Abstract
Colon cancer remains a significant health concern due to limited efficacy and high toxicity of current chemotherapeutic agents. The quest for novel anticancer therapies has led to exploration of natural compounds, including chrysin, which exhibits promising anticancer properties. Chrysin, a flavone found in various natural sources, has been extensively studied for its biological effects, particularly its pro-apoptotic activity and ability to inhibit tumor angiogenesis. In preclinical studies, chrysin has demonstrated efficacy against various cancer cell lines including cervical, breast, prostate, liver, ovarian, lung, leukemic, and colon cancer. However, challenges remain in translating these findings into effective treatments, necessitating further research into chrysin's mechanism of action and optimization of its formulation for enhanced bioavailability. This review summarizes the role of chrysin in colon cancer treatment, highlighting recent experimental data on different chrysin formulations and their efficacy in vitro and in vivo. The findings underscore the potential of chrysin as a therapeutic key for colon cancer and emphasize the need for advanced formulations to improve its solubility and absorption, thereby enhancing its therapeutic efficacy.
Keywords
Colon, Gene Defects, Colorectal Cancer, TNM Staging, Mediterranean Diet, Chrysin.
Introduction
The latest assessment of the International Agency for Research on Cancer (IARC) on the analysis of cancer statistics worldwide by world region for the year 2022 showed presence of 9.3% colorectal cancer of all cancers globally. In 2022, colorectal cancer accounted for about 2.5 million new cases, of which 609,216 were male and 533,006 were female. It was the third most often diagnosed cancer.1 The rate of incidence of colon cancer can be seen constantly rising in highly developed countries due to their changed lifestyles specifically speaking in the South region of Central and Eastern Asia along with the Europe and America.2the study, which gave an idea of the prevalence of colorectal cancer in 204 nations and provinces on a national, regional, and worldwide scale between 1990 and 2019, showed the number of CRC cases worldwide increased from 842,098 to 2,166,168. With a 95% uncertainty interval, linked deaths climbed from 518,126 to 1,085,797 between 1990 and 2019. In 2019, CRC was accountable for 24,284,087 DALYs. From 1990 to 2019, there was a 157% increase in CRC incidence, an 110% increase in fatalities, and a 96% increase in disability-adjusted life years. The highest rates of CRC occurred in East Asia, Equatorial Guinea, and Southern Latin America; globally, men were more likely to report having CRC than women.3Depending on where the metastatic growth is located, surgery and chemotherapeutic administration are part of the traditional treatment for colon cancer. However, in this type of cancer, the risk of death for the patient stays higher because the current chemotherapeutic agents have high toxicity and severe negative effects.4One of the most crucial therapies for cancer patients is chemotherapy. Nonetheless, for the majority of solid tumours, the efficacy of the chemotherapeutic medications is still restricted.5
More than a thousand different medications have been shown to have notable anticancer effects. Yet there is still a need for scientific research to generate novel anticancer medications from a wide range of natural, biological, and synthetic components. Predictions suggest that leveraging natural goods could significantly decrease cancer-related deaths in the 21st century. These products have emerged as crucial sources of anti-cancer agents.6 Because of their pro-apoptotic properties, natural compounds are promising anti-cancer agents.7Many phytochemicals including polyphenols and flavonoids with potential applications in medicines are found in plants. Flavonoids are a family of compounds that are found in meals derived from fruits and veggies and consist of a broad variety of natural pigments. A broad class of polyphenolic compounds, found in many herbal products and food, make up chysin. These compounds have been linked for a long time to a number of significant pharmacological & biochemical actions in the prevention of cancer and maintenance of health.8Chrysin has been demonstrated in several studies to possess a variety of biological properties.9Furthermore, in different cancer cell lines, namely Cervical cancer Hela cells, leukaemia cells L1210 along with HL-60 and U937 have been shown to undergo apoptosis when exposed to chrysin. Additionally, this plant flavonoid has the ability to prevent tumor angiogenesis, a crucial stage in the spread of cancer cells.10In vitro, chrysin has been shown to have an impact on human colon cancer cells' viability.11 There are number of bioactives which have anticancer activity but very few of them are formulated as an effective dosage forms due to the need of thorough study on their mechanism of action. The present review is based on understanding mechanism of the bioactive chrysin and its impact on colon cancer.
COLON OR COLORECTAL CANCER
Colorectal cancer (CRC) typically initiates with the non-cancerous proliferation of mucosal epithelial cells. These growths termed polyps undergo gradual development over a span of 10 to 20 years before progressing to a cancerous state. This sequence of progression is often referred to as the adenoma-carcinoma sequence.12
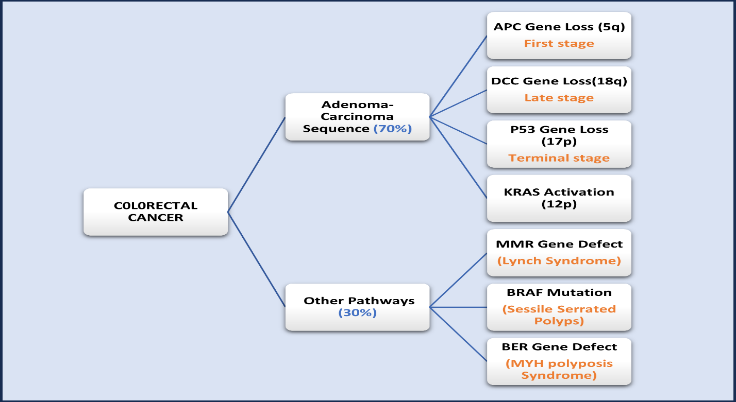
Fig. 1. Colo/ colorectal cancer development pathways through gene defects
In 35–45% of CRC cases, the KRAS oncogene is activated. It is linked to poor survival rates, increased aggressiveness of colorectal cancer, and lower reactivity to several chemotherapeutic drugs, especially those which are epidermal growth factor targeting agents.13 The process of fixing errors created during DNA replication involves MMR genes. Numerous DNA mutations seen in MMR-deficient cells have the potential to cause cancer and MSI. 12–15% of all cases of CRC in the USA are sporadic, involving MSI and somatic mutations in the MMR gene. They almost invariably occur in the right colon. they primarily affect fair toned-middle aged groups with no family history for cancer. a common feature of CRC is methylation of DNA with CpG island and its phenotype methylator. MLH1 methylation linked to CIMP results in isolated instances of MMR deficiency. 3-6% of total CRC cases in the United States are caused by mutations in hMSH2, hMLH1, hPMS1, and hPMS2 genes which are found in hereditary non-polyposis colorectal cancer.14 In CRC cases, the genetic alteration in the BRAF gene causes the emergence of both classic serrated adenoma (TSA) and sessile serrated adenoma (SSA). BRAF-mutated colorectal cancers (CRCs) are typically right-sided, develop in higher age group commonly in women.15 Sporadic microsatellite instability is caused by the CpG island methylator phenotype, which is closely linked to the BRAF mutation in colorectal cancer. CIMP-positive BRAF-mutated colorectal cancer patients shows predominance.16 Thus, tumors that are CIMP-positive are typically the result of serrated adenoma. CIMP-positive tumours have a microsatellite instability rate of about 50%. Ten to twelve percent of total CRCs are CIMP-positive and microsatellite unstable as well as CIMP positive in nature.17
TNM STAGING SYSTEM OF COLON CANCER BY AMERICAN JOINT COMMITTEE ON CANCER (AJCC)
According to latest updated version( 8th Edition published in 2017) under AJCC colon cancer initiates from stage 0 and develops with the progression to stage IVC. It also incorporates quantifiable changes or biologic and genetic markers that personalizes cancer staging.18,19
Table 1. Stages of colorectal cancer by prognostic groups and their criteria (AJCC)19,20,21,22,23,24,25
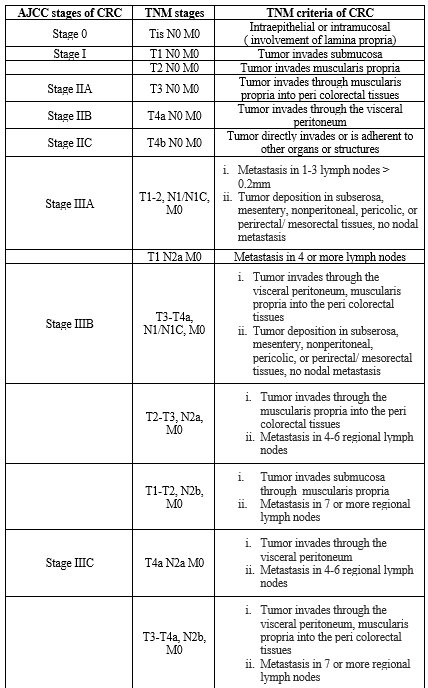
*TNM: Tumor/Nodes/Metastases; Stage 0- Stage IVC; Anatomic stages of colon/ colorectal cancer (CRC); Tis- T4b: prognostic groups representing stages of tumor (T); N0- N2b: prognostic groups representing stages of lymph nodes (N); M0- M1c: prognostic groups representing stages of metastasis (M).
RISK FACTORS RESPONSIBLE FOR COLON CANCER
All the risk factors of colon or colorectal cancer basically alter the pathophysiology of the colonic cells by different mechanisms.26 the most common risk factors are the family history, genetic alterations, obesity, gender, race and ethnicity , High risk genetic syndromes, consumption of alcohol as well as red processed or burned meat , smoking, physical activities and exercises, rapid industrialization and continuously changing life style and diseases like diabetes and inflammatory bowel disease.27,28 Some rare but high-risk factors are acromegaly, hereditary hemochromatosis, ureterosigmoidostomy, childhood cancer, and occupational exposures in firefighting.29 Again, there are multiple severe root causes for development of this type of cancer which may include dietary habits like low-fibre, high-fat diets, insulin resistance, and long-term immunosuppression post renal transplantation. Additionally, androgen deprivation therapy for extended period or family history of colorectal cancer or adenoma, and genetic syndromes such as Serrated Polyposis Syndrome (SPS), familial adenomatous polyposis (FAP), Muir-Torre syndrome, Peutz-Jeghers syndrome, Cowden syndrome, Lynch syndrome, Cronkhite-Canada syndrome, Juvenile Polyposis Syndrome, and non-inherited polyposis syndromes contribute to increased risk.30,31 As per the data published in 2020, their study was documented on 3058 CRC cases which showed that the different parts from colon to rectum pauses varied risk of occurrence of the cancer. It involved sigmoid colon (750 cases), ascending colon (633 cases), rectum (528 cases), cecum (474 cases), transverse colon (250 cases), descending colon (221 cases), rectosigmoid junction (202 cases). the favourable correlations between age and a family history of colorectal cancer with cancer risk were ascertained to be declined from the cecum to the rectum. On the other hand, the inverse relationship with respect to intake of whole grains, cereal fibre and processed red meat was found by endoscopic surveillance from the cecum to the rectum was seen. The correlation between colorectal cancer (CRC) and female sex exhibited a noteworthy non-linear pattern. The risks ratios for ascending and sigmoid colon cancer varied from 1.73 to 0.54. From the cecum to the transverse colon, there was a greater correlation between alcohol use and smoking prior to the age of thirty and proximal colon malignancies. The positive correlation between men's waist circumference and distal colon cancers was stronger for descending than for sigmoid colon cancers.32 Previous research indicates that while male sex and increasing age were found to be significantly associated with increased risk, aspirin use turned out to be strongly linked with lower risk of EOCRC in a case-control study of veterans; the same characteristics were found to alter the risk for CRC onset beyond age 50. One of the early clinical symptoms of EOCRC may be a weight loss.33
Fig. 2. Parts of colon
MEDITERRANEAN DIET (MD)
In case of the colon cancer age around 50 to 70 years is considered to be a common risk factor but the most significant one is the diet.29 the MD is a blend of several dietary behaviours of population regarding their religious, economic, cultural lifestyle and food customs that have dwelled in the Mediterranean region for thousands of years. Extensive research conducted over many decades has examined the MD as a potential to offer protection against numerous chronic illnesses and cancers, such as colorectal cancer.34
TREATMENT FOR COLON CANCER
Chemotherapy, radiation therapy, surgery, cryosurgery are the major treatment options available for colon cancer. The conventional treatments like monotherapy, combined immunotherapy and combined immunotherapy in combination with chemotherapy they all have their possible side effects as they deliver drug to the non-targeted sites which damages both cancerous as well as non-cancerous cells and produces drug resistance during treatments over a long period of time leading to the death of the patients. Since 1957, 5-Fluorouracil (5-FU) continues to be the primary therapeutic approach for colorectal cancer despite of its high toxicity with very low efficacy.35 To overcome these underlying side effects, the replacement of conventional drugs with herbal bioactives has become active research to discover the activity of herbs and their bioactives for their diagnostic and therapeutic values in cancer.36 As conventional chemotherapy for colon cancer has many limitations, the natural phytochemicals have demonstrated benefits over conventional chemotherapy in the treatment of colon cancer.37currently, vinca alkaloids, roscovitine, camptotecin, irinotecan, paclitaxel, docetaxel. podophyllotoxin and colchicine are some of the most commonly used plant-based compounds for their anticancer activities.38
CHRYSIN
A flavone found in honey, propolis, passion fruit and blue passion flower is a chrysin (5, 7-dihydroxyflavone). The two premium, high-quality sources of chrysin are considered to be the propolis and honey. A dried root of the Scutellariae baicalensis Georgi (Radix scutellariae)39, bitter melon (Momordica charantia)40 and an edible mushroom (Pleurotus ostreatus)41, wild Himalayan pear (Pyrus pashia)42 can be utilized for extraction of chrysin.
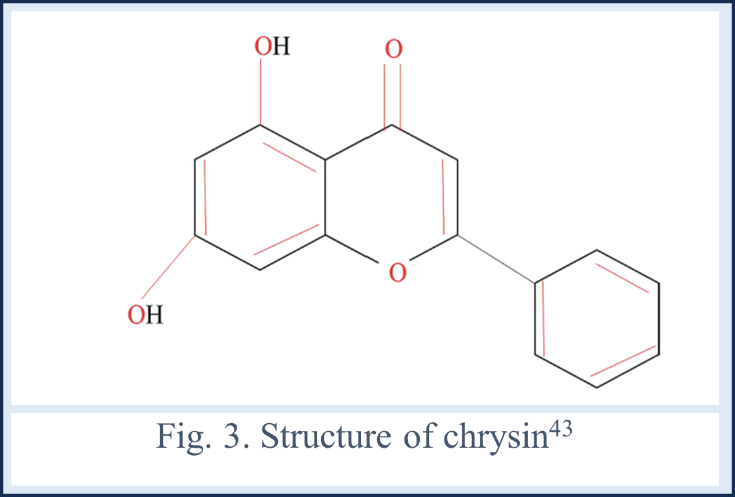
Structurally, the chrysin is classified under the subclass of flavonoids, possesses a distinctive molecular structure with benzene ring A and B along with one central oxygen holding heterocyclic ring. its configuration features a carbonyl group attached to 4rth carbon atom with double bonds, distinguishing it from the other flavonoids with the missing C3H7OH.
Fig. 4. Schematic of major sources and pharmacological activities of chrysin44
Notably, chrysin bears (-OH) group on 5th & 7th carbon, distinguishing it further within its class. Unlike many flavonoids, it does not display oxygenation in B ring, contributing to its unique chemical profile and potentially distinct biological activities.44
DOSE AND TOXICITY
Most people agree that moieties on the A and C rings, where the hydroxyl can have antitoxic effects, are linked to chrysin's pharmacological activity In theory, a moiety like pan-assay interference compounds might be formed by the carbonyl coupled with the double bond at 2nd and 3rd place which could possibly result in toxicities.45Chrysin, however, has been shown in preclinical models to be reasonably safe. A safe dose of chrysin for human is considered 0.5–3.0?g.46 Chrysin was given orally or intravenously, either as a pure molecule or in herbal extracts, at doses ranging from 0.23 to 100 mg/kg in preclinical PK investigations including mice and rats. as a result, herbal extract produced a Tmax of less than one-hour, pure chrysin showed a Tmax of two to six hours, perhaps because of the slower absorption of higher doses. Tmax was considerably reduced by formulations with enhanced water solubility.it is possibly the result of the higher pure chrysin doses than its maximum absorbable dose.47 the half-life of chysin varied from 3 to 11 hours, indicating enterohepatic recycling.48
KINETICS AND BIOAVAILABILITY
according to previous studies on human volunteer with 400 mg chrysin administration, chrysin sulphate was majorly produced in plasma and as compare to this its systemic exposure was 20 folds lower having 64?±?33?ng/mL·hr. of AUC. Chrysin shows elimination through feces and urine. the 90 % and more of it can be seen in form of aglycon in feces. which could be due to the low aqeous solubilty or enterohepatic recycling of chrysin. through urine in shows elimination as a chrysin-glucuronide as well as in its original form without any change in its structure chemically.49 The excreation of chrysin occurs via bile in the form of conjugates like glucuronide and sulfate. this conjugates further in the colon get hydrolyzed to chrysin again due to the action of intestinal microbiome which makes chrysin to have a poor oral bioavailability. while talking about the solubility of chrysin it belongs to BCS class II, poses high permeability but low aqueous solubility giving a poor intestinal absorbtion. Showing / making a need of advanced formulations like nanoparticles, etc.50
ROLE OF CHRYSIN IN COLON CANCER
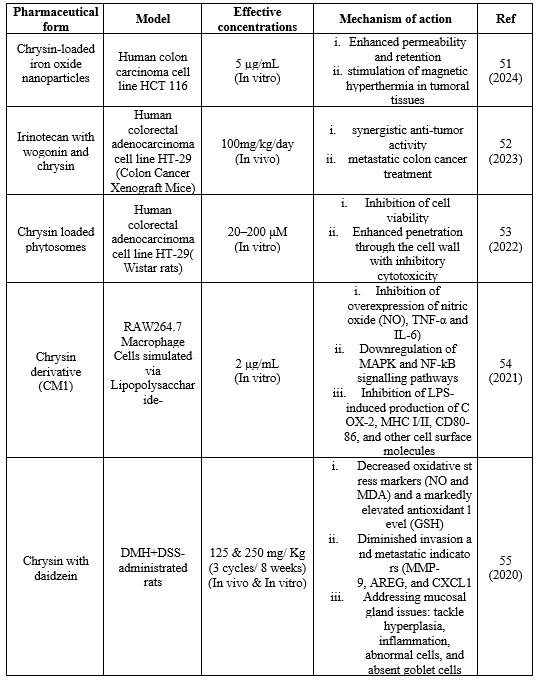
The recent experimental data of chrysin, its derivatives and chrysin in different pharmaceutical forms from year 2019- 2024 was collected, understood and summarized in the present review.
Table 2. Collective data of efficacy of different chrysin formulations on in vitro/ in vivo colorectal cancer models and their doses
Chrysin, a natural flavone, shows potential in treating colon cancer through varied formulations and combinations. Iron oxide nanoparticles loaded with chrysin improve tumor permeability and retention, and irinotecan and chrysin and wogonin together have synergistic anti-tumor actions. Phytosomes loaded with chrysin enhance cell penetration, while chrysin derivatives such as CM1 suppress signalling pathways and inflammatory indicators. As previous findings chrysin poses anticancer activity for number of cancers. But findings suggest that their derivatives also have similar mechanisms of action on colon cancer cells. In recent experimental study, among the novel set of seventeen chrysin derivatives that synthesized in the recent experimental study, compound 33A, a derivative of chrysin, had showed the greatest efficacy against HCT116 cell lines, with an IC50 value of 4.83 ?M.59 5,7- dimethoxychrysin, 5,7-dimethoxy-8-iodochrysin, 5,7-dimethyl-8-trifluoromethylchrysin, 5,7-diacetoxychrysin, 6,8-diiodo-5-hydroxy-7-acetoxychrysin, 6,8-ditrifluoromethyl-5-hydroxy-7-acetoxychrysin, 8-bromo-5-hydroxy-7-methoxychrysin, 5,7-dihydroxy-8-nitrochrysin, 8-iodo-5- hydroxy-7-methoxy-6-nitrochrysin are the derivatives of chrysin found to have anticancer activity against colon cancer cell lines. 8-bromo-5-hydroxy-7-methoxychrysin was the most potent anticancer chrysin derivative on human colon adenocarcinoma cell line HT29.60
CONCLUSION
This comprehensive review provides a structured overview of all possible studies of herbal bioactive chrysin focusing on the anticancer efficacy of b bioactive chrysin specifically on colon cancer cells from 2019 to 2024 with inclusion of studies with their respective models, effective concentrations and mechanisms of action. it indeed would provide a thorough understanding of chrysin's potential by enabling present scenario and clear evidence for effective treatment of colon/ colorectal cancer using a bioactive chrysin indicating a strong need of advancement in chrysin formulation which can increase its aqueous solubility and colonic absorption.
REFERENCES
- Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2024;74(3):229–63.
- Arnold M, Abnet CC, Neale RE, Vignat J, Giovannucci EL, McGlynn KA, et al. Global burden of 5 major types of gastrointestinal cancer. Gastroenterology 2020;159(1):335-349.e15.
- Wu Z, Chen K, Li J, Dai X. The global, regional, and national burden of colorectal cancer in 204 countries and territories from 1990 to 2019. Z Gesundh Wiss 2024;32(4):609–18.
- Ohe Y. Treatment-related death from chemotherapy and thoracic radiotherapy for advanced cancer. Panminerva Med 2002;44(3):205–12.
- Mushiake H, Tsunoda T, Nukatsuka M, Shimao K, Fukushima M, Tahara H. Dendritic cells might be one of key factors for eliciting antitumor effect by chemoimmunotherapy in vivo. Cancer Immunol Immunother 2005;54(2):120–8.
- Mukherjee A, Basu S, Sarkar N, Ghosh A. Advances in cancer therapy with plant based natural products. Curr Med Chem 2001;8(12):1467–86.
- Engel N, Falodun A, Kühn J, Kragl U, Langer P, Nebe B. Pro-apoptotic and anti-adhesive effects of four African plant extracts on the breast cancer cell line MCF-7. BMC Complement Altern Med 2014;14(1).
- Khoo BY, Chua SL, Balaram P. Apoptotic effects of chrysin in human cancer cell lines. Int J Mol Sci 2010;11(5):2188–99.
- Liu Y, Song X, He J, Zheng X, Wu H. Synthetic derivatives of chrysin and their biological activities. Med Chem Res 2014;23(2):555–63.
- Ren J, Cheng H, Xin WQ, Chen X, Hu K. Induction of apoptosis by 7-piperazinethylchrysin in HCT-116 human colon cancer cells. Oncol Rep 2012;28(5):1719–26.
- Ronnekleiv-Kelly SM, Nukaya M, Díaz-Díaz CJ, Megna BW, Carney PR, Geiger PG, et al. Aryl hydrocarbon receptor-dependent apoptotic cell death induced by the flavonoid chrysin in human colorectal cancer cells. Cancer Lett 2016;370(1):91–9.
- Leslie A, Carey FA, Pratt NR, Steele RJC. The colorectal adenoma–carcinoma sequence. Br J Surg 2002;89(7):845–60.
- Dinu D, Dobre M, Panaitescu E, Bîrl? R, Iosif C, Hoara P, et al. Prognostic significance of KRAS gene mutations in colorectal cancer--preliminary study. J Med Life. 2014;7(4):581–7.
- Nicholas C, Nicolaides S, James KV, Willson RE, Parsons J, Jen N, et al. Mismatch repair gene defects in sporadic colorectal cancers with microsatellite instability Bo Liu. Stanley R, Hamilton KW, editors. Vol. 9. Albert de la Chapelle; 1995.
- Barras D. BRAF mutation in colorectal cancer: An update: Supplementary issue: Biomarkers for colon cancer. Biomark Cancer 2015;7s1:BIC.S25248.
- Weisenberger DJ, Siegmund KD, Campan M, Young J, Long TI, Faasse MA, et al. CpG island methylator phenotype underlies sporadic microsatellite instability and is tightly associated with BRAF mutation in colorectal cancer. Nat Genet 2006;38(7):787–93.
- Ahmed M. Colon cancer: A clinician’s perspective in 2019. Gastroenterology Res 2020;13(1):1–10.
- Charmley S. What is the AJCC TNM staging system for colorectal cancer? Medicalnewstoday. com. Medicalnewstoday.com. Julie Scott, editor. AOCNP2022; September. 28.
- Kim K, Lee DJ. The updated AJCC/TNM staging system (8th edition) for oral tongue cancer. Transl Cancer Res 2019;8(S2):S164–6.
- Sloothaak DAM, Sahami S, van der Zaag-Loonen HJ, van der Zaag ES, Tanis PJ, Bemelman WA, et al. The prognostic value of micrometastases and isolated tumour cells in histologically negative lymph nodes of patients with colorectal cancer: a systematic review and meta-analysis. Eur J Surg Oncol 2014;40(3):263–9.
- Shimada K, Ajani JA. Adjuvant therapy for gastric carcinoma patients in the past 15 years: A review of Western and Oriental trials. Cancer 1999;86(9):1657–68.
- Freeman HJ. Early stage colon cancer. World J Gastroenterol 2013;19(46):8468.
- Gato M, Reis AN, Silvério S, Ito F, Lopes FT. Oncological Diseases. In: The Golden Guide to Oncologic Pharmacy. Cham: Springer International Publishing; 2022.
- Di dottorato redatta con il contributo finanziario di T. Recellularized colorectal patient-derived scaffold as in vitro pre-clinical 3D model for drug screening Unipd.it. [cited 2024 May 14].
- Yu Y, Anwar M, Chung HT. Colorectal cancer. In: Handbook of Evidence-Based Radiation Oncology. Clin Oncol. 2018;9.
- Nouri Nojadeh J, Behrouz Sharif S, Sakhinia E. Microsatellite instability in colorectal cancer IfADo - Leibniz Research Centre for Working Environment and Human Factors, Dortmund; 2018.
- Banerjee A, Pathak S, Subramanium VD, G D, Murugesan R, Verma RS. Strategies for targeted drug delivery in treatment of colon cancer: current trends and future perspectives. Drug Discov Today 2017;22(8):1224–32.
- Sninsky JA, Shore BM, Lupu GV, Crockett SD. Risk factors for colorectal polyps and cancer. Gastrointest Endosc Clin N Am 2022;32(2):195–213.
- Ahmed M. Colon cancer: A clinician’s perspective in 2019. Gastroenterology Res 2020;13(1):1–10.
- Mork ME, You YN, Ying J, Bannon SA, Lynch PM, Rodriguez-Bigas MA, et al. High prevalence of hereditary cancer syndromes in adolescents and young adults with colorectal cancer. J Clin Oncol 2015;33(31):3544–9.
- Mitra S, Paramaguru R, Das P, Katti SV. Preneoplastic lesions and polyps of the gastrointestinal tract. In: Surgical Pathology of the Gastrointestinal System. Singapore: Springer Singapore; 2022. p. 593–698.
- Wang L, Lo C-H, He X, Hang D, Wang M, Wu K, et al. Risk factor profiles differ for cancers of different regions of the colorectum. Gastroenterology 2020;159(1):241-256.e13.
- Low EE, Demb J, Liu L, Earles A, Bustamante R, Williams CD, et al. Risk factors for early-onset colorectal cancer. Gastroenterology 2020;159(2):492-501.e7.
- Donovan MG, Selmin OI, Doetschman TC, Romagnolo DF. Mediterranean diet: Prevention of colorectal cancer. Front Nutr 2017;4.
- Olguin JE, Mendoza-Rodriguez MG, Sanchez-Barrera CA, Terrazas LI. Is the combination of immunotherapy with conventional chemotherapy the key to increase the efficacy of colorectal cancer treatment? World J Gastrointest Oncol 2023;15(2):251–67.
- Chavda VP, Patel AB, Mistry KJ, Suthar SF, Wu Z-X, Chen Z-S, et al. Nano-drug delivery systems entrapping natural bioactive compounds for cancer: Recent progress and future challenges. Front Oncol. 2022;12:867655.
- Telang N. Drug-resistant stem cells: Novel approach for colon cancer therapy. Int J Mol Sci 2022;23(5):2519.
- Garcia-Oliveira P, Otero P, Pereira AG, Chamorro F, Carpena M, Echave J, et al. Status and challenges of plant-anticancer compounds in cancer treatment. Pharmaceuticals (Basel) 2021;14(2):157.
- Li C, Lin G, Zuo Z. Pharmacological effects and pharmacokinetics properties ofRadix Scutellariaeand its bioactive flavones. Biopharm Drug Dispos 2011;32(8):427–45.
- Lopes AP, Galuch MB, Petenuci ME, Oliveira JH, Canesin EA, Schneider VVA, et al. Quantification of phenolic compounds in ripe and unripe bitter melons (Momordica charantia) and evaluation of the distribution of phenolic compounds in different parts of the fruit by UPLC–MS/MS. Chem Pap 2020;74(8):2613–25.
- Rahimah SB, Djunaedi DD, Soeroto AY, Bisri T. The the phytochemical screening, total phenolic contents and antioxidant activities in vitro of white oyster mushroom (Pleurotus ostreatus) preparations. Open Access Maced J Med Sci 2019;7(15):2404–12.
- Sharma P, Kumari A, Gulati A, Krishnamurthy S, Hemalatha S. Chrysin isolated fromPyrus pashiafruit ameliorates convulsions in experimental animals. Nutr Neurosci 2019;22(8):569–77.
- Shahbaz M, Naeem H, Imran M, Ul Hassan H, Alsagaby SA, Al Abdulmonem W, et al. Chrysin a promising anticancer agent: recent perspectives. Int J Food Prop 2023;26(1):2294–337.
- Naz S, Imran M, Rauf A, Orhan IE, Shariati MA, Iahtisham-Ul-Haq, et al. Chrysin: Pharmacological and therapeutic properties. Life Sci 2019;235(116797):116797.
- Baell JB, Nissink JWM. Seven year itch: Pan-assay interference compounds (PAINS) in 2017—utility and limitations. ACS Chem Biol 2018;13(1):36–44.
- Shahbaz M, Naeem H, Imran M, Ul Hassan H, Alsagaby SA, Al Abdulmonem W, et al. Chrysin a promising anticancer agent: recent perspectives. Int J Food Prop 2023;26(1):2294–337.
- Gao S, Siddiqui N, Etim I, Du T, Zhang Y, Liang D. Developing nutritional component chrysin as a therapeutic agent: Bioavailability and pharmacokinetics consideration, and ADME mechanisms. Biomed Pharmacother 2021;142(112080):112080.
- Roberts MS, Magnusson BM, Burczynski FJ, Weiss M. Enterohepatic circulation: Physiological, pharmacokinetic and clinical implications. Clin Pharmacokinet 2002;41(10):751–90.
- Walle T, Otake Y, Brubaker JA, Walle UK, Halushka P. aDepartment of Cell and Molecular Pharmacology and Experimental Therapeutics, Division of Clinical Pharmacology. British Journal of Clinical Pharmacology. 2001;51(2).
- Noh K, Oh DG, Nepal MR, Jeong KS, Choi Y, Kang MJ, et al. Pharmacokinetic interaction of chrysin with caffeine in rats. Biomol Ther (Seoul) 2016;24(4):446–52.
- Karimova A, Hajizada S, Shirinova H, Nuriyeva S, Gahramanli L, Yusuf MM, et al. Surface modification strategies for chrysin-loaded iron oxide nanoparticles to boost their anti-tumor efficacy in human colon carcinoma cells. J Funct Biomater 2024;15(2):43.
- Godavarthi JD, Williams-Villalobo A, Etim I, Ting D, Gao S, Zhang Y. The synergistic antitumor effect of irinotecan and flavonoids on human colon cancer xenograft mice. In: ASPET 2023 Annual Meeting Abstract - Cancer Pharmacology. American Society for Pharmacology and Experimental Therapeutics; 2023.
- Kudatarkar N, Jalalpure S, Kurangi B. Formulation and characterization of chrysin loaded phytosomes and its cytotoxic effect against colorectal cancer cells. Ind J Pharm Educ 2022;56(3s):s407–12.
- Byun E-B, Song H-Y, Kim WS, Han JM, Seo HS, Park WY, et al. Chrysin derivative CM1 and exhibited anti-inflammatory action by upregulating toll-interacting protein expression in lipopolysaccharide-stimulated RAW264.7 macrophage cells. Molecules 2021;26(6):1532.
- Salama AAA, Allam RM. Promising targets of chrysin and daidzein in colorectal cancer: Amphiregulin, CXCL1, and MMP-9. Eur J Pharmacol 2021;892(173763):173763.
- Marzec E, ?witalska M, Winiewska-Szajewska M, Wójcik J, Wietrzyk J, Maciejewska AM, et al. The halogenation of natural flavonoids, baicalein and chrysin, enhances their affinity to human protein kinase CK2. IUBMB Life 2020;72(6):1250–61.
- Ghamkhari A, Pouyafar A, Salehi R, Rahbarghazi R. Chrysin and docetaxel loaded biodegradable Micelle for combination chemotherapy of cancer stem cell. Pharm Res 2019;36(12).
- Song H-Y, Kim H-M, Mushtaq S, Kim WS, Kim YJ, Lim S-T, et al. Gamma-irradiated chrysin improves anticancer activity in HT-29 colon cancer cells through mitochondria-related pathway. J Med Food 2019;22(7):713–21.
- Yu Q, Huang B, Ling Y. Design, synthesis, and in vitro anticancer activity of novel chrysin derivatives. Lett Drug Des Discov 2023;20(7):854–62.
- Zheng X, Meng W-D, Xu Y-Y, Cao J-G, Qing F-L. Synthesis and anticancer effect of chrysin derivatives. Bioorg Med Chem Lett 2003;13(5):881–4


 Chetna K. Dhone* 1
Chetna K. Dhone* 1
 Shagufta A. Khan 2
Shagufta A. Khan 2





 10.5281/zenodo.11348372
10.5281/zenodo.11348372