified egg hatch assay: This assay is frequently used to assess the effectiveness of plant items against eggs of Haemonchus contortus or other trichostrongylids. Larval development assay and larval motility assay: Modified versions of these assays are used to test parasite resistance to anthelmintics and have been employed in in-vitro studies (38). Novel technology using housefly worms: Researchers have produced worms from houseflies that mimic parasitic pinworms found in humans under laboratory settings. These housefly worms and earthworms, have been used to examined the anthelmintic effects of several medications (39). The effectiveness of several plant materials as anthelmintics has additionally been tested in vivo. The criteria for this kind of activities included the evacuation of worms from their hosts or a decrease in the quantity of eggs per gram of faeces (EPG) that the hosts with the infection passed when compared to animals that had been given commercial anthelmintics. As an example, oral feeding of Indonesian papaya (Carica papaya) decreased parasite burden up to 100% within seven days after treatment for pigs who were infected with Ascaris suum. Similar to this, various other plant extracts discovered with anthelmintic qualities were examined for their effectiveness against gastrointestinal nematodes in experimentally infected sheep (38).
Plants studied for anthelmintic activity
Adhatoda vasica (Family- Acanthaceae) – Showed anthelmintic activity against Haemonchus contortus in-vitro and in-vivo (40). Aerva lanata Linn Juss (Family- Amaranthaceae) – Methanolic and aqueous extracts showed anthelmintic activity against Pheritima postuma (41). Annona squamosa (Family- Annonaceae) -Leaf extracts showed significant anthelmintic activity (42). Azadirachta indica (Family- Meliaceae)- Neem leaf extracts showed effectiveness against Fasciola (43). Butea monosperma Lam. (Family-Fabaceae) – Seeds showed anthelmintic activity against ascarids, with palasonin being the active compound (44). Buchholzia coriaceae (Family- Capparidacea) – Leaves and stems showed concentration dependent anthelmintic activity (45). Calotropis procera (Family-Apocynaceae) – Floral extracts showed time dependent anthelmintic efficacy against Haemonchus contortus (46). Cassia tora L. (Family- Fabaceae) – Ethyl acetate fraction showed potent anthelmintic activity (47). Coriandrum sativum (Family- Umbellifers) – Aqueous and hydroalcoholic extracts showed anthelmintic activity against Haemonchus contortus (48). Cucurbita maxima Duch. (Family-Cucurbitaceae) – Seeds extracts showed anthelmintic activity particularly against tapeworms (49). Capparis decidua Edgew. (Family-Capparidaceae) – Ethanolic extract of root bark showed dosed dependent anthelmintic activity (50). Carica papaya Linn. (Family-Caricaceae) – Seed extract showed anthelmintic activity with benzyl isothiocyanate as the active compound (51). Erythrina indica Lam (Family- Papilionaceae) – Leaf extracts showed anthelmintic activity due to tannins (52). Eucalyptus globulus (Family Myrtaceae) – Oil showed anthelmintic potential due to phytoconstituents like borneol and cineol (53). Evolvulus alsinoides Linn. (Family-Convolvulaceae) – Ethanolic extract showed anthelmintic activity against earthworms (54). Gynandropsis gynandra (Family- Capparidacea)- Methanol extract of leaves showed toxicity to earthworms (45). Hugonia mystax (Family- Linaceae) – Extracts showed anthelmintic activity, validating its traditional use (55). Juglans regia L. (Juglandaceae) – Extracts showed anthelmintic activity due to flavonoids and polyphenolic compounds (56). The tannins known as chemically polyphenolic substances have anthelmintic action (57). Melia azedarach Linn. (Family-Meliaceae) – Ethanol extract of drupes showed anthelmintic activity against tapeworms and earthworms (58). Mussaenda frondose (Family- Rubiaceae) – Plant extract showed anthelmintic activity (59). Murraya koenigii (Family- Rutaceae)- Aqueous and methanolic extracts showed dose dependent anthelmintic activity (60). Neolamarckia cadamba (Family- Rubiaceae)- Fruit extract showed anthelmintic activity due to terpenoids and phenolic compounds (61). Nyctanthes arbor-tristis (Family- Oleaceae)- Extracts showed anthelmintic activity, making it potential alternative to commercial anthelmintics (62). Punica granatum Linn. (Family-Punicaceae)- Stem bark extract showed anthelmintic activity and prevent egg development (63). Psidium guajava (Family- Myrtaceae)- Leaf extract showed strong anthelmintic activity at high concentrations (64). Spigelia anthelmia Linn (Family- Loganiaceae)- Aqueous extract showed anthelmintic activity (65). Additionally, rats exposed to the extract have considerable anthelmintic activity towards Nippostrongylus braziliensis (66). Spermacoce articularis L.f. (Family- Rubiaceae)- Methanolic extract showed dose- dependent anthelmintic activity (67). Trachyspermum ammi Linn. (Family-Apiaceae) – (68) Seed extracts showed ovicidal effectiveness against Haemonchus contortus eggs (69). Trifolium repens Linn. (Family-Fabaceae)- Aerial shoot extract showed anticestodal effectiveness against Hymenolepis diminuta (70). Trigonella foenum-graecum (Family- Leguminosae)- Aqueous and alcoholic extracts showed anthelmintic activity against earthworms (71). Zingiber officinale (Family- Zingiberaceae)- Ginger powder showed anthelmintic activity, reducing parasite loads in pigs (72).

Epidemiology
Helminthiasis poses a major public health challenge in developing countries, where it spreads common parasitic infections. More than 1 million people are affected with helminthiasis, which is comparable to the number of people affected by AIDS and malaria. The infections are caused by various parasitic worms such as Ascaris lumbicoides, Trichuris trichiura, Necator americanus, Ancylostoma duodenale, Schistosomes and filarial worms. Helminthiasis has severe consequences including morbidity, malnutrition, decreased productivity and low socioeconomic status. Helminths have immunomodulatory effects on host immune cells, which can affect the severity of their infections. In endemic areas, helminthiasis is recognized as a contributing factor to the development of AIDS, malaria and tuberculosis. Helminthiasis can also induce severe hypersensitivity reactions, leading to chronic allergic reactions known as anaphylaxis (73). In the United States, about 1.3 to 2.8 million people have serological evidence of Toxocara species infection. 4 million people are infected with soil transmitted helminths. Between 41,400 and 169,000 people are infected with cysticercosis. Approximately 8,000 people are infected with schistosomiasis diseases. A recent study in Chicago found that 12% of immigrant participants had evidence of current or prior infection with a pathogenic parasite species. The most common infections were Toxocara (6.4%) and s. stercorali s(4%). The affected rate was 8% for females and 7% for males (74). Globally prevalence of parasitic infections has decreased between 1990 and 2016. Ascariasis prevalence decreased from 1.089 billion to 799.68 million. Trichuriasis prevalence has decreased from 524.62 million to 435.09 million. Hookworm incidence decreased from 497.52 million to 450.68 million. Schistosomiasis distribution decreased from 195.36 million to 189.77 million. Lymphatic filariasis incidence decreased from 44.14 million to 29.38 million. Onchocerciasis incidence decreased from 24.13 million to 14.65 million (35). About 2 billion people are infected with soil transmitted helminthiasis worldwide (9). Helminthiasis is a significant public health concern worldwide, particularly in tropical and subtropical regions: Global distribution: Helminthiasis is found in all regions of the world, but it is more common in tropical and subtropical regions. High prevalence in rural areas: Helminthiasis is more common in rural areas where sanitation and hygiene are poor. High prevalence in children and pregnant women: Children and pregnant women are at higher risk of helminthiasis due to their weaker immune systems. Seasonal variation: Helminthiasis can vary seasonally, with higher transmission rates during the rainy season (75).
Etiology
Intestinal parasites often result in significant morbidity and mortality, especially among children. The key risks factors for helminthiasis are: Rural areas: limited availability to clean water, sanitation and healthcare. Low socioeconomic status: Poverty and poor living conditions, leading to inadequate nutrition and limited education. Poor sanitation: Contaminated water, inadequate waste disposal. Poor personal hygiene: Lack of handwashing and nail trimming (76).
Causes of helminthiasis
Contaminated food and water: Contaminated food and water are common sources of helminth infection. Food and water can become contaminated with helminth eggs or larvae through: Poor sanitation and hygiene practices, Inadequate waste disposal, Inadequate sewage treatment, Poor sanitation and hygiene: Poor sanitation and hygiene practices, such as: Not washing hands regularly, Not using proper waste disposal methods, Not treating sewage properly, Close contact: Close contact with an infected person can also spread helminthiasis. Vector-borne transmission: Some helminths, such as hookworms, can be transmitted through the bite of an infected vector, such as a mosquito, Inadequate waste disposal: Inadequate waste disposal can contribute to the spread of helminthic infection, Lack of proper sewage treatment: Lack of proper sewage treatment can contribute to the spread of helminthic infection, Walking barefoot: Walking barefoot in areas where the soil is contaminated with helminth eggs or larvae can lead to infection, Ingesting contaminated soil: Ingesting contaminated soil can lead to helminthic infection, Consuming undercooked or raw meat: Consuming undercooked or raw meat can lead to helminthic infection, Drinking contaminated water: Drinking water contaminated with helminth eggs or larvae can lead to infection, Poor hand hygiene: Poor hand hygiene can contribute to the spread of helminthic infection, Inadequate cleaning of fruits and vegetables: Inadequate cleaning of fruits and vegetables can lead to helminthic infection, Contact with contaminated feces: Contact with contaminated feces can lead to helminthic infection, Inadequate disposal of human waste: Inadequate disposal of human waste can contribute to the spread of helminthic infection, Lack of proper food handling: Lack of proper food handling can lead to helminthic infection, Eating food from street vendors: Eating food from street vendors can raise the risk of helminthic infection, Drinking untreated water: Drinking untreated water can increase the risk of helminthic infection, Walking in areas with poor sanitation: Walking in areas with poor sanitation can raise the risk of helminthic infection, Contact with contaminated animals: Contact with contaminated animals can lead to helminthic infection, Inadequate use of protective gear: Inadequate use of protective gear, such as gloves and masks, can increase the risk of helminthic infection (77).
Transmission routes
Fecal oral route: Intake of contaminated food, water or soil containing parasite eggs or larvae. e.g. A. lumbricoides (roundworm) and T. trichiura (whipworm) (7). Skin penetration: Parasites penetrate the body via skin contact with contaminated soil or water. e.g. A. duodenale and N. americanus (hookworms) (78). Uncooked meat: Poorly cooked food meat from infected animals. e.g. Diphyllobothriasis (79). Percutaneous and oral: Parasites can enter the body through skin contact or ingestion of contaminated food or water. e.g. S. stercoralis. Contact with contaminated water: Schistosomiasis (80).
Specific parasites
There are several species of helminths that can infect humans are: Roundworms(Ascaris lumbricoides), Tapeworms (Taenia saginata), Hookworms(Ancylostoma duodenale) and Whipworms (Trichuris trichiura). Helminthic infections are classified into two main categories i.e. intestinal helminthiasis and extraintestinal helminthiasis : Intestinal helminthiasis- This type of infection occurs when worms live in intestine and affect the intestinal tract. Extraintestinal helminthiasis – This type of infection occurs when worms infect vital organs including liver, lungs, and brain. Nematodes (Roundworms) – These worms are round in shape and reside in the hosts intestine , lungs and other organs. Cestodes (Tapeworms) – These worms are long and thin in shape and reside in the hosts intestine. Trematodes (flukes) – These worms are flat in shape and reside in the hosts liver, lungs or other organs (77).
Life cycle of helminths
Infection – Helminths enter in the hosts body leading to the infection. This infection can cause due to intake of contaminated food, water, poor health, soil, vegetables and poor sanitation and hygiene. Larval stage – Helminths larvae develop in the host’s body. Larvae adapt the environment of its surroundings and develop its growth. Adult stage – Helminths larva develops in to mature into adults. Adult helminths absorb nutrients from its hosts body and develop its growth and reproduction Egg laying – Adult helminths lay eggs which are excreted from the hosts’ body. Eggs enter in the host body through contaminated food, water, vegetables and soil (73).
Helminthic disease pathogenesis
Direct injury caused by worms: Helminths can cause direct damage to the human body through various mechanism i.e. Blockage of internal organs: Large nematodes (e.g. Ascaris) or tapeworms (e.g. Taenia, Diphyllobothrium) can physically block the intestine, Pressure effects: Growing parasites can exert pressure on surrounding tissues, leading to pathological changes, Granuloma formation: Schistosome eggs can trigger granuloma formation, blocking blood flow through the liver, Cyst formation: Tapeworms cysts (Echinococcus multilocularis) can develop in vital organs like the brain, liver or lungs causing abnormal organ enlargement, organ metastasis and necrosis due to pressure exerted by the cysts (81).
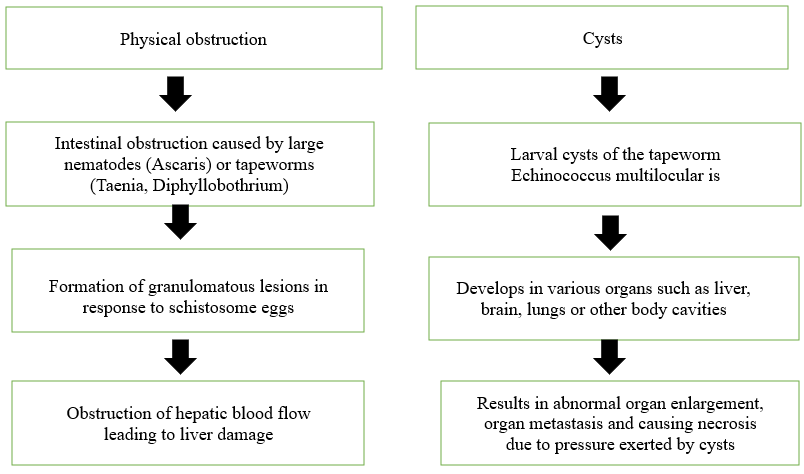
Figure no.3 Direct tissue damage due to helminth infestation
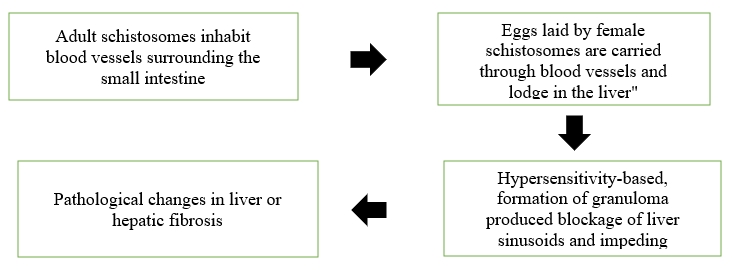
Figure no.4 Immune-mediated collateral damage
Indirect damage caused by host responses: Indirect damage is seen in the pathology related with schistosome infections, especially with Schistosoma mansoni (3). Hypersensitivity reactions: Hosts immune response to parasites (e.g. Schistosoma mansoni) lead to granuloma formation, blockage of liver sinusoids and impaired blood flow, which leads to changes in liver pathology. Figure 4. Indirect damage caused by immunopathologic response. Hypersensitivity based inflammatory changes probably also contribute to the lymphatic blockage related with filarial infections (81).
Pathophysiology
The pathophysiology of helminthiasis involves several mechanisms: Helminthiasis is a parasitic infection caused by helminths (worms). These worms reside in the hosts body and absorb nutrients, leading to adverse effects on the hosts health. The pathophysiological changes that occur in helminthiasis are malabsorption, immune response, hormonal imbalance, metabolic changes, cardiovascular changes, neurological changes (3). Helminth invasion: Helminths invade the human body through contaminated food, water, or soil, and migrate to their preferred site of infection, Immune response: The immune system responds to the helminth infection by producing antibodies and activating immune cells, Tissue damage: Helminths can cause tissue damage and inflammation, leading to a range of health problems, Nutrient uptake: Helminths can interfere with nutrient uptake, leading to malnutrition and other health problems, Hormonal imbalance: Helminths can cause hormonal imbalances, leading to a range of health problems, Malabsorption : Nutrient deficiency – Helminths absorb nutrients from the host leading to malabsorption, Weight loss – Malabsorption can cause weight loss, fatigue and weakness Impaired growth – Malabsorption can impair growth and development in children (82).
Inflammation: Cellular Damage – Helminths can cause cellular damage leading to tissue destruction, Organ dysfunction – Tissue damage can lead to organ dysfunction such as intestinal obstruction or liver damage, Fibrosis – Chronic tissue damage can lead to fibrosis and scarring, Immune response: Host defense – The host immune system responds to the presence of helminths, Inflammation – The immune response can lead to inflammation which can cause tissue damage, Antibody production – The immune system produces antibodies against the helminths, Hormonal imbalance: Endocrine disruption – Helminth’s can disrupt the hosts endocrine system leading to hormonal imbalances, Reproductive dysfunction – Hormonal imbalances can lead to reproductive dysfunction such as infertility or abortion, Metabolic changes – Hormonal imbalances can also lead to metabolic changes such as weight gain or loss, Metabolic changes: Metabolic rate – Helminth can alter the hosts metabolic rate leading to weight changes, Glucose metabolism – Helminths can disrupt glucose metabolism leading to diabetes or insulin resistance, Lipid metabolism – Helminths can also disrupt liquid metabolism leading to changes in cholesterol or triglyceride levels, Cardiovascular changes: Cardiac dysfunction – Helminths can cause cardiac dysfunction leading to heart failure or arrhythmias, Vascular changes – Helminths can cause vascular changes leading to hypertension or atherosclerosis, Thrombosis – Helminths can also cause thrombosis leading to stroke, Neurological changes: Neuroinflammation – Helminths can cause neuroinflammation leading to cognitive impairment or seizures, Neurodegeneration – Helminths can cause neurodegeneration leading to Parkinson disease or Alzheimer’s disease, Neuropsychiatric changes – Helminths can also cause neuropsychiatric changes leading to depression, anxiety or psychosis (83).
Clinical Manifestations
The clinical manifestations of helminthiasis determined depending on the type of helminth and the burden of the infection: Gastrointestinal symptoms: Helminthiasis can cause gastrointestinal symptoms like pain, diarrhea, and vomiting, Weight loss: Helminthiasis can cause weight loss due to malnutrition and decreased appetite, Fatigue: Helminthiasis can cause fatigue due to anemia and decreased energy levels, Skin lesions: Helminthiasis can cause skin lesions such as rashes and itching, Respiratory symptoms – Cough , wheezing, shortness of breath, Systemic symptoms – Fever , fatigue, weight loss, Neurological symptoms – Some helminths can cause neurological symptoms such as seizures and headaches, Allergic reactions – Certain individuals may experience allergic reactions to helminthic infection such as hives and itching (84).
Precautions from helminthiasis
Precautions from helminthiasis are personal hygiene, food and water safety, environment precautions.
Personal Hygiene -Wash hands regularly: Wash your hands regularly with soap and water especially before eating, after using the toilet, and after coming in contact with soil or feces. Keep fingernails clean: Keep fingernails clean and short to avoid dirt and parasites from accumulating. Shower or bathe regularly: Shower regularly to keep the body clean and prevent the spread of parasites. Food and Water Safety Drink safe water: Drink safe and clean water to prevent the intake of parasites, Eat cooked food: Eat cooked food to prevent the intake of parasites, Avoid raw or undercooked meat: Avoid eating raw or undercooked meat, especially pork, to prevent the intake of parasites. Environmental Precautions - Use proper waste disposal: Use of proper waste disposal methods to avoid the spread of parasites, Keep surroundings clean: Keep the surroundings clean and free of feces and urine, Avoid contact with contaminated soil: Avoid contact with contaminated soil to prevent the intake of parasites. Other Precautions - Wear protective clothing: Wear protective clothing, such as gloves, while handling soil or feces, Avoid sharing personal items: Prevent sharing personal items, such as towels or utensils, to avoid the spread of parasites, Get regular check-ups: Regular medical check-ups can help identify and manage helminthiasis in its early stages (85).
Diagnosis
The diagnosis of helminthiasis is based on:
Parasitological tests: Diagnosis of parasite microscopically.
Serological assays: Diagnosis of parasite specific antibodies in serum sample.
Antigen tests: Diagnosis of parasite biomarker
Molecular diagnosis: Diagnosis of parasite nuclei acid.
Stool examination: Stool examination is the most common method of diagnosing helminthiasis.
Blood tests: Blood tests can be used to diagnosis antibodies against helminths.
Imaging tests: Imaging tests such as X-rays and CT scans can be used to diagnosis helminth infections in the lungs or other organs.
Other specific tests: Diagnosis in the intermediate hosts (85).
Treatment
The treatment of helminthiasis depends on the type of helminth and the severity of the infection: Anthelmintic drugs: Anthelmintic medications such as albendazole and mebendazole are commonly used to treat helminthiasis (86). Surgical treatment: Surgical treatment may be necessary in some cases, such as when the helminth infection has caused intestinal obstruction. Supportive care: Supportive care such as rest, hydration (3).
Anthelmintic drugs and Traditional medicines
Herbal and allopathic medicine systems have been used for centuries to cure, treat and prevent diseases. Herbal Treatment is the oldest, traditional and most accessible medicine system especially for low-income communities. Combining herbal and allopathic medicines can lead to better treatment outcomes and faster recovery. Herbal medicines can help to reduce the side effects of allopathic medicines (87). Traditional medicines are chosen over anthelmintic drugs because of easy accessibility, affordability, cultural significance, perceived safety, local knowledge but traditional medicines have some disadvantages i.e. unproven efficacy, quality control, interactions and side effects, overuse and misuse (26). Anthelmintic drugs are not chosen because of high cost, more side effects, limited availability, resistance, lack of awareness but anthelmintic drugs have some advantages i.e. proven efficacy, target action, quick relief, wide availability (88).
Table no.2 Difference between Anthelmintic drugs and Traditional medicines (89)
|
S. No. |
Parameters |
Anthelmintic Drugs |
Traditional Drugs |
|
1. |
Origin |
Synthetic |
Natural |
|
2. |
Efficacy |
High |
Variable |
|
3. |
Quality control |
Strict |
Less |
|
4. |
Dosage |
Standard |
Variable |
|
5. |
Interactions |
May interact with other medicines |
Less interact with other medicines |
|
6. |
Availability |
Require prescription |
Available over the counter |
|
7. |
Integration |
Integrated with conventional medicine |
May not integrated with conventional medicine |
|
8. |
Side effects |
More side effects like nausea, vomiting |
Less side effects |
|
9. |
Cost |
Expensive |
Inexpensive |
|
10. |
Research |
Ongoing research and development |
Less research and development |
|
11. |
Environmental impact |
High environmental toxicity, waste generation |
Low environmental toxicity, biodegradable |
|
12. |
Patient adherence |
Easy to take, more side effects |
More frequent, less side effects |
Origin
Anthelmintic Drugs - These are man-made drugs which are developed through scientific research and technology.
Traditional Medicines - These are formed from the natural sources such as plants, herbs, and other natural products.
Efficacy
Anthelmintic Drugs - These drugs are highly effective against the parasites.
Traditional Medicines - These medicines have variable efficacy against the parasites.
Quality Control
Anthelmintic Drugs - These drugs undergo testing and inspection, to ensure their safety and efficacy.
Traditional Medicines - These medicines often have less stringent quality control measures, which can compromise their safety and efficacy.
Dosage
Anthelmintic Drugs - These drugs come in standardized dosages, making them easier to use. These drugs are given to patients according to their age, weight and body surface area
Traditional Medicines - These medicines often have variable dosages, making them more difficult to use In this the dose given to patients according to their health condition.
Interactions
Anthelmintic Drugs - These drugs can interact with other medications, making their use more complex.
Traditional Medicine - These medicines can interact with other herbs and supplements, making their use more complex.
Availability
Anthelmintic Drugs - These drugs are often available by prescription only, controlling their use.
Traditional Medicines - These medicines are often available over-the-counter (OTC), making them more accessible.
Integration
Anthelmintic Drugs - These drugs are often integrated with modern medicine, ensuring their safe use.
Traditional Medicines - These medicines are often integrated with traditional medicine, ensuring their safe use.
Side Effects
Anthelmintic Drugs - These drugs generally have more side effects, but it can be more severe. Common side effects are - Nausea Vomiting Diarrhea Stomach cramps Headache and fatigue and severe side effects are Liver damage, Kidney damage
Traditional Medicines These medicines can have less side effects, but can be less severe common side effects are Digestive issues (Such as nausea, vomiting and diarrhea) Interactions with other medications and serious side effects may involve liver and kidney damage.
Cost
Anthelmintic Drugs - These drugs are often more expensive, making them less accessible.
Traditional Medicines - These medicines are often less expensive, making them more accessible.
Research
Anthelmintic Drugs - These drugs are developed through scientific research and technology, ensuring their safety and efficacy.
Traditional Medicines - These medicines are developed through traditional knowledge and experience, which can be less reliable.
Environmental Impact
Anthelmintic Drugs - These drugs can have a high environmental impact, contributing to pollution.
Traditional Medicines - These medicines can have a lower environmental impact, being more biodegradable.
Patient Adherence
Anthelmintic Drugs: These drugs are easier to use, with fewer side effects, making patient adherence better.
Traditional Medicines: These medicines can be more difficult to use, with more side effects, making patient adherence poorer.
Why traditional medicines are chosen over the anthelmintic drugs?
Traditional medicines are chosen over the anthelmintic drugs because of easy accessibility, affordability, cultural significance, perceived safety, local knowledge.
Easy accessibility: Traditional medicine is often available and accessible as it is based on local plants and herbs.
Affordability: Traditional medicine is often cheaper than anthelmintic drugs, making it more affordable option for low-income communities. Cultural significance: Herbal medicine is often firmly established in cultural beliefs and practices making it preferred choice for many. Perceived safety: People often perceive traditional medicine as being safer and more natural with less side effects. Local knowledge: Local communities have more knowledge of traditional medicine(90). Anthelmintic drugs are not chosen because of high cost, more side effects, limited availability, resistance, lack of awareness. High cost: Anthelmintic drugs are expensive they are inaccessible to low-income communities. Side effects: Anthelmintic drugs have more side effects includes as diarrhea , nausea and allergic reactions than traditional medicine. Limited availability: Anthelmintic drugs are not available in local areas. Lack of awareness: Peoples may not be aware of the existence or benefits of anthelmintic drugs and they beliefs that traditional medicines are most effective and good than anthelmintic drugs. Resistance: The overuse of anthelmintic drugs can lead to resistance in worms making them less effective (91). But traditional medicine has some disadvantage’s also i.e. unproven efficacy, quality control, interactions and side effects, overuse and misuse. Unproven efficacy: The effectiveness of traditional medicines is often not scientifically proven. Quality control: The quality control of traditional medicines can be poor. Interactions and side effects: The interactions and side effects of traditional medicines are often unknown. Overuse and misuse: Traditional medicines can be overused or misuse
Advantages of Anthelminthic Drugs
Proven efficacy: The effectiveness of anthelminthic drugs is scientifically proven.
Targeted action: Anthelminthic drugs target specific parasites.
Quick relief: Anthelminthic drugs can provide quick relief.
Wide availability: Anthelminthic drugs are widely available (92).
Therapy for helminthiasis
Anthelmintic drugs are medicines that help remove worms from the body. They work in two ways i.e. locally expelling worms from the gut and systemically killing worms that have invaded organs and tissues. These anthelmintic drugs work by paralyzing worms i.e. making them unable to move or function, damaging worm skin i.e. causing the worms outer layer to break down, leading to its death and disrupting worm metabolism i.e. interfering with the worm’s internal processes, making it hard for them to survive. Different worms have different needs, so a drug that works for one type of worm may not work for another. This is why we have various anthelmintic drugs, each targeting specific worms. Detail about anthelmintic drugs is given below in the tables (93).
Table no.3 Drugs name with their class
|
S. No. |
Class |
Drug Name |
|
1. |
Benzimidazoles |
Albendazole Mebendazole Thiabendazole Fenbendazole Oxfendazole Albendazole sulfoxide Mebendazole sulfoxide |
|
2. |
Imidazothiazoles |
Levamisole Levamisole hydrochloride |
|
3. |
Tetrahydro pyrimidines |
Pyrantel Pyrantel pamoate |
|
4. |
Macrocyclic lactones |
Ivermectin Ivermectin sulfoxide |
|
5. |
Isoquinolines |
Praziquantel Praziquantel sulfoxide |
|
6. |
Salicylanilides |
Niclosamide Niclosamide sulfoxide |
|
7. |
Piperazine |
Diethylcarbamazine Diethylcarbamazine citrate |
|
8. |
Quinolines |
Quinfamide |
|
9. |
Halogenated hydrocarbons |
Tetrachloroethylene |
|
10. |
Aminoglycosides |
Paromomycin |
|
11. |
Thiazolides |
Nitazoxanide |
|
12. |
Oxamniquine |
Oxamniquine Oxamniquine hydrochloride |
|
13. |
Metrifonate |
Metrifonate Metrifonate hydrochloride |
|
14. |
Hydrochlorides |
Thiabendazole hydrochloride Fenbendazole hydrochloride Oxfendazole hydrochloride |
Table no.4 MOA of Anthelminthic drugs
|
S. No. |
Anthelminthic Drugs |
Mechanism of Action (MOA) |
|
1. |
Albendazole |
Inhibits microtubule polymerization disrupts parasite’s cellular structure and function ultimately leading to parasites death |
|
2. |
Mebendazole |
Interferes with microtubule function, inhibits parasite's glucose uptake, transport, aur metabolism, ultimately leading to parasite death |
|
3. |
Pyrantel |
Acts as a nicotinic acetylcholine receptor agonist, causing parasite muscle contraction aur paralysis, ultimately leading to parasite expulsion |
|
4. |
Ivermectin |
Acts as a glutamate-gated chloride channel agonist, causing parasite muscle contraction aur paralysis, ultimately leading to parasite death |
|
5. |
Praziquantel |
Acts as a calcium channel agonist, causing parasite muscle contraction aur paralysis, ultimately leading to parasite death |
|
6. |
Niclosamide |
Uncouples oxidative phosphorylation, inhibiting parasite's energy production aur ultimately leading to parasite death |
|
7. |
Levamisole |
le: Acts as a nicotinic acetylcholine receptor agonist, causing parasite muscle contraction aur paralysis, ultimately leading to parasite expulsion |
|
8. |
Diethylcarbamazine |
Interferes with microfilarial function, inhibiting parasite's reproduction aur ultimately leading to parasite death |
|
9. |
Oxamniquine |
Inhibits succinate dehydrogenase, inhibiting parasite's energy production aur ultimately leading to parasite death |
|
10. |
Metrifonate |
Inhibits cholinesterase, causing parasite muscle contraction aur paralysis, ultimately leading to parasite death |
|
11. |
Thiabendazole |
Interferes with microtubule function, inhibiting parasite's glucose uptake, transport, aur metabolism, ultimately leading to parasite death |
|
12. |
Fenbendazole |
Fenbendazole: Interferes with microtubule function, inhibiting parasite's glucose uptake, transport, aur metabolism, ultimately leading to parasite death |
|
13. |
Oxfendazole |
Oxfendazole: Interferes with microtubule function, inhibiting parasite's glucose uptake, transport, aur metabolism, ultimately leading to parasite death |
|
14. |
Albendazole sulfoxide |
Inhibits microtubule polymerization, disrupts parasite's cellular structure aur function, ultimately leading to parasite death |
|
15. |
Mebendazole sulfoxide: |
Interferes with microtubule function, inhibiting parasite's glucose uptake, transport, aur metabolism, ultimately leading to parasite death |
|
16. |
Pyrantel pamoate |
Acts as a nicotinic acetylcholine receptor agonist, causing parasite muscle contraction aur paralysis, ultimately leading to parasite expulsion |
|
17. |
Ivermectin sulfoxide |
Ivermectin sulfoxide: Acts as a glutamate-gated chloride channel agonist, causing parasite muscle contraction aur paralysis, ultimately leading to parasite death |
|
18. |
Praziquantel sulfoxide |
Acts as a calcium channel agonist, causing parasite muscle contraction aur paralysis, ultimately leading to parasite death |
|
19. |
Niclosamide sulfoxide: |
Uncouples oxidative phosphorylation, inhibiting parasite's energy production aur ultimately leading to parasite death |
|
20. |
Levamisole hydrochloride |
Acts as a nicotinic acetylcholine receptor agonist, causing parasite muscle contraction aur paralysis, ultimately leading to parasite expulsion |
|
21. |
Diethylcarbamazine citrate |
Interferes with microfilarial function, inhibiting parasite's reproduction aur ultimately leading to parasite death |
|
22. |
Oxamniquine hydrochloride |
Inhibits succinate dehydrogenase, inhibiting parasite's energy production aur ultimately leading to parasite death |
|
23. |
Metrifonate hydrochloride |
Inhibits cholinesterase, causing parasite muscle contraction aur paralysis, ultimately leading to parasite death. |
|
24. |
Thiabendazole hydrochloride |
Interferes with microtubule function, inhibiting parasite's glucose uptake, transport, aur metabolism, ultimately leading to parasite death |
|
25. |
Fenbendazole hydrochloride |
Interferes with microtubule function, inhibiting parasite's glucose uptake, transport, aur metabolism, ultimately leading to parasite death |
|
26. |
Oxfendazole hydrochloride |
Interferes with microtubule function, inhibiting parasite's glucose uptake, transport, aur metabolism, ultimately leading to parasite death |
Table no.5 Anthelmintic drugs with causes and symptoms
|
S. No. |
Anthelmintic Drugs |
Causes |
Symptoms |
|
1. |
Albendazole |
Hookworm, round worm, tapeworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
2. |
Mebendazole |
Hookworm, round worm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
3. |
Mebendazole sulfoxide |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
4. |
Oxfendazole |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
5. |
Albendazole sulfoxide |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
6. |
Thiabendazole |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
7. |
Fenbendazole |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
8. |
Ivermectin |
Strongyloidiasis, onchocerciasis |
Diarrhea, abdominal pain, skin rashes, itching, swelling |
|
9. |
Metrifonate |
Schistosomiasis |
Diarrhea, abdominal pain, blood in stool |
|
10. |
Pyrantel |
Hookworm, roundworm |
Diarrhea, abdominal pain, weight loss |
|
11. |
Pyrantel pamoate |
Hookworm, roundworm |
Diarrhea, abdominal pain, weight loss |
|
12. |
Praziquantel sulfoxide |
Schistosomiasis, tapeworm |
Diarrhea, abdominal pain, blood in stool |
|
13. |
Ivermectin sulfoxide |
Strongyloidiasis, Onchocerciasis |
Diarrhea, abdominal pain, skin rash |
|
14. |
Niclosamide sulfoxide |
Tapeworm |
Diarrhea, abdominal pain, weight loss |
|
15. |
Levamisole hydrochloride |
Hookworm, roundworm |
Diarrhea, abdominal pain, weight loss |
|
16. |
Diethylcarbamazine citrate |
Filariasis |
Swelling of legs, genital areas |
|
17. |
Oxamniquine hydrochloride |
Schistosomiasis |
Diarrhea, abdominal pain, blood in stool |
|
18. |
Metrifonate hydrochloride |
Schistosomiasis |
Diarrhea, abdominal pain, blood in stool |
|
19. |
Thiabendazole hydrochloride |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
20. |
Fenbendazole hydrochloride |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
21. |
Praziquantel |
Schistosomiasis, tapeworm |
Diarrhea, abdominal pain, blood in stool |
|
22. |
Niclosamide |
Tapeworm |
Diarrhea, abdominal pain, weight loss |
|
23. |
Levamisole |
Hookworm, roundworm |
Diarrhea, abdominal pain, weight loss |
|
24. |
Diethylcarbamazine |
Filariasis |
Swelling of legs, genital areas |
|
25. |
Oxamniquine |
Schistosomiasis |
Diarrhea, abdominal pain, blood in stool |
|
26. |
Albendazole oxime |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
q27. |
Mebendazole oxime |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
28. |
Pyrantel tartrate |
Hookworm, roundworm |
Diarrhea, abdominal pain, weight loss |
|
29. |
Nitazoxanide |
Helminthic infection, cryptosporidiosis, giardiasis |
Diarrhea, abdominal pain, weight loss |
|
30. |
Paromomycin |
Helminthic infection, cryptosporidiosis, giardiasis |
Diarrhea, abdominal pain, weight loss |
|
31. |
Quinfamide |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
|
32. |
Tetrachloroethylene |
Hookworm, roundworm, whipworm |
Diarrhea, abdominal pain, weight loss |
Table no.6 Anthelmintic drugs with sign and side effects
|
S. No. |
Anthelmintic Drugs |
Signs |
Side Effects |
|
1. |
Albendazole |
Eggs in stool, blood in stool |
Headache, nausea, vomiting, allergic reactions |
|
2. |
Mebendazole |
Eggs in stool, blood in stool, anemia, malnutrition |
Headache, nausea, vomiting, allergic reactions |
|
3. |
Mebendazole sulfoxide |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
4. |
Oxfendazole |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
5. |
Albendazole sulfoxide |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
6. |
Thiabendazole |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
7. |
Fenbendazole |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
8. |
Ivermectin |
Eggs in stool, skin rash, fever |
Headache, nausea, vomiting, allergic reactions |
|
9. |
Metrifonate |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
10. |
Pyrantel |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
11. |
Pyrantel pamoate |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
12. |
Praziquantel sulfoxide |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
13. |
Ivermectin sulfoxide |
Eggs in stool, skin lesions |
Headache, nausea, vomiting |
|
14. |
Niclosamide sulfoxide |
Eggs in stool, segments of tapeworm in stool |
Headache, nausea, vomiting |
|
15. |
Levamisole hydrochloride |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
16. |
Diethylcarbamazine citrate |
Microfilariae in blood |
Headache, nausea, vomiting |
|
17. |
Oxamniquine hydrochloride |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
18. |
Metrifonate hydrochloride |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
19. |
Thiabendazole hydrochloride |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
20. |
Fenbendazole hydrochloride |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
21. |
Praziquantel |
Eggs in stool, blood in stool |
Headache, nausea, vomiting, allergic reactions |
|
22. |
Niclosamide |
Eggs in stool, segments of tapeworm in stool |
Headache, nausea, vomiting |
|
23, |
Levamisole |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
24. |
Diethylcarbamazine |
Microfilariae in blood |
Headache, nausea, vomiting |
|
25. |
Oxamniquine |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
26. |
Albendazole oxime |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
27. |
Mebendazole oxime |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
28. |
Pyrantel tartrate |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
29. |
Nitazoxanide |
Abnormal stool |
Headache, nausea, vomiting |
|
30. |
Paromomycin |
Nephrotoxicity, ototoxicity |
Headache, nausea, vomiting |
|
31. |
Quinfamide |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
|
32. |
Tetrachloroethylene |
Eggs in stool, blood in stool |
Headache, nausea, vomiting |
Herbal remedies for worm infection
Most existing anthelmintic drugs have side effects like abdominal pain, loss of appetite, nausea, vomiting, headache and diarrhea. Even well tolerated drugs like mebendazole can cause gastrointestinal side effects and dizziness in some patients. Prolonged use can lead to more side effects like headache, fever, alopecia, jaundice and neutropenia (94). To eliminate the harmful side effects of synthetic anthelmintic drugs, it’s essential to promote the study of traditionally used anthelmintic plants. This can lead to the development of new anthelmintic substances with ease of availability and lesser side effects (95). According to the World Health Organization (WHO), 80% of the world’s population relies on traditional medicines, mostly plant-based, for primary healthcare needs. Even modern pharmacopeia contains at least 25% of plant derived drugs(15). Phytoconstituents in plants help fight against worm’s infections naturally and fewer side effects. Phytoconstituents like alkaloids, saponins, polyphenols and tannins are present in the plants exhibit anthelmintic properties and play a significant role in combating helminthiasis. Alkaloids block worms’ glucose uptake, affecting their production. Saponins damage worm outer layer, causing their death. Polyphenols and tannins interfere with worm energy generation, leading to their death (96).
Table no. 7 Medicinal plants with family and properties
|
S. No. |
Medicinal Plants |
Family |
Properties |
|
1.
|
Sophora interrupta
|
Fabaceae
|
Abortifacient, antibacterial, anti-inflammatory, anti-spasmodic, diuretic, emetic, emollient, febrifuge, hypotensive, purgative, styptic, tonic, anthelmintic |
|
2.
|
Jussiaea hyssopifolia |
Onagraceae
|
Astringent, carminative, laxative, diuretic, anthelmintic |
|
3.
|
Cucurbita mexicana |
Cucurbitaceae
|
Anti-inflammatory, antioxidant, antimicrobial, anthelmintic |
|
4.
|
Nerium oleander
|
Apocynaceae
|
Anti-inflammatory, anthelmintic, antioxidant, antimicrobial |
|
5. |
Luffa cylindrica |
Cucurbitaceae |
Anti-inflammatory, anthelmintic, antioxidant, antimicrobial |
|
6.
|
Clitoria ternatea
|
Fabaceae
|
Anti-inflammatory, anthelmintic, antioxidant, antimicrobial |
|
7.
|
Guazuma ulmifolia |
Sterculiaceae
|
Anti-inflammatory, antioxidant, antimicrobial, anthelmintic |
|
8.
|
Thespesia lampas |
Malvaceae
|
Anti-inflammatory, antioxidant, antimicrobial, Anthelmintic |
|
9.
|
Madhuca indica
|
Sapotaceae
|
Anti-inflammatory, antioxidant, antimicrobial, anthelmintic |
|
10.
|
Leontis nepetiifolia |
Lamiaceae
|
Anti-inflammatory, antioxidant, antimicrobial, anthelmintic |
|
11.
|
Ficus benghalenis |
Moraceae
|
Anti-inflammatory, antioxidant, antimicrobial |
|
12.
|
Pongamia glabra
|
Fabaceae
|
Anti-inflammatory, anthelmintic antioxidant, antimicrobial |
|
13.
|
Ficus relgiosa
|
Moraceae
|
Anti-inflammatory, antioxidant, antimicrobial, anthelmintic |
|
14. |
Ginger |
Zingiberaceae |
Anthelmintic, antioxidant, anti-inflammatory, digestive |
|
15.
|
Neem
|
Meliaceae
|
Anthelmintic, antimicrobial, insecticidal, anti-inflammatory |
|
16.
|
Garlic
|
Amaryllidaceae
|
Anthelmintic, antioxidant, anti-inflammatory, antimicrobial |
|
17.
|
Clove
|
Myrtaceae
|
Anthelmintic, antioxidant, anti-inflammatory, antimicrobial, analgesic |
|
18.
|
Turmeric
|
Zingiberaceae
|
Anthelmintic, antioxidant, anti-inflammatory, antimicrobial |
|
19. |
Papaya seeds |
Caricaceae |
Anthelmintic, antibacterial, antifungal |
|
20.
|
Areca nuts
|
Arecaceae
|
Anthelmintic, antioxidant, anti-inflammatory, antimicrobial |
|
21.
|
Senna alata
|
Fabaceae
|
Anthelmintic, laxative, anti-inflammatory, antimicrobial |
Table no. 8 Medicinal plants with bioactive compounds
|
S. No.
|
Medicinal Plants
|
Bioactive Compounds Present |
Bioactive Compounds Responsible for Anthelmintic Activity |
|
1. |
Sophora interrupta |
Alkaloids, flavonoids, glycosides |
Alkaloids (Sophoramine, Sophocarpine) |
|
2. |
Jussiaea hyssopifolia |
Flavonoids, phenolic acids, terpenoids |
Flavonoids (Quercetin, Kaempferol) |
|
3. |
Allium sativum |
Allicin, sulphur compounds, flavonoids |
Allicin |
|
4. |
Zingiber officinal |
Gingerols, shogaols, flavonoids |
Gingerols (Gingerol, Shogaol) |
|
5. |
Cucurbita mexicana |
Cucurbitacin’s, flavonoids, phenolic acids |
Cucurbitacin’s (Cucurbitacin E) |
|
6. |
Ficus religiosa |
Flavonoids, terpenoids, phenolic acids |
Flavonoids (Quercetin, Kaempferol) |
|
7.
|
Nerium oleander |
Cardiac glycosides, flavonoids, phenolic acids |
Cardiac glycosides (Oleandrin) |
|
8. |
Luffa cylindrica |
Saponins, flavonoids, phenolic acids |
Saponins (Luffa saponin A) |
|
9. |
Thespesia lampas |
Flavonoids, terpenoids, phenolic acids |
Flavonoids (Quercetin, Kaempferol) |
|
10. |
Clitoria ternatea |
Flavonoids, terpenoids, phenolic acids |
Flavonoids (Quercetin, Kaempferol) |
|
11. |
Guazuma ulmifolia |
Flavonoids, terpenoids, phenolic acids |
Flavonoids (Quercetin, Kaempferol) |
|
12. |
Leontis nepetiifolia |
Flavonoids, terpenoids, phenolic acids |
Flavonoids (Quercetin, Kaempferol) |
|
13.
|
Madhuca indica |
Flavonoids, terpenoids, phenolic acids |
Flavonoids (Quercetin, Kaempferol) |
|
14. |
Ficus benghalensis |
Flavonoids, terpenoids, phenolic acids |
Flavonoids (Quercetin, Kaempferol) |
|
15. |
Pongamia glabra |
Flavonoids, terpenoids, phenolic acids |
Flavonoids (Quercetin, Kaempferol) |
|
16. |
Clove |
Eugenol, beta-caryophyllene |
Eugenol |
|
17. |
Neem |
Azadirachtin, nimbin |
Azadirachtin |
|
18. |
Turmeric |
Curcumin, demethoxycurcumin |
Curcumin |
|
19. |
Papaya seeds |
Papain, carpaine |
Carpaine |
|
20. |
Areca nuts |
Arecoline, guvacine |
Arecoline |
|
21. |
Senna alata |
Anthraquinones, sennosides |
Anthraquinones |
Table no. 9 Medicinal plants parts have anthelmintic activity
|
S. No. |
Medicinal Plants |
Plants Part Used for Anthelmintic Activity |
References |
|
1. |
Sophora interrupta |
Leaves |
97 |
|
2. |
Jussiaea hyssopifolia |
Whole plant |
98 |
|
3. |
Allium sativum |
Bulb |
99 |
|
4. |
Zingiber officinale |
Rhizome |
100 |
|
5. |
Cucurbita mexicana |
Fruit |
99 |
|
6. |
Ficus religiosa |
Leaves |
99 |
|
7. |
Nerium oleander |
Flowers |
95 |
|
8. |
Luffa cylindrica |
Leaves |
101 |
|
9. |
Thespesia lampas |
Root |
102 |
|
10. |
Clitoria ternatea |
Leaves |
103 |
|
11. |
Guazuma ulmifolia |
Leaves |
103 |
|
12. |
Leontis nepetiifolia |
Leaves |
104 |
|
13. |
Madhuca indica |
Leaves |
103 |
|
14. |
Ficus benghalensis |
Leaves |
105 |
|
15. |
Pongamia glabra |
Seeds, leaves, bark |
106 |
|
16. |
Syzygium aromaticum (Clove) |
Flower buds |
107 |
|
17. |
Azadirachta indica (Neem) |
Leaves |
108 |
|
18. |
Curcuma longa (Turmeric) |
Rhizome |
100 |
|
19. |
Carica papaya (Papaya seeds) |
Seeds |
109 |
|
20. |
Dypsis lutescens (Areca nuts) |
Seeds |
110 |
|
21. |
Senna alata |
Pods, leaves |
111 |


CONCLUSION
Helminthiasis a prevalent parasitic infection, poses a significant health risk globally. The conventional treatment approach relies heavily on anthelmintic drugs, which although are effective are often plagued by issues of resistance, toxicity and side effects. This review highlights the potential of plant-based remedies as an alternative or complementary approach to helminthiasis. The comparison between traditional medicines and anthelmintic drugs reveals that while conventional drugs offer rapid relief, they often come with adverse effects. In contrast, plant-based remedies, with their natural origin and multifaceted mechanisms of action, present a promising an avenue for treatment. The phytochemicals present in these plants such as alkaloids, saponins, polyphenols and tannins exhibit anthelmintic properties, disrupting worm metabolism, interfering with energy generation and damaging teguments. The review of various plants with anthelmitic properties including neem, garlic, turmeric and ginger underscores their potential in helminthiasis treatment. These plants with their rich phytochemical’s profiles offer a natural and relatively safe approach to managing parasitic infections. Helminthiasis is a serious public health problem that requires a comprehensive approach to control and prevent. A comprehensive approach including public health education, improved sanitation and hygiene practices and development of new anthelmintic drugs and vaccines is necessary to effectively reduce the burden of this disease and improve the lives of those affected.
Future Aspects
The future research directions for helminthiasis control and treatment include several key areas:
Development of new anthelmintic drugs: New drugs are being developed to overcome resistance. and improve efficacy. Validation of traditional plants: Traditional plants with anthelmintic properties are being studied for their potential use in treatment. Vaccine development: Vaccines are being developed to prevent helminthiasis, particularly in endemic areas. Public health education: Public health education is crucial in preventing helminthiasis, particularly in endemic areas. Improved sanitation and hygiene: Improved sanitation and hygiene practices can help reduce the transmission of helminthiasis.
REFERENCES



Niharika Singh*, Kamal Kishore Maheshwari, Zafar Akbar, Helminthiasis: Natural Remedies, Anthelmintic Drugs and Medicinal Plants, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 6, 3471-3501. https://doi.org/10.5281/zenodo.15719236
 10.5281/zenodo.15719236
10.5281/zenodo.15719236