Abstract
A simple structure found in naturally occurring compounds, chalcone has been utilised in medicinal chemistry to generate drugs that show promise in treating a range of illnesses. Here, we discuss the synthesis, properties, and structural diversity of chalcones. We next go over how chalcones are made and their biological properties, including their antibacterial, anti-inflammatory, anti-cancer, and antioxidant properties, highlighting the relationships between structure and activity. Chalcone derivatives continue to enthral medical scientists due to their straightforward synthesis, straightforward chemistry, ease of working with hydrogen atoms, and a number of intriguing biological functions. Chalcones have not yet gotten the recognition they merit despite their enormous potential as chemical feedstock for the creation and development of new, potent drugs.
Keywords
Chalcone, Claisen-Schmidt condensation, Antioxidant, Anticancer, Anti-inflammatory, Anti-bacterial.
Introduction
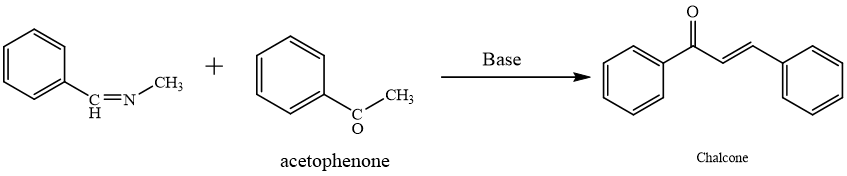
Chalcones are naturally occurring substances in various plants, including vegetables, fruits, drinks, and more.1 Term "chalcone" derives from Greek word "chalcos," which translates to "bronze." referring to the colors found in most natural chalcones. 2 These include common in consumable plants and serve as precursors of flavonoids as well as isoflavonoids.3 Chalcones possess conjugated double bonds as well as a delocalized pi-electron system spanning both benzene rings. Molecules with this structure often have lower redox potentials and are more likely to conduct electron transfer processes. 4 Pure chalcones from several plants had licensed for medical studies to treat cancer and viral, and cardiovascular illnesses, demonstrating potential pharmacological action.5
Chemistry: -
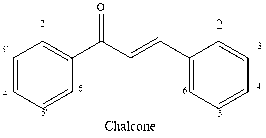
Many chalcones found in nature have aryl rings that are polyhydroxylated. There are two isomeric forms of chalcones: cis and trans, with the transform being thermodynamically advantageous.6 However, as bioactivity is lost when this structural characteristic is removed, the common alpha-beta unsaturated ketone moiety is probably in charge of the many biological activities shown.7 Novel bioactive chalcone analogs compounds had created by altering both rings while leaving this moiety unaltered.8 Chalcones are often created via “Claisen-Schmidt condensation” of aldehyde as well as ketone, which can be catalyzed by acid or base and then dehydrated to produce chalcones.9
Synthesis: -
General Procedure:-
With stirring, 5 mmol of acetophenone and 5 mmol of benzaldehyde derivatives were dissolved in 10 ml of methanol. To produce a blood-red solution, quantities of potassium hydroxide (15 mmol) were added.10 For eight to twelve hours, resultant solution was swirled, causing chalcone to precipitate as potassium salt.11 After adding more concentrated HCl until solution became acidic, solution or suspension was poured into 10 milliliters of cold 1 N HCl.12 Product was obtained by filtering the resultant yellow solid, washing it with two 20 ml batches of water, and then recrystallizing it from appropriate solvent.13
Claisen-Schmidt condensation: -
The most popular and widely used method for creating chalcone is Claisen-Schmidt condensation, which condenses ketone and aldehyde with less work-up and fewer byproducts.14 Schmidt condensation is the most dominating method employing a diversity of catalysts such as lithium aluminum hydride, Potassium carbonate, and Pot. Hydroxide, Sodium Hydroxide, Aluminium Chloride, and Silica-Sulphuric acid.15 Here aldehyde and ketone react in presence of base in organic solvent and produce Chalcone with 60-80% yield.16
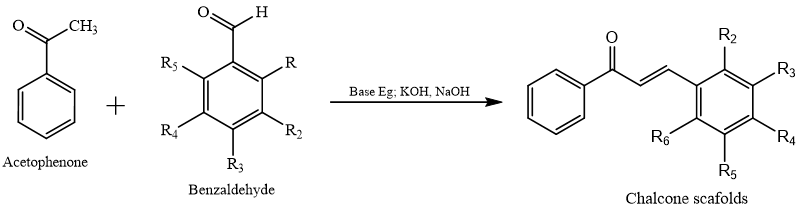
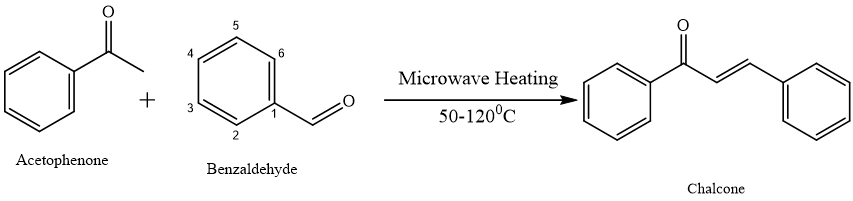
Microwave-assisted synthesis of chalcone:-17
Microwave-assisted synthesis is an efficient and eco-friendly method for preparing chalcones, which are important intermediates in medicinal and material chemistry.18,19 The reaction involves condensing an aromatic aldehyde with acetophenone in presence of a base catalyst like NaOH or KOH.20 Mix reactants with a base, optionally in ethanol or under solvent-free conditions.21 Irradiate mixture at 50–120°C for 2–10 minutes.[20] Cool, precipitate, and recrystallize product by using the ethanol.22, 23
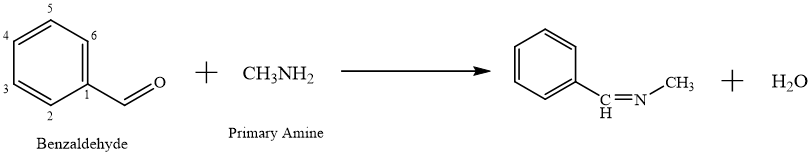
Synthesis of chalcones using Schiff bases: -
Chalcones synthesis via Schiff bases is an alternative approach in organic synthesis, often leveraging the pre-formed Schiff base as a starting material or an intermediate.24 Schiff bases, Imine derivatives produced from condensation of primary amines with aldehydes or ketones, can react further to form chalcones under suitable conditions.25 it is a two-step process where an aromatic aldehyde with a primary amine forms a Schiff base.26 Schiff base undergoes a reaction with ketone in presence of a basic catalyst like NaOH and KOH, undergoing an aldol condensation to produce the chalcone.27
Recrystallization Process: -
Recrystallization is process where we isolate desired compound from a racemic mixture or mixed compound.28 Here we recrystallized chalcone derivative in various organic solvents based on their solubility.29, 30 Compounds 001 002 and 003 were recrystallized in methanol and compound 004 was recrystallized in ethanol and chloroform and compound 005 was recrystallized on methanol and Dichloromethane.31
Pharmacological Activities: -
- Antioxidant Activity: -
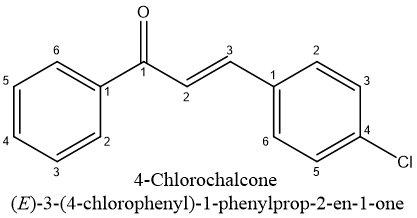
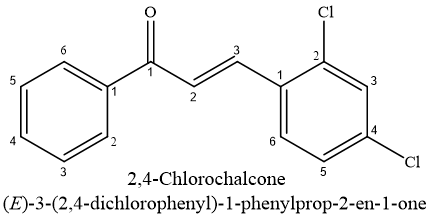
Many antioxidants are produced by plants to protect molecules against reactive oxygen species, as well as main class of antioxidants obtained from plants is made up of phenolics.31,33 The flavonoids are arguably the most significant class of phenolic chemicals.34 They are capable of scavenging free radicals as well as to stop lipid peroxidation.35 The antioxidant potential is present in a large number of hydroxy chalcones with isoprenyl substituents on their phenyl rings.36 According to induction period measurements, chalcones, including 4-Chlorochalcone (001) and 2,4 Dichlorochalcone (002), are very potent antioxidants within concentration range of 0.025–0.1%.37
Anticancer Activity: -
Recent publications highlighting anti-proliferative as well as tumor-reducing properties of certain chalcones have heightened interest in this family of chemicals. Additionally, it has been established that substantial consumption of plant-based dietary flavonoids seems safe yet not linked to negative health effects.38,39 Chalcones represent a notable class of naturally occurring small molecules that were beneficial in cancer chemotherapy among currently recognized anticancer agents.40,41 According to sulforhodamine staining, 2,3-Dichlorochalcone might be useful as chemopreventive medication during prostate carcinogenesis, prostate hyperplasia, as well as decreased proliferation(IC50 between 26 & 12?M for 24,48, & 72 h).42 Studies on the toxicity of compounds to animals suggested that compound 002 might not be dangerous to people.43 Several chlorochalcones for their modes of action and anticancer properties. Compound 002 had strongest growth inhibitory efficacy of five chalcone derivatives examined, exhibiting IC50 values 1.5 & 0.6M in colony formation assay & (MTT) assay, correspondingly.44,45

Anti-inflammatory Activity: -
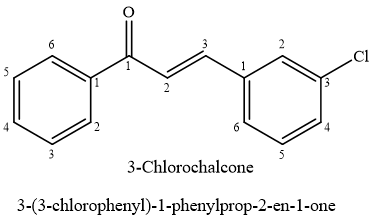
Inflammation constitutes multifaceted biological reaction to damaging factors that may result in various pathological conditions.46 Upon exposure to inflammatory triggers, macrophages produce pro-inflammatory substances, such as the transient free radical nitric oxide.47,48 Compound 001 was one of various chalcone derivatives evaluated for possible anti-inflammatory properties.49,50 It effectively scavenges superoxide anion created through activated human neutrophils or hypoxanthine/xanthine oxidase system(HX/XO), exhibiting IC50 values, 0.1 & 0.3M, correspondingly.51 Investigated the inhibitory effects of dimethyl amino-chalcones on creation of NO & PGE2 mediators within RAW 264.7 macrophage cells treated with lipopolysaccharide (LPS).52
Anti-bacterial Activity: -
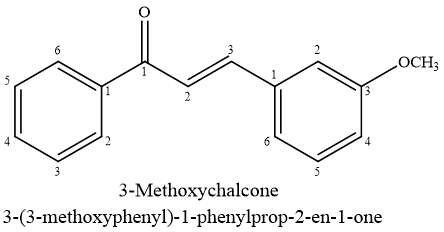
Rapid resistance in Gram-positive bacteria presents a considerable safety risk.53 Methoxy-chalcone and chalcone exhibited strong antimicrobial activity against Staphylococcus aureus, Micrococcus luteus, as well as Bacillus subtilis, exhibiting MIC values varying from 3.13 to 12.5 g/mL.54,55 Antibacterial action was seen on MRSA strains(OM481,OM505) exhibit MIC values 16~g/mL, while MRSA strains (K3& ST28) demonstrate MIC values 6.25~g/mL.56,57 The antibacterial activity of methoxy-chalcone analogs against S. aureus was found to be dependent on free hydroxyl group at 4-position in ring A. Elimination of lipophilic prenyl group leads to complete lack of activity.58
CONCLUSION: -
Present study shows that Chalcone derivatives have many Pharmacological activities that include anti-inflammatory, anti-cancer, antimicrobial, as well as antioxidant activities. This study also shows that synthesis of chalcone is done majorly via the Claisen-Schmidt condensation method. Chlorochalcone gives antioxidant activity while methoxy chalcone gives anti-bacterial activity. This study also shows that position of Chloro group in chalcone structure poses the anticancer activity. Moreover, significant actions, including anti-obesity, anti-hypertensive, as well as hepatoprotective effects, suggest that chalcone may serve as promising source of biologically active chemicals for these conditions.
REFERENCES
- Zhuang C, Zhang W, Sheng C, Zhang W, Xing C, Miao Z. Chalcone: a privileged structure in medicinal chemistry. Chemical reviews. 2017 Jun 28;117(12):7762-810.
- K Sahu N, S Balbhadra S, Choudhary J, v Kohli D. Exploring pharmacological significance of chalcone scaffold: a review. Current medicinal chemistry. 2012 Jan 1;19(2):209-25.
- Rahman A, Qureshi R, Kiran M, Ansari FL. Electron affinities, solvation energies and redox potentials of some chalcones: substituentseffect and correlation with semi-empirical MO energies. Turkish Journal of Chemistry. 2007;31(1):25-34.
- Patil CB, Mahajan SK, Katti SA. Chalcone: A versatile molecule. Journal of Pharmaceutical sciences and research. 2009 Sep 1;1(3):11.
- K Sahu N, S Balbhadra S, Choudhary J, v Kohli D. Exploring pharmacological significance of chalcone scaffold: a review. Current medicinal chemistry. 2012 Jan 1;19(2):209-25.
- Guida A, Lhouty MH, Tichit D, Figueras F, Geneste P. Hydrotalcites as base catalysts. Kinetics of Claisen-Schmidt condensation, intramolecular condensation of acetonylacetone and synthesis of chalcone. Applied Catalysis A: General. 1997 Dec 23;164(1-2):251-64.
- Romanelli G, Pasquale G, Sathicq Á, Thomas H, Autino J, Vázquez P. Synthesis of chalcones catalyzed by aminopropylated silica sol–gel under solvent-free conditions. Journal of Molecular Catalysis A: Chemical. 2011 Apr 23;340(1-2):24-32.
- Elias DW, Beazely MA, Kandepu NM. Bioactivities of chalcones. Current medicinal chemistry. 1999;6(12):1125.
- Batovska DI, Todorova IT. Trends in utilization of the pharmacological potential of chalcones. Current clinical pharmacology. 2010 Feb 1;5(1):1-29.
- Farooq S, Ngaini Z. Recent synthetic methodologies for chalcone synthesis (2013-2018). Current Organocatalysis. 2019 Sep 1;6(3):184-92.
- Jung JC, Lee Y, Min D, Jung M, Oh S. Practical synthesis of chalcone derivatives and their biological activities. Molecules. 2017 Nov 1;22(11):1872.
- Barnard DL, Smee DF, Huffman JH, Meyerson LR, Sidwell RW. Antiherpesvirus activity and mode of action of SP-303, a novel plant flavonoid. Chemotherapy. 1993 Sep 11;39(3):203-11.
- Tran TD, Park H, Ecker GF, Thai KM. 2’-Hydroxychalcone Analogues: Synthesis and Structure-PGE2 Inhibitory Activity Relationship. In12th International Electronic Conference on Synthetic Organic Chemistry (ECSOC-12) 2008 Nov 20 (pp. 1-30).
- Alam S, Mostahar S. Studies of antimicrobial activity of two synthetic 2?, 4?, 6?-trioxygenated flavones. Journal of Applied Sciences. 2005 Feb;5(2):327-33.
- Capasso A, Pinto A, Mascolo N, Autore G, Capasso F. Reduction of agonist?induced contractions of guinea?pig isolated ileum by flavonoids. Phytotherapy Research. 1991 Apr;5(2):85-7.
- Ram VJ, Saxena AS, Srivastava S, Chandra S. Oxygenated chalcones and bischalcones as potential antimalarial agents. Bioorganic & medicinal chemistry letters. 2000 Oct 2;10(19):2159-61.
- Alcaraz MJ, Jimenez MJ. Flavonoids as anti-inflammatory agents.
- Abe I, Sano Y, Takahashi Y, Noguchi H. Site-directed mutagenesis of benzalacetone synthase: the role of Phe215 in plant type III polyketide synthases. Journal of Biological Chemistry. 2003 Jul 4;278(27):25218-26.
- Adole VA, Jagdale BS, Pawar TB, Sagane AA. Ultrasound promoted stereoselective synthesis of 2, 3-dihydrobenzofuran appended chalcones at ambient temperature. South african journal of chemistry. 2020;73(1):35-43.
- Avila-Villarreal G, Hernández-Abreu O, Hidalgo-Figueroa S, Navarrete-Vázquez G, Escalante-Erosa F, Peña-Rodríguez LM, Villalobos-Molina R, Estrada-Soto S. Antihypertensive and vasorelaxant effects of dihydrospinochalcone-A isolated from Lonchocarpus xuul Lundell by NO production: Computational and ex vivo approaches. Phytomedicine. 2013 Nov 15;20(14):1241-6.
- Awasthi SK, Mishra N, Kumar B, Sharma M, Bhattacharya A, Mishra LC, Bhasin VK. Potent antimalarial activity of newly synthesized substituted chalcone analogs in vitro. Medicinal Chemistry Research. 2009 Jul;18:407-20.
- Balu P, Jas JS, Govindaraj M. Design and evaluation of chalconeimine derivatives as ?-amylase inhibitors. Bioinformation. 2019;15(7):523.
- Bandgar BP, Gawande SS, Bodade RG, Totre JV, Khobragade CN. Synthesis and biological evaluation of simple methoxylated chalcones as anticancer, anti-inflammatory and antioxidant agents. Bioorganic & medicinal chemistry. 2010 Feb 1;18(3):1364-70.
- Selepe MA, Van Heerden FR. Application of the Suzuki-Miyaura reaction in the synthesis of flavonoids. Molecules. 2013 Apr 22;18(4):4739-65.
- Daskiewicz JB, Comte G, Barron D, Di Pietro A, Thomasson F. Organolithium mediated synthesis of prenylchalcones as potential inhibitors of chemoresistance. Tetrahedron letters. 1999 Sep 24;40(39):7095-8.
- Babu MA, Shakya N, Prathipati P, Kaskhedikar SG, Saxena AK. Development of 3D-QSAR models for 5-lipoxygenase antagonists: Chalcones. Bioorganic & medicinal chemistry. 2002 Dec 1;10(12):4035-41.
- Wilhelm A, Bonnet SL, Twigge L, Rarova L, Stenclova T, Visser HG, Schutte-Smith M. Synthesis, characterization and cytotoxic evaluation of chalcone derivatives. Journal of Molecular Structure. 2022 Mar 5;1251:132001.
- Pérez-González A, Castañeda-Arriaga R, Guzmán-López EG, Hernández-Ayala LF, Galano A. Chalcone derivatives with a high potential as multifunctional antioxidant neuroprotectors. ACS omega. 2022 Oct 18;7(43):38254-68.
- Tran TD, Park H, Ecker GF, Thai KM. 2’-Hydroxychalcone Analogues: Synthesis and Structure-PGE2 Inhibitory Activity Relationship. In12th International Electronic Conference on Synthetic Organic Chemistry (ECSOC-12) 2008 Nov 20 (pp. 1-30).
- Ohkatsu Y, Satoh T. Antioxidant and photo-antioxidant activities of chalcone derivatives. Journal of the Japan Petroleum Institute. 2008;51(5):298-308.
- Iqbal H, Prabhakar V, Sangith A, Chandrika B, Balasubramanian R. Synthesis, anti-inflammatory and antioxidant activity of ring-A-monosubstituted chalcone derivatives. Medicinal Chemistry Research. 2014 Oct;23:4383-94.
- Mazzone G, Malaj N, Galano A, Russo N, Toscano M. Antioxidant properties of several coumarin–chalcone hybrids from theoretical insights. Rsc Advances. 2015;5(1):565-75.
- Haider K, Haider MR, Neha K, Yar MS. Free radical scavengers: An overview on heterocyclic advances and medicinal prospects. European Journal of Medicinal Chemistry. 2020 Oct 15;204:112607.
- Karthikeyan C, SH Narayana Moorthy N, Ramasamy S, Vanam U, Manivannan E, Karunagaran D, Trivedi P. Advances in chalcones with anticancer activities. Recent patents on anti-cancer drug discovery. 2015 Jan 1;10(1):97-115.
- Brglez Mojzer E, Knez Hrn?i? M, Škerget M, Knez Ž, Bren U. Polyphenols: Extraction methods, antioxidative action, bioavailability and anticarcinogenic effects. Molecules. 2016 Jul 11;21(7):901.
- Ahmed FF, Abd El-Hafeez AA, Abbas SH, Abdelhamid D, Abdel-Aziz M. New 1, 2, 4-triazole-Chalcone hybrids induce Caspase-3 dependent apoptosis in A549 human lung adenocarcinoma cells. European journal of medicinal chemistry. 2018 May 10;151:705-22.
- Ketelut-Carneiro N, Fitzgerald KA. Apoptosis, pyroptosis, and necroptosis—Oh my! The many ways a cell can die. Journal of molecular biology. 2022 Feb 28;434(4):167378.
- Jiang M, Qi L, Li L, Li Y. The caspase-3/GSDME signal pathway as a switch between apoptosis and pyroptosis in cancer. Cell death discovery. 2020 Oct 28;6(1):112.
- Zhuang C, Zhang W, Sheng C, Zhang W, Xing C, Miao Z. Chalcone: a privileged structure in medicinal chemistry. Chemical reviews. 2017 Jun 28;117(12):7762-810.
- Herencia F, Ferrandiz ML, Ubeda A, Domínguez J, Charris JE, Lobo GM, Alcaraz MJ. Synthesis and anti-inflammatory activity of chalcone derivatives. Bioorganic & medicinal chemistry letters. 1998 May 19;8(10):1169-74.
- Goyal K, Kaur R, Goyal A, Awasthi R. Chalcones: A review on synthesis and pharmacological activities. Journal of Applied Pharmaceutical Science. 2021 Feb 11;11(1):001-14.
- Yang S, Liu X, Wang S, Song J, Wu J, Shen B, Jia H, Guo S, Wang Y, Yang Y, Jiang Y. Chalcone derivatives as dual-sensitive fluorescent probe for protein aggregation investigation. Sensors and Actuators B: Chemical. 2023 Nov 15;395:134514.
- Russell AE, Gines BR. Chalcones: potential chemotherapeutic compounds and educational tools for closing the loop in STEM. Accounts of Chemical Research. 2023 Jan 25;56(11):1256-62.
- Xu M, Wu P, Shen F, Ji J, Rakesh KP. Chalcone derivatives and their antibacterial activities: Current development. Bioorganic Chemistry. 2019 Oct 1;91:103133.
- Dan W, Dai J. Recent developments of chalcones as potential antibacterial agents in medicinal chemistry. European journal of medicinal chemistry. 2020 Feb 1;187:111980.
- Rajendran G, Bhanu D, Aruchamy B, Ramani P, Pandurangan N, Bobba KN, Oh EJ, Chung HY, Gangadaran P, Ahn BC. Chalcone: a promising bioactive scaffold in medicinal chemistry. Pharmaceuticals. 2022 Oct 11;15(10):1250.
- Bhukal A, Kumar V, Kumar L, Lal K. Recent advances in chalcone-triazole hybrids as potential pharmacological agents. Results in Chemistry. 2023 Dec 1;6:101173.
- Zembower DE, Zhang H. Total synthesis of robustaflavone, a potential anti-hepatitis B agent. The Journal of Organic Chemistry. 1998 Dec 11;63(25):9300-5.
- Baker W, Ollis WD. A New Synthesis of iso Flavones. Nature. 1952 Apr 26;169(4304):706-.
- Tsoi YT, Zhou Z, Chan AS, Yu WY. Palladium-catalyzed oxidative cross-coupling reaction of arylboronic acids with diazoesters for stereoselective synthesis of (E)-?, ?-diarylacrylates. Organic Letters. 2010 Oct 15;12(20):4506-9.
- Shou QY, Tan Q, Shen ZW. Hirtellanines A and B, a pair of isomeric isoflavonoid derivatives from Campylotropis hirtella and their immunosuppressive activities. Bioorganic & medicinal chemistry letters. 2009 Jul 1;19(13):3389-91.
- Li Y, Qi Z, Wang H, Fu X, Duan C. Palladium-catalyzed oxidative Heck coupling reaction for direct synthesis of 4-arylcoumarins using coumarins and arylboronic acids. The Journal of Organic Chemistry. 2012 Feb 17;77(4):2053-7.
- Zhang L, Meng T, Fan R, Wu J. General and efficient route for the synthesis of 3, 4-disubstituted coumarins via Pd-catalyzed site-selective cross-coupling reactions. The Journal of Organic Chemistry. 2007 Sep 14;72(19):7279-86.
- Garazd MM, Garazd YL, Khilya VP. Neoflavones. 2. Methods for synthesizing and modifying 4-arylcoumarins. Chemistry of natural compounds. 2005 May;41:245-71.
- Khare T, Anand U, Dey A, Assaraf YG, Chen ZS, Liu Z, Kumar V. Exploring phytochemicals for combating antibiotic resistance in microbial pathogens. Frontiers in pharmacology. 2021 Jul 21;12:720726.
- Qin HL, Liu J, Fang WY, Ravindar L, Rakesh KP. Indole-based derivatives as potential antibacterial activity against methicillin-resistance Staphylococcus aureus (MRSA). European journal of medicinal chemistry. 2020 May 15;194:112245.
- Faleye OS, Boya BR, Lee JH, Choi I, Lee J, Page C. Halogenated antimicrobial agents to combat drug-resistant pathogens. Pharmacological Reviews. 2024 Jan 1;76(1):90-141.
- Verma R, Verma SK, Rakesh KP, Girish YR, Ashrafizadeh M, Kumar KS, Rangappa KS. Pyrazole-based analogs as potential antibacterial agents against methicillin-resistance staphylococcus aureus (MRSA) and its SAR elucidation. European journal of medicinal chemistry. 2021 Feb 15;212:113134


 Sukanta Debnath*
Sukanta Debnath*










 10.5281/zenodo.14750375
10.5281/zenodo.14750375