Herbal medicines are widely used across the world due to their safety, cultural acceptance, and significant therapeutic potential. However, poor bioavailability remains a major limitation for many phytoconstituents. Most plant-derived active compounds, including flavonoids, glycosides, polyphenols, tannins, and alkaloids, exhibit low absorption and reduced systemic availability because of their large molecular size, poor lipid solubility, and limited membrane permeability. As a result, higher doses are often required to achieve therapeutic effects, which may reduce patient compliance. Phytosome technology has emerged as an advanced drug delivery system designed to overcome these challenges by complexing phytoconstituents with phospholipids, thereby improving their compatibility with biological membranes. The formation of a phospholipid–phytoconstituent complex enhances absorption, increases bioavailability, improves stability, and prolongs therapeutic activity. This review highlights the fundamental concept of phytosomes, various preparation methods, characterization techniques, advantages, limitations, and diverse applications in herbal drug delivery. Additionally, recent advancements and commercially available phytosomal formulations are discussed to emphasize their growing significance in modern phytopharmaceutical research.
Phytosomes, Herbal Drug Delivery, Phytoconstituents, Bioavailability Enhancement, Phospholipid Complexes
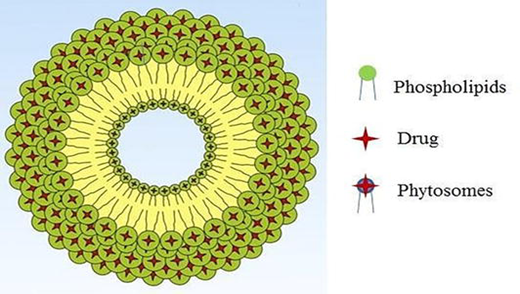
4. COMPONENTS OF PHYTOSOME
- Phytoconstituent / plant extract (hydrophilic part)
- Standardized plant extract or pure phytochemical, most often polyphenols such as flavonoids, terpenoids, tannins, xanthones
- Examples: Silymarin, Quercetin, Catechin, Curcumin, Green Tea Polyphenols, Ginsenosides 2,17,18
- Phospholipid (lipid component)
- Core structural component; usually phosphatidylcholine (PC) from soy or egg3,19
- Other phospholipids that can be used: phosphatidylethanolamine, phosphatidylserine, phosphatidylinositol3,20.
Phytosomes are formed by hydrogen bonding between the polar head of the phospholipid and polar groups (–OH, –COOH, –NH?) of the phytoconstituent2,21,22
- Solvent system (for complex formation)
- Complex is produced by reacting extract and phospholipid in aprotic or non?polar organic solvents such as dioxane, acetone, ethanol, methanol, chloroform, dichloromethane, or hexane, depending on method 2,11,21,23
- Solvent is later removed (rotary evaporation, lyophilization), leaving the solid phytosomal complex2,21.
- Optional excipients (formulation dependent)
- Stabilizers / antioxidants (e.g., vitamin E, butylated hydroxytoluene) to protect lipids and polyphenols from oxidation 24,25
- Cryoprotectants like mannitol or sugars when freeze?drying26
- Surfactants or polymers in advanced systems to adjust surface charge or stability 24,25
5. METHODS OF PREPARATION OF PHYTOSOMES FOR HERBAL DRUG DELIVERY
Phytosomes are prepared by reacting herbal extracts or purified phytoconstituents with phospholipids (mainly phosphatidylcholine) in suitable organic solvents, then removing the solvent and processing the resulting phyto?phospholipid complex into a dosage form. The main methods used for herbal drug delivery are solvent evaporation / thin?film hydration, anti?solvent precipitation, solvent ether injection, co?solvent lyophilization / freeze?drying and spray?drying6,27–30
- Solvent Evaporation/ Thin?Film Hydration
In the solvent evaporation / thin film method, the extract and phospholipid are dissolved in a common organic solvent (e.g., methylene chloride, dioxane, ethanol), often in a 1:1 or 1:2 molar ratio, then the solvent is removed under reduced pressure to form a thin film or solid complex that can be hydrated to give phytosomal vesicles6,29,31 This is the most commonly reported technique for phytosomes of silymarin, ginkgo, green tea, and many other herbal drugs 2,6,32
- Anti Solvent Precipitation
In anti solvent precipitation, the extract–phospholipid solution in a miscible organic solvent is added to a non solvent (e.g., n hexane), leading to precipitation of the complex, which is then filtered and dried28,30,31
- Solvent Ether Injection
Solvent ether (or solvent) injection involves injecting an organic solution of phospholipid and phytoconstituent into an aqueous phase, where rapid diffusion of solvent induces spontaneous formation of nano sized phytosomes 27,28
- Freeze Drying / Co Solvent Lyophilization
To improve stability and handling, these primary complexes can be further processed by freeze drying or co solvent lyophilization, in which the organic solvent is removed and the dispersion is lyophilized, often with cryoprotectants27–29
- Spray Drying / Vacuum Drying
Spray drying or vacuum drying are also used to obtain free flowing phytosomal powders suitable for solid oral dosage forms. 29,30Choice of method affects particle size, entrapment efficiency, yield and release behaviour, and is selected according to the herbal drug’s solubility and the intended route (oral, topical, transdermal).28,33,34
- KEY CHARACTERISATION / EVALUATION PARAMETERS FOR PHYTOSOMES
For herbal phytosome products, characterisation focuses on confirming complex formation, nanosystem quality, and in vitro performance.
1. Basic physicochemical characterisation
Core critical quality attributes:
-
- Particle size & size distribution (PDI)
Dynamic light scattering is routinely used to determine mean size and polydispersity; size strongly influences stability, skin/oral absorption and biodistribution2,9,35
-
- Zeta potential (surface charge)
Indicates electrostatic stability; sufficient magnitude (positive or negative) helps prevent aggregation and predicts shelf stability and behavior at biological interfaces 2,35,36
-
- Morphology and lamellarity
Transmission and scanning electron microscopy (TEM/SEM) provide information on vesicle shape, surface, and uni-/multilamellar structure, important for release behavior and stability.2,9,37
-
- Chemical composition and complex formation
FTIR, DSC, NMR and X ray diffraction are used to confirm interactions (H bonding, ionic interactions) between phytoconstituents and phospholipids and to distinguish true phytosomes from simple physical mixtures.9,19,37
2. Encapsulation and drug loading
-
- Encapsulation efficiency (EE%) and drug loading
Quantified (typically by HPLC/UV) after separating free drug (dialysis, ultracentrifugation); EE% is a key indicator of formulation success and strongly affects dose and bioavailability.9,35,36
3. In vitro performance tests
Dialysis or diffusion studies in biorelevant media are used to determine release kinetics; sustained or improved release vs. crude extract is usually targeted.9,12,35
Monitoring size, PDI, zeta potential, EE%, and chemical integrity over time under different temperatures, pH, and light conditions evaluates physical/chemical stability and suitability for scale up.2,9,36,38
4. Biological evaluation (preclinical/clinical)
-
- Permeation/uptake studies
Cell or skin models (for topical) and Caco 2/intestinal models (for oral) assess enhanced permeability versus plain extracts.7,9,39
-
- Pharmacokinetics and bioavailability
Animal or human studies measuring Cmax, AUC, and t½ show consistently higher systemic exposure for phytosomes of curcumin, silymarin, quercetin and others compared with non complexed extracts.6,9,24,29
-
- Pharmacodynamic/therapeutic endpoints
Disease specific models (anti inflammatory, anticancer, hepatoprotective, metabolic, etc.) compare efficacy and required dose versus conventional herbal formulations.8,9,12,24
Cytotoxicity assays and in vivo toxicity profiles confirm that phospholipid complexes remain biocompatible and do not introduce new adverse effects relative to the parent herb.24,36,38
- ADVANTAGES AND LIMITATIONS PHYTOSOMES FOR HERBAL PRODUCTS
Phytosomes are phospholipid–plant extract complexes designed to overcome poor bioavailability of many herbal actives.
Advantages
- Greatly enhanced bioavailability and absorption of poorly absorbed polyphenols (e.g., silymarin, curcumin, quercetin, resveratrol) versus crude extracts or simple capsules 9,35,40
- Improved solubility and permeability across lipid?rich biological membranes (gut, skin), giving higher tissue levels at lower doses7,8,12,35
- Protection from degradation (acid, enzymes, oxidation), leading to better stability and shelf life than conventional herbal formulations7,24,35
- Dose sparing and fewer side?effects: higher pharmacological activity at reduced doses, with good biocompatibility due to use of physiological phospholipids 9,34,35,40,41
- Versatile dosage forms: can be formulated as capsules, tablets, syrups, gels, creams and cosmetics 7,29,35
- Food?grade/ nutraceutical positioning and existence of successful marketed products (e.g. Siliphos, Meriva, Greenselect, Casperome) support translational potential 7,9,35
Limitations and challenges
- Formulation and stability issues: some systems are pH?sensitive, prone to instability or rapid drug leakage, complicating large?scale production and storage 7,9,12,42,43
- Scale?up and manufacturing control: maintaining particle size, loading and reproducibility during industrial production is difficult; quality control must be rigorous9,42
- Limited and heterogeneous clinical evidence: many data are preclinical; few well?designed head?to?head clinical trials versus non?phytosomal extracts exist, so superiority is not fully established across indications9,40
- Regulatory and cost barriers: classification between drug/nutraceutical, lack of specific guidelines, and higher production costs may restrict broad adoption24,42,43
- Safety still needs long?term validation: components are generally regarded as safe, but more chronic?use and high?dose clinical studies are required38,43
- MARKETED PHYTOSOMAL PRODUCTS FOR HERBAL DRUGS
A number of phytosome?based herbal products are already commercialized, mainly through the Indena “phytosome/ herbosome/ planterosome” platforms and similar technologies.


 Rahana Raveendran*
Rahana Raveendran*
 10.5281/zenodo.18817283
10.5281/zenodo.18817283