Post-marketing surveillance plays a pivotal role in ensuring the safety and efficacy of medicines after their approval for clinical use. In India, adverse drug reaction (ADR) reporting remains a major challenge due to underreporting, lack of awareness, inadequate training, and limited integration of pharmacovigilance into healthcare practice. This study highlights the current status of ADR monitoring in India, the functioning of the Pharmacovigilance Programme of India (PvPI), and the barriers faced by healthcare professionals in spontaneous reporting. It emphasizes the importance of strengthening reporting systems, improving regulatory frameworks, and encouraging active participation of physicians, pharmacists, nurses, and patients in ADR reporting. Enhancing digital reporting platforms, integrating pharmacovigilance into medical curricula, and conducting awareness campaigns are key strategies to improve ADR documentation. Strengthening post-marketing surveillance will not only improve drug safety but also build public trust in the healthcare system and contribute to evidence-based therapeutic action.
Pharmacovigilance; post-marketing surveillance; Adverse Drug Reactions (ADR); Drug safety; Regulatory framework; Healthcare professionals; India; Spontaneous reporting; Patient safety
An adverse drug reaction (ADR) is defined as an unintended, harmful, or undesirable effect caused by a medication when used at normal doses for prevention, diagnosis, or treatment of a disease, or for modifying physiological function. The World Health Organization (WHO) formally defines an ADR as "a response to a drug which is noxious and unintended, and which occurs at doses normally used in man." ADRs are a significant public health concern, contributing to patient morbidity, mortality, and increased healthcare costs. They are a leading cause of hospital admissions and can complicate medical treatment, reduce patient adherence, and affect quality of life.1
Adverse reactions of drugs continue to remain as an important public health issue. Safety monitoring of medicines is the responsibility of all stakeholders of the healthcare system since it continues to be an important cause of morbidity and mortality.2 In some countries adverse drug reactions are among the leading causes of mortality. The safety of patients and the safe use of medicines are crucial for health policy development and delivery of the best healthcare. To prevent or reduce harm to patients thereby improving public health, the safety of medicines in clinical use must be monitored and evaluated through specialised systems. This requires a well-organised pharmacovigilance system to be established. Thus, a pharmacovigilance system is defined as a system used by an organisation to monitor the safety of authorised medicinal products and detect any change to their benefit-risk balance. A pharmacovigilance system is characterised by its structures, processes and outcomes. To run an effective pharmacovigilance system, a protocol is required for reporting adverse reactions associated with drug use. Therefore, National Coordination Centre (NCC) aims to ensure the systematic and effective functioning of PvPI by publishing and implementing its guidance document for reporting Adverse Drug Reactions (ADRs).2
This guidance document lays down requirements and guidance for reporting ADR and significant safety issues related to drugs regulated by the Central Drugs Standard Control Organization (CDSCO). This document does not establish legally enforceable responsibilities. This has been prepared by the NCC and approved by Working Group. The purpose of this document is to present the importance of pharmacovigilance in India, to record the growth and potential as a significant discipline within medical science, and to describe its impact on patient welfare and public health. This document also highlights the importance of collaboration and communication at local, regional and international levels to ensure that pharmacovigilance delivers its full benefits.2
It also provides guidance to stakeholders on good pharmacovigilance practices, assessment of data regarding drugs including vaccines and blood products.2 post-marketing surveillance (PMS) ensures that medicines approved for public use are safe and effective. It monitors real-world drug interactions and identifies previously unknown adverse effects. In India, the Pharmacovigilance Programme of India (PvPI) oversees PMS, with ADR reporting being its core component. However, India's ADR reporting rates are significantly lower than global benchmarks due to systemic challenges, including underreporting and lack of awareness among healthcare professionals (HCPs). This report investigates these gaps and offers practical solutions to improve pharmacovigilance in India, ensuring safer healthcare practices.2
Need of Study :
India is one of the largest producers of pharmaceuticals worldwide and the market is expanding with a pace. The PV in India continues to evolve, grow, and improve. These tools such as ADR-reporting forms, PvPI helpline, and the “ADR PvPI” mobile application are now not only limited to the AMCs under PvPI, but as a new initiative, these tools could be displayed by pharmacy stores, private-sector hospitals, and corporate hospitals to enhance the reach to the patients/caregivers.
Further, the efforts are being done to make these tools available to every district hospital across the country. Provision of filling the ADR forms downloaded through the website of IPC is a suitable practice as it is less time consuming and more efficient, however, it is still a challenge to provide a convenient e-reporting system in India similar to that available in the USA and other countries. The “ADR PvPI” mobile application and toll-free helpline being the most convenient ways to report ADRs are also the most technically advanced sources available in India; however, efforts are being made to get this mobile app available in official languages other than English to enhance user-acceptance and convenience.
The convenience of availability of package inserts (PIs) or patient information leaflets (PILs) to a PV-official must also be encouraged as a huge amount of drugs are marketed in India but the information about the PIs/PILs is not easily available to the PV-officials, and hence, efforts are required from the regulatory authority, to make these PIs/PILs available on the website of CDSCO, India.
This will not only help the PV-officials but will encourage consumers to refer these PIs/PILs before taking any medicines. The ambit of PvPI may be expanded to cover veterinary drugs, herbal drugs, and cosmetics based on feasibility. Recently, through S.O. 648 (E) dated February 11, 2020, as notified by the MoHFW, the “devices” definition has been specified, therefore, the scope of determining the safety of medical devices is also expanding.
PvPI has special focus in conducting various kinds of trainings and capacity-building workshops, and SDPs will not only help train HCPs, budding PV professionals and students but help strengthening the consumers' knowledge about ADRs and their reporting system in India.
Aim and Objective :
Aim:
Strengthening Post-Marketing Surveillance in India : A Study on Adverse Drug Reaction reporting.
Objective:
- To analyze the current status of ADR reporting in India under the Pharmacovigilance Programme of India (PvPI).
- To identify the barriers and challenges faced by healthcare professionals in ADR reporting.
- To assess the level of awareness, knowledge, and attitudes regarding ADR reporting among healthcare providers, including physicians, pharmacists, and nurses.
- To compare India's ADR reporting system with global best practices in pharmacovigilance.
- To suggest actionable recommendations and strategies for improving ADR reporting rates and post-marketing surveillance in India.
- Out of all conventional methods for PV, spontaneous ADR reporting is one of the most cost effective and time tested method and it remains as the cornerstone of any successful PV program.
- Unfortunately, under-reporting is a major drawback inherent with it. The effect of educational and other interventions on ADR reporting has been studied in The effectiveness of such interventions is highly variable and is confounded by several factors. To our knowledge, our study is one of the first in India to assess the impact of some of the globally suggested IT on the quantum and quality of ADR reporting.
- Since the applicability and suitability of some of the proposed interventions in an Indian context may differ, the present study was designed to evaluate the effects of a few of the suggested IT on ADR reporting in a tertiary care hospital (TH).
- A quantitative and qualitative analysis of the impact of these IT on ADR reporting and KAP of the prescribers was performed.
Plan of Work :
Strengthening ADR reporting requires a multi-pronged approach. India must prioritize education, digital transformation, and policy reforms. The success of global models demonstrates that robust systems not only improve public health but also enhance healthcare system efficiency.
Challenges of ADR and AEFI reporting
This scoping review revealed that perceptions among HCPs about ADR and AEFI influence their reporting of ADR and AEFI. These findings concur with the results of a study conducted in Thailand which revealed that the negative perceptions about ADR reporting by the majority of the HCPs led to poor adverse events reporting (Srisuriyachanchai et al., 2022). The negative perception may be attributed to the time required to complete the ADR and AEFI forms which increase the HCPs’ workload (Katusiime et al., 2015). This study also revealed that there was low awareness of ADR and AEFI reporting procedures among HCPs. Similar findings were reported from a study conducted in Pakistan, which revealed that the majority of HCPs in the study did not know how to report an ADR at their workplace (Hussain et al., 2022). These findings may be an indication that HCPs do not receive adequate training on ADR and AEFI reporting. This scoping review revealed that low clinical knowledge of ADR and AEFI among HCPs is a challenge to ADR and AEFI reporting. A study conducted in South Africa among HCPs to evaluate their knowledge, attitudes, and practices toward ADR reporting alsorevealed that a lack of knowledge about adverse events was a discouraging factor in reportingthe events. This lack of knowledge may lead to a lack of appreciation of the importance of ADR and AEFI reporting, uncertainty about the outcome of reporting, and a lack of confidence in discussing ADR and AEFI. This scoping review revealed that fear of blame and litigation by HCPs leads to poor ADR and AEFI reporting. These findings concur with the results of a study conducted in Australia, which revealed that the fear of blame and litigation acts as a barrier to the creation of a positive ADR reporting culture. HCPs, therefore, need to be assured that there will be no repercussions associated with ADR and AEFI reporting so that they can feel free to report them.
This study revealed that a lack of training on ADR and AEFI contributes to poor reporting. The problem of a lack of training on ADR and AEFI is not only seen in SSA. A study conducted in Finland revealed that almost half of the participants in the study had not received training on ADR (Sandberg et al., 2022). Lack of training on ADR and AEFI among HCPs makes it difficult for them to confirm them, resulting in the ADR and AEFI not being reported. In addition, if HCPs are not trained on ADR and AEFI, they might not be aware of ADR and AEFI reporting procedures. This study also revealed that other challenges faced when reporting ADR and AEFI include the unavailability of ADR and AEFI reporting forms and the forms not being user-friendly. A study conducted in India also revealed that a lack of ADR reporting was associated with the unavailability of ADR reporting forms at the hospital (Kiran et al., 2014). Where ADR and AEFI reporting forms are not easily available, HCPs may not try to look for the forms since they are usually busy. In addition, HCPs might have forgotten about the ADR and AEFI by the time the reporting forms become available.
This scoping review revealed that challenges associated with the activities of national pharmacovigilance organizations contribute to challenges in ADR reporting. Some of the challenges include the shortage of personnel, an inadequate budget, a lack of a pharmacovigilance centre, and difficulties in communicating with the pharmacovigilance centre. A comparative assessment of the national pharmacovigilance systems in East Africa revealed that pharmacovigilance units were understaffed in all countries included in the study, and Ethiopia and Rwanda did not have a designated budget for pharmacovigilance activities (Barry et al., 2020). A baseline analysis of pharmacovigilance activities in four countries in SSA revealed that Ethiopia, Eswatini, and Nigeria’s pharmacovigilance activities were not directly funded by the governments, while Eswatini di” not have a medicine regulatory authority or general pharmacovigilance guidelines (Tiemersma et al., 2021). Without a pharmacovigilance centre, enough personnel, and an adequate budget, it is difficult to have guidelines and organize training of HCPs on ADR and AEFI. This study also revealed that the lack of feedback from the pharmacovigilance centre posed a challenge to ADR and AEFI reporting. This was also reported in a study conducted in Africa to evaluate pharmacovigilance systems (Sabblah et al., 2022). It is therefore important that HCPs who report ADR and AEFI receive feedback so that they are motivated to continue reporting the events.
Strategies to address the challenges of ADR and AEFI reporting
This review revealed that HCPs require training and mentoring to improve ADR and AEFI reporting. This recommendation was also suggested in a review conducted for Africa. The review suggested that HCPs should be trained in pharmacovigilance, ADR and AEFI during their training as regular in-service training .Training on ADR and AEFI should focus on awareness, knowledge, and reporting. Once HCPs are aware of ADR and AEFI and have knowledge of ADR and AEFI, they are more likely to report them. Regulatory staff training should also be strengthened since they are the ones who monitor ADR and AEFI reporting. Reporting of ADR and AEFI should also be made mandatory to increase the rates of reporting.
This review revealed that ADR and AEFI reporting may be improved by the use of electronic reporting tools, adopting user-friendly ADR and AEFI reporting forms, and improving access to ADR and AEFI reporting forms. A study conducted in East Africa also recommended the use of electronic reporting systems and mobile phone reporting applications as this may increase the number of reports. The study also recommended that national pharmacovigilance systems should establish a mechanism to capture medicine utilization, weigh the drug risk at the population level, and prioritize safety signals (Barry et al., 2020). Where electronic reporting forms are not being used, paper forms should be easily available at all healthcare facilities so that any HCP who needs to report an ADR and AEFI can easily access them.
This scoping review revealed that ADR and AEFI reporting can be improved by the establishment of a national pharmacovigilance centre, national pharmacovigilance guidelines and regulations, adequately funding the pharmacovigilance activities, improving feedback and collaboration, and decentralizing the activities. For regulatory authorities to execute their mandate, they require the necessary infrastructure and resources, including laws, systems, structures, human resources, and financial resources. Human resources should be adequate in terms of numbers, knowledge, and skills. The development of strong and sustainable pharmacovigilance systems that ensure improved reporting of ADR and AEFI requires strong political will and financial support from governments and partners. It is also essential that comprehensive guidelines on ADR and AEFI reporting are developed and implemented (Abiri & Johnson, 2019). Decentralization of pharmacovigilance activities will require healthcare facilities to have policies for universal and inclusive reporting (Adenuga et al., 2020a, 2020b).
This review revealed that community-level strategies that can be used to improve ADR and AEFI reporting include creating awareness and promoting self-reporting, using posters at healthcare facilities, and establishing toll-free telephone lines for ADR and AEFI reporting.
Involving patients in ADR and AEFI reporting is important as patients are the first to notice any problems associated with the medications they are taking. However, for them to be able to recognize ADR and AEFI, they should be provided with information about ADR and AEFI when taking different types of medications. Apart from toll-free telephone lines, mobile applications for ADR and AEFI reporting should be developed to make it easier for patients to report adverse events.
Strengths and limitations of the study
One of the strengths of the study is that it followed PRISMA-P guidelines, which makes it easy for the results to be reproducible. The other strength is that two reviewers independently extracted and synthesized the data, then compared their results, which makes the results believable. The study, however, had several limitations. One of the limitations is that only articles published in English were included in this review, and this may have resulted in language bias. The other limitation is that only three databases were used.
Recommendations include:
- Making ADR reporting a legal requirement.
- Increasing collaboration between PvPI and international organizations.
- Regular audits to monitor the effectiveness of pharmacovigilance initiative.
METHODOLOGY
- A prospective observational study was conducted at Madras Medical College (MMC) and Rajiv Gandhi Government General Hospital (RGGGH), Chennai.
- Study duration: January 2019 to March 2021.
- Used suspected adverse drug reaction (sADR) reporting form from Pharmacovigilance Program of India (PvPI).
- Included all spontaneous ADRs associated with antiepileptic drugs (AEDs) reported by HealthCare Professionals (HCPs).
- Collected patient demographic details, suspected drug(s), and ADR-related information from sADR forms.
- Study was retrospective and record-based, started after Institutional Ethics Committee approval.
- Analyzed ADR reports from August 2023 to July 2024 from various clinical departments.
- Total of 532 ADRs were evaluated for key parameters per IPC SOP, Ghaziabad.
- Studied ADRs using Individual Case Safety Reports (ICSRs) and supporting documents like prescriptions and investigation reports.
- After analysis results are categorized into:
- Demographic details
- Organ systems involved
- Types of ADRs
- Suspected drugs
- Causality assessment
- Severity assessment
- ADR outcomes
- Reporters' qualifications.
Demographic Details
This includes patient-specific information such as age, gender, and sometimes weight. It helps identify which groups are more prone to adverse drug reactions and supports risk stratification.
- Age, gender, and weight of the patients
- Geographic location or hospital unit
- Relevant medical history
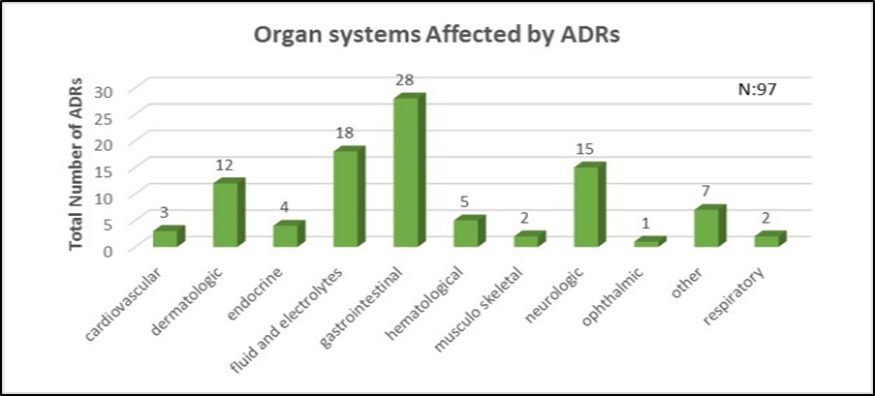
Organ Systems Involved
Refers to the physiological systems affected by the ADR. Categorization (e.g., skin, liver, nervous system) helps understand drug toxicity patterns and guides clinical monitoring.
Body systems affected by the ADRs, categorized using systems like:
- Nervous system
- Gastrointestinal system
- Dermatological system
- Hematologic system, etc.
Types of ADRs
These are the specific manifestations of the reactions, like rash, nausea, or dizziness. Classifying ADRs helps in evaluating the frequency and seriousness of reactions linked to specific drugs.
Classification based on nature of the reaction, such as:
- Rash
- Nausea
- Dizziness
- Liver enzyme elevation
- Seizure aggravation
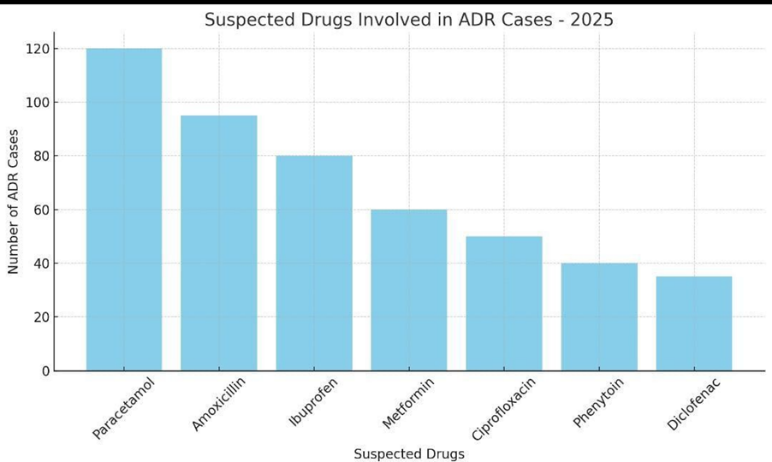
Suspected Drugs
This includes the names of antiepileptic drugs (AEDs) reported to be associated with the ADRs. Understanding which drugs are frequently implicated helps improve prescribing safety.
Specific antiepileptic drugs (AEDs) implicated in the ADRs, e.g.:
- Phenytoin
- Carbamazepine
- Valproate
- Levetiracetam, etc.
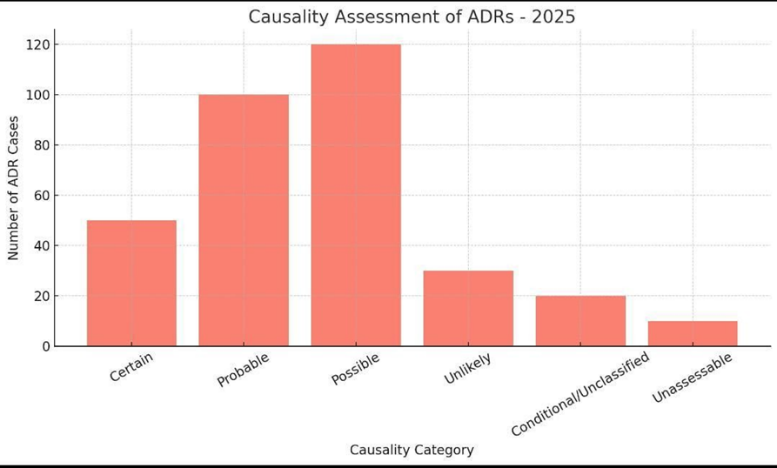
Causality Assessment
An evaluation of the likelihood that a drug caused the reaction. Methods like the WHO-UMC scale or Naranjo algorithm are used to classify ADRs as certain, probable, possible, or unlikely.
Evaluation of the likelihood that the drug caused the ADR. Usually done using WHO-UMC scale or Naranjo Algorithm, classified as:
- Certain
- Probable
- Possible
- Unlikely
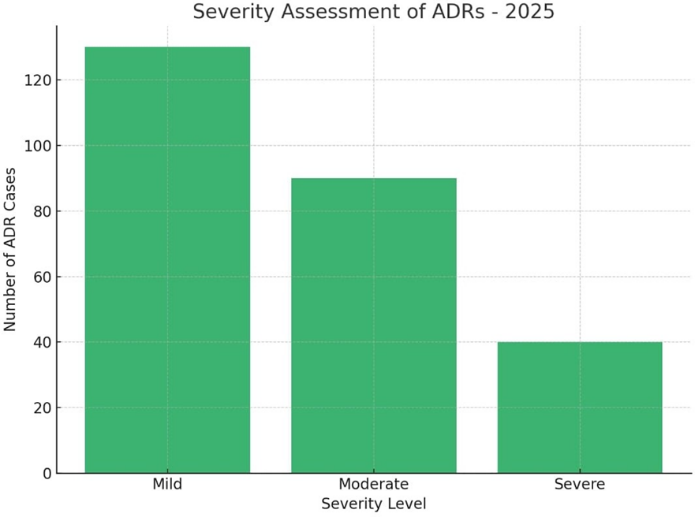
Severity Assessment
This assesses how serious the reaction is—mild (self-limiting), moderate (requires treatment), or severe (life-threatening or requiring hospitalization). Tools like Hartwig’s scale are often used.
Classification of ADRs based on intensity or impact:
- Mild
- Moderate
- Severe
- Can also use tools like Hartwig’s Severity Scale
ADR Outcomes
Refers to the patient's recovery status post-ADR—whether the patient recovered, is recovering, has not recovered, or the outcome was fatal. This information helps assess overall impact and recovery trends. Final status of the ADR after medical intervention:
- Recovered
- Recovering
- Not recovered
- Fatal
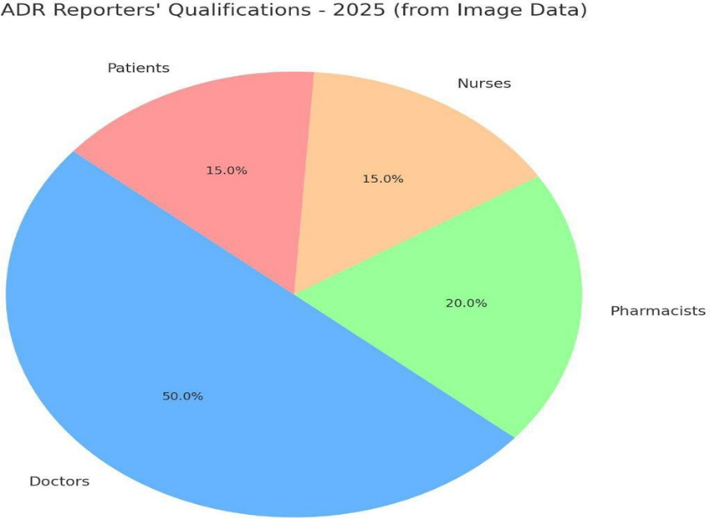
Reporters Qualifications
Denotes who reported the ADR—doctors, nurses, pharmacists, or other healthcare professionals. Reporter qualifications can affect the detail and quality of reports submitted.
Professional background of the individuals who reported the ADRs:
- Doctors
- Pharmacists
- Nurses
- Medical students or interns
RESULTS AND DISCUSSION
- The study focused on ADR reports categorized under WHO-ART SOC, specifically on fetal disorders.
- There has been a significant increase in ADR reports related to fetal disorders, likely due to improvements in the reporting system over the past 20 years.
- An increase in the number and quality of ADR reports has been observed in the last 3 months.
- 87% of the suspected drugs had sufficient informational content in the reports, indicating good report quality.
- In approximately 60% of cases, only one suspected drug was reported, aiding in the evaluation of drug-ADR associations.
- Drugs acting on the immune system were most frequently associated with fetal disorder reports (47% of all drugs).
- This frequent reporting does not necessarily imply these drugs are the most dangerous.
- Immune system-targeting drugs are the most commonly reported anatomical main group in the general population.
- These drugs are of primary interest in ADR studies due to their widespread usage and relevance.
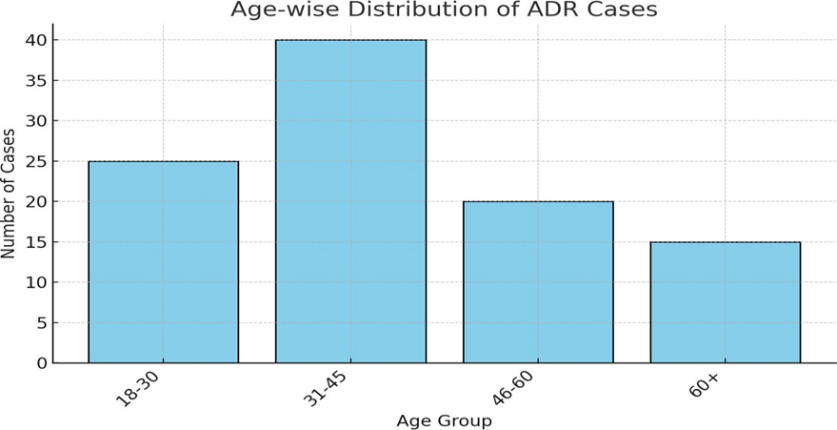
Demographic Details.
- Majority of ADRs occurred in the 31–45 age group (40%).
- Followed by 18–30 years (25%) and 46–60 years (20%).
- Least ADRs were seen in patients aged 60+ (15%).
- Younger adults and middle-aged patients often receive multiple prescriptions, increasing exposure to drugs. Elderly populations may have reduced metabolic capacity but report fewer ADRs due to under-reporting.


 Vishal Rathod *
Vishal Rathod *
 Dr. Vijay Navghare
Dr. Vijay Navghare
 10.5281/zenodo.16926159
10.5281/zenodo.16926159