
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmaceutics, Konkan Gyanpeeth Rahul Dharkar College of Pharmacy and Research Institute, Karjat, Maharashtra.
The microbead is a potential multiparticulate drug delivery platform for controlled release system with excellent performance profile compared to the conventional dosage forms based on uniform drug delivery, low dosing schedule, and increased bioavailability. It reviews the development and testing of drug-loaded microbeads, emphasizing the potential of such microbeads for sustained, targeted, and site-specific drug release. Crucial sections describe the complex pathways of controlled release, such as diffusion, erosion, swelling, and osmotic pressure-mediated kinetics as described in mathematical models, such as Korsmeyer–Peppas, and zero-order profiles. Different approaches to preparation are examined, including ionotropic gelation, emulsification, coacervation, spray drying, extrusion, and their influence on particle size, drug entrapment efficiency (usually between 44-90%), and release profiles, are elucidated. Comparison to other dosage forms, such as for instance tablets, capsules or nanoparticles, highlights the lower incidence of dose dumping and increased adherence of the patient. In future perspectives stimulus-responsive smart microbeads, nanotechnology hybrids, personalized medicine applications all show promise over the forthcoming horizon. But questions of scale and regulatory obstacles remain to be answered. Finally, in light of this information, drug-loaded microbeads would provide for a next-generation controlled release system that can provide patients with powerful chronic therapies. This article, in synthesis of recent developments, directs researchers of pharmaceutical sciences to the development of an enhanced version of the drug for optimal therapeutic outcomes.
Microbeads, discrete spherical microparticles ranging from 1-1000μm, have emerged as a cornerstone in controlled drug release systems, enabling precise modulation of drug pharmacokinetics. Unlike single-unit dosage forms, microbeads distribute evenly in the gastrointestinal tract, minimizing local irritation and inter-subject variability. Their matrix or reservoir structure incorporates biocompatible polymers like alginate, chitosan, and pectin, facilitating entrapment of hydrophilic or hydrophobic drugs. Pioneered in the 1970s with alginate gels for insulin delivery, microbeads evolved through ionotropic gelation advancements in the 1990s. Recent milestones include FDA-approved multiparticulates like Nucleon capsules (1990s) and nanocomposite hybrids post-2010, shifting from synthetic to natural polymers for biocompatibility. (1–4)Microbeads in pharmaceuticals are classified primarily by structure, release profile, functional properties, and targeting mechanism, distinguishing them from microcapsules or nanoparticles for optimized controlled drug delivery.(5,6)

Figure No. 01 Structural & Functional Classification of Microbeads(4,7, 8,9)
Table No. 01 Release Based Classification of Microbeads(10)
|
Type |
Mechanism |
Example Polymers ? |
|
Sustained release |
Diffusion/swelling |
HPMC, xanthan gum |
|
Pulsatile release |
Coating rupture |
Ethylcellulose rupturable |
|
Delayed release |
Enteric coating |
Eudragit S100 |
|
Extended release |
Erosion/matrix |
PLGA, natural gums |
Smaller sizes enhance absorption and targeting, while larger improve handling and compliance; measured via optical microscopy/laser diffraction.
Table no. 02- Detailed Classification Size-Based Classification of Microbeads(8,11–14)
|
Size Range (μm) |
Designation |
Key Characteristics |
GI Transit Behaviour |
Typical Applications |
Examples/ Notes |
|
1-100 |
Nano microbeads |
Ultra-fine, high surface area; often hybrid with NPs; PDI <0.2 via microfluidics |
Rapid systemic absorption; colon reach |
Targeted delivery (cancer, ocular); high bioavailability boost |
Doxorubicin-loaded chitosan (burst <20%) |
|
100-500 |
Small microbeads |
Optimal for uniform dispersion; high entrapment (70-95%); ionotropic gelation yields |
Even GI spread; minimal dose dumping |
Sustained oral release; mucoadhesive |
Alginate-nebivolol (427-697 μm); floating gellan |
|
500-1000 |
Large microbeads |
Discrete, free-flowing; robust matrices; spray-dried or extrusion |
Gastric retention; slower transit |
Gastroretentive; taste-masked suspensions |
Pectin-HPMC (792-961 μm); sprinkleable up to 2.5 mm FDA limit |
|
>1000 (up to 2500) |
Mini-pellets/beads |
Larger, chewable; lower surface: volume ratio |
Phased release; sprinkle dosing |
Paediatric/geriatric; extended release |
Eudragit-coated (1-2.4 mm safe per FDA) |
Common polymers in microbead formulations enable controlled drug release by forming matrices that modulate diffusion, swelling, erosion, or ionotropic gelation. (15)
Sodium alginate stands out as the most widely used due to its ability to form rigid gels via Ca²? cross-linking, achieving high entrapment (70-95%) and pH-independent sustained release over 8-24 hours for drugs like simvastatin or aceclofenac. Chitosan, a cationic polymer, provides mucoadhesion and enhances permeability, often combined with alginate for polyelectrolyte complexes that reduce burst release. Other naturals include pectin, gellan gum, and guar gum for biodegradable matrices; albizia, cissus, irvingia, and khaya gums offer natural alternatives with varying viscosities for sustained profiles.
Recent advances in natural gum-based microbeads have focused on enhancing controlled release through improved gastric retention, site-specific targeting, and biocompatibility, leveraging gums like gellan, karaya, guar, xanthan, and alginate blends.(14)
Key Innovations(11,18)
Gellan gum-based floating microbeads for acyclovir delivery use ionotropic emulsion-gelation with oil entrapment, achieving >90% entrapment efficiency, sustained release over 12 hours, and prolonged gastric retention (up to 6 hours in rabbits), boosting bioavailability by 2-3-fold via upper GIT absorption. Colon-targeted gellan gum microbeads exploit divalent cation gelation (Ca²?/Mg²?) for pH/enzyme-sensitive swelling in colonic fluids, minimizing gastric loss and enabling zero-order kinetics for therapeutics like 5-ASA; spray drying scales production while preserving mechanical strength. Hydrogel networks of sodium alginate-gellan gum (varying ratios) for valacyclovir show diffusion-controlled release (Higuchi model fit), with SEM confirming porous matrices ideal for prolonged oral delivery and reduced dosing frequency. Guar and xanthan gums in glipizide microspheres via orifice ionic gelation yield mucoadhesive particles (size 200-500μm) with smoother surfaces at optimal gum: drug ratios (1:0.25-0.5), sustaining release up to 10 hours.
Table no. 03 Comparison of Gums(12,14)
|
Gum Type |
Key Advance |
Release Profile |
Application |
|
Gellan |
Floating/colon-targeted |
Sustained (12-24h) |
Antivirals |
|
Guar/ Xanthan |
Mucoadhesive microspheres |
8-10h controlled |
Hypoglycemics |
|
Karaya |
Oil-entrapped buoyancy |
Zero-order |
Gastric retention |
These developments emphasize eco-friendly, scalable methods for chronic therapies, addressing scale-up and in vivo correlation challenges.
Hydroxypropyl methylcellulose (HPMC) and ethylcellulose act as release retardants in coatings, creating diffusion barriers for zero-order kinetics. Eudragit polymers (e.g., Eudragit S100, RL/RS) provide pH-sensitive release for colon targeting. Poly (lactic-co-glycolic acid) (PLGA) and polylactic acid (PLA) enable erosion-based release, ideal for hydrophobic drugs.
Table no. 04 Comparison Table of Various Polymers.(16,20)
|
Polymer Type |
Examples |
Key Properties |
Common Applications |
|
Natural Anionic |
Sodium alginate, pectin |
Ionotropic gelation, swelling |
Oral sustained release |
|
Natural Cationic |
Chitosan, gellan gum |
Mucoadhesion, biocompatibility |
Buccal/colon delivery |
|
Cellulosic |
HPMC, ethyl-cellulose |
Viscosity control, coatings |
Matrix systems |
|
Acrylic/ Synthetic |
Eudragit, PLGA |
pH-sensitivity, biodegradability |
Targeted release |
These polymers are selected based on drug solubility, desired kinetics, and biocompatibility, often blended for optimized performance.
Preparation methods for pharmaceutical microbeads include conventional techniques like ionotropic gelation and advanced scalable processes, selected based on drug properties, polymer type, and desired release profile.
Table no. 05 Comparison of various methods(25)
|
Method |
Pros |
Cons |
Yield/Size (μm) |
Best For |
|
Ionotropic Gelation |
Simple, biocompatible, high EE |
Size variability, pH-sensitive |
70-98%/200-1000 |
Hydrophilic drugs |
|
Emulsion Gelation |
Handles hydrophobics, smooth |
Emulsifier residues |
60-90%/100-800 |
Lipophilic actives |
|
Polyelectrolyte |
No heat/chemicals, strong adhesion |
Limited polymers |
80%/300-900 |
Mucoadhesive |
|
Spray Drying |
Scalable, uniform |
Heat-sensitive drugs |
90%/<100 |
Mass production |
Table no.06 Comparison with Other Dosage Forms(26)
|
Dosage Form |
Advantages over Microbeads |
Disadvantages vs. Microbeads |
Release Control |
Patient Compliance |
|
Tablets |
High dose uniformity |
Dose dumping risk, GI transit variability |
Matrix/ diffusion |
Lower (swallowing) |
|
Capsules |
Taste masking |
Single-unit failure, burst release |
Coating-based |
Moderate |
|
Nanoparticles |
Smaller size, targeting |
Stability issues, toxicity; harder scale-up |
Diffusion/erosion |
High, but IV often |
|
Pellets |
Similar multiparticulate |
Larger size (1-2 mm), less uniform |
Coating |
Comparable |
Microbeads offer significant advantages in drug delivery by providing controlled and sustained release, ensuring even distribution across the GI tract to minimize dose dumping and inter-subject variability, while reducing dosing frequency, side effects, and peak-trough fluctuations through mechanisms like diffusion or erosion in polymer matrices such as alginate or chitosan. They enhance bioavailability for poorly soluble drugs (BCS II/IV), enable targeting (e.g., colon via pH-sensitive coatings), improve patient compliance via taste masking and compatibility with liquids/suspensions, and support combination therapies in multiparticulate form.
Table no. 07 Comparison Table(27,28)
|
Aspect |
Advantages |
Disadvantages |
|
Release Profile |
Predictable, sustained (up to 24h) |
Potential burst (10-30% initial) |
|
Patient Factors |
High compliance, reduced side effects |
Variable GI transit affects absorption |
|
Production |
Versatile polymers, targeted options |
Scale-up, reproducibility issues |
|
Cost/Efficacy |
Bioavailability boost for insoluble drugs |
Higher cost, excipient needs |
Involves diffusion, swelling/erosion, osmotic pressure, and stimuli-responsive processes, each tailored by polymer matrix design to achieve zero-order or predictable kinetics.
Diffusion-Controlled
Drug molecules diffuse through polymer pores or matrices following Fick's law or Higuchi model: Q=D(2C-Cs)Cst
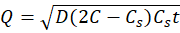
, where Q is cumulative release, D diffusion coefficient, C total concentration, and Cs
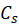
solubility. Common in reservoir systems (coated cores) or dense matrices like ethylcellulose/alginate microbeads; initial lag followed by steady release up to 24h.
Swelling/Erosion-Controlled
Hydrophilic polymers (e.g., HPMC, gellan gum) imbibe fluid, swell to enlarge mesh size (Peppas: Mt/M∞=ktn
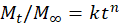
, n>0.45 anomalous), then erode via hydrolysis/enzymatic degradation. Surface erosion yields zero-order; bulk erosion shows burst. Aceclofenac alginate microbeads exemplify pH-dependent swelling in neutral media.
Osmotic Pressure-Controlled
Semi-permeable coatings (e.g., cellulose acetate) allow water influx, generating hydrostatic pressure that ruptures pores or expands core, expelling drug at constant rate. Nifedipine osmotically active microbeads maintain steady release independent of pH.
Stimuli-Responsive
Hybrid mechanisms predominate in natural gum microbeads, optimized via coatings to minimize burst (<20%).
FUTURE ASPECTS
Future aspects of drug-loaded microbeads for controlled release are set to transform pharmaceutical delivery by integrating nanotechnology, smart materials, and personalized medicine. Stimuli-responsive "smart" microbeads—featuring pH-sensitive Eudragit blends, thermosensitive PNIPAAm copolymers, or enzyme-triggered chitosan-gelatin hybrids—enable precise site-specific activation, such as colon-targeted gellan gum microbeads that dissolve only in IBD microenvironments to minimize systemic exposure and boost efficacy for chronic conditions. Nanohybrid systems combining microbeads with liposomes, solid lipid nanoparticles, or metal-organic frameworks (MOFs) achieve >95% drug loading and dual-release kinetics, exemplified by doxorubicin-alginate-MOF conjugates for redox-triggered cancer therapy. 3D printing and microfluidics facilitate patient-specific dosing with monodisperse beads (PDI<0.1), using AI-optimized designs to tailor size (100-500 μm) and polymer ratios, addressing GI transit variability. Sustainable biodegradable exudate gums (karaya, khaya) and marine polysaccharides (agarose, fucoidan) replace synthetics, providing mucoadhesion for oral peptides and vaccines, while layered microbeads support combo therapies for diabetes-hypertension and floating-magnetic hybrids extend gastric retention >12h. Innovations like core-shell architectures and FDA QbD frameworks tackle GMP scale-up, IVIVC, and burst release challenges, with Phase II trials for PLGA-biologics-loaded microbeads signaling market entry by 2030 and ICH Q12 enabling chronotherapy, gene delivery, and implantable depots.
CONCLUSION
Drug-loaded microbeads have widely regarded themselves as an innovative tool in the field of controlled drug release, which can close the distance between traditional dosage form and more modern therapeutics due to their multi-particle structure, high entrapment efficiencies (70-98%) and the flexibility in release mechanisms ranging from diffusion-based Higuchi kinetics, through stimulus-responsive zero-order profiles, and beyond. Through providing a uniform GI distribution, dose-reducing characteristics and enhanced bioavailability for BCS Class II/IV medications, microbeads have superior patient compliance and therapeutic consistency to tablets and capsules, whereas natural polymers like alginate, gellan gum, and chitosan offer new eco-friendly solutions with mucoadhesive and gastroretentive properties. Preparation steps such as ionotropic gelation and spray-drying allow for scalable solutions, despite burst release and scale-up reproducibility problems that should be resolved in future by improvements of nanohybrids, 3D printed personalized beads and QbD scaffolds. With rapid clinical translation as clinical translation speeds up—demonstrated by trials of microbeads now under way, through Phase II: biologics and oncology application is part of a new era, and emerging phase II trials for biologic technologies like microbeads represent a significant advancement in precision medicine, chronotherapy and combination therapies for treating chronic diseases with precision treatment strategies to enhance the outcomes of chronic diseases and reduce the side effects and healthcare costs. The article highlights their central importance in the pharmaceutical landscape of drug development as they provide a basis for formulation scientists toward the next-gen, multiparticulate system of such a wide set of next generation multi-drug solutions in pharmaceutical sciences.
REFERENCES




Sakshi Divate, Dr. Sanghadeep Sukhadeo Gajbhiye, Dr. Swapnil Phalak, Dr. Mohan Microbead Innovations in Controlled Drug Release: Advanced Formulation Strategies and Therapeutic Potential Int. J. of Pharm. Sci., 2026, Vol 4, Issue 4, 908-917 https://doi.org/10.5281/zenodo.19437694
 10.5281/zenodo.19437694
10.5281/zenodo.19437694