
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Channabasweshwar pharmacy college latur
Heart failure with preserved ejection fraction (HFpEF) has emerged as the predominant phenotype of heart failure globally, largely due to aging populations and the rise in cardiometabolic diseases. Once considered primarily a mechanically driven disorder characterized by impaired ventricular relaxation, HFpEF is now understood as a complex multisystem syndrome encompassing structural, metabolic, inflammatory, and electrophysiological abnormalities. Among these emerging aspects, QT interval prolongation has garnered significant clinical attention as an indicator of delayed ventricular repolarization and arrhythmic vulnerability. QT prolongation is an integrated manifestation of myocardial remodeling, autonomic imbalance, ion channel dysfunction, electrolyte disturbances, and pharmacological exposure. Increasing clinical evidence indicates that patients with HFpEF exhibit heightened susceptibility to both spontaneous and drug-induced QT prolongation compared to individuals without heart failure. Despite preserved systolic function, these patients frequently experience ventricular arrhythmias and sudden cardiac death. This comprehensive review synthesizes current mechanistic, clinical, and pharmacological evidence linking HFpEF to QT prolongation. Particular emphasis is placed on electrophysiological remodeling, microvascular disease, polypharmacy, monitoring strategies, precision medicine approaches, and the expanding role of clinical pharmacists in cardiotoxicity prevention[1, 2].
Heart failure continues to be a predominant contributor to global morbidity and mortality. Over the past two decades, there has been a significant shift in epidemiological trends, with heart failure with preserved ejection fraction (HFpEF) now constituting approximately half of all heart failure cases. In contrast to heart failure with reduced ejection fraction (HFrEF), HFpEF is characterized by preserved left ventricular systolic function, yet impaired diastolic relaxation and increased ventricular stiffness. Initially conceptualized as a purely hemodynamic disorder, HFpEF is now recognized as a systemic inflammatory syndrome, encompassing endothelial dysfunction, metabolic dysregulation, obesity-related inflammation, renal impairment, and neurohormonal activation. [3]These processes collectively facilitate myocardial fibrosis and electrical remodeling. The QT interval, which represents the duration of ventricular depolarization and repolarization, has emerged as a significant electrophysiological biomarker. Prolongation of the QT interval indicates delayed myocardial recovery and heightens susceptibility to torsades de pointes and malignant ventricular arrhythmias. Although extensively investigated in HFrEF, QT abnormalities in HFpEF have only recently garnered attention, highlighting a substantial gap in cardiotoxicity research.[4]
Among electrocardiographic markers, the QT interval has emerged as a significant indicator of ventricular electrical stability. The QT interval reflects the duration required for ventricular depolarization and repolarization, thereby representing the period of electrical systole. Prolongation of this interval signifies delayed recovery of myocardial cells following contraction, thereby increasing susceptibility to life-threatening arrhythmias..
Numerous clinical conditions are known to prolong the QT interval, including electrolyte imbalances, genetic channelopathies, myocardial ischemia, and drug exposure. Recent studies have identified heart failure with preserved ejection fraction (HFpEF) as a potential independent risk factor for QT prolongation. Structural myocardial remodeling, fibrosis, and ion channel alterations associated with HFpEF may disrupt the intricate balance of ionic currents essential for ventricular repolarization[5]. Patients with HFpEF are frequently prescribed multiple medications, including diuretics, antiarrhythmics, antibiotics, psychotropic agents, and proton pump inhibitors, many of which have the potential to prolong the QT interval. The combination of polypharmacy, renal dysfunction, and electrolyte imbalance further exacerbates the risk of drug-induced QT prolongation. From a pharmacological perspective, recognizing HFpEF as a QT-vulnerable phenotype has significant implications for medication safety, drug development, and clinical monitoring strategies. Clinicians must judiciously weigh the therapeutic benefits against potential cardiotoxic risks when prescribing medications that affect ventricular repolarization. [6]Advances in digital health technologies, artificial intelligence-assisted electrocardiography, and wearable cardiac monitoring devices present new opportunities for the earlier and more accurate detection of QT abnormalities. These innovations may ultimately facilitate precision medicine approaches for arrhythmia prevention in patients with HFpEF. This review aims to provide a comprehensive and integrative discussion of QT prolongation risks in HFpEF. By synthesizing evidence from clinical studies, electrophysiological research, and pharmacological investigations, this article underscores the increasing significance of repolarization abnormalities as determinants of cardiovascular outcomes in HFpEF.[4]
.2. GLOBAL EPIDEMIOLOGY AND CLINICAL BURDEN
Heart failure with preserved ejection fraction (HFpEF) predominantly affects older adults, particularly women with comorbid conditions such as hypertension, obesity, diabetes mellitus, atrial fibrillation, and chronic kidney disease. These comorbidities collectively constitute the cardiovascular-kidney-metabolic (CKM) syndrome, which is a primary factor in the development of HFpEF. Despite the preservation of ejection fraction, hospitalization rates remain elevated. The impairment of quality of life, exercise intolerance, and frequent hospital readmissions contribute significantly to the healthcare burden. Notably, sudden cardiac death represents a substantial portion of mortality, challenging previous assumptions that HFpEF is associated with a low risk of arrhythmias. [7]Recent observational studies have identified increased ventricular ectopy and nonsustained ventricular tachycardia in HFpEF populations, underscoring electrical instability as a critical factor influencing outcomes..[5]
3. PHYSIOLOGY OF VENTRICULAR REPOLARIZATION
Ventricular repolarization is contingent upon the coordinated activity of sodium, calcium, and potassium channels. The rapid (IKr) and slow (IKs) delayed rectifier potassium currents are instrumental in restoring membrane potential following depolarization. Disruptions in these currents can lead to a prolonged action potential duration and an extended QT interval. QT correction formulas are employed to adjust for heart rate variability. Notably, QTc values exceeding 500 ms are associated with a significantly increased risk of torsades de pointes. It is particularly important to note that patients with heart failure with preserved ejection fraction (HFpEF) frequently exhibit autonomic dysfunction, thereby necessitating precise QT interpretation.[4]
4. PATHOPHYSIOLOGY OF QT PROLONGATION IN HFpEF4.1 Structural Remodeling
The myocardium in heart failure with preserved ejection fraction (HFpEF) exhibits concentric hypertrophy and widespread interstitial fibrosis. This fibrosis disrupts electrical continuity, resulting in heterogeneous conduction velocities and dispersion of repolarization. Such heterogeneity creates a substrate conducive to the formation of re-entry circuits[8]
.4.2 Ionic Remodeling
Reduced expression of potassium channels decreases the repolarization reserve. Concurrently, impaired calcium reuptake results in intracellular calcium overload, which subsequently triggers early after depolarizations..[9]
4.3 Autonomic imbalance Chronic
Sympathetic activation extends the duration of action potentials via beta-adrenergic signaling pathways. A reduction in vagal tone further destabilizes electrical recovery..[10]
4.4 Inflammation and oxidative stress Inflammatory
Cytokines influence the expression of ion channels and the energetics of mitochondria. Oxidative stress compromises membrane stability, thereby increasing susceptibility to arrhythmias.[11].
4.5 Microvascular Dysfunction
Coronary microvascular disease results in chronic ischemia in the absence of epicardial obstruction. The recurrent ischemic injury facilitates fibrosis and electrophysiological remodeling[12].
5. ELECTRICAL DIASTOLE AND NOVEL CONCEPTS .6. Patients with DRUG?INDUCED QT PROLONGATION HFpEF
Patients frequently receive multiple medications, thereby increasing the risk of drug-induced QT prolongation. Common contributing factors include class III antiarrhythmics, fluoroquinolone and macrolide antibiotics, psychotropic medications, antiemetics, and proton pump inhibitors. Additionally, drug-drug interactions via cytochrome P450 inhibition further elevate plasma drug concentrations[13].
7. ELECTROLYTE DISTURBANCES
Loop diuretics continue to serve as the primary therapeutic approach for heart failure with preserved ejection fraction (HFpEF); however, they are associated with an increased risk of hypokalemia and hypomagnesemia. Even minor electrolyte imbalances can substantially elevate the risk of torsades de pointes. It is therefore recommended to maintain potassium levels above 4.0 mEq/L and magnesium levels above 2.0 mg/dL to prevent arrhythmias[14].
8. ARRHYTHMIC CONSEQUENCES
QT prolongation increases the risk of torsades de pointes, syncope, ventricular tachycardia, and sudden cardiac death. Patients with HFpEF demonstrate a combination of arrhythmic mechanisms, including bradyarrhythmias and ventricular tachyarrhythmias..[15]
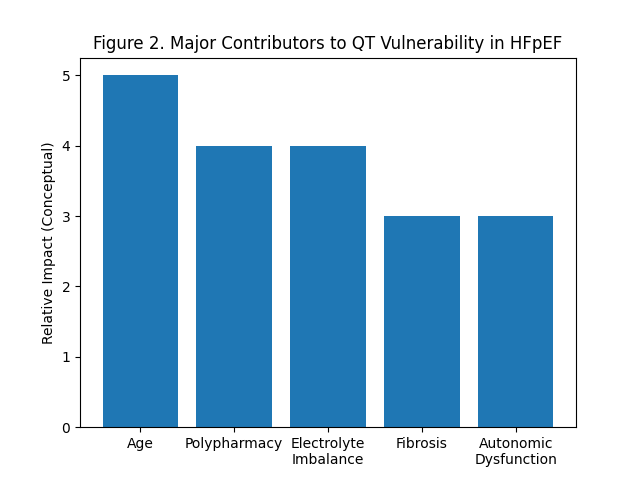
9. Risk factor clustering
Heart failure with preserved ejection fraction (HFpEF) is characterized by the presence of several risk factors for QT interval prolongation, including advanced age, predominance of females, polypharmacy, renal dysfunction, atrial fibrillation, and diuretic therapy. Risk assessment models developed for general cardiac populations may underestimate the susceptibility of individuals with HFpEF to these risks[5].
10. Monitoring strategies Baseline
Before the initiation of QT-prolonging pharmacological agents, an electrocardiogram (ECG) assessment is imperative. It is advisable to conduct serial ECG monitoring during periods of hospitalization and when adjustments to medication are made. Ambulatory ECG devices are capable of detecting transient QT interval changes that may not be captured during routine assessments. Furthermore, implantable devices can offer continuous monitoring for selected individuals at high risk..[16]
11. Artificial intelligence and digital cardiology
AI-driven ECG interpretation facilitates automated QT interval measurement with a high degree of accuracy. Wearable ECG technologies enable remote monitoring and early identification of repolarization abnormalities. The incorporation of AI monitoring into outpatient care for heart failure with preserved ejection fraction (HFpEF) has the potential to decrease hospitalizations and sudden cardiac events.[17]
12. Pharmacotherapy Interactions
Therapies for HFpEF, including diuretics, MRAs, ARNI, and SGLT2 inhibitors, indirectly affect repolarization through electrolyte and renal pathways. The optimization of medication requires a multidisciplinary approach. Clinical pharmacists play a crucial role in identifying QT risk, preventing drug interactions, and guiding dose adjustments.[18]
13. PRECISION MEDICINE APPROACH
Future management of HFpEF is anticipated to integrate biomarker-guided therapy, genetic risk profiling, and electrophysiological phenotyping. The creation of QT-prediction models specific to HFpEF is a significant research priority.[19]
14. RESEARCH GAPS
Key unresolved issues include the absence of randomized QT-guided therapy trials, scarcity of mechanistic human electrophysiology studies, and lack of HFpEF-specific monitoring guidelines.[20]
FUTURE DIRECTIONS
Prospective cohort studies that incorporate electrocardiography (ECG) analytics, imaging biomarkers, and pharmacogenomics are essential. AI-supported remote monitoring has the potential to transform the prevention of cardiotoxicity.[21]
CONCLUSION
Heart failure with preserved ejection fraction (HFpEF) should no longer be considered electrically benign. The interplay of structural stiffness, ionic remodeling, inflammation, and pharmacological exposure culminates in a phenotype vulnerable to QT interval prolongation. Recognizing QT prolongation as a fundamental aspect of HFpEF pathophysiology facilitates enhanced risk stratification and more informed therapeutic decision-making. A multidisciplinary approach integrating cardiology, clinical pharmacy, electrophysiology, and digital health technologies represents the next frontier in preventing cardiotoxicity and improving patient outcomes in HFpEF.
REFERENCES



Shruti Kore, Janhavi Kaile, Swarupa Dongare, QT Prolongation Risks in HFpEF: The Next Frontier in Cardiotoxicity, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 3625-3631, https://doi.org/10.5281/zenodo.19253316
 10.5281/zenodo.19253316
10.5281/zenodo.19253316