
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
SIMS College of Pharmacy, Affiliated to Acharya Nagarjuna University, Guntur, Andhra Pradesh, India.
Carica papaya, commonly referred to as papaya, is a member of the Caricaceae family, boasting a diverse array of compounds and biomolecules, notably papain, with significant industrial and medicinal value. This comprehensive review meticulously examines the properties, extraction techniques, and pharmacological importance of papain, coupled with an exploration of the pharmacological activities inherent in C. papaya. Extensive research data from reputable sources such as Google Scholar, PubMed, Scopus, and Web of Science were scrutinised to collate pertinent information on the plant and its enzyme. Within C. papaya, a spectrum of bioactive compounds were identified, encompassing alkaloids, tannins, phenolics, flavonoids, saponins, terpenoids, sugars, glycosides, amino acids, steroids, and the pivotal enzyme papain. Papain extraction methods and their activity modulation in response to pH, temperature, and other physicochemical parameters were examined in detail. This review provides a nuanced analysis of papain, addressing both its pharmacological implications and potential side effects. Its far-reaching applications in the medical, industrial, and pharmaceutical sectors were meticulously discussed, unveiling its market potential. Furthermore, C. papaya emerged as a reservoir of pharmacological activities, exhibiting antioxidant properties, anti-cancer potential, wound healing capabilities, digestive disorder management, and anxiolytic effects. The research underscores the promising potential of C. papaya and papain for diverse biological applications, prompting a call for further studies to unlock their therapeutic benefits, particularly in the realm of public health. Looking ahead, continued advancements in drug development hold the key to unravelling the intricate bioactive components of papaya and its papain cysteine proteases. These insights are poised to catalyse their development and application, promising substantial contributions to the betterment of humanity. The screening of antioxidant of plant extracts indicates plant potential as therapeutic agent, which could play a very important role in drug development and as a health supplement. This study was designed to explore the antioxidant property of Carica papaya leaf extracts. The extraction was performed by the process of cold maceration using solvent methanol, water and chloroform. The antioxidant activity was evaluated by using DPPH scavenging method. The phytochemical tests revealed the presence of alkaloids, glycoside, saponin, resins, flavonoids, phenols and tannins. In DPPH radical scavenging method, the scavenging of C. papaya extract reached to 96% in methanol,96% in water and 98% in chloroform and the standard gallic acid was found to be 83%. The extract showed with highly moderate antioxidant activity which signifies that papaya leaf is a good source of health promoting constituents that can be used for therapeutic purposes.
The Carica papaya plant belongs to the Caricaceae family and is commonly called papaya. It is also known as papaw, paw-paw, kapaya, lapaya, tapaya, papayao, papaya, papaia, papita, lechosa, fruta bomba, mamon, mamona, mamao and tree melon in different parts of the world. Interestingly, a separate plant Asimina triloba of the Annonaceae family is also known as pawpaw. Papaya is native to the Caribbean Coast of Central America and is grown in tropical and subtropical parts of the world [1]. Among the 31 species that make up the botanical family Caricaceae and the genus Carica, the papaya species is the most economically significant and frequently grown species [2].
During the early years of growth, papaya develops a single stem, which can develop into heavy lateral branches in highly fertile soil, promoting favourable growth conditions [3]. The leaves of a mature papaya plant are palmate with deep lobes and are supported by smooth, hollow petioles. The stems, fruits, and leaves of the papaya plant contain a copious amount of latex, although the source is unclear [1].
Ripe papaya fruits have a slight resemblance to melons, and they are rich in retinol and ascorbic acid. Every part of the plant has been found to be important (Fig. 1). They have been reported to be used in the treatment of diseases just like every other plant. Every part of the papaya plant, including its leaves, fruits, seeds, bark, stem, and root, can be processed for nutritional and pharmaceutical purposes (Fig. 1). They are rich sources of antioxidants and mineral ions, and they also contain phytochemicals and B vitamins [4]. Papaya enzymes have been reported to treat arthritis and colon cancer. C. papaya contains biologically active compounds and enzymes [1].
The protease is utilised as a commercial meat tenderiser because it can disintegrate dead tissue without harming living cells. We reported its usage for wound care [8]. Papain is used in the management and treatment of allergies and injuries [1]. According to some studies indicate that the enzyme helps in the prevention of diabetic-dependent-heart-disease. Papain is known for its efficiency and less destructive nature compared to other proteases, making them commonly used in cell isolation procedures on no certain tissues [6]. It is a constituent of contact lens solution and is used in clarifying beer, and in the pharmaceutical industry. In a study traditional medicine through the administration of papain extract from papaya leaves is shown to be effective against the nematodes as papain causes damage to the cuticles of the nematodes [7]. Papain is generally a sulfhydryl protease obtained from C. papaya. Several studies have focused on the extraction and application of papaya plant enzymes [8]. The enzyme is known for its ability to tenderise flesh and enhance wound healing. This is particularly useful for people who experience poor wound healing, as they can avoid expensive and complex materials that may not be readily available. This report presents an in-depth analysis of papain, focusing on its properties, extraction methods, and pharmacological significance. Additionally, it explores the pharmacological activities associated with Carica papaya. In this review, the pharmacological side-effect of papain, as well as its medical, industrial, and pharmaceutical importance while exploring its market potential. Hence, knowledge from this review offers intelligence into the pharmacological potential of C. papaya and may find its application in alternative therapeutics towards the treatment of infectious as well as terminal diseases [9]. More industrial and research effort and investment on the plant subject is able to contribute immensely towards areas of agri-business, health, and science. In addition, the relevance of this study gives a perspective toward the achievement of Sustainable Development Goal. The knowledge and application of the papain enzyme and other bioactive components of papaya in this study will guide the pharmaceutical and research industries to ensure healthy lives and promote the well-being of all at all ages. Hence, this present study will also be important to pharmaceutical industries, medical industries, researchers, policymakers, governments of nations, and students [9].
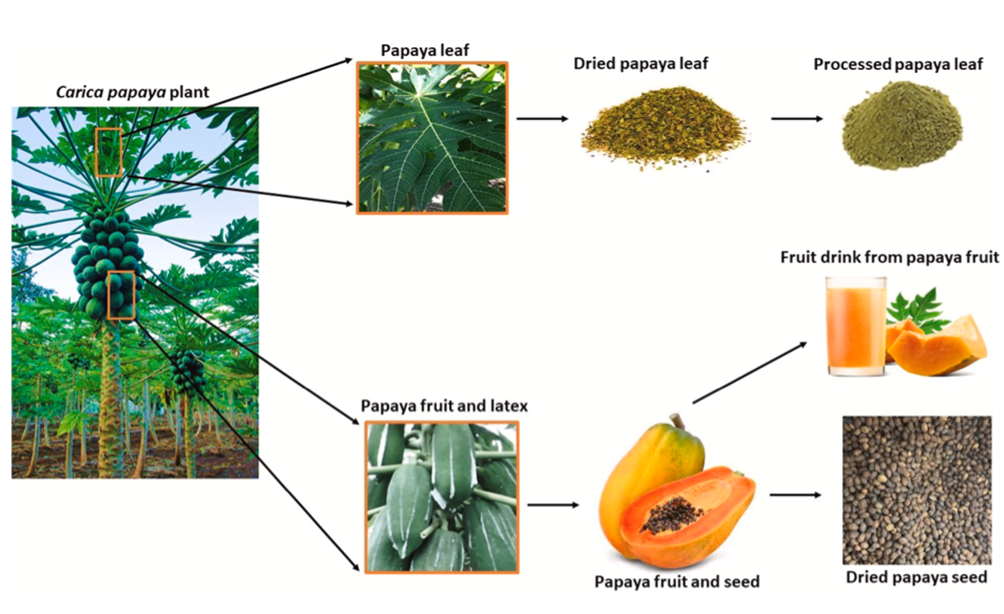
Fig. 1. Papaya plant and its processing. Different parts of the plant including the leaves, fruits, latex, and seed can be processed for different use. This may be for medicinal, nutraceutical or industrial purposes. [10]
Nutritional Profile
Papaya is a rich source of essential nutrients and bioactive compounds that vary by plant part:
Therapeutic and Biological Activities
Research highlights a broad spectrum of health benefits associated with different parts of the papaya plant:
Industrial and Pharmaceutical Applications
The versatility of C. papaya extends into several commercial sectors:
Phytochemical Composition of Papaya Leaves:
Phytochemicals are chemical components, naturally found in different parts of plants, which make many species beneficial for therapeutic uses. Indeed, leaves of papaya are known to have various health-promoting phytochemical, as it arose from chemical analysis performed in various studies which clearly illustrated the presence of significant amounts of alkaloids, saponins, glycosides, flavonoids, phenolic compounds, enzymes, amino acids, lipids, carbohydrates, vitamins, and minerals [11]. There were seven flavonoids found in papaya leaves, which were named as quercetin, kaempferol 3-rutinoside, quercetin3-(2g-rham- nosylrutinoside), quercetin 3-rutinoside, kaempferol 3-(2G- rhamnosylrutinoside), myricetin 3-rhamnoside. Caffeic acid, protocatechuic acid, quercetin, 5,7-dimethy coumarin, p-coumaric acid, and chlorogenic acid are among the phenolic substances found in the leaves [12]. There is evidence to suggest that leaves contain a wide range of phytochemicals, 3 including carpaine, kaempferol 3-(2G-glucosylrutinoside), kaempferol 3-(2″rhamnosylgalactoside), 7-rhamnoside, kaempferol 3-rhamnosyl-(1->2)-galactoside-7-rhamnoside, luteolin 7-galactosyl-(1->6)-galactoside, orientin 7-O-rham-noside, 11-hydroperoxy-12, 13epoxy-9-octadecenoic acid, palmiticamide, and 2-hexaprenyl-6-methoxyphenol [13]. Due to these potent bioactive components, extracts of the aforementioned leaves can be used to prepare nutraceuticals and herbal medicinal formulations. Chemical constituent and structure of some important compounds of C. papaya leaves are illustrated in Figure 2. There were reports that C. papaya leaves were used with other herbs to heal ailments. Traditional doctors in Nigeria use it to treat diabetes, while in Cameroon, they combine it with other herbs to treat malaria and other fungal infections and aboriginal Australians’ record using decoctions of the leaf as an anticancer remedy [14]. The functional bioactive components of leaves of papaya can elevate the overall antioxidant potential of blood. The leaves of papaya plant are well known to have papain, cystatin, chymopapain, tocopherol, phenolic acids, cyanogenic glucosides, glucosinolates, and vitamin C as main phytochemical [15]. Mainly alkaloids, saponins, glycosides, phenolic compounds, and flavonoids are responsible for the anti-inflammatory and anti-cancerous properties of papaya leaves [16]. Vitamins, minerals, and amino acids of papaya leaves are quite helpful to improve the total haemoglobin, proteins, and immunity of human system [17]. Carpaine along with dehydrocarpaine-I and dehydrocarpaine-II are most important health-promoting and major bioactive components found in the leaves of papaya. Due to the presence of carpaine, these herbal leaves are utilised in Ayurveda formulations for treating various physical disorders and viral fevers such as dengue and chikungunya. The aforementioned alkaloid has the ability to calm high blood pressure and fast heart rate and is effective for the uterus marked relaxation, the bronchioles dilatation, and movement of the intestinal strips along with anti-plasmodial properties [18]. Carpaine has also been reported to have potent anti cancerous and anthelminthic properties [19]. Its concentration has been reported the highest in mature leaves of papaya, i.e., 9.30 mg/g, followed by fruit pulp, i.e., 4.90 mg/100 g, fruit peel, i.e., 1.99 mg/100 g, and seeds, i.e., 0.65 g/g [20]. Leaves constitute different components in varying proportions such as 8.3% of carbohydrates, 38.6% of vitamin C, 5.6% of pro- and 0.23% of phosphoric acid. A good amount of tannin (0:85 ± 10−3 ± 1:76 ± 10−4 M, 0.824%) in papaya leaf extract has been reported by a few researchers [21]. Papaya leaves have been found to have highest total phenolic compounds as 424:89 ± 0:22 mg GAE/ 100 g of the dry sample followed by the 339:91 ± 9:40 in unripe papaya, 272:66 ± 1:53 in ripe papaya, and 30:32 ± 6:90 mg GAE/100 g in seeds [22]. Due to the aforementioned bioactive compounds, a very good antioxidant potential of 90% has been recorded in its leaves already. Researchers also reported a good concentration of calcium and magnesium, i.e., 3480 mg/kg and 5928 mg/kg, respectively; other minerals like iron (558 mg/kg), zinc (33.4 mg/kg), manganese (22.88 mg/kg), chromium (7.50 mg/hg), and copper (2.16 mg/kg) were also found in fair amount [14].

Figure 2: Chemical constituents and structure of some important compounds of C. papaya leaves. (a) Constituents of C. papaya leaves along with functional uses; (b) chemical structures of important bioactive compounds present in C. papaya leaves. [23]
MATERIALS AND METHODS:
Plant materials
The leaves were collected. The leaf’s were washed and cleaned, preserved in small polythene bags with 70% alcohol which was poured into these bags to kept the leaves fresh and then dried in shade condition.
Preparation of extract
The extraction was performed by the process of cold maceration using methanol, water and chloroform as solvents. The marc of the extracts were collected by filtering through Whatman No.1 filter paper, dried on water bath, covered, abed and subjected towards the screening of various phytochemicals and tests.
Phytochemical screening
Phytochemical evaluation has been carried out for all the extracts according to standard protocols [24,25].
In-vitro Antioxidant assay:
DPPH scavenging method:
The antioxidant activity of plant extract and standard antioxidant were measured in-vitro in terms of hydrogen donating or free radical scavenging ability by using the 1,1 diphenyl-2-picryl hydrazyl (DPPH). DPPH stable free radical method is an easy, rapid and sensitive way to survey the antioxidant activity of a specific compound or plant extract [26]. The purple DPPH becomes yellow diphenyl picryl hydrazine while reduced. Extract solution was prepared by dissolving 1mg of dry plant extract in 1ml methanol. An aliquot of 2.5ml of 75 µM DPPH solution in methanol and 0.5ml of plant extract in methanol at various concentrations were mixed and incubated at room temperature for 90 minutes and absorbance of the test mixture was read at 517 nm using a UV-VIS spectrophotometer (Systronic 119) against a DPPH control containing 1ml of methanol measurements. The experiment was done in triplicate. Gallic acid was used as standard.
Scavenging % = [(AC-AS)/AC] ×100
Where, AC is Absorbance of control
AS is Absorbance of Sample
Scavenging of hydrogen peroxide:
H2O2 itself is not very reactive, it is a weak oxidising agent, but sometimes it can be toxic to cell because it may give rise to hydroxyl radical in the cells. It can cross cell membrane rapidly, once inside the cell, H2O2 can probably react with Fe2+ and possibly Cu2+ ions to form hydroxyl radical and this may be the origin of many of its toxic effects. A solution of hydrogen peroxide (40mM) was prepared in phosphate buffer solution (pH- 7.4) (0.6 ml, 40mM). Absorbance of hydrogen peroxide was determined 10 minutes later at 230 nm against a blank solution containing the phosphate buffer without hydrogen peroxide [27].
Scavenging % = [(AC-AS)/AC] ×100
Where AC is Absorbance of control
AS is Absorbance of Sample
ABTS Radical Scavenging Assay:
The ABTS scavenging determination was carried out [28]. To generate ABTS radical cation (ABTS+), a 7 mM solution of ABTS was reacted with 140 mM potassium phosphate at room temperature in the dark for 12–16 hours before use. The solution was diluted with ethanol to give an absorbance of 0.70 ± 0.02 at 732 nm. Then, the sample solution (25 µL) was fully mixed with the ABTS solution (2 mL) and reacted in darkness for 6 min. The results were expressed as the Trolox equivalent.
FRAP Assay:
The FRAP determination method was carried out using Benzie and Strain’s methodology (1996).
The FRAP test solution contained 10 mM 2,4,6-Tri(2-pyridyl)-s-triazine, 20 mM ferric chloride and 300 mM sodium acetate. The FRAP solution (1.8 mL) was mixed with the sample solution (10 µL) and incubated at 37°C for 10 min. The absorbance of the coloured reaction product was measured at 593 nm, and Trolox was considered as the standard [29].
Total Phenolic Content Assay:
The Folin-Ciocalteu technique was used to determine the total phenolic content. The Folin-Ciocalteu reagent (0.5 mL) was mixed with distilled water (7.9 mL) and the sample extract (0.1 mL), and the mixture was neutralized with sodium carbonate (1.5 mL, 20%). After 2 hours in the dark, the absorbance at 765 nm was measured, and gallic acid was used as the standard [30].
Total Flavonoid Content Assay:
The total flavonoids content was determined using ferric chloride coloration. Briefly, distilled water (5 mL) and the sample extract (1 mL) were combined and reacted with sodium nitrite (0.15 mL, 5%) for 6 min. The aluminium chloride (0.15 mL, 10%) was added and reacted for further 6 min. It was neutralised with sodium hydroxide (2 mL, 4%), and the mixture was reacted for another 15 min. The absorbance was measured at 420 nm with rutin as the standard, and the result was represented as a percentage [31].
Statistical Analysis:
All tests were repeated three times, and the results were expressed as the mean ± standard deviation (SD). All data were examined with SPSS software and Duncan’s test (P < .05) was used for mean comparisons. The Pearson method was used to determine the correlation between antioxidant activity and total flavonoids and total phenols. The final results were obtained using a detailed evaluation of the Technique for Order Preference by Similarity to Ideal Solution (TOPSIS) comprehensive evaluation analysis [32,33]. The evaluation indexes in the TOPSIS approach are rated according to how close they are to the idealised target.
RESULTS AND DISCUSSIONS:
The plant Carica papaya leaves has been investigated in systematic way covering initial pharmacognostical, phytochemical study to rationalise its use as a drug.
Phytoconstituents:
Successful evaluation of botanical phytocompounds from plant material is largely dependent on the type of solvent used in the extraction procedure. Hence, extracts were screened to find out the presence of various phytoconstituents in Carica papaya leaves. The results of qualitative screening of phytochemical components in leaves of Carica papaya revealed the presence of alkaloids, glycoside, saponin, resins, flavonoids, phenols and tannins presented in Table 2.
Table 2: Phytochemical screening of Carica papaya leaf extracts [34]
|
Constituents |
Tests |
Extracts |
||
|
|
|
Water |
Methanol |
Chloroform |
|
Alkaloids |
Dragendorff`s Test |
+ |
+ |
- |
|
Mayer`s Test |
+ |
+ |
- |
|
|
Carbohydrates |
Benedict`s Test |
- |
- |
- |
|
Fehling`s Test |
- |
- |
- |
|
|
Flavonoids |
Lead Acetate Test |
+ |
+ |
- |
|
Phenol |
Ferric Chloride Test |
+ |
- |
- |
|
Glycoside |
Legal Test |
+ |
+ |
- |
|
Killer- Killani Test |
+ |
+ |
- |
|
|
Protein |
Xanthoproteic Test |
- |
- |
- |
|
Warming Test |
- |
- |
- |
|
|
Tannin |
Gela&n Test |
+ |
+ |
- |
|
Ferric Chloride Test |
+ |
+ |
- |
|
|
Lead Acetate Test |
+ |
+ |
- |
|
|
Steroids |
Salkowski`s Test |
+ |
- |
- |
|
Saponin |
Foam Test |
+ |
- |
- |
Antioxidant study:
In DPPH free radical scavenging method, the scavenging activity of Carica papaya leaf extract at concentration of 1mg/ml reached to 96%, 96% and 98% in methanol, water and chloroform extracts respectively. On the other hand, the Gallic acid at same concentration had 83% of scavenging activity. The data presented in Table 3 and figure 2 depicts that the extracts possess the scavenging character in accordance with the standards and showed their activities at different concentration. The study reveals that the extract of Carica papaya exhibits the proton-donating ability and could serve as free radical inhibitors or scavengers, acting possibly as primary antioxidants.
Hydrogen peroxide scavenging activity of the Carica papaya extract on hydroxyl radical is shown in Table 3 and figure 2. H2O2 is highly important because of its ability to penetrate biological membranes. The correlation between total flavonoids content and hydrogen peroxide in this study was found positive (r = 0.932). The percentage of H2O2 scavenging activity of Carica papaya extracts reached to 82%, 95% and 66% in methanol, water and chloroform respectively. And the standard used i.e. Gallic acid was found to be 95%.m It is therefore biologically advantageous for cells to control the amount of hydrogen peroxide that is allowed to accumulate. C. papaya extract also showed hydrogen peroxide decomposition activity in a concentration dependent manner. The decomposition of H2O2 by methanol, water and chloroform extracts of C. Papaya and free radical scavenging activity resembles with the report described [35].
CONCLUSIONS
It can be concluded by saying that the plant Carica papaya is a very precious gift for human being for the bearing of some important phytoconstituents. The phytochemical tests confirmed that the plant is a store house of glycoside, alkaloids, tannins, flavonoids, saponin, phenol and resin etc. The result suggests that the methanolic and aqueous extracts show the presence of these constituents more distinctly than the chloroform extract. At various concentrations of the extracts the scavenging activity of C. Papaya extract reached to 96% in methanol, 96% in water and 98% in chloroform in DPPH radical scavenging method against the standard Gallic acid found to 83%.
REFERENCES









Syed Sabreen, Thangjam Billgates Singh, T. Sachin Meitei, Muppuri Venkata Gopi, Yalagala Jaya Manoj, Manitha Kumari, Sk. Nousheen, Kakani Nagajyothi, Thangabalan B., Review Article on Antioxidant Activity of Carica Papaya Leaf Powder, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 3, 3280-3290. https://doi.org/10.5281/zenodo.19229408
 10.5281/zenodo.19229408
10.5281/zenodo.19229408