The present study investigated the hepatoprotective activity of the ethanolic root extract of Tectona grandis against paracetamol-induced hepatotoxicity in Wistar rats. Roots of T. grandis were extracted using a Soxhlet apparatus and subjected to physicochemical and preliminary phytochemical evaluation. Acute oral toxicity was determined in accordance with OECD guideline 423. Hepatotoxicity was induced by oral administration of paracetamol (2 g/kg). Animals were divided into five groups: normal control, paracetamol control, standard control treated with silymarin (100 mg/kg), and test groups receiving T. grandis root extract treated at doses of 100 and 200 mg/kg. Serum biochemical parameters, including SGPT, SGOT, ALP, total bilirubin, and total protein, were estimated followed Paracetamol exposure resulted in a substantial increase in liver enzymes and bilirubin levels with a reduction in total protein, indicating hepatic injury. Treatment with the extract produced dose-dependent hepatoprotection, with the higher dose showing effects comparable to silymarin. Histopathological findings corroborated biochemical results by demonstrating restoration of normal hepatic architecture. The study confirms the hepatoprotective potential of Tectona grandis root extract.
Tectona grandis; hepatoprotection; paracetamol; liver enzymes; medicinal plants
The liver is a multifunctional organ essential for maintaining metabolic balance, detoxifying xenobiotics, synthesising vital proteins, regulating nutrient metabolism, and producing bile required for digestion. Because it receives blood directly from the gastrointestinal tract and is the primary site of drug metabolism, it is highly vulnerable to toxic injury from pharmaceuticals, chemicals, alcohol, environmental contaminants, infectious agents, and unhealthy lifestyle practices. Liver diseases, including fatty liver disease, hepatitis, cirrhosis, and hepatocellular carcinoma, remain major global health burdens and are often associated with increased morbidity and mortality. Drug-induced liver injury (DILI) represents one of the most significant causes of acute liver failure, occurring either through predictable dose-dependent mechanisms, as in paracetamol toxicity, or through unpredictable idiosyncratic reactions associated with antibiotics, anti-epileptics, NSAIDs, anti-tubercular therapy, chemotherapeutic agents, and even certain herbal supplements¹. Hepatotoxicity arises through several interconnected pathways, including oxidative stress, mitochondrial dysfunction, bile flow disruption, formation of reactive metabolites, inflammatory responses, and immune-mediated destruction of hepatocytes13. To investigate these mechanisms and evaluate hepatoprotective interventions, experimental models such as paracetamol-induced toxicity, carbon tetrachloride (CCl?)-induced oxidative damage, and aflatoxin B1-mediated DNA injury are widely employed in preclinical research2. Their predictable pathological outcomes—ranging from necrosis and inflammation to fibrosis—make them suitable for screening therapeutic agents3.
Assessment of liver injury in these models relies on biochemical, histological, and oxidative stress parameters. Elevations in serum biomarkers such as alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and bilirubin serve as indicators of hepatocellular damage and cholestasis4. Histopathological evaluation of liver tissue provides direct evidence of structural alterations, including hepatocyte degeneration, fatty changes, necrosis, haemorrhage, and inflammatory cell infiltration. Furthermore, quantifying indices of oxidative stress like malondialdehyde (MDA) and evaluating oxidative stress–reducing defences like the activity of superoxide dismutase (SOD) and glutathione (GSH) offer insight into the role of reactive oxygen species in liver pathology5. Together, these analyses provide a comprehensive understanding of hepatic injury and recovery. Within this framework, medicinal plants—such as Tectona grandis—are increasingly being explored for their potential hepatoprotective effects. Their bioactive constituents may modulate oxidative stress, enhance antioxidant capacity, stabilise cellular membranes, and inhibit the formation of toxic metabolites6. Thus, integrating biochemical, histological, and antioxidant assessments is crucial for validating the therapeutic potential of plant-based interventions against experimentally induced hepatotoxicity7.
Liver diseases remain a major global health concern, and drug-induced hepatotoxicity is a major etiological factor in acute liver injury. Paracetamol (acetaminophen) widely used as an analgesic and antipyretic drug, but at high doses it forms a highly reactive metabolite, N-acetyl-p-benzoquinone imine (NAPQI), which exhausts glutathione stores, generates oxidative stress, results in centrilobular hepatic necrosis. Because currently available hepatoprotective drugs may have limitations or adverse effects, there is a continuous need to explore safer, plant-based therapeutic alternatives8.
Tectona grandis (Family: Lamiaceae), commonly known as teak, is an important medicinal plant traditionally used in Ayurveda for treating inflammation, skin disorders, and liver ailments. Different parts of the plant contain bioactive constituents such as flavonoids, tannins, phenolics, and naphthoquinones (including tectoquinone and lapachol), which possess strong antioxidant, membrane-stabilising, and anti-inflammatory activities. Although some studies have reported hepatoprotective properties of T. grandis, research focusing specifically on its root extract, supported by chemical marker identification, biochemical profiling, and detailed histopathology, remains limited9.
Considering the phytochemical richness and traditional claims of T. grandis, the present study was designed to evaluate the hepatoprotective effect of its ethanolic root extract against paracetamol-induced liver injury in Wistar rats. The study further aimed to correlate biochemical outcomes with histopathological observations, thereby providing scientific validation for the therapeutic potential of Tectona grandis root in liver protection10.
Materials and equipment
All chemicals and reagents used in the study were of analytical grade. Paracetamol, silymarin, ethanol, methanol, chloroform, and carboxymethyl cellulose were obtained from standard suppliers. Major equipment included a Soxhlet apparatus, heating mantle, digital weighing balance, water bath, muffle furnace, oral gavage needle, and standard laboratory glassware12.
Physicochemical Evaluation and Preliminary Phytochemical Screening
The crude drug's recognition, purity, and quality were determined using a physicochemical investigation that met all WHO standards. Important assessments included water-soluble ash by boiling, acid-insoluble ash with hydrochloric acid, and total ash value by incineration. Using 90% ethanol for alcohol-soluble extractives and chloroform-water for water-soluble extracts, the extractive values were used to estimate the solubility of active ingredients. By heating to a constant weight, loss on drying indicated the moisture content. The plant's therapeutic value was brought to light by the discovery of secondary metabolites as flavonoids, alkaloids, phenolic compounds, and saponin glycosides after preliminary phytochemical screening11.
Collection and authentication of plant material
Roots of Tectona grandis were collected from local areas of Allahabad, Uttar Pradesh, India, during the month of March. The plant material was authenticated by the Botanical Survey of India, Allahabad, and a voucher specimen was preserved for future reference.
Preparation Of Plant Extract
The collected roots were washed thoroughly, shade-dried, and pulverized to obtain a coarse powder. Extraction was carried out using ethanol in a Soxhlet apparatus at 50–75 °C until exhaustive extraction was achieved. The extract was concentrated in a water bath and dried to obtain the crude ethanolic extract.
Experimental animals
Healthy adult Albino Wistar rats (150–200 g) of either sex were housed in a controlled environment with a 12-hour light–dark cycle and maintained at a temperature of 25 ± 2 °Celsius, and relative maintained at a relative humidity of approximately 55%. Animals had free access to a standard pellet diet and water. The study protocol was reviewed and approved by the Institutional Animal Ethics Committee and conducted in accordance with CPCSEA guidelines.
Acute toxicity study
Acute oral toxicity of the ethanolic root extract was evaluated according to OECD guideline 423. Animals were observed for mortality and behavioural changes for 14 days following administration.
Preparation of Extract Dosage Form
For in vivo experiments, the crude ethanolic extract of Tectona grandis root was suspended in an aqueous solution of carboxymethyl cellulose (CMC) to obtain the required concentrations for oral administration. The freshly prepared suspension was administered by oral gavage.
Biochemical and Histopathological Assessment
At the end of the experimental period (5th day of treatment), blood was collected from rats of all groups by cardiac puncture under appropriate conditions. Serum was separated and used for the estimation of hepatic biochemical markers, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and total bilirubin (hematoidin), and related parameters.
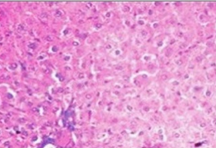
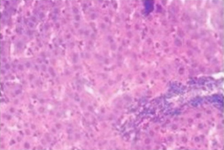
After blood collection, the animals were sacrificed, and the livers were carefully excised, washed with normal saline, and fixed in 10% formalin. The fixed tissues were processed for histopathological examination to assess structural changes associated with hepatotoxicity and the hepatoprotective effect of the Tectona grandis extract.
RESULTS
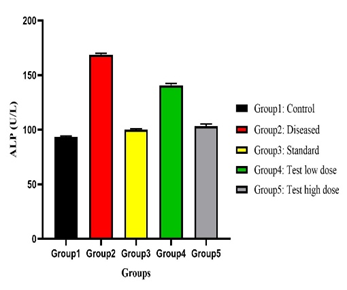
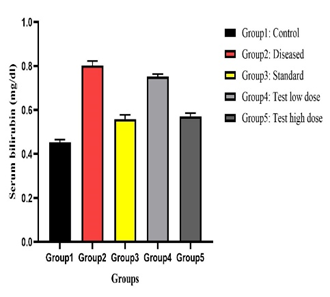
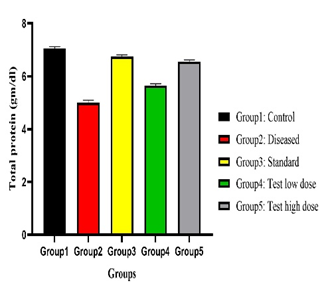
Physicochemical analysis confirmed the quality and purity of the plant material. Preliminary phytochemical screening revealed the presence of flavonoids, tannins, alkaloids, phenolic compounds, and saponins, which are known to exert antioxidant activity and protect hepatic tissue. Paracetamol administration resulted in significant elevation of serum SGPT, SGOT, ALP, and total bilirubin levels, accompanied by a reduction in total protein, indicating hepatocellular damage. Treatment with Tectona grandis root extract produced a dose-dependent normalisation of these biochemical parameters. The higher dose (200 mg/kg) showed hepatoprotective effects comparable to silymarin.
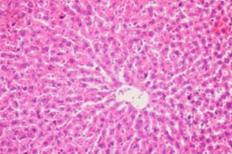
Histopathological examination supported the biochemical findings. Paracetamol-treated rats exhibited centrilobular necrosis, fatty degeneration, and inflammatory infiltration. In contrast, extract-treated groups showed marked restoration of hepatic architecture, particularly in the high-dose group, which demonstrated near-normal liver morphology.
Effect of Tectona grandis on Serum Maker Enzymes (SGOT, SGPT, ALP), Total Bilirubin, Total Protein on paracetamol-induced Hepatotoxicity on Rats


 Shruti Shukla*
Shruti Shukla*
 Arvind Kumar Shrivastava
Arvind Kumar Shrivastava
 Rishita Shrivastava
Rishita Shrivastava
 Preeti Maurya
Preeti Maurya
 Siddharth Kesharwan
Siddharth Kesharwan




 10.5281/zenodo.18810732
10.5281/zenodo.18810732