Abstract
Gel is a partial preparation with an external solvent that can be non-polar ( organogels) or polar (hydrogels), existing in a three-dimensional network structure of different sizes. Organogels are bicontinuous systems formed by a gelling agent and a nonpolar solvent, which may or may not contain water molecules in the self-assembled structure of the gelling agent. Gelling agents may have a physical or chemical effect when used at a rate of 15% in the environment, they cause self-assembly of interconnected fibrous structures, thus creating a three-dimensional network structure. The resulting three-dimensional network structure blocks the flow of external non-polar phases. Sterols, sorbitan monostearate, lecithin, and cholesterol anthraquinone derivatives are examples of gelling agents. Special properties such as thermal reversibility, viscoelasticity and versatility provide organogels with a longer shelf life and ease of preparation. The ability of the organogel to capture hydrophilic and hydrophobic compounds in its structure also expands the scope of the organogel used in various delivery products. These properties ensure that excellent matrices can deliver effective medications over a long period of time, thus increasing the patient's chances of compliance. Hybridization with other materials may also change their drug delivery capacity. Organogels have many potential uses; so this article talks about many aspects of them.
Keywords
Organogels, Organogelators, Drug delivery, Lecithin Organogel, Organogel Method, Pluroic Lecithin Organogel.
Introduction
A gel is defined as a semisolid, cross-linked system consisting of agglomerated particles interpenetrated by a liquid. Gels can be called hydrogels or organogels and can be distinguished based on the polarity the gel has; For example, if the liquid phase in the gel is water, it is called hydrogel, whereas if the liquid phase in the gel is the liquid phase, it is. water is called hydrogel. Organic solvents are called organogels. Organogels are carriers used to deliver drugs to the desired site. Organogels consist of gelling agents, which are their building blocks. Gelling agents are generally some high molecular weight products (such as sorbitan derivatives, lecithin, fatty acid derivatives, biuret compounds, etc.). The gelling agent helps to form the 3D structure of the mesh network due to the integration of self-generated fibrous structures, which are formed by some physical or chemical interactions when one stops the body gel used at <15>
Advantages
- The organogels are easy to prepare.
- The organogels are more stable than other gel types.
- Organogels are increases the penetration of the drug into the skin.
- Do not disturb the metabolism.
- Not sensitive to moisture.
- Thermodynamically stable.
- Use drugs with short half-lives.
- Provide Controlled drug release, longer shelf life and longer working time.
- Reduce the dose frequency.
- Are Less oily and can be easily removed from the skin.
- In nature both lipophilic and hydrophilic substances can be combined.[2,3]
Disadvantages
- The drug must have suitable distribution coefficient, otherwise the drug will not penetrate the skin.
- This method is not suitable for chemicals that cause irritation or sensitize the skin.
- Lecithin must be in pure form, otherwise gelation will not occur.
- Lecithin is expensive and cannot be used on a large scale.
- It should have a good storage space.
- The gel tends to shrink after standing for a while and some of the liquid gets squeezed out, this is called syneresis.[2,3]
TYPES OF ORGANOGELS
- Lecithin Organogels:
Lecithin organogels have excellent physicochemical properties, making them suitable for use in cosmetics, and they are mainly used in cosmetics. These can be used for transdermal delivery of various hydrophilic and lipophilic drugs. Lecithin is a valuable substance that can be isolated from many animals and plants (except egg yolk) and is therefore biocompatible, safe and stable. It is a potential carrier for many bioactive agents. Lecithin is a phosphatidyl choline derivative of phospholipids. It has been observed that lecithin will not form a gel if its phospholipid content is less than 95%. The idea of forming organogels with lecithin was first proposed by Luisi and Scartazzini in 1988. Lecithin only gels when used in its pure form (for example, hydrogenated forms of soy lecithin do not cause gelling). Therefore, the unsaturated fatty acids found in natural lecithin are very important.[1]
- Pluronic lecithin Organogels (PLO):
The PLO is a tool developed by pharmacists in the United States in the 1990s. Pluronic Lecithin Organogel is an opaque yellow gel made from isopropyl palmitate, soy lecithin, water and the hydrophilic polymer Pluronic F127. The difference between PLO and its precursor lecithin gel is the presence of Pluronic F127, a hydrophilic polymer that gels water, and more water compared to oil. Therefore, PLO is not a true organogel but can be considered "organogel" due to its name. Pluronic F127 is added to the original lecithin organogel to stabilize the gel formulation. Example- PLO is often used as an ointment or antidote for haloperidol, prochlorperazine, secretin, and some hormones. PLO is also being studied/approved as a tool for oral and mucosal use. [4]
- Sorbitan Monosterate Organogels:
A mixture of sorbitol monostearate (Span 60) and sorbitol monopalmitate (Span 40) that has been found to gel more organic solvents at low pressure. Span 60 gel was found to be more stable than Span 40 gel and more studies were conducted on it. Thermo-reversible gels are prepared by heating the gelling agent/liquid mixture in a water bath at 60 °C (which causes the gelling agent to disperse in the liquid medium) and cooling the resulting suspension until it then solidifies to an opaque white, semi-solid state. solid gel. Cooling causes the affinity of the solvent and gelator molecules to decrease and the gelator molecules themselves to aggregate into small tubes. X-ray diffraction and freeze-fracture studies show that sorbitol monostearate molecules are arranged in an inverted bilayer in the tube. Sorbitan monostearate organogels are opaque, thermally reversible semisolids whose microstructure consists of surfactant tubes dispersed in a continuous organic phase. Inverted ring vesicles are the precursors of surfactant tubules. Using hot-phase optical microscopy, a gelation process was observed as the isotropic sol phase of sorbitan monostearate in isopropyl myristate was cooled. It was determined that the difference in the structure of the ring shaped vesicle at the gelation temperature grew in the organic phase. These toroids are thought to be similar to other vesicles, liposomes, and liposomes, except for their toroidal (rather than spherical) shape and different properties.[5]
- Eudragit Organogels:
Eudragit organogels are made from a mixture of polyols (propylene glycol, glycerol), high concentrations (30-40%) of Eudragit (L or S) and liquid PEG. To prepare Eudragit organogels, the drug is first dissolved in PEG and this drug is added to Eudragit powder. Mix this mixture with a mortar and pestle for about a minute. This study showed that the concentration of Eudragit and the amount of drug directly affected the concentration of the gel. Gel viscosity increased with Eudragit concentration and decreased with increasing dosage. At low drug concentration, the gel has high hardness and stability. [6]
- Micro/Nano-emulsion based Organogels:
Microemulsions are defined as thermodynamically stable, transparent, optically isotropic liquid systems composed of water, oil, and surfactants, often combined with appropriate cosurfactants. Microemulsions are known to increase drug bioavailability through both local and systemic pathways. Microemulsions appear to be able to act more topically on the mucosa than gels and creams. Using microemulsion gels as carriers can increase the penetration into the system by many methods, more molecules will be dissolved in microemulsions. In addition, microemulsions have a positive effect on the penetration of drugs by changing their distribution coefficients by causing a change in the thermodynamic activity of the drugs they contain. stratum corneum Additionally, surfactants reduce the activity of the stratum corneum. Nanoemulsions are thermodynamically stable, transparent (translucent) oil and water dispersions stabilized by an interfacial film composed of surfactant and co-surfactant molecules with droplet sizes less than 100 nm. [5]
- Limonene GP1/PG Organogels:
GP1 (dibutylauroylglutamide)/PG (propylene glycol) limonene, a terpene, was found to be a highly permeable organogel that could be prepared by mixing appropriate amounts of the enhancer and thus various transdermal GP1 was placed in limonene and PG and then incubated. Formulations at 120 °C to increase the permeability of bioactive substances. As the mixture cools, it forms a translucent layer on top, increasing the bioavailability of the white gel. Bioactive substances in dermal tissue. Limonene has been shown to be present in GP1/PG. In addition to limonene, many other terpene based compounds (e.g., linalool, farnesol, and cineole) were also incorporated into the organogels, causing changes in rheological properties, but there were no significant changes in GP1/PG organogels. The success of the adhesive. This type of penetration increases the penetration rate of bioactive substances.[2]
- Poly (ethylene) Organogels:
The only two systems that have been extensively tested for drug delivery applications are polyethylene and P(MAAco-MMA) organogels. A study dating back to the 1950s and involving 300 patients showed that polyethylene organogel (PO) patches were non-irritating and hypoallergenic. In a related study, 326 patients treated with Spectrocin-containing PO were compared with patients treated with petrolatum-based Spectrocin alone. Both topical antibiotics clear pyoderma and secondary infections within 3-5 days, but PO has been shown to provide faster and more effective release. Poly(ethylene) is also used in the formulation of 5-iodo-2'-deoxyuridine for the treatment of oral herpes simplex lesions. The 10% drug-loaded formulation demonstrated resolution of herpetic lesions within 3 days of initiation of treatment compared to 1-2 weeks in untreated patients.[5]
- Supramolecular Organogels:
Recently, molecules with various structures, from simple alkanes to complex phthalocyanines, have been found to be gelling agents. Recently, there has been interest in studying gels obtained with low molecular gelling agents (supramolecular or simple molecular gels). The motivation for this is not only to understand the aggregation patterns of gels of different sizes, but also to explore their potential for future technological applications. By incorporating spectrally active or receptor units as part of the gelling agent molecule, the gel is made sensitive to external stimuli such as light and chemicals. This makes them suitable for applications such as hearing and processing. The diversity of the gel process allows it to be used as a model for the preparation of new inorganic systems for use in catalysis and separation. Gels derived from liquid crystals (anisotropic gels) have been prepared as functional materials for (rewritable) recording, for example. Supramolecular gels are used in controlled release applications, oil recovery, gelation of cryogenic fuels, etc. It is very important. They can also serve as advertisements for various applications. Example: cyanochalcone. [8]
METHOD FOR PREPARATION OF ORGANOGELS
Heating the oil-surfactant mixture at 60°C produces a clear liquid that turns into an organogel upon cooling. According to the phase diagram, firstly, lecithin solution was prepared by mixing lecithin in organic solvent using a magnetic stirrer. The organogel was formed by adding water using a micropipette syringe. Sometimes heat may be used to complete the treatment. Lecithin and organic solvents are mixed together to form an oil phase and then allowed to stand overnight to ensure complete destruction. To prepare the aqueous (polar) phase, add Pluronic to ice cold water and stir to ensure complete mixing. The resulting PLO is mixed with the aqueous phase of Pluronic using high shear mixing technology with a magnetic stirrer. Fatty acid gelling agents can also be used to form organogels by first dissolving them in a water-in-oil emulsion at a higher temperature and then lowering the temperature. As the temperature increases, the solubility of the gelling agent decreases, causing the gelling agent to precipitate and self-assemble into a network of intermingling tubules, forming a gel pattern.[7,10]

Fig 1 : Method of formation of organogels by fluid-filled fiber mechanism[10]
Fluid-Filled Fiber Method:
This is a well-known process for making organogels, where reverse micelles are formed by dissolving surfactants and cosurfactants in nonpolar solvents. After water is added, the reverse micelles transform into tubular reverse micelles. Elongated reverse micelles entangle toc form a 3D network that immobilizes nonpolar solvents.[14]

Fig 2 : Method of formation of organogels by fluid-filled fiber mechanism[10]
Solid Fiber Method:
In the solid fiber process, the nonpolar solvent and the organogelator are heated together to form a nonpolar solution of the organogelator. After cooling at room temperature, the organic gelling agent precipitates in the form of fibers that physically interact with each other to form a three-dimensional network structure that processes non-polar heavy substances.[6]

Fig 3 : Method of formation of organogels by solid fiber mechanism[11]
Hydration Method:
Gels can be prepared by direct immersion of inorganic substances, which allows the separation of the dispersion. In addition to water carriers, other substances such as propylene glycol, propyl gallate, and hydroxypropylcellulose can be used to improve gel formation.[2]
Novel Methods:
The organogel preparation process generally requires a long heating time and moderate. The organogel is prepared using new equipment, homogenized at high speed, and then heated with a microwave. Efren et al. Triclosan organogels were prepared using Carbopol 974 NF in PEG 400. Different fractions (2-4%) of carbomer were dispersed in PEG 400. The resulting dispersion was homogenized at 24,000 rpm. Two methods were used to heat the dispersion. The first involves heating at 80 °C with stirring at 200 rpm. In the second method, the crack was subjected to micro irradiation (1200W/1 hour) for 2 minutes. The results show that microwave heating is necessary to prepare carbomer organogel. Due to the reduction in time and energy, this method has good prospects for use in industry.[15]
Organogel Formulations For Drug Delivery:
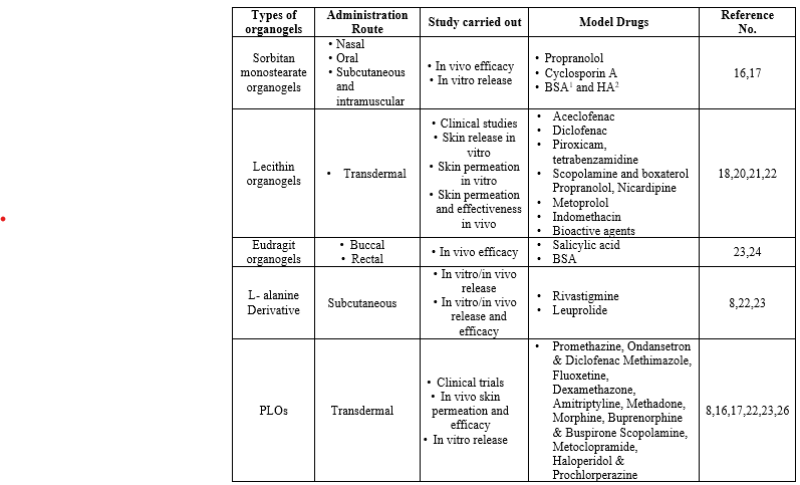
TYPES OF ORGANOGELATOR
- Aryl Cyclohexanol Derivatives:
These are 4-tert-butyl-1-arylcyclohexanol derivatives. The properties they express in gels will vary depending on the nature of the nonpolar solvent contained in the organogels. They have low solubility in nonpolar solvents, so they can appear as cloudy or clear preparations depending on the nature of the nonpolar solvent involved. Their bodies are strong at room temperature. They can gel only when the phenyl groups in their structure are in an axial configuration. Derivatives with phenyl groups in the equatorial configuration cannot form gels. They help to obtain organogels with the desired thermal reversibility. Some examples of this group are CCL4, benzene, cyclohexane, etc.[1]
- Gemini Organogelator:
"Gemini" means "Twins". This word comes from Latin. The first L-lysine Gemini organogelator was developed by Suzuki. It consists of two L-lysine chains of different lengths connected by amide bonds. The length of this chain is proportional to the gelling capacity of the gelling agent. They have good gelling properties. They can immobilize many non-polar solvents. A good example of this class is ketones, alcohol, etc. It is bis (N-lauroyl-L-lysine ethyl ester) acetamide that can handle solvents such as.[10,25,28,29]
- Polymer organogels:
These are gelling substances containing long chains. These are gelling substances that have the ability to gel. Their molecular size is over 2 kilodaltons. They can form gels even when used in very low concentrations. They can appear in different shapes (straight, leg, etc.). If you change their chemical formula slightly, you can change the effectiveness of their gelation. They can also be classified as organic or organogelants. They are considered physical organogels if they form antibodies in the organogel network, causing cross-linking; They are considered organogels if they form non-covalent bonds that lead to cross-linking in the network. The transition temperature from the gel state to the sol state is also very low. They have a better gel than other LMOGs. These usually include L-lysine derivatives and polyethylene, polycarbonate, polymethylmethacrylate, polyester, etc. Includes other examples such as.[10,25,27,28,31]
- Low-Molecular –Weight Organogelators (LMWOs):
These are gelling agents of small weight (?3000 Daltons). The assembly is a support for gel-forming ability. They form thermoreversible transparent gels. Nonpolar solvents in which they can be immobilized include benzene. These are the most commonly used organogelators. They can weaken the water level even when used in low concentrations (<2>
CONCLUSION
Organogels, whether nonpolar (organogels) or polar (hydrogels), provide a variety of three-dimensional connections with applications ranging from drug delivery to cosmetics. Organogels, especially those produced by two systems of gelling agents and nonpolar solvents, have special properties such as thermal reversibility, viscoelasticity, and ease of customization. These properties make them attractive for drug formulations, extending shelf life and increasing drug delivery. Various organogelators, including sterols, sorbitan monostearate, lecithin, and anthraquinone derivatives, contribute to this difference. Special types, such as lecithin organogels, Pluronic lecithin organogels (PLO), sorbitan monostearate organogels, and Eudragit organogels, have unique physicochemical properties that expand their use in drug delivery and cosmetic applications. New organogel preparation methods, such as microwave heating and liquid fiber, are being developed that offer better options and save time. Meanwhile, various organogelators, from arylcyclohexanol derivatives to polymeric organogelators, expand the tools for the production of gels with good properties. Organogels are widely used in drug release control, oil recovery and many other applications. Ongoing research into supramolecular organogels and their adaptability combined with advanced preparation techniques make them key players in future advances in medicine and materials science. Overall, extensive research on organogels demonstrates their importance in problem solving and offers opportunities in many fields.
REFERENCES
- Hemlata D. Hire, 2Kunal S Surwade, 3Dipti G. Phadtare. A REVIEW ON ORGANOGELS: AS A NEW FORMULATION. Journal of Emerging Technologies and Innovative Research. January 2023, Volume 10, Issue 1.
- Garg T, Bilandi A, Kapoor B, Kumar S and Joshi R. Organogels: Advanced and Novel Drug Delivery System. International Research Journal of Pharmacy. 2011;2 (12):15-21.
- Choudhary P, Agrawal S, Choukse R and Chaturvedi P. A Review of Novel Drug Deleivery System of Pluronic lecithin organogel. International Journal of Pharmaceutical Erudition. 2013;3(2):65- 80.
- R, Gupta P and Yadav A. Organogels: A Review International Journal of Research in Pharmacy and Life Sciences. 2013;1(2):125-130.
- Nigar Kadar Mujawar, Sangramsinh Laxman Ghatage and Veerendra C. Yeligar . Organogel: factors and its importance. International journal of pharmaceutical, chemical and biological sciences .2014, 4(3), 758-773
- Mujawar NK, Ghatage SL, Yeligar VC. Organogel: Factors And Its Importance. Int J Biol Chem Sci [Internet]. 2014;4(3):758–73. Available from: https://www.researchgate.net/publication/305359032
- Rahman M, Hussain A. Lecithin-microemulsion based organogels as topical drug delivery system (TDDS). IJCRR: International Journal of Current Research and Review. 2011;3(3):22-33.
- Anda V, Leroux J, Organogels and their use in the drug delivery- A Review, Journal of Controlled Release, Accepted 27 September 2007,p 18-59.
- Chang JY, Lim GS, Jung BM, Lee SJ, Song HH and Chulhee Kim. Synthesis of Polycatenar-Type Organogelators Based on Chalcone and Study of Their supramolecular Architectures. Chemistry of Materials. 2007;19:460-467.
- Sahoo S, Kumar N, Bhattacharya C, Sagiri SS, Jain K, Pal K, et al. Organogels: Properties and applications in drug delivery. Designed Monomers and Polymers. 2011;14(2):95-108.
- Jadhav NK, Patil KA, Patil JK, Patil PA and Pawar SP. A Review on Organogels: Lipid Based Carrier Systems. Pharma Science Monitor An International Journal of Pharmaceutical Sciences. 2012;3(4):3132-3143.
- Zeng L, Lin X, Li P, Liu F-Q, Guo H, Li W-H. Recent advances of organogels: From fabrications and functions to applications. Prog Org Coatings. 2021;159:106417.
- Rahman M, Hussain A. Lecithin-microemulsion based Organogels as Topical Drug Delivery System (TDDS). Int J Curr Res Rev. 2011;3(3):22–33.
- Thakur VK, Thakur MK, Voicu SI. Polymer Gels: Perspectives and Applications. Gels Horizons: From Science to Smart Materials. Singapore: Springer; 2018. pp. 419
- Gökçe EH, Yurdasiper A, Korkmaz E, Özer Ö. A novel preparation method for organogels: High-speed homogenization and micro-irradiation. AAPS PharmSciTech. 2013;14(1):391-397
- Pisal S, Shelke V, Mahadik K, Kadam S. Effect of organogel components on in vitro nasal delivery of propranolol hydrochloride. AAPS PharmSciTech. 2004;5(4):92-100
- Liu H, Wang Y, Han F, Yao H, Li S. Gelatin-stabilised microemulsion-based organogels facilitates percutaneous penetration of Cyclosporin A In Vitro and dermal pharmacokinetics In Vivo. Journal of Pharmaceutical Sciences. 2007;96(11):3000-3009
- Varshosaz J, Andalib S, Tabbakhian M, Ebrahimzadeh N. Development of lecithin nanoemulsion based organogels for permeation enhancement of metoprolol through rat skin. Journal of Nanomaterials. 2013;2013:1-10
- Jean-Christophe L and Anda V. Review on: Organogel and Their Use in Drug Delivery. Science Direct Journal of Controlled Release. 2008:179-192.
- Raut S, Bhadoriya SS, Uplanchiwar V, Mishra V, Gahane A, Jain SK. Lecithin organogel: A unique micellar system for the delivery of bioactive agents in the treatment of skin aging. Acta Pharmaceutica Sinica B. 2012;2(1):8-15
- Shaikh IM, Jadhav KR, Gide PS, Kadam VJ, Pisal SS. Topical delivery of aceclofenac from lecithin organogels: Preformulation study. Current Drug Delivery. 2006 Oct;3(4):417-427
- Kumar R, Katare OP. Lecithin organogels as a potential phospholipid-structured system for topical drug delivery: A review. AAPS PharmSciTech. 2005;6(2):E298-E310
- Murdan S. Organogels in drug delivery. Expert Opinion on Drug Delivery. 2005;2(3):489-505
- Goto S, Kawata M, Suzuki T, Kim N-S, Ito C. Preparation and evaluation of Eudragit gels. I: Eudragit organogels containing drugs as rectal sustained-release preparations. Journal of Pharmaceutical Sciences. 1991;80(10):958-961.
- Mehta C, Bhatt G, Kothiyal P. A Review on organogel for skin aging. Indian J Pharm Biol Res. 2016;4(3):28.
- Shehipunov YA. Lecithin organogel: a micellar system with unique properties, Colloids and Surfaces: A Physicochemical and Engineering Aspects, 183-185, 2001, p. 541-554.
- Bonam SP. Preparation and evaluation of pluronic lecithin organogel containing ricinoleic acid for transdermal drug delivery. University of Toledo; 2013.
- Aiswarya Madathil, Nethaji Ramalingam, Neethu Krishna, Mishahal TM BG. No Title. SSRG Int J Pharm Biomed Eng. 2021;8.
- Sagiri SS, Behera B, Rafanan RR, Bhattacharya C, Pal K, Banerjee I, et al. Organogels as matrices for controlled drug delivery: a review on the current state. Soft Mater. 2014;12(1):47–72.
- Vintiloiu A, Leroux J-C. Organogels and their use in drug delivery - A review. J Control Release [Internet]. 2008;125(3):179 -92. Available from:
- Martinez RM, Rosado C, Velasco MVR, Lannes SC da S, Baby AR. Main features and applications of organogels in cosmetics. Int J Cosmet Sci. 2019;41(2):109–17.
- Reddy G, Reddy D. ORGANOGELS-A REVIEW INTRODUCTION. 2010 Dec 1; Garg T, Bilandi A, Kapoor B, Kumar S, Joshi R. Organogels: advanced and novel drug delivery system. Int Res J Pharm. 2011;2(12):15–21.
- Madamwar D, Thakar A. Entrapment of enzyme in water-restricted microenvironment for enzymemediated catalysis under microemulsion-based organogels. Appl Biochem Biotechnol. 2004;118(1):361–9.
- Rees GD, Robinson BH. Microemulsions and organogels: properties and novel applications. Adv Mater. 1993;5(9):608–19.
- Esposito CL, Kirilov P. Preparation, characterization and evaluation of organogel-based lipstick formulations: Application in cosmetics. Gels. 2021;7(3):97.
- Mosquera Narvaez LE, Ferreira LM de MC, Sanches S, Alesa Gyles D, Silva-Júnior JOC, Ribeiro Costa RM. A Review of Potential Use of Amazonian Oils in the Synthesis of Organogels for Cosmetic Application. Molecules. 2022;27(9):273


 Rushikesh Khemnar* 1
Rushikesh Khemnar* 1
 Sonia Singh 2
Sonia Singh 2
 Nikhil Sutar 3
Nikhil Sutar 3
 Pallavi Kaple 4
Pallavi Kaple 4
 Avadhut Kulkarni 5
Avadhut Kulkarni 5
 vitthal Dherange 6
vitthal Dherange 6
 Pandurang Chavan 7
Pandurang Chavan 7




 10.5281/zenodo.10815894
10.5281/zenodo.10815894