
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Shree Panchavati Education Society Institute of Pharmacy, Nashik, Maharashtra 422003.
Buccal administration of drugs provides a convenient route of administration for both systemic and local drug action. Buccal bioadhesive films, releasing topical drugs in the oral cavity at a slow and predetermined rate, provide distinct advantages over traditional dosage forms for treatment of many diseases. Buccal drug delivery leads direct access to the systemic circulation through the internal jugular vein bypasses drugs from the hepatic first pass metabolism leading to high bioavailability. The main aim of this article is to formulate a herbal buccal patches for treatment of various diseases.
Buccal administration of drugs provides a convenient rate of administration for both systemic and local drug action.[1] Buccal bioadhesive films, releasing topical drugs in the oral cavity at a slow and predetermined rate, provide distinct advantages over traditional dosage forms for treatment of many diseases. The main aim of this article is to formulate a herbal buccal patches for treatment of various diseases. [2] Buccal patches are described as laminates comprised of an impermeable backing layer, a drug-containing reservoir layer which releases the drug in a controlled manner, and a mucoadhesive surface for mucosal attachment. Patches may be used to deliver drugs directly to a mucosal membrane.[3] A well-defined definition of bioadhesion is a substance's propensity to stick for an extended period of time to biological tissues, whether it be synthetic or biological. The biological surface can be a mucous layer covering a tissue's surface or it can be epithelial tissue. Mucoadhesive is the term used to describe a phenomenon where adhesion is to a mucous coat. There are more applications for mucoadhesive polymers in buccal drug delivery. There are many mucoadhesive products that have recently been developed, including tablets, films, patches, disks, strips, ointments, and gels. But compared to other devices, buccal patches offer more comfort and flexibility.[4]
Buccal patch is a non-dissolving thin matrix modified release dosage form composed of one or more polymer films or layers containing the drug and/or other excipients.[5]
Buccal drug delivery is a highly effective way to increase bioavailability. This is because the buccal mucosa has a rich in blood supply which facilitates the direct entry of the drug into the systemic circulation. [6]
In addition, buccal dosage forms allow drug absorption to be rapidly terminated in case of an adverse reaction. Formulations of buccal dosage forms include- tablets, gels and patches of which patches are preferable in terms of flexibility and comfort [7].
Structure of Buccal mucosa :-
Physiological aspects and functions of Oral Cavity: -
The buccal mucosa is composed of an outermost layer of stratified squamous epithelium (Figure 1). Below this lies a basement membrane, a lamina propria followed by the submucosa as the innermost layer. The epithelium is similar to stratified squamous epithelia found in the rest of the body in that it has a mitotically active basal cell layer, advancing through a number of differentiating intermediate layers to the superficial layers, where cells are shed from the surface of the epithelium. The epithelium of the buccal mucosa is about 40-50 cell layers thick, while that of the sublingual epithelium contains somewhat fewer. The epithelial cells increase
in size and become flatter as they travel from the basal layers to the superficial layers. [5,6,7]
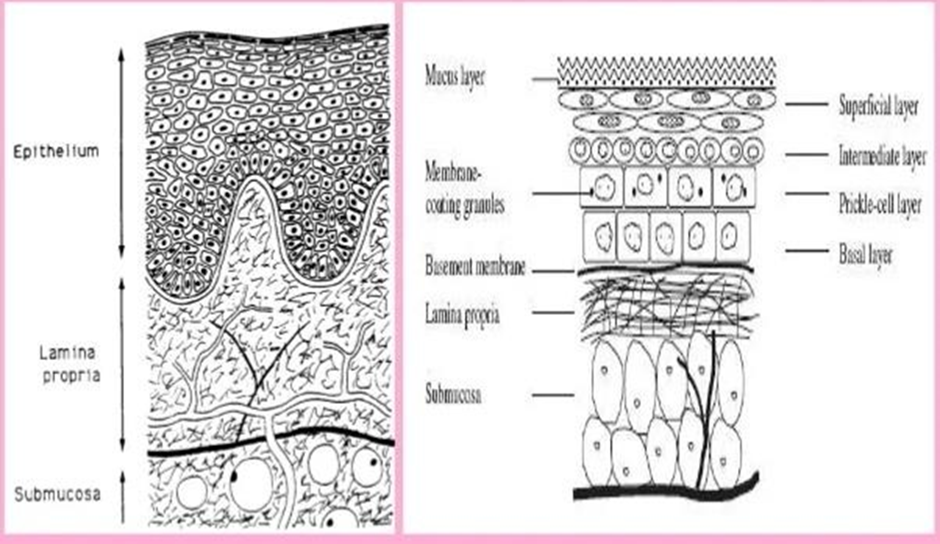
Fig. 1: - Structure of buccal mucosa
Schematic cross section through the oral mucosa showing the epithelium, basal lamina, and connective tissue [5,6, 7] There is need to develop a dosage form that bypasses first pass metabolism and GI degradation. Oral cavity provides route for the administration of a therapeutic agent for local as well as systemic delivery, so that first pass metabolism and GI degradation can be avoided. For the preparation of patches commonly used technique is solvent casting technique.[8] The oral cavity is easily accessible for self-administration, stopping of drug is feasible if required, safe and, hence is well accepted by patients [9]. To avoid the swallowing of dosage form or dose dumping, bioadhesive polymers have received considerable attention for platforms of buccal controlled delivery. Due to bioadhesion, the immobilization of drug carrying particles at the mucosal surface would result in, a prolonged residence time at a site of absorption or action, a localization of the drug delivery system at a given target site and Increase in the drug concentration gradient due to the instant contact of the particles with mucosal surface. [9]
Types :-
1.Matrix type (Bi-directional): The buccal patch designed in a matrix configuration contains drug, adhesive, and additives mixed together. [10]
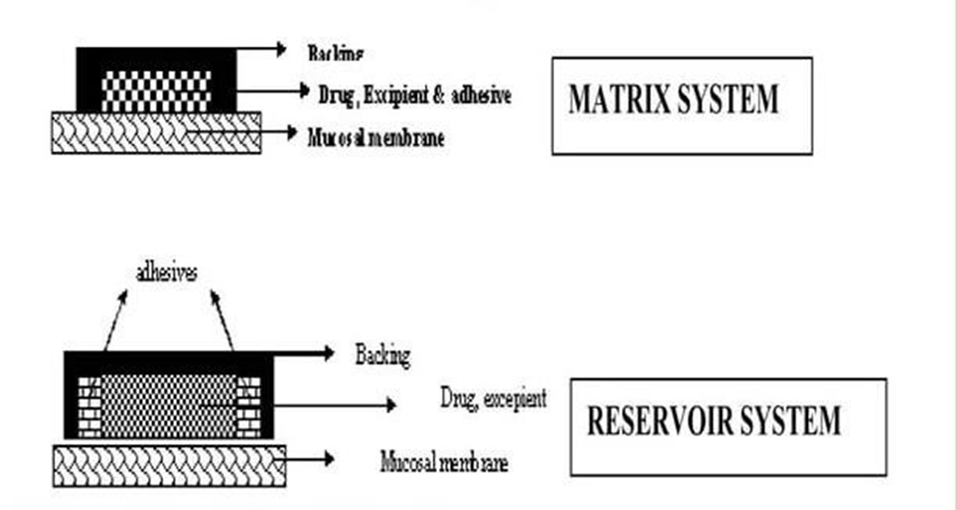
Fig. 2: Buccal Patch designed for Bidirectional drug release.
2.Reservoir type (Unidirectional):
The buccal patch designed in a reservoir system contains a cavity for the drug and additives separate from the adhesive. Anti permeable backing is applied to control the direction of drug delivery; to reduce patch deformation and disintegration while in the mouth; and to prevent drug loss.[11] Bucca
3.Buccal Patch designed for unidirectional drug release:-
Bioadhesive Delivery of Drug System in Oral Cavity :-
1]Sublingual delivery :- This is systemic delivery of drugs through the mucosal membranes lining the floor of the mouth.
2] Buccal delivery:- This is drug administration through the mucosal membranes lining the cheeks (buccal mucosa).
3]Local delivery:-This is for the treatment of conditions of the oral cavity, Principal ulcers, fungal conditions and periodontal disease. These oral mucosal sites differ greatly from one another in terms of anatomy, permeability to an applied drug and their ability to retain a delivery system for a desired length of time. [12]
Classification Of Buccal Bioadhesive Dosage Forms:-
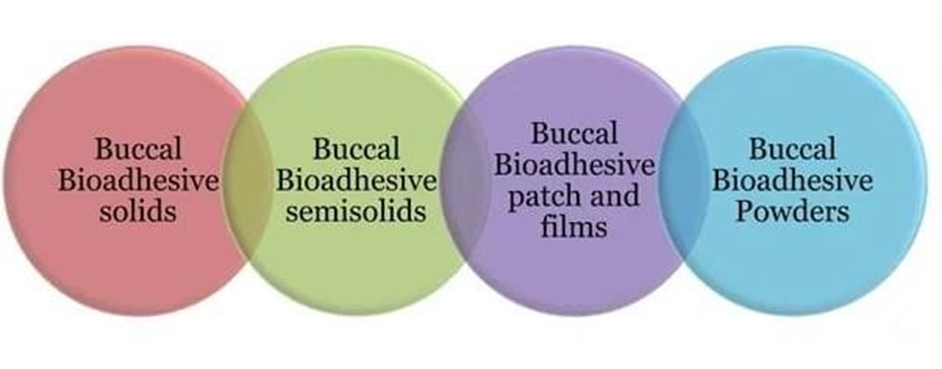
Fig. 3: Classification of Buccal Bioadhesive Dosage Forms
i) Tablets for the buccal mucosa: -
That are buccal bioadhesive are dry dosage forms that must be moistened before use. Currently, double and multilayered tablets are made using bioadhesive polymers and additives. The type of additives contained in the dosage form will determine whether the tablets, which are solid dosage forms created by direct compression of powder, can be placed in contact with the oral mucosa and allowed to adhere or dissolve. This dosage form has multiple routes for delivering medication to the oral cavity or mucosal surface.[13]
ii) Buccal Bioadhesive Semisolid Dosage Forms: -
These dosage forms are semisolid and contain natural or synthetic polymers in powdered form that is dispersed in polyethylene or water. [14]
iii) Buccal Bioadhesive Patches and Films: -
These films or patches include multilayered thin film or two poly laminates that are oval or round in shape, containing primarily of bioadhesive polymeric layer and impermeable backing layer to allow unidirectional flow of drug across buccal mucosa. To create these films, bioadhesive polymers are dissolved in alcohol before being mixed with the medication.[15]
Method Of Preparation: -
1] Solvent casting:-
It involves dispersing all of the patch excipients, including the medication, in an organic solvent before coating the mixture on a sheet of release liner. The coated release liner sheet is laminated with a thin layer of protective backing material after the solvent has evaporated. This laminate is then die-cut into patches with the desired size and geometry. A boiling tube was filled with weighed-out HPMC E15. (Hydroxypropyl methylcellulose) This was then mixed with 20 ml of the solvent solution (1:1 dichloromethane: methanol). An adequate amount of care was taken to avoid lump formation.[28] To give the polymer time to swell, the boiling tube was left idle for six hours. Propylene glycol was added in a precise amount after swelling, and the mixture was vortexed. The final CPH amount was weighed out, and 5 ml of the solvent mixture was used to dissolve it. It was then added to the polymer solution and thoroughly mixed. It was transferred into a Petri plate that had already been cleaned after being set aside for a while to let any trapped air escape. These patches were dried in an oven that was positioned over a flat surface for 8 hours. With no plasticizer added, the process is repeated for HPC EF.[29]
2] Direct milling (Solvent free):-
In this process, patches are made without the use of solvents. Direct milling or kneading, typically without the use of any liquids, is used to mechanically combine the drug and excipients. The final product of the mixing process is rolled on a release liner until the desired thickness is reached. Following that, the backing material is laminated as before delivery of drugs possible.[30]
Advantages of Buccal Patches :-
1.The oral mucosa has a rich blood supply. Drugs are absorbed from the oral cavity through the oral mucosa, and transported through the deep lingual or facial vein, internal jugular vein and bracio cephalic vein into the systemic circulation.[16]
2.Buccal administration, the drug gains direct entry into the systemic circulation thereby bypassing the first pass effect. Contact with the digestive fluids of gastrointestinal tract is avoided which might be unsuitable for stability of many drugs like insulin or other proteins, peptides and steroids. In addition, the rate of drug absorption is not influenced by food or gastric emptying rate. [17]
3.The area of buccal membrane is sufficiently large to allow a delivery system to be placed at different occasions, additionally; there are two areas of buccal membranes per mouth, which would allow buccal drug delivery systems to be placed, alternatively on the left and right buccal membranes.[18]
4.Buccal patch has been well known for its good accessibility to the membranes that line the oral cavity, which makes application the oral cavity, which makes application painless and with comfort.[19]
5.Patients can control the period of administration or terminate delivery in case of emergencies. The buccal drug delivery systems easily administered into the buccal cavity. The novel buccal dosage form exhibits better patient compliance.[20]
Limitations of buccal patches:-
i. The buccal route cannot be used to administer mucosa-both ring or strongly flavored medications.
ii. Small dose medications can only be given.
iii. Saliva production is constant, causing drugs to be quickly eliminated.
iv. Little area for absorption.
v. Involuntary salivation gulping causes a sizable portion of the delivered drug to disintegrate or suspend and be removed from the site of retention. The delivery system itself could also be swallowed, which is another risk.[21]
Factors affecting Buccal Absorption :-
The oral cavity is a complex environment for drug delivery as there are many interdependent and independent factors which reduce the absorbable concentration at the site of absorption;
1.Membrane Factors:-
This involves degree of keratinization, surface area available for absorption, mucus layer of salivary pellicle, intercellular lipids of epithelium, basement membrane and lamina propria. In addition, the absorptive membrane thickness, blood supply/ lymph drainage, cell renewal and enzyme content will all contribute to reducing the rate and amount of drug entering the systemic circulation.[22]
2. Environmental Factors:-
A] Saliva:-The thin film of saliva coats throughout the lining of buccal mucosa and is called salivary pellicle or film. The thickness of salivary film is 0.07 to 0.10 mm. The thickness, composition and movement of this film affect the rate of buccal absorption. [23]
B] Salivary glands: -The minor salivary glands are located in epithelial or deep epithelial region of buccal mucosa. They constantly secrete mucus on surface of buccal mucosa. Although, mucus helps to retain mucoadhesive dosage forms, it is potential barrier to drug penetration.[24]
C] Movement of buccal tissues:- Buccal region of oral cavity shows less active movements. The mucoadhesive polymers are to be incorporated to keep dosage form at buccal region for long periods to withstand tissue movements during talking and if possible during eating food or swallowing.[25]
Benefits of Buccal Drug Delivery System Drug:-
1.Buccal films provide large surface area that leads to rapid disintegration and dissolution in the oral cavity due to which it promotes the systemic absorption of Active pharmaceutical ingredient.
2.No need of chewing and swallowing.
3.No risk of choking.
4.The film increases the systemic bioavailability of the drugs, as it bypasses the hepatic first pass metabolism.
5.Drug can be protected from degradation by GI enzymes and the acidic environment.
6.Rapid onset of action and minimum side effects.
7.Self-administration is possible.
8.Accurate dosing compared to liquid dosage forms.
9.Taste masking is possible.
10.Prolongs the residence time of the dosage form at the site of absorption, hence increases the bioavailability.
11.Ease of administration to pediatric, geriatric patients, and also to the patients who are mentally retarded, disabled or non-cooperative.
12.Good mouth feel and good stability. [26,27]
Evaluation Parameters: -
CONCLUSION
Drugs are simply and consistently delivered through the buccal mucosis extensive vascular and lymphatic system. Patches are a safe and convenient way to administer medication in the buccal space because buccal medication can be stopped any time in case of toxicity. It has been found that the buccal route of administration is the easiest and fastest effective way to deliver the medication.
More over the possibility of self-administration makes it easy even to the serious patients. Compared to the existing medications available for treatment of Heart burn, acidity, Herbal Buccal patches are safer, with less side effects and easily available. This will definitely cause the reduction in side effects along with less financial burden to patient population taking into consideration the price of herbal drugs.
REFERENCES



Harshali Chaudhari, Pooja Kumbhar, Rupali Dhikale, Herbal Buccal Patches: A Novel Approach to Drug Delivery, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 3007-3014, https://doi.org/10.5281/zenodo.19202463
 10.5281/zenodo.19202463
10.5281/zenodo.19202463