Abstract
A simple, sensitive and accurate Visible Spectrophotometric method was developed for the determination of abacavir sulphate in pharmaceutical dosage forms. Abacavir sulphate was diazotized in acidic medium at 0-5 ºC temperature. The diazonium salt was reacting with coupling reagent ?-naphthol to form orange red colored azo dye in an alkaline medium. The orange red colored solution was measured at 470 nm against reagent blank. The orange red color azo dye is stable for about 6 hours. The calibration curve is found to be linear over a concentration range of 20 to 100 µg/ml of abacavir
Keywords
Abacavir sulphate, ?-naphthol, Visible spectroscopy
Introduction
Abacavir sulphate [1] is chemically {(1S, 4R)-4-[2-Amino-6- (cyclopropylamino)-9H-purin-9- yl]-2-cyclopentene-1-methanol}. It is a nucleoside reverse transcriptase inhibitor with antiretroviral activity against HIV. It is administered alone or in combination therapy with other antiretrovirals. The structure of abacavir sulphate is in fig.1. Survey of literature reveals that the abacavir sulphate was determined by using various methods which includes Spectrophotometric method1-11, High Performance Liquid Chromatography12 and RP-HPLC method13. The present study describes simple, sensitive, accurate, rapid and economical spectrophotometric methods for the estimation of abacavir sulphate in tablet dosage forms.
Fig.1. Structure of abacavir sulhate
Determination of Maximum wave length of Abacavir sulphate
The wavelength of maximum absorbance of the diazotised abacavir sulphate treated with ?-naphthol solution is ascertained by the following procedure. 1.0 ml of abacavir solution (100µg/ml) is transferred into a 10 ml volumetric flask. To this, 2.0 ml of 0.1N hydrochloric acid and 1.0 ml of cold 0.1N sodium nitrite solution are added. The resultant solution is well mixed, and then allowed to stand for five minutes at 0-5oC temperature for diazotization. To this solution 1 ml of 1% urea solution is added and shaken frequently for nitrogen gas to escape. Then 1.0 ml of 0.5N sodium carbonate and 1ml of 1% ?-naphthol solution are added and the volume is made to 10 ml with methanol. The absorbance of the orange colour formed is measured in the wavelength range of 400 to 560 nm, against the reagent blank. The overlain spectrum of abacavir sulphate is given in fig.2.

Fig.2: Spectrum of abacavir sulphate
The above overlain Spectrum has maximum absorbance at 470 nm and all further studies are made at 470 nm.
MATERIALS AND METHODS
Instrumentation
Spectronic 1000 plus UV Visible Spectrophotometer with 1 cm matched quartz cells was used for all spectral and absorbance measurements.
Hydrochloric acid (0.1 N):
Hydrochloric acid solution (0.1N) is prepared by diluting the requisite volume of concentrated AR hydrochloric acid with distilled water and standardized by usual procedure.
Sodium nitrite (0.1N):
0.69 g of Sodium nitrite is dissolved in distilled water and the resulting solution is made up to the mark in 100 ml standard flask with distilled water. This solution is standardized by the usual analytical procedure.
?-Naphthol solution(1%):
Accurately weighed 1.0 g of ?-Naphthol solution is dissolved in methanol and the volume adjusted to 100 ml with methanol.
Urea solution (1%):
Accurately weighed 1 gm of urea is dissolved in double distilled water and the volume made up to 100 ml with double distilled water.
Preparation of abacavir sulphate solution:
50 mg of abacavir sulphate was dissolved in 50 ml methanol. 1.0 ml of the above stock solution is further diluted to with methanol to get working concentration of 100 µg/ml.
Assay Procedure
Various aliquots of the standard abacavir solution ranging from 0.2-1.0 ml are transferred into a series of 10 ml volumetric flasks. To each flask, 2.0 ml of 0.1N hydrochloric acid solution and 1.0 ml of cold 0.1N sodium nitrite solution are added. The resultant solution in each flask is well shaken and allowed to stand for five minutes at 0-50C temperature for diazotization to complete. 1.0 ml of 1% urea solution is added to each flask and the solution is shaken frequently to allow nitrogen gas to escape. Then 1.0 ml of 0.5N sodium carbonate solution and 1.0 ml of 1% ?-naphthol solution are added and the volume in each flask is made up to 10 ml with methanol. An orange colour is formed. The maximum absorbance of the orange coloured solution is measured at 470 nm against the reagent blank. Calibration graph is obtained by plotting absorbance values against the concentration of abacavir solution. The calibration curve is found to be linear over a concentration range of 20 to 100 µg/ml of abacavir. The amount of abacavir present in the sample is estimated from the calibration graph. The results are presented in fig.2.

Preparation of the sample solution:
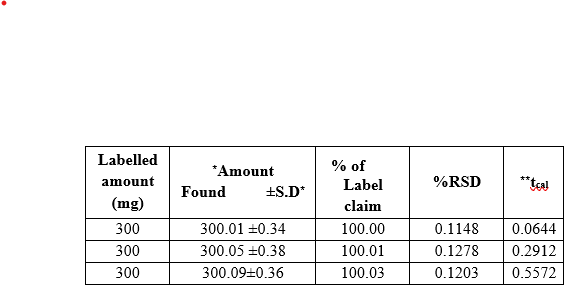
Powdered tablet equivalent to 50 mg of the drug is weighed accurately and transferred into a 50 ml beaker and mixed well with 30 ml of methanol. The solution is filtered and transferred into a 50 ml volumetric flask and the volume is made up to 50 ml with methanol. The concentration of the drug solutions is now 1mg/ml. This stock solution is further diluted to obtain the working concentration of 100 µg/ml and analysed as under the assay procedure for bulk drug samples. The results are presented in Table.2.
Table.1: Optical characteristics of proposed method
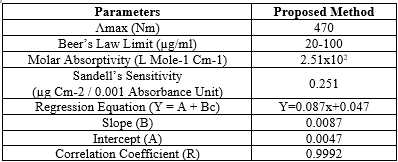
Table: 2 Assay of abacavir in pharmaceutical formulations
RESULTS AND DISCUSSIONS
Abacavir undergoes diazotisation when treated with sodium nitrite and hydrochloric acid. The excess nitrous acid during the diazotisation is removed by the addition of urea solution. The solution was shaken frequently to allow the nitrogen gas to escape. The diazonium cation reacts with the coupling reagent, ?- naphthol to produce an orange azo coloured solution. The orange coloured solution is measured at 470 nm against reagent blank.. The colour of the product is stable for more than 24 hours. The calibration curve is linear over the range of 20-100 ?g/ml of abacavir. The optical characteristics of the proposed method such as absorption maxima, Beer´s law limits, molar absorptivity and Sandell´s sensitivity are presented in Table1. The molar absorp¬tivity and Sandell´s sensitivity values show that method is sensitivity. The regression analysis using method of least squares was made for the slope (b), intercept (a) and correlation (r) obtained from different concentrations and results are summarized in the Table 1. The value of correlation coefficient was 0.999, which indicated the good linearity of calibration lines. The percent relative standard deviation calculated from the five measurements of abacavir shown in Table.1. The % RSD is less than 2, which indicates that the method has good reproducibility. The standard deviation values are low indicates high accuracy and reproducibility of the method. The‘t’ calculated values are compared well with the theoretical value of 2.78 there by indicating that the precision of the method is good. There no effect of additives and excipients such starch, calcium lactose and glucose in the concentrations those present in general pharmaceutical preparations. The proposed method is simple, accurate, precise and selective for the estimation of abacavir in bulk and in dosage forms.
REFERENCES
- Appala Raju, Venkateswar Rao J, Vanitha Prakash and Mukkanti K, Spectrophotometric estimations of abacavir sulphate in pharmaceutical formulations. E-Journal of Chemistry. 2008;5(3), 511-514.
- Naresh kumar K, Appala Raju N and Mukkanti K.Spectrophotometric estimation of abacavir sulphate in pharmaceutical formulations. Int. J. Chem. Sci., 2008;6(4): 2066-2072.
- Amudhavalli V, Lakshmi KS, Dheeraj Varma Kalidindi, Ramya Sree Surapaneni, Rudraraju Raju SRK and Vamsi Kumar Pichikala. Difference Spectrophotometric Estimation of Abacavir sulphate in Bulk and Tablet dosage form J. Chem. Pharm. Res., 2011;3(3):122 -119.
- Veldkamp AI, Sparidans RW, Hoetelmans RM, Beijnen JH. Quantitative determination of abacavir (1592U89), a novel nucleoside reverse transcriptase inhibitor, in human plasma using isocratic reversed-phase high-performance liquid chromatography with ultraviolet detection. J Chromatogr B Biomed Sci Appl. 1999;736(1-2):123-8.
- Devmurari VP, Patel RB. Simultaneous spectrophotometric determination of lamivudine and abacavir in the mixture. Int J Pharm Sci Res. 2010;1:82-86.
- Prasada Rao CH, Seshagiri Rao JVCN, Dhachina Moorthi D, Lakshmi Aswini G and K. Ashok K. Visible spectrophotometrric determination of abacavir sulphate in bulk drugs and pharmaceutical formulations. Research J. Pharmacy and t Technol., 2011; 4(2): 234-236.
- Venkatamahes D and Hachina D M.. Visible spectrophotometrric determination of Abacavir Sulphate in bulk drug and tablet dosage forms. International J. Pharma Tech Res., 2011; 3(1): 356-359.
- Venkatamahesh R, Dhachinamoorthi D. Visible spectrophotometric determination abacavir sulphate bulk drug tablet dosage form. International Journal of Pharm Tech Research. 2011;3(1):356-367.
- Sudhakar Reddy J, Prabhavathi K. and Chakravarthi !E. A simple spectrophotometric determination of abacavir sulphate in pharmaceutical formulations, Asaian Journal of pharmaceutical and Health Sciences, 2011;1(3): 130-132.
- Srihari G, Rami Reddy Nand Chakravarthi IE. Spectrophotometric Method for the Determination of Abacavir Sulphate in Pharmaceutical Preparations. Global Journal of Pharmacology2011; 5 (3):172-175.
- Akhilesh S, Tokey, Mughar, Suryawnshi, Pranav P. Develoment and validation of UV Spectrophotometric method for estimation of abacavir sulphate in tablet dosage forms. International Journal of Current Pharmaceutical Research. 2022; 14(5):112-116.
- Ravitch JR, Moseley CG. High-performance liquid chromatographic assay for abacavir and its two major metabolites in human urine and cerebrospinal fluid. J Chromatogr B Biomed Sci Appl. 2001;762(2):165-73.
- Sudha T, kumar RVR, Hemalatha PV. RP-HPLC method for simultaneous Estimation of lamivudine and abacavir sulfate in tablet form. International Journal on Pharmaceutical Biomedical Res. 2008;1(4):108-13.


 N. Rami Reddy*
N. Rami Reddy*
 J. Kalpana
J. Kalpana





 10.5281/zenodo.13333311
10.5281/zenodo.13333311