
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
A. M. Reddy Memorial College of Pharmacy, Narasaraopet, India.
Chroman (benzopyran) derivatives, comprising 2H- and 4H- isomers, are recognized as privileged scaffolds in medicinal chemistry due to their wide spectrum of pharmacological activities and low toxicity. Various synthetic approaches have been developed for the preparation of chroman derivatives, including acid-catalysed cyclization, intramolecular Oxa-Michael reactions, transition-metal-catalysed reactions, and multicomponent reactions. Recent research has focused heavily on sustainable synthesis, utilizing multicomponent reactions (MCRs), green solvents(water/ethanol), and heterogeneous catalysts (e.g., ZnO, Fe3O4) for highly efficient, one-pot preparation of functionalized 2-amino-4H-chromanes and benzo chromones. These compounds demonstrate potent anticancer, antimicrobial, anti-inflammatory, and enzyme-inhibitory activities. Many natural compounds, such as tocopherols (vitamin E derivatives), contain the chroman nucleus and contribute significantly to their pharmacological properties.
The chromone ring system, 1benzopyran4one, is the core fragment in several flavonoids, such as flavones, flavanols and isoflavones (1). The word chromones is derived from the Greek word chroma, meaning “color”, which indicates that many chromone derivatives exhibit a broad variation of colors. The rigid bicyclic chromone fragment has been classified as a privileged structure in drug discovery, due to its use in a wide variety of pharmacologically active compounds such as anticancer (2), anti-HIV, antibacterial and anti-inflammatory agents (3-12). Several chromone derivatives have also been reported to act as kinase inhibitors, to bind to benzodiazepine receptors and as efficient agents in the treatment of cystic fibrosis (13-15).
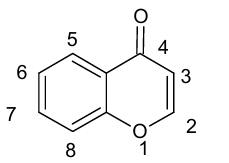
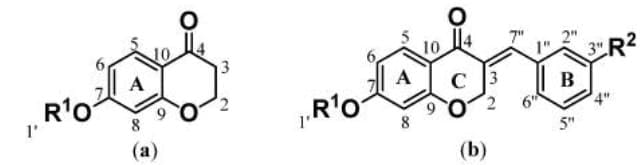
The general structure and numbering of chromanes
Although there are a large number of chroman derivatives known for their pharmacological properties there are only a few examples that have been or that are used as therapeutic agents today. Khellin as an example extracted from the seeds of the plant Ammi visnaga, was the first chroman in clinical practice and it has been used for centuries in the Mediterranean area as a diuretic to relieve renal colic (16). Around the 1950s, khellin was used as a smooth muscle relaxant in the treatment of angina pectoris and asthma (17). However, present use of khellin as a therapeutic agent focuses on the treatment of vitiligo, a pigmentation disorder (18). Other current medical treatments with chroman derivatives are exemplified by sodium cromoglycate (Lomudal) used as a mast cell stabilizer in allergic rhinitis, asthma and allergic conjunctivitis; diosmin (Daflon) for the treatment of venous diseases; flavoxate as smooth muscle relaxant to treat urge incontinence (19-23).
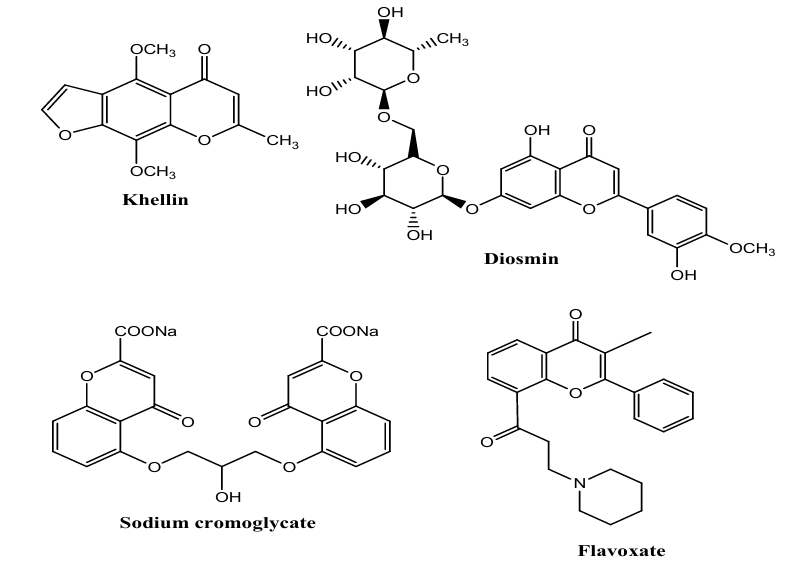
Examples of chromane-based compounds that have been or that are used as pharmaceutical agents
Synthesis of chromanes by Triflimide-catalyzed annulation:
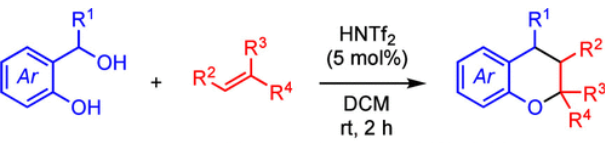
The convergent synthesis of a series of chroman derivatives by a triflimide-catalyzed annulation of o-hydroxy benzylic alcohols with alkenes is reported. The Bronsted acid-catalyzed reaction proceeds from simple starting materials under mild conditions and provides chroman products of varying substitution patterns. (24)
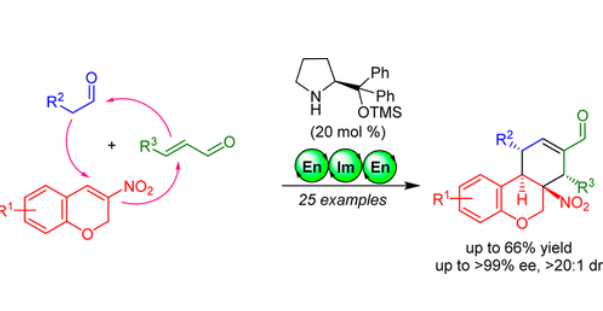
Asymmetric Synthesis of Functionalized Tricyclic Chromanes via an Organocatalytic Triple Domino Reaction
A highly stereoselective triple domino reaction for the synthesis of functionalized tricyclic chroman scaffolds has been developed. A secondary amine-catalyzed domino Michael/Michael/aldol condensation reaction between aliphatic aldehydes, nitro-chromanes, and α, β-unsaturated aldehydes lead to the formation of synthetically important tricyclic chromanes bearing four contiguous stereo genic centers including a tetrasubstituted carbon in good yields (20–66%) and excellent stereoselectivities (>20:1 dr and >99% ee).(25)
Aceto acetic-Ester Condensation Claisen Condensation
The Claisen Condensation between esters containing α-hydrogens, promoted by a base such as sodium ethoxide, affords β-keto esters. The driving force is the formation of the stabilized anion of the β-keto ester. If two different esters are used, an essentially statistical mixture of all four products is generally obtained, and the preparation does not have high synthetic utility.
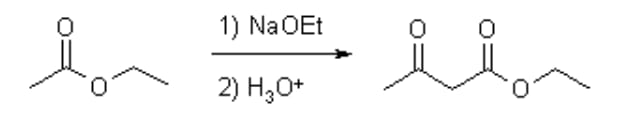
However, if one of the ester partners has enolizable α-hydrogens and the other does not (e.g., aromatic esters or carbonates), the mixed reaction (or crossed Claisen) can be synthetically useful. If ketones or nitriles are used as the donor in this condensation reaction, a β-diketone or a β-Ket nitrile is obtained, respectively. The use of stronger bases, e.g. sodium amide or sodium hydride instead of sodium ethoxide, often increases the yield. The intramolecular version is known as Dieckmann Condensation. (26)
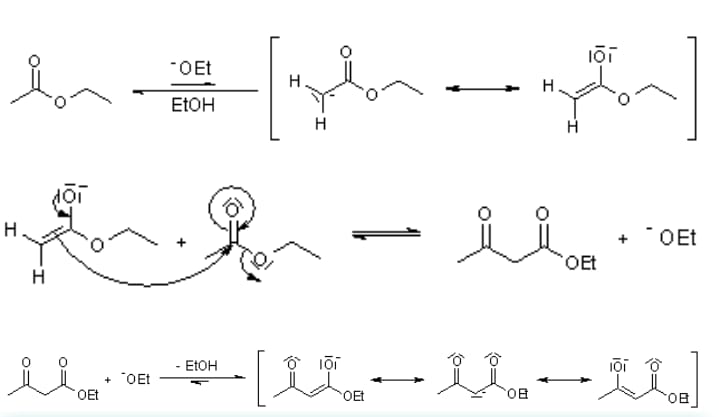
Mechanism of the Claisen Condensation
Chroman ring closure under the microwave irradiation
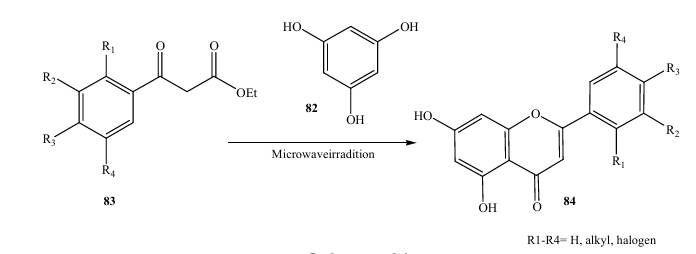
Recently, microwave irradiation 80 offers a considerable advantage over conventional heating because it results in substantial rate enhancements in a wide range of organic reactions. Cleaner reactions are also commonly achieved, together with improvements in yield and selectivity. The increasing demand for clean and ‘‘green’’ chemical syntheses has resulted in increased use of microwave irradiation, so there have been several recent reports, describing the application of microwave irradiation to the synthesis of flavonoids. In 2005, Seijas et al. (27) reported an ecofriendly direct solvent free synthesis of functionalized flavones 84 under microwave irradiation (Scheme). This method was valid for flavones with or without substitution in the B ring. Thus, the flavonoids were prepared from the corresponding ethyl benzoyl acetates 83 and phloroglucinol for 2–12 min of irradiation in 66–96% yields. The successful use of microwave irradiation in providing this rapid and direct route to flavones in comparison to classical procedures contributes to confirming the participation of specific effects in some microwave assisted organic synthesis.
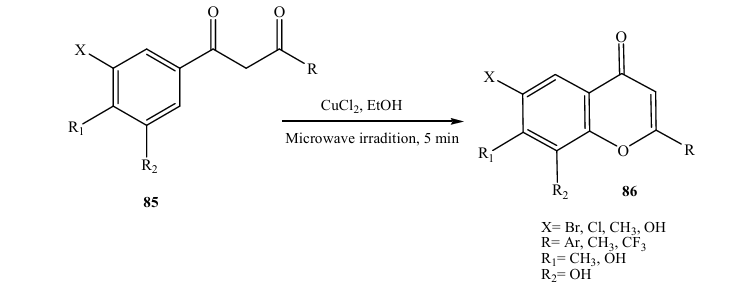
In 2005, Kabalka and Mer eddy (28) reported a facile microwave synthesis of functionalized flavones and chromans via the cyclization of 1(2hydroxyaryl)3aryl1,3propanedione (Scheme). In their study, the intermediate 1,3propanediones 85 were synthesized in 5 min via dehydrative cyclization to the corresponding flavones and chromans 86 in ethanol, in the presence of CuCl2 under microwave irradiation.
In 2009, Luthman and coworkers (29) reported a base promoted condensation between 2 hydroxy acetophenones 87 and aliphatic aldehydes 88 (Scheme); they optimized the reaction to afford 2alkylsubstituted 4chromanones 89 in an efficient manner using microwave heating. Performing the reaction using diisopropylamino in EtOH at 170 0C for 1 h gave high yield in 88%. The 4chromanones could be further converted into highly functionalized 2,3,6,8tetrasubstituted chromones in which a 3substituent (acetate, amine, or bromine) was introduced via straightforward chemical transformations.
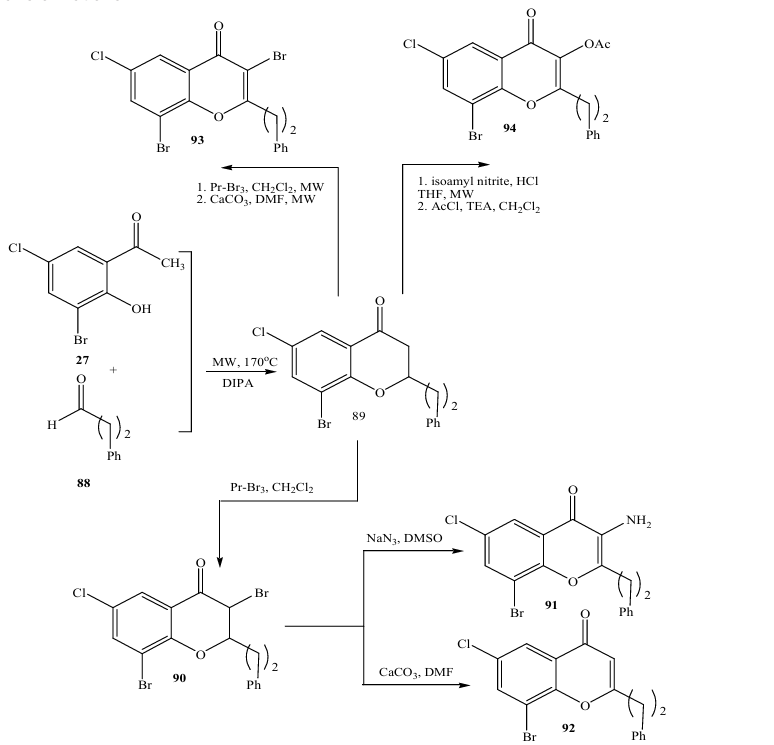
Iodine as a catalyst:
In 2004, Tome´ and coworkers (31) reported the synthesis of chromane139 through iodine as a catalyst (Scheme)
BIOLOGICAL SIGNIFICANCE
Heterocycles play an important role in the design and discovery of new physiological/pharmacologically active compounds (32). Chemically, chromones (4Hchromen4ones) are heterocyclic compounds with the benzopyrone framework. Molecules containing the chromone or benzopyran one ring have a wide range of biological activities. They have been shown to be tyrosine and protein kinase inhibitors (33), as well as anti-inflammatory (34), antiviral (35), antioxidant (36,37), antihypertensive agents (35).
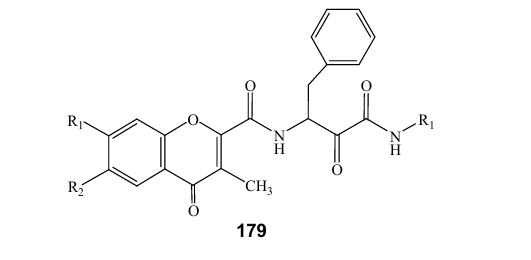
1.Antioxidant: Lee and coworkers (2011), reported that new chromone carboxamide derivatives 179 were synthesized as conformationally constrained structural variants of MDL, to provide alternative calpain inhibitors and antioxidant activities in DPPH scavenging and lipid peroxidation inhibitory effects (38).
2.Chroman derivative as Antimicrobial:
Antimicrobials are the drugs which are use to kill or inhibit the growth of micro-organism. [39-44] R.B. Patil et al. synthesized aromatic ketone-antimicrobial by the condensation of 7- hydroxy-3-formyl chromen-4-one with substituted acetophenones by base catalyzed reaction followed by dehydration using Claisen Schmidt reaction and Schiff bases were prepared. The synthesized Schiff bases and chalcones were tested for their antimicrobial activity on bacteria Staphylococcus aureus & Bacillus subtilis Gm+ organisms and two Gm-ve organisms Escherichia coli & Pseudomonas aeruginosa.[45]
(12E)-N-((7-methyl-4-methylene-4H-chromen-3-yl) methylene)-4-nitrobenzenamine
(1Z)-1-((7-methyl-4-methylene-4H-chromen-3-yl) methylene) thiosemicarbazide
3.As oral contraceptive:
Oral contraceptives are the drugs which are administered orally and it is used to prevent pregnancy in females. Estrogen and progestin are sex hormones secreted in females. Oral contraceptives are the combinations of estrogen and progestin. These drugs are inhibiting the release of eggs from the ovaries. Contraceptives changes the lining of the uterus for the inhibition of mucus at the opening of the uterus called cervix, for prevention of sperm entering. Oral contraceptives are a very effective birth control method. Oral contraceptives are well known as mini pill.[46]
Ormeloxifene oral contraceptive agent developed by the Indian Central Drug Research Institute Lucknow, India, a nonsteroidal selective estrogen receptor modulator and once-a-week. Ormeloxifene oral contraceptives under the brand name Shahel.
Ormeloxifene: trans-7-methoxy-2,2-dimethyl-3-phenyl-4-[4-(2pyrrolidinoethoxy)phenyl] chroman hydrochloride.
1-(2-(4-(7-methoxy- 2, 2-dimethyl-3-phenyl-2H-chroman-4-yl)phenoxy)ethyl)pyrrolidine
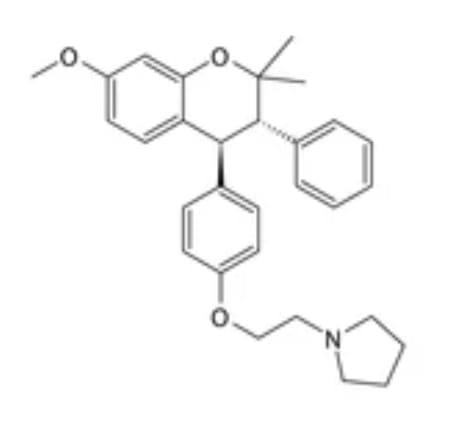
Ormeloxifene
4.As antiepileptic
Epilepsy is a common neurological disorder affecting approximately 45–100 million people globally. Though many antiepileptic drugs are available in clinical use, neurotoxicity and distinctive adverse effects restrict their clinical use. Therefore, it is necessary to discover new chemical pharmacophores as more effective antiepileptics with less neurotoxicity. [47-49] Azolylchromane derivatives were prepared as conformationally work as analogs of (arylalkyl)azole anticonvulsants. The anticonvulsant activities of the Azolylchromane derivatives are evaluated by pentylenetetrazol (PTZ)-induced convulsions in mice by determining seizure latency and protective effect. Among these compounds, 7-chloro-3-(1H-imidazol-1-yl)chroman-4-one and 3-(1H-1,2,4-triazol-1-yl)chroman-4-one exhibited significant action in decrease seizures and also effective against PTZ-induced seizures.
If the Schiff base is present in chroman nucleus, causes increase in antiepileptic activity. There is following examples of Schiff base with chroman nucleus.
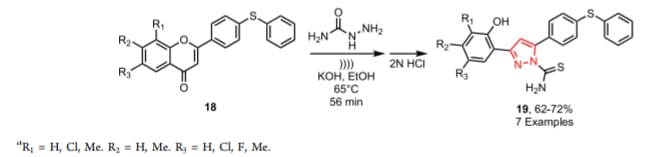
5.As Anti-inflammatory and analgesic:
Inflammation is the first response of the immune system to infection, irritation or foreign substance. The chroman pharmacophore represents a novel class of COX-2 selective inhibitors (coxibs) in non-steroidal anti-inflammatory drugs (NSAIDs) which provide higher potency, efficacy, and selectivity over the existing coxibs (e.g.: celecoxib, valdecoxib, rofecoxib, and etoricoxib) for the treatment of inflammation. The chroman coxib clinical candidates are SD-8381 and SC-75416. SC-75416 provides a fast onset of action and higher efficacy compared to ibuprofen. Other examples for chroman cyclooxygenase-2 selective inhibitors: [50-53] Inflammatory stimuli stimulate to secrete pro-inflammatory cytokine Factor α. Factor α is also known as TNF α and is responsible for the tumor Necrosis. Chroman molecule blocks the secretion of TNF α production. It is found that in chroman moiety the substitution in benzene ring increases the ability to block Factor α or TNF α production as response to inflammatory stimuli. The methoxy group present at 7th position and 3,4,5-trimethoxy phenyl group in chroman are very potent blockers of TNF α production. For example, the compound 7-methoxy-2-(2,4,5-trimethoxyphenyl)-2H-chromene shows the good activity against the TNF α production.
6.As dopamine antagonist:
A dopamine antagonist is also known as antidopaminergic drug. Antidopaminergic drugs cause receptor antagonism by blocking dopamine receptors. Generally antipsychotic drugs are dopamine antagonists and use in treating schizophrenia, bipolar disorder and some other antidopaminergic drugs are used for treatment of nausea and vomiting and also use as antiemetic. A series of chromen-2-one discovered with selective affinity for the dopamine (DA) D4 receptor is described. The target compounds were tested for binding to cloned human DA D2l, D3, and D4 receptor subtypes expressed in Chinese hamster ovary K1 cells. The compound 7-[(2-phenylaminoethylamino) methyl]chromen-2-one is responsible for accumulation of rat brains at dose given 20 mg/kg orally. Because it causes increased DOPA (l-3,4-dihydroxyphenylalanine) level in rat brain.[54]
7.Anticancer Activity of Chromans:
The disease cancer is characterized by uncontrolled growth of abnormal cell and causes second highest mortality in the world. Today’s scenario programmed cell death induced by cytotoxic anticancer agents commonly use to treat the cancer. Chroman moieties have been reported with anticancer activity found in many of the natural compounds. These compounds are isolated from naturally occurring herbal plants and animal origin like sea fish etc. [55-61] Tephrosia (lung cancer), calanone (leukemia and cervical carcinoma), acronycine (lung, colon and ovary cancer), are the examples of natural anticancer agents which contains chroman ring.
A novel class of microtubule inhibitors belongs to Substituted 4-aryl-4Hchromene compounds. Substitution of aryl group at the 4th position increases the anticancer activity of the compound.
2,3-diaryl chromanones derivatives was synthesized under microwave irradiation technique and synthesized compound was evaluated for their cytotoxicity using MTT(3-[4,5-dimethylthiazole-2-yl]-2,5-diphenyltetrazolium bromide) assay using HL60 cells and Peripheral blood mononuclear cells (PBMC). Lipid peroxidation assay was use for antioxidant activity. A series of newly synthesized compounds N-substituted-3-(2-oxo-2H-chroman-6-ylimino)thioureas, N-substituted-3-(2-oxo-2H-chroman-6-ylimino)thiazolidin-4-ones was prepared and the activity of compound was tested against PTZ and strychnine induced convulsion. The newly synthesized compound shows good anticonvulsant activity
8.Antitubercular Activity:
Tuberculosis (TB) is caused by several species of Mycobacterium (Mycobacterium tuberculosis complex, which includes Mycobacterium tuberculosis itself, Mycobacterium microti, Mycobacterium pinnipedian Mycobacterium Bovis, Mycobacterium carpet, Mycobacterium africanus, and Mycobacterium Canetti), TB is an acute or chronic infectious disease. Generally TB infected the lungs but can also attack on the central nervous system, lymphatic system, circulatory system, genitourinary system, gastrointestinal system, bones, joints, and even the skin etc.[62-65]
Rezayan and co-workers et al. describe synthesis of coumarin & its derivatives and antimycobacterial activity was evaluated by the broth microtiter dilution method invitro against the bacteria M. Bovis. The activity results are compared with ethambutol. Some of the synthesized derivatives found active against M. Bovis.
Rashmi Tandon1, Prija Ponnan, Neha Aggarwal et al. synthesized the compound 7-amino-4-methylcoumarin and the antitubercular activity was tested against H37Rv with the help of broth dilution method. The synthesized compound shows good anti-tubercular activity. The compound inhibits the growth of TB bacteria.
Silvia H Cardoso, Milena B. Barreto et al synthesized the derivatives of compound 2-oxo-2H-chromene-3-carbohydrazide by the refluxing of compound coumarin-3-carboxylateethyl ester with 80% ethanolic solution of hydrazine. Finally aldehyde in ethanol gives the required compound. The synthesized compound screened for anti-tubercular activity and was found the compound shows good anti-tubercular activity.
RESULT AND DISCUSSION
The present review represents a board description for the methods used in the synthesis of chromans and some of its derivatives. The rigid bicyclic chromans fragment has been classified as a privileged structure in drug discovery, due to its use in a wide variety of pharmacologically active compounds; few examples as therapeutic agents chromans are used as scaffolds for the development of bioactive compounds, the application in medicinal chemistry, such as preparation of fluorescence probes, due to photochemical properties of chromans have been also mentioned.
REFERENCES





Dr. K. Narsu kumari*, Y. Nagalakshmi, S. k. Shahin kousar, M. Gowtham Reddy, M. Venkateshwarlu, A Review on Synthesis and Biological Significance on Chromane Derivatives, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 4, 557-569. https://doi.org/10.5281/zenodo.19410446
 10.5281/zenodo.19410446
10.5281/zenodo.19410446