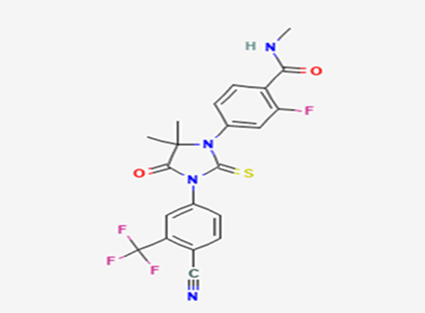
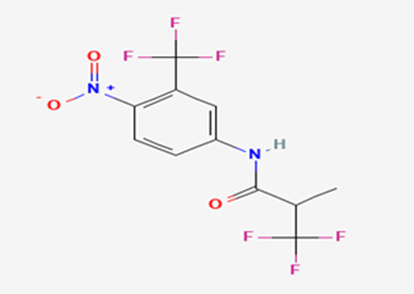
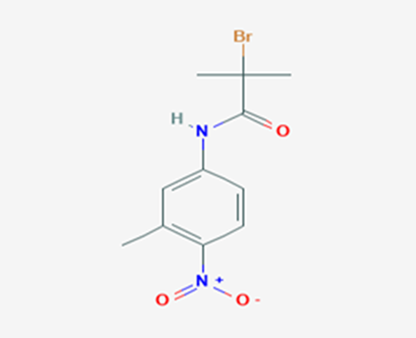
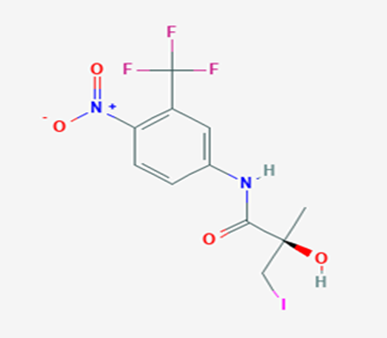
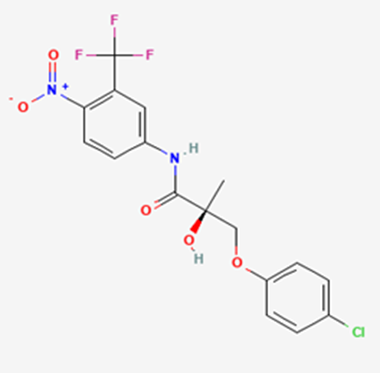
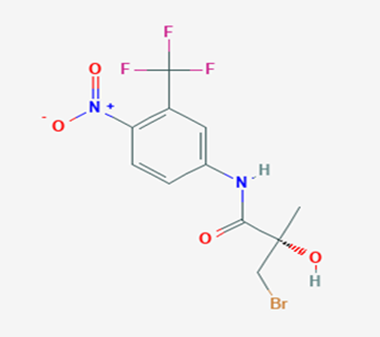
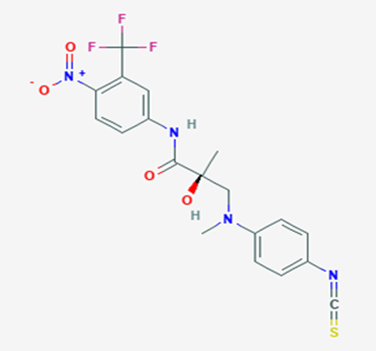
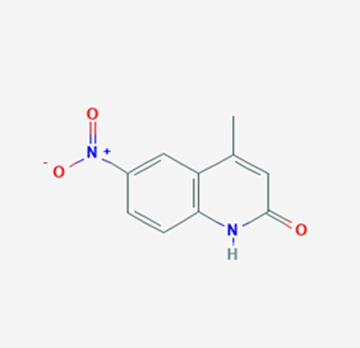
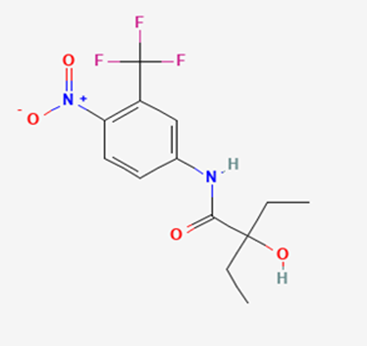
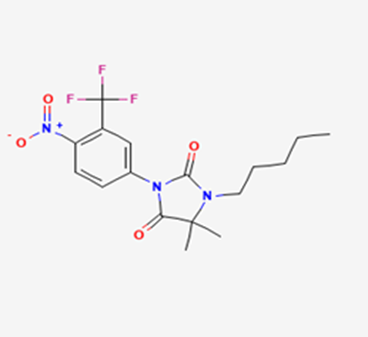
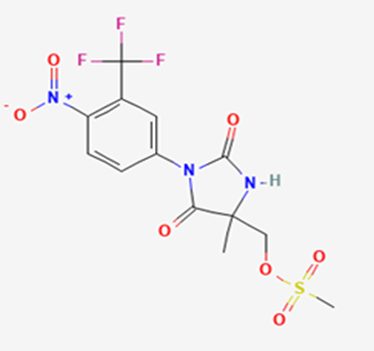
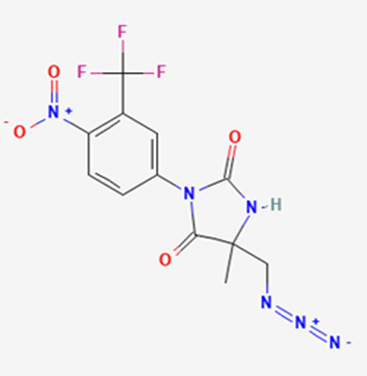
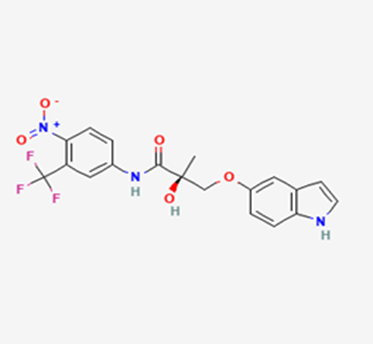
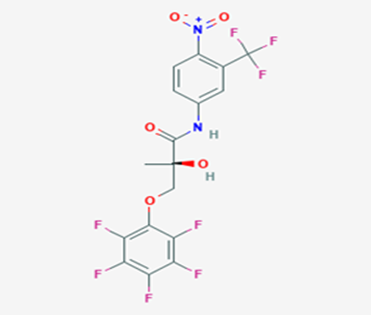
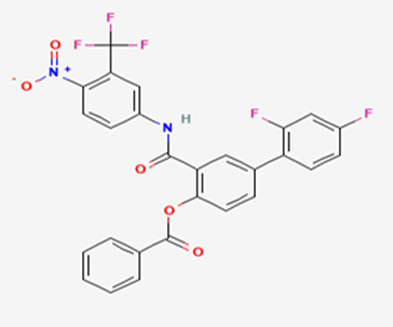
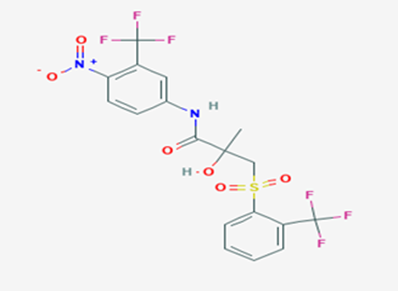
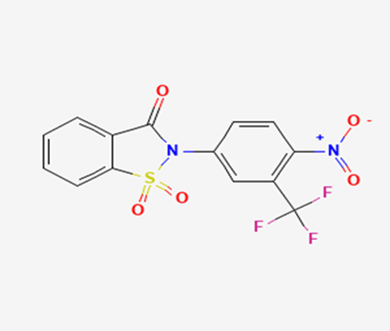
Cancer is now a days very common in all over the World. The mortality rate of cancer is increasing day by day. Prostate cancer is one of them for old men. Sometimes this cancer is symptomless. During the progress of the diseases the symptoms will shown in body like painful urination, bleeding during urination, lower abdominal pain, blood in semen, very light urination in daytime, frequent urination at night, etc. The diseases can be detected by PSA analysis in blood as well as different scans and USG in prostate or lower abdomen. If the PSA test will be very high then doctor will suggest to take biopsy from prostate for detection of prostate cancer. Early detection is very safe to treat this deadliest disease. Sometime hormonal changes and low immunity due to different co-morbidic conditions can create prostate enlargement. Several existing drugs like Flutamide, Nilutamide etc are available to treat this disease at early stages. Androgen receptor is the target to treat this disease. These molecules can work over this androgen receptor. These molecules are having the complex structure of N-[4-nitro-2-(trifluoromethyl) phenyl] amide derivatives, imidazolidine-2,4-dione derivatives. Based on the standard molecules we are trying to find newer potent molecules which will be worked safely on androgen receptors through different in silico drug design tools. The present study focuses that the existing molecules, targets of this disease, Mortality rate, screening of molecules through molecular docking and ADMET generation.
Prostate Cancer, Androgen receptor, Flutamide, Nilutamide, Docking, Pharmacophore, Molecular descriptors, ADMET.
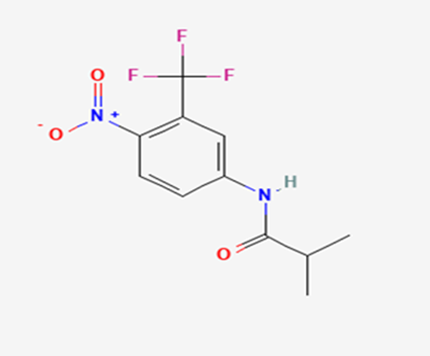
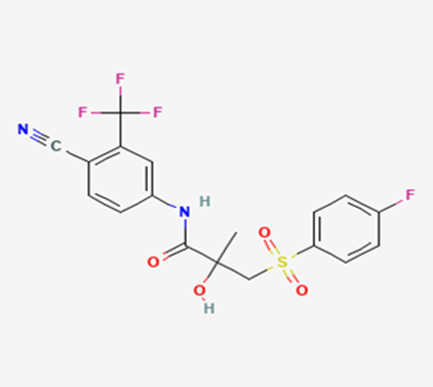
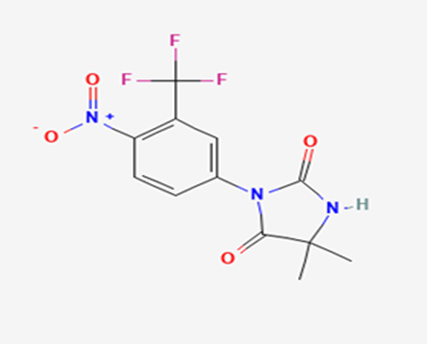
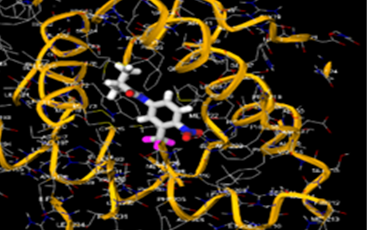
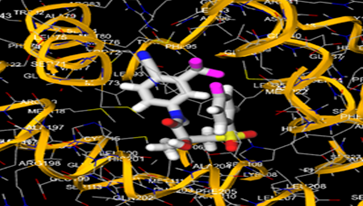
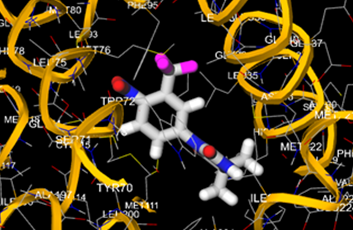
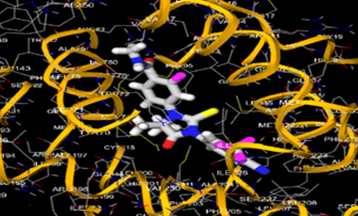
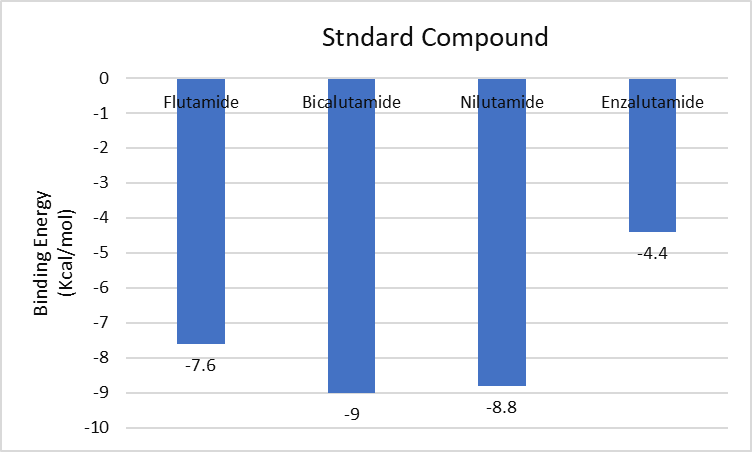
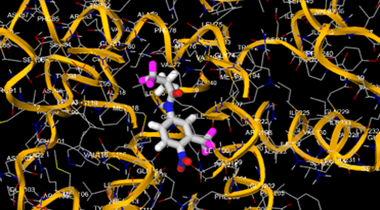
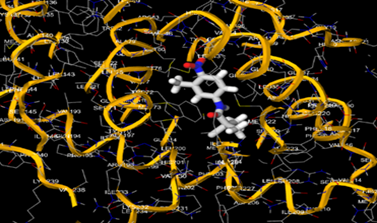
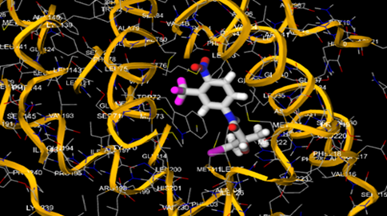
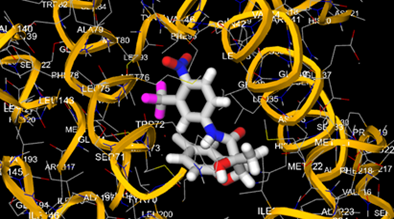
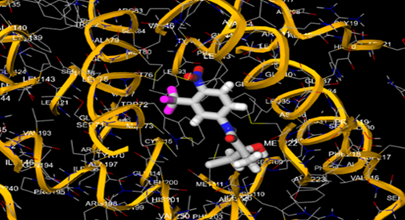
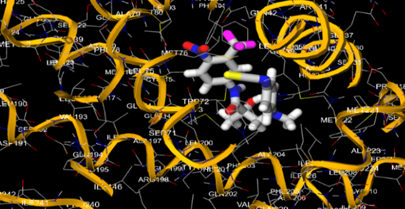
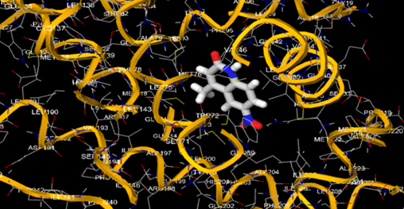
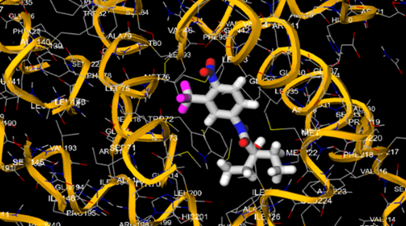
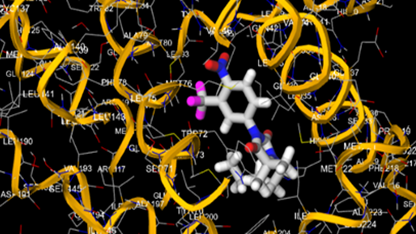
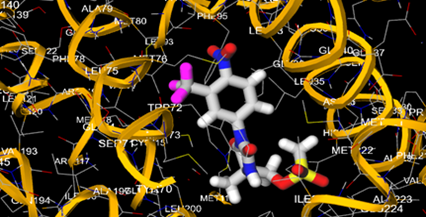
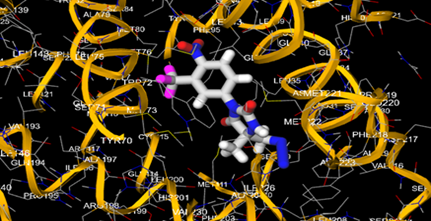
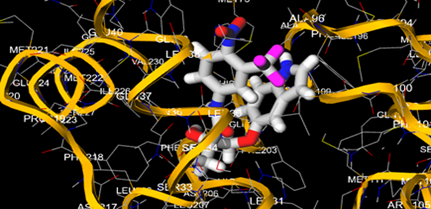
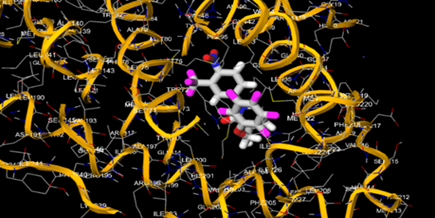
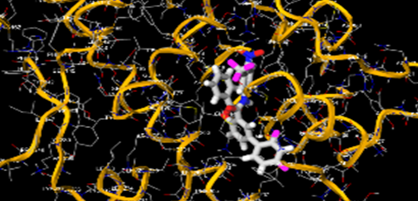
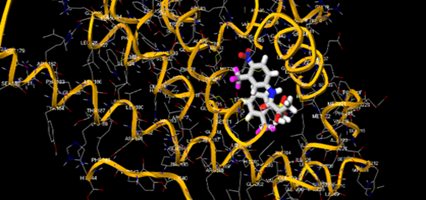
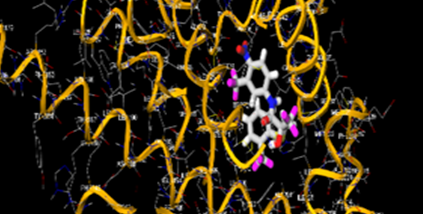
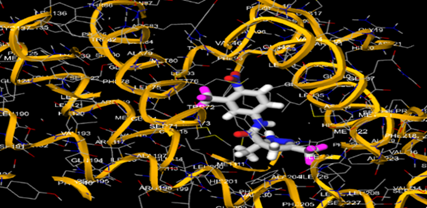
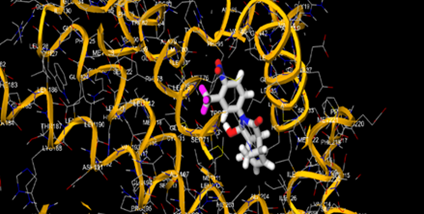
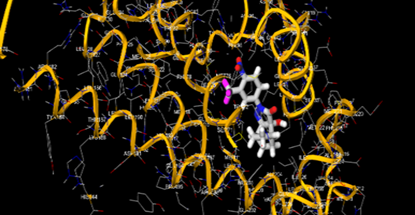
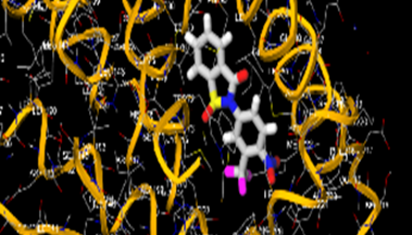
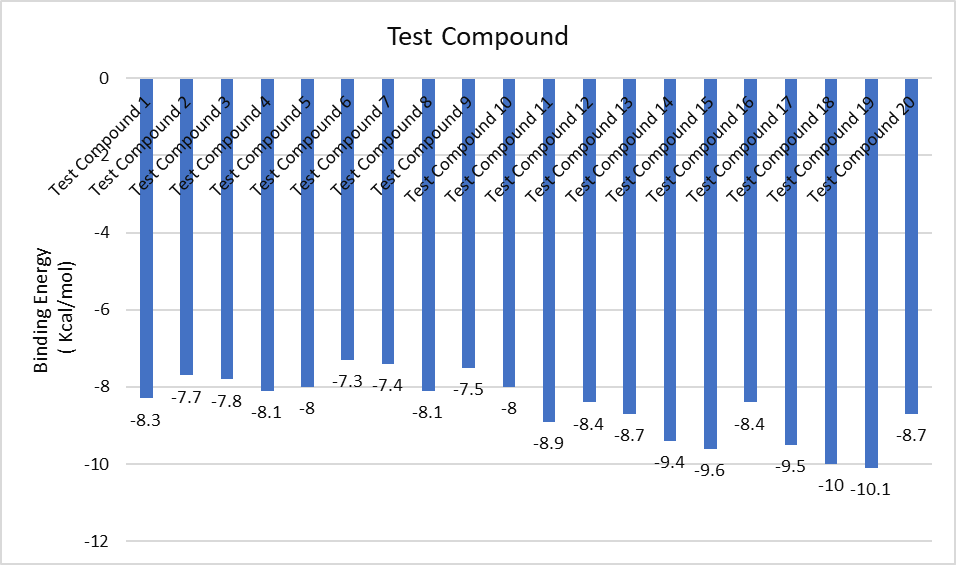
The most prevalent cancer among men in northern and western Europe is prostate cancer (PC). [1]. Among the most common malignancies diagnosed in males cum specially older aged males, it counts as the most leading cause of cancer-related deaths worldwide. [2] It primarily affects older males, with a sharp rise in occurrence during the seventh decade of life. The population's aging is a contributing factor in the growing incidence, although the age-adjusted incidence has also gone up. A reversal may have occurred recently, maybe as a result of better treatment and enhanced identification through PSA (Prostate specific antigen) screening. [3] Prostate development and optimal prostate function depend on androgens, which act through the androgen receptor (AR) [1]. Testicular production of testosterone, its delivery to target tissues, and its conversion by 5-reductase to the more active metabolite 5-dihydrotestosterone (DHT) are all thought to be involved in the axis via which androgen action occurs. Testosterone and DHT both have biological effects through binding to AR and inducing AR transcriptional activity. [4] To present, epidemiologic studies have yielded the most information about prostate cancer risk. However, epidemiology is a quite unrefined method of studying what can turn out to be an exceptionally complicated cause. Most of these studies have serious problems with exposure and disease characterization. [5] These days, high blood PSA levels are used to detect a growing proportion of prostate malignancies. Several cases were identified by digital rectal examination (DRE) or clinical symptoms prior to PSA testing becoming common place. [3] Activation of the androgen receptor (AR) pathway abnormally is the primary mechanism driving the advancement of PCA. As a result, the conventional treatment for PCA and metastatic hormone-sensitive prostatic carcinoma (mHSPC), often referred to as castration-sensitive cancer (mCSPC), is androgen deprivation therapy (ADT), which can be done surgically or chemically. [2] This deadliest illness can be safely treated with early discovery. Prostate enlargement can occasionally result from hormonal changes and weakened immunity brought on by other co-morbid disorders. Numerous medications, such as Flutamide, Nilutamide, and others, are already on the market to treat this illness in its early stages. Treatment for this condition focuses on the androgen receptor. As per analogue-based drug design, here we have study lots of Flutamide and Nilutamide analogues and these compounds are collected from the different molecular libraries. All the compound has the structural similarities and possess a lead structure that is N-[4-nitro-2-(trifluoromethyl)phenyl] amide group. Then all molecules are docked through in silico docking tools, and after perform the docking screened them based on their binding affinity. Generate the pharmacophore modelling and molecular descriptor for their QSAR analysis. Finally, if all the analysis has the positive result, then go for the ADMET prediction of the molecule to establish as a newer drug molecule.
RISK FACTORS
To present, epidemiologic studies have yielded the most information about prostate cancer risk. However, epidemiology is a quite unrefined method of studying what can turn out to be an exceptionally complicated cause. Most of these studies suffer from serious problems with exposure and sickness development.
Endogenous Risk Factors
- Family history
In epidemiologic studies, family history is strongly linked to the risk of prostate cancer, albeit detection bias may have an impact. Cancers that are familial and those that are not have many clinical and pathologic characteristics.
- Hormones
Prostate cancer growth rates are greatly influenced by androgens, and changes in androgen metabolism may contribute to the development of prostate cancer from preclinical to clinically relevant forms. Although results have been mixed, elevated levels of testosterone and its metabolite, dihydrotestosterone, over several decades may raise the risk of prostate cancer.
- Race
Three reasons might be responsible for racial disparities in prostate cancer risk: genetic differences (endogenous factors); differences in detection (reflecting exogenous factors); and differences in exposure, such as dietary variances (exogenous factors).
Exogenous Risk Factors
- Diet
Geographic variations, temporal studies, and descriptive epidemiologic studies of migrants have all linked a wide range of dietary parameters to the development of prostate cancer. Prostate cancer incidence and mortality are strongly positively correlated with fat consumption, particularly polyunsaturated fat. This may be due to changes in hormone profiles brought on by fat, the impact of fat metabolites as protein or DNA-reactive intermediates, or the increase in oxidative stress caused by fat. Vitamin A is one retinoid that helps control the differentiation and proliferation of epithelial cells and has a favourable correlation with the risk of prostate cancer.
- Occupation and other factors
The biggest concerns are farming and, to a lesser extent, working in the rubber sector. Numerous industrial and occupational exposures have been investigated in connection to prostate cancer risk, but the results are conflicting. Numerous other factors, such as smoking, energy intake, sexual activity, marital status, vasectomy, social factors (lifestyle, socioeconomic factors, and education), physical activity, and anthropometry, have either produced conflicting results, negative associations, or very little data with regard to prostate cancer risk. [6]
ANATOMY OF PROSTATE GLAND
McNeal’s conception of zonal deconstruction has developed from the original two-zone conception to a description of four introductory anatomic regions [7,8] a) the supplemental zone b) the central zone c) the zone of transition & d) the fibro muscular stroma anteriorly. The anatomical course of the prostatic urethra is a crucial to the zonal deconstruction of the prostate. The urethra angulates sprucely forward at the midpoint of its course through the prostate. It's at this point that the verumontanum merges with the posterior urethral wall, but the bulk of the verumontanum lies in the distal urethral member. A double row of tubes passes indirectly into the prostatic mesenchyme to the distal urethral member. These tubes drain the area of the supplemental zone, which makes up -75 of the glandular towels of the prostate. The supplemental zone forms a large, boat- shaped area and incompletely surrounds the central zone at the base. The supplemental zone also forms utmost of the glandular towel distal to the verumontanum. Tubes addict out into the supplemental zone in the coronal aeroplane and taper in size as they approach the capsule. The supplemental zone glands are slightly small and round and their cells are small with dark capitals located basally in invariant columnar cells. The tubes which enter the urethra proximal to the ejaculatory tubes drain the central zone. This wedge-shaped area suckers out from the verumontanum toward the base of the prostate. The ejaculatory tubes are girdled by central zone towel throughout their intraprostatic course. The central zone comprises 20- 25 of the glandular towel of the prostate. The central zone, while fused to the supplemental zone, is separated from it by a distinct anatomical boundary line. The central zone glands are larger than those of the supplemental zone and have cells with relatively high, irregular capitals and grainy cytoplasm. In contrast to the supplementary zone and urethra, which are of urogenital sinus origin, McNeal suggests that the central zone is of Wolffian conduit origin, highlighting the stark histologic differences between the two zones. [9]
ANDROGEN RECEPTOR SIGNALING IN PROSTATE CANCER DEVELOPMENT AND PROGRESSION
The mortal AR gene is a nuclear recap factor and a member of the steroid hormone receptor superfamily of genes. It is located on the X chromosome (q11–12) and has eight exons. It produces a protein that weighs 110 kDa and has 919 amino acids. The AR consists of four structurally and functionally distinct disciplines, a inadequately conserved N-terminal sphere (NTD), a largely conserved deoxyribonucleic acid (DNA)- binding sphere (DBD) and a relatively conserved ligand- binding sphere (LBD). A short amino acid sequence called the ‘hinge region’ separates the LBD from the DBD and also contains part of a dual ligand-dependent nuclear localization signal (NLS) for AR nuclear transport. AR sphere structure and function has been considerably reviewed away. [10-12] Testosterone is the prototype of androgenic steroids, which are 19-carbon steroids. In males, the testes create the majority of it, with the adrenal glands contributing only a little. Numerous physiological and developmental reactions are influenced by androgens. The prostate and vaginal tissues have significant levels of the cytochrome P450 enzyme 5α-reductase, which changes testosterone into DHT. [13,14] Under physiological settings, both DHT and testosterone may bind to and activate AR; however, DHT has a far higher affinity for AR and can activate target genes at lower concentrations than testosterone. [15,16] When there is no ligand present, the AR is mostly found in the cytoplasm where it interacts with cytoskeletal proteins, various chaperones, and heat shock proteins (HSP)-90, -70, and -56. [17] It is thought that cytoskeletal proteins like FlnA are linked to HSPs. FlnA interacts with AR and influences transcriptional activity, nuclear translocation and dependency of androgen. [18,19,20] Additionally, this complex activates focal adhesion kinase (FAK) and Rac1, which both regulate cell motility, and attracts integrin beta 1. [21] This complex seems to serve as a bridge between the actin cytoskeleton and androgen signalling, promoting cell migration and perhaps influencing the course of prostate cancer. Helices 3, 4, and 12 inside the LBD create the AF-2 binding surface when a ligand binds to the AR ligand-binding pocket, causing a conformational shift in AR. Nuclear receptors employ AF-2 because their primary protein-protein interaction surface to influence co-activators that have the LxxLL motif. [22] The pocket in the LBD interacts poorly with the LxxLL motifs frequently seen in coactivators and binds preferentially to the FxLF motifs present in the NTD. [23,24] Consequently, the hydrophobic pocket in the AR LBD makes it easier for the AR NTD and its carboxy-terminal domain (CTD) to interact both intramolecularly and intermolecularly, which causes AR to dimerize. The majority of this NTD/CTD interaction takes place when AR is not attached to DNA. [25] The isolation of a number of AR-associated co-activators with FxLF motifs [26] indicates that these regulatory proteins and the NTD compete with one another for binding to the AF-2. Although the importance of this is unknown, it implies that distinct types of co-activators may interact with different AR surfaces and that AR might engage with its co-activators through other binding sites outside of this well-defined co-activator pocket. [27] The nuclear targeting of AR and the creation of AR homo-dimers are facilitated by these interactions. AR attaches itself to certain recognition sequences in the target gene's enhancer and promoter regions called androgen response elements (AREs) after entering the nucleus. The recruitment of co-regulators completes the AR transcriptional complex, which in turn modifies gene expression. [28]
STAGES OF PROSTATE CANCER
The International Union against Cancer (UICC) and the American Joint Committee on Cancer (AJCC) approved a unified tumour, node, and metastasis (TNM) staging approach for prostate cancer, which marked the introduction of the TNM staging system. The modifications included the inclusion of extra prostatic extension and microscopic bladder neck invasion in the T3a category, the acceptance of Gleason score as the preferred grading system, and the incorporation of preoperative prostate-specific antigen (PSA) and Gleason score as prognostic factors into stage grouping. [29]
ANDROGEN DEPRIVATION THERAPY FOR PROSTATE CANCER
To counteract the effects of androgens, a number of different types of medications are employed in therapeutic settings. To counteract the effects of androgens, androgen receptor antagonists like Flutamide, Bicalutamide, and Nilutamide are frequently used either by themselves or in conjunction with castration. Cytochrome P450 enzymes, which are essential for the synthesis of androgens and other hormones, are suppressed by Ketoconazole and other adrenal ablating medications. Dihydrotestosterone, a stronger androgen receptor activator than testosterone, is produced from the testes when testosterone is released. The enzyme that causes this conversion, 5α-reductase, is inhibited by Finasteride. Although its function in the routine treatment of individuals with prostate cancer is unclear, Finasteride may have a part in prevention. [30]
MATERIAL & METHODOLOGY
- Software and Web server:
- Pubchem
- Protein data bank (PDB)
- PDBSUM
- UCSF Chimera
- Rasmol
- Padel Descriptors
- Chemdes
- Pharmagist
- Zinc pharmer
- CB-DOCK2
- Auto Dock Tools
- Swiss ADME
- Pharmit [31-38]
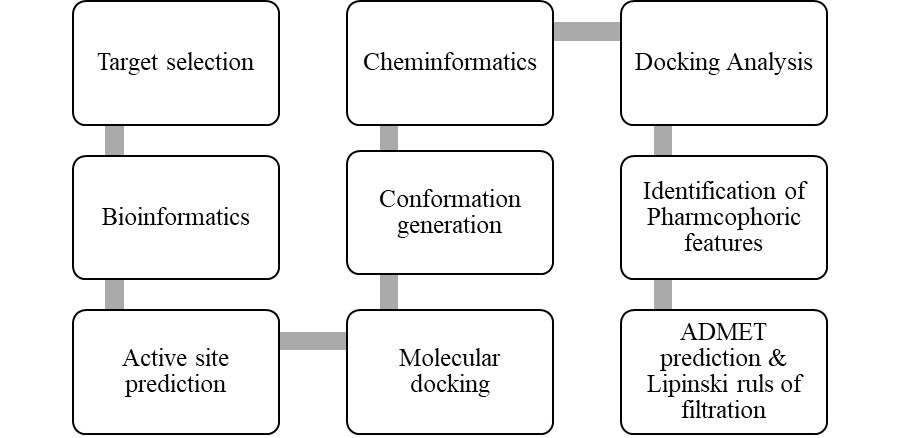
B) Methodology:


 Soumallya Chakraborty*
Soumallya Chakraborty*
 Somenath Bhattacharya
Somenath Bhattacharya
 Santu Mandal
Santu Mandal
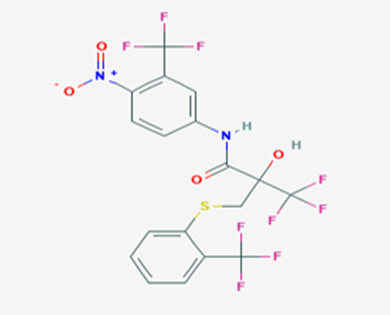
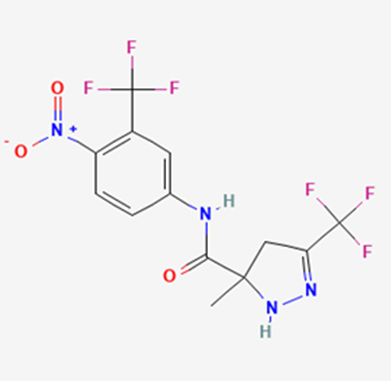
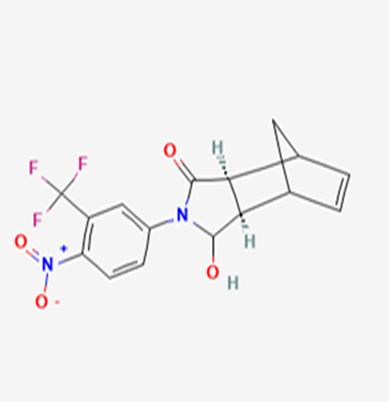
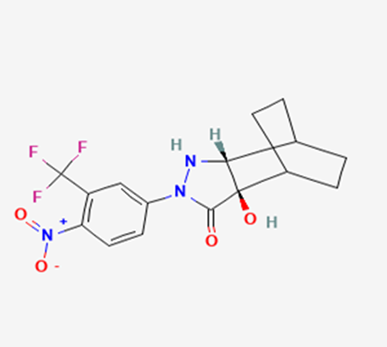
 10.5281/zenodo.15835726
10.5281/zenodo.15835726