
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
KLE College of Pharmacy Belagavi, KLE Academy of Higher Education and Research (KAHER), Belagavi, Karnataka, India.
Urolithiasis, or kidney stone disease, is a multifactorial disorder characterized by crystalline deposits in the urinary system. Its global prevalence has risen significantly over the past two decades, making it a major public health concern. The etiology involves genetic, metabolic, dietary, and environmental factors. Kidney stones, including calcium oxalate, uric acid, struvite, and cystine stones, form through distinct mechanisms. The stone formation process includes supersaturation, nucleation, crystal growth, aggregation, and retention in the kidneys. Hormonal influences, particularly sex hormones, impact stone formation, while the gut and urinary microbiome are emerging regulators of urolithiasis susceptibility. Additionally, immune responses to urinary crystals trigger inflammatory cascades that promote stone development. Preventive measures focus on dietary modifications, hydration, and pharmacological approaches. Treatment options range from minimally invasive procedures, such as extracorporeal shock wave lithotripsy, to surgical interventions. Recent research explores novel therapeutic strategies, including microbiome modulation and immunotherapy, to enhance management and prevention. This review comprehensively examines the mechanisms underlying stone formation, current treatment strategies, and future directions for improved urolithiasis management.
Urolithiasis, or kidney stones, is a common condition caused by mineral buildup in the kidneys, bladder, or urethra, posing a global health challenge(1). Kidney stones are primarily calcareous, such as calcium oxalate (over 80% of cases), or non-calcareous, like uric acid (5-10%) or struvite. In the U.S., they affect 10.6% of men and 7.1% of women (2). In India, 12% of people face this issue yearly, especially in states like Gujarat and Rajasthan (3). Without treatment, stones may recur at a 10% annual rate (4). Men (12%) are affected more than women (6%), with peak cases between ages 20-40 (5). The exact causes of stone formation remain unclear (6). Stones form through crystal nucleation and growth when urine is oversaturated with insoluble substances(7,8). No drug fully treats urolithiasis; preventive measures like thiazide diuretics are used, but surgical options such as shock wave lithotripsy or ureteroscopy are frequent, with a 50% recurrence rate (9). Surgery can also cause issues like high blood pressure or kidney damage (10). The kidneys, reddish-brown and bean-shaped, lie in the retroperitoneal space, regulating fluids, electrolytes, and acid-base balance while clearing toxins. Blood enters through renal arteries, exits via veins, and urine drains to the bladder through ureters. The glomerulus filters 20% of blood, reabsorbing nutrients like glucose and amino acids, while expelling waste. Human kidneys contain about one million nephrons (11). Urine forms as filtrate passes through tubules, with the proximal tubule reabsorbing most nutrients and the loop of Henle concentrating urine. Urine is 95% water, 2.5% urea, and contains minerals and enzymes, while key molecules are reabsorbed (12). The urinary system—kidneys, ureters, bladder, and urethra—filters blood and produces urine. The proximal convoluted tubule handles nutrient reabsorption, the loop of Henle dilutes filtrate, and the distal tubule balances sodium and pH. The collecting duct finalizes urine by removing ions and reabsorbing water (11).
Aetiology of Kidney Stones
Kidney stone formation stems from low fluid intake, high animal protein, oxalate-rich diets (e.g., spinach, nuts), and excess salt (13). Hydration producing 2.5L urine daily (water, coffee) is protective; high-oxalate juices (e.g., cranberry) increase risk. Citrate inhibits crystal aggregation, with hypocitraturia in 60% of calcium stone cases (14). Low calcium intake raises oxalate absorption, while vitamin C and fish oil elevate stone risk (15).
Types of Kidneys Stones:
Stones include calcium-based (75-90%) and non-calcium types (uric acid, struvite, cystine) (16,17); Table 1).
Table 1 Mineral composition kidney stone
|
Name of stone |
Approximate incidence |
Constituents |
|
Calcium oxalate, Calcium phosphate |
75 - 90 % of all stones |
Calcium, oxalate, phosphate |
|
Uric acid |
3-10 % of all stone |
Uric acid |
|
Struvite |
10 – 15 % of all stones |
Calcium, ammonia, phosphate |
|
Cystine |
Less than 1% of all stone |
Cystine |
|
Medication induced stones |
1-2% of all stone |
Composition depends on medication or herbal (examples Product include azanavir, sulfadiazine, acetazolamide |
Calcium Stones:
Comprising 80% of cases, these include calcium oxalate (50%) and phosphate (5%), with mixed types (45%) (Coe, 2005a). Calcium oxalate monohydrate (COM) is stable and prevalent, forming at pH 4.5-5.5; phosphate stones favor pH >7.5 (4).
Struvite Stones:
Linked to UTIs from urease-producing bacteria (e.g., Klebsiella), these form 10-15% of stones, more in women (2).
Uric Acid Stones:
3-10% of cases, tied to low pH and hyperuricosuria, common in men (18).
Cystine Stones:
<2% of cases, from cystinuria, an autosomal recessive disorder (19).
Drug-Induced Stones:
1-2% of cases, from drugs like atazanavir (20).
Kidney Stone Compositions:
Stones consist of crystals and an organic matrix (proteins 64%, sugars 9.6%, lipids 10.3%) that modulates formation (21). Phospholipids (8.6% of lipids) aid calcium stone development. Brushite, a resistant phosphate mineral, occurs in 25% of calcium phosphate stones (22)

Figure 1: Kidney stones and their types (23).
Pathophysiology: Several risk factors have been identified that increase susceptibility to urolithiasis.
Demographic Factors: Men (10.6%) exceed women (7.1%) in prevalence, with 50% recurrence; women’s risk rises with obesity (2). Stones are rare in youth but increase in the elderly (10-12% of cases), with infection stones post-60 (24). Caucasians (5.9%) outpace Blacks (1.7%), though diet aligns rates (25).
Genetics: 25% of patients have a family history; hypercalciuria (35-65%) often hereditary (15), Table 2).
Hormonal Factors: Testosterone increases oxalate via glycolic acid oxidase, while estrogen boosts protective osteopontin (26).
Metabolic Conditions: Diabetes lowers urine pH via insulin resistance, aiding uric acid stone . Obesity (BMI >30) raises oxalate and uric acid excretion (2). Pregnancy increases hypercalciuria, though citrate counters risk (27).
Lifestyle: High fluid intake dilutes urine, reducing crystal formation (28).
Other mechanisms by which crystals remain in the kidney:
Calcium oxalate monohydrate (COM) crystals stick fast to kidney epithelial cells because their positive charge pulls in the cells' negatively charged surface. Once attached, a crystal becomes a hub for more crystals to latch on. The cell then engulfs the crystal, making the nearby membrane stickier for other crystals. This stickiness lasts at least 24 hours, boosting the number or strength of crystal attachment spots (29,30).
Table 2 Genes Associated with Hypercalciuria And Kidney Stone Formation.
|
S.NO. |
Gene |
Gene function |
Renal tubular expression |
Renal phenotype |
|
1 |
DR |
Vitamin D receptor |
DCT, CD |
Decreased calcium reabsorption. |
|
2 |
CLCN5 |
Cl/H antiporter |
PT, TAL, αIC |
Inactivating mutations |
|
3 |
CASR |
Calcium sensing receptor |
PT, MCD, TAL, DCT |
Gain of function mutation |
|
4 |
CLDN16 |
Tight junction protein |
TAL, DCT |
Hypercalciuria, magnesium-wasting |
|
5 |
NPT2a/c |
Sodium phosphate co-transporter |
PT |
Hypercalciuria, magnesium-wasting |
|
6 |
TRPV5 |
Calcium selective transient receptor potential channel |
DCT, connecting tubule |
Hypercalciuria |
|
7 |
sAC |
Soluble adenylate cyclase/ bicarbonate exchanger |
DCT, TAL, CD |
Hypercalciuria |
|
8 |
KLOTHO |
Aging suppression protein/ regulator of calcium homeostasis |
DCT |
Hypercalciuria |
Physicochemical Processes in Kidney Stone Formation:
Urinary supersaturation and crystallization, driven by renal impairment, urine pH, and excess specific substances (e.g., CaOx, CaP, uric acid, struvite, cysteine, purines, and certain drugs), contribute to intrarenal crystal precipitation. Modulator molecules, including, promoters, and inhibitors, further influence crystal formation shown in (Figure 2) (31,32).
Urine Supersaturation:
Kidney stones form when urine becomes supersaturated, causing dissolved salts to exceed their solubility and crystallize into solids (Figure 3). Supersaturation is the key driver of this phase transition in urine (33).
Promoters of stone formation:
Promoters of stone formation include receptors and molecular mechanisms vital for crystal-cell interactions, aiding stone retention (34,35). COM-treated HK-2 cells show 1,141 DEPs, including CD44, nucleolin, HA, HSP90 , annexin II (36), and OPN (37,38), all involved in lithogenesis (39). Crystal attachment is facilitated by phosphatidylserine and acidic protein side chains (40). Calcium, oxalate, urate, and phosphate ions promote crystallization. Dysregulated calcium and vitamin D metabolism, with elevated serum calcium and 1,25(OH)2D, contributes to stone formation via CaSR pathways (41,42). Urate and phosphate also aid crystal nucleation (42,43). Urine pH affects crystallization: low pH favors CaOx crystallization, while high pH enhances nucleation (44). Lysozyme and lactoferrin promote COM crystal growth (45).
Inhibitors of stone formation:
Urine contains various inhibitors—anions (e.g., citrate), metallic cations (e.g., magnesium), and macromolecules (e.g., OPN, THP, UPTF?1, Nc, IαI subunits)—that competitively and cooperatively reduce crystallization, aggregation, and adhesion (46,47). Citrate inhibits crystal growth above 0.1 mM, and its reduced excretion in nephrolithiasis is managed with alkali supplements (48–50). Hydroxycitrate, a citrate analog, also prevents crystallization (51,52). Magnesium enhances citrate’s effect in acidic urine (53). Macromolecules potently inhibit stone-forming processes (33,44). Crystalluria depends on the supersaturation-inhibitor balance (54)
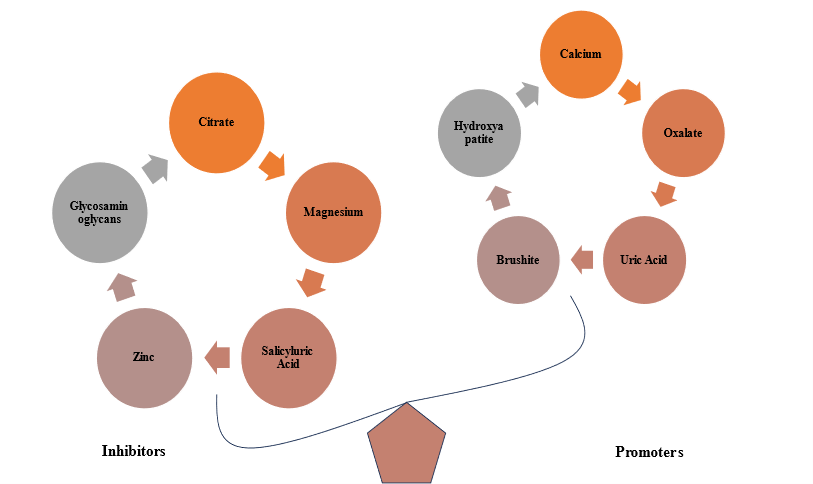
Figure.2 Imbalance Between Promoters and Inhibitors in Urolithiasis: A Key Factor in Kidney Stone Formation.
Randall's plaque and calcium oxalate stone formation:
Randall’s plaques (RPs), first identified by Alexander Randall (55), are subepithelial CaP-rich mineral deposits at the papillary tip near the ducts of Bellini (39), composed of calcified tubules and CaP plugs (56), embedded in a protein-, lipid-, and ECM-rich organic matrix (57). RPs are proposed as the nidus for kidney stones (58). LncRNA H19 promotes osteogenic differentiation via Wnt/β-catenin signaling (59) and induces oxidative stress and injury through the miR-216b/HMGB1/TLR4/NF-κB pathway (60). MALAT1 sponges miR-320a-5p to upregulate Runx2, enhancing osteogenic traits in hRIFs (61), highlighting the need to explore RP formation in stone pathogenesis.
Role of sex hormones in urolithiasis:
Testosterone plays a significant role in kidney stone formation through multiple mechanisms. (62), reported that testosterone induces apoptosis and necrosis in renal tubular epithelial cells via the HIF-1α/BNIP3 pathway, creating a favorable environment for calcium oxalate monohydrate (COM) crystal formation. Additionally, (63) found that testosterone enhances kidney stone disease by increasing the expression of α-enolase on kidney epithelial cells, which facilitates COM crystal adhesion, a crucial step in stone formation. Furthermore, (64) demonstrated that testosterone weakens the immune system's ability to clear COM crystals by suppressing colony-stimulating factor 1 (CSF-1) through miR-185-5p upregulation, reducing macrophage recruitment and phagocytic activity. These combined mechanisms—kidney cell damage, increased crystal adhesion, and impaired crystal clearance—help explain the higher incidence of kidney stones in men compared to women and suggest potential therapeutic targets for nephrolithiasis prevention shown in (Figure 4).
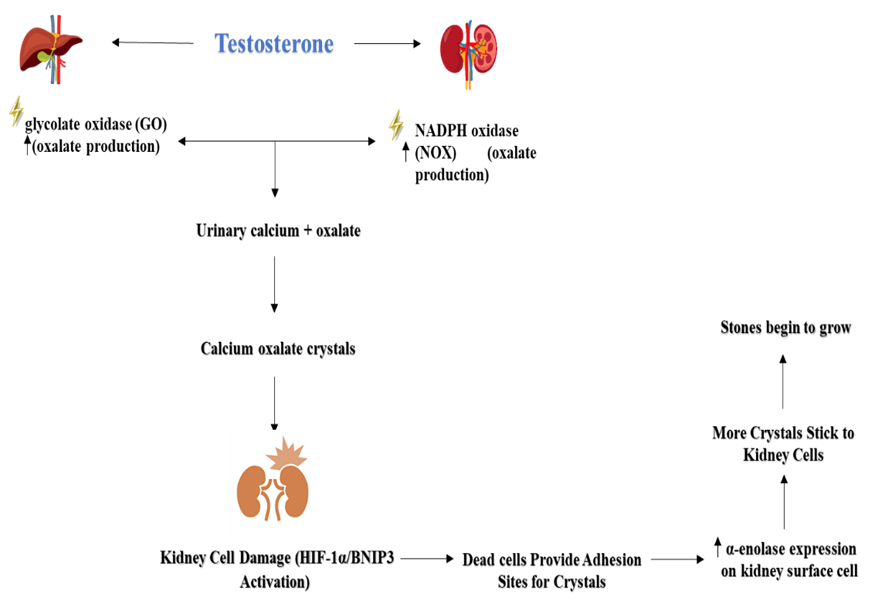
Figure 4. Role of sex hormones in calcium oxalate urolithiasis
The role of the microbiome in kidney stone formation:
Oxalobacter formigenes is a Gram-negative, anaerobic bacterium that aids in oxalate metabolism in the intestine (65). This bacterium is susceptible to certain antibiotics, though its full sensitivity is not well established (66). Its absence may increase intestinal oxalate absorption and reduce endogenous secretion (67), leading to higher urinary oxalate levels and a greater risk of calcium oxalate (CaOx) stone formation show in (Figure 5). Studies show that individuals with kidney stones or hyperoxaluria have a lower prevalence of O. formigenes in their stool than healthy controls (68).Additionally, nephrolithiasis patients with O. formigenes excrete lower urinary oxalate levels than those without it (69).
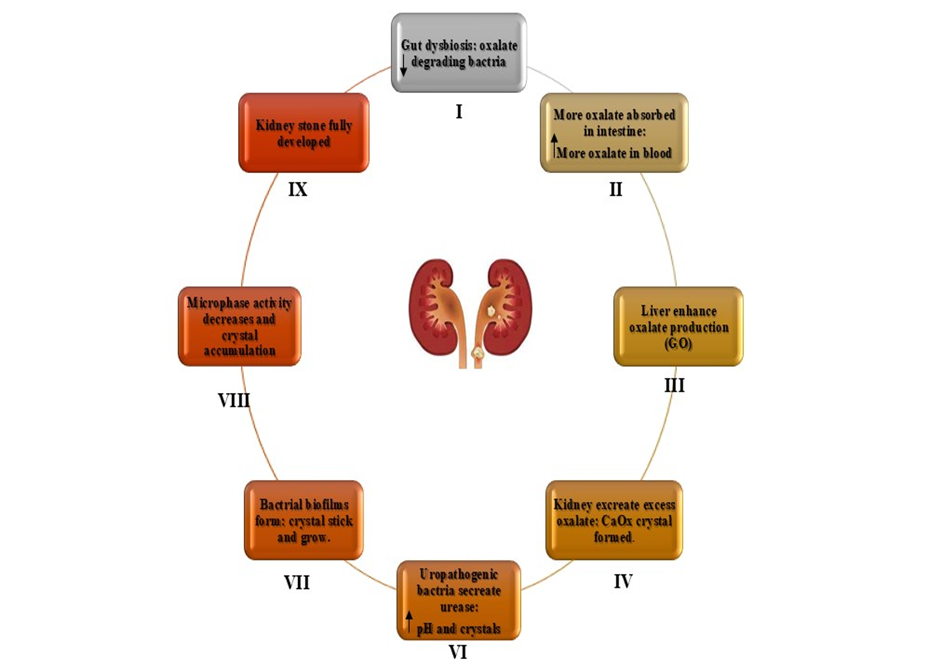
Figure 5. Role of oxalate?degrading bacteria in stone formation
Oxidative stress:
Kidney stone formation is linked to increased ROS production, primarily via NADPH oxidase activation by angiotensin II (70). CaOx crystal formation upregulates renin and angiotensin II, enhancing ROS, which activate transcription factors through pathways like P38 MAPK/JNK, AP-1, NF-kB, TGF, RUNX-2, and Obsterix. This promotes inflammation, with NF-kB inducing CRP production. The resulting cellular damage and inflammation create a feedback loop that further activates NADPH oxidase and ROS generation, impairing endothelial function (71).
Crystal Nucleation:
Nucleation kicks off kidney stone formation when stone salts in supersaturated urine clump together, shifting from liquid to a stable form. These loose clusters grow by adding more particles or merging with others, forming crystals with a unique structure. In urine, this usually happens on surfaces like epithelial cells, red blood cells, or other crystals, known as heterogeneous nucleation, which needs less saturation than homogeneous nucleation. Once a nucleus forms, it can grow quickly at lower chemical pressures. Damage to kidney tubules boosts calcium oxalate (CaOx) crystallization by supplying components, like membrane vesicles from injured cells, that spark nucleation. (21).
Crystal Growth:
Crystal growth is the stage that follows nucleation in the formation of urinary stones. During this phase, crystals present in urine combine and aggregate, gradually forming a stone. This process can be difficult to manage, as it leads to the development of larger stones. Proteins play essential roles in the crystallization process of nephrolithiasis, where certain urinary stone matrix protein modulators contribute to regulating and influencing the growth and formation of these stones (72).
Crystal Aggregation:
Crystal aggregation is a key step in the formation of urinary stones. A small, challenging mass of crystals in a solution binds together through this process, gradually forming a larger stone. The aggregation occurs when individual crystals stick together, growing in size and complexity. This process is considered the most critical stage in stone formation, as it significantly contributes to the development of larger, more stable stones (72).
Crystal-cell interaction:
Crystal-cell interaction involves the binding of calcium oxalate monohydrate (COM) crystals to renal tubular epithelial cells, especially in hyperoxaluria. Oxalate and COM crystals damage epithelial cells, promoting crystal retention and CaOx crystallization. Some crystals evade clearance by macrophages or lysosomes. Damaged cells release membrane vesicles and anionic proteins like renal prothrombin fragment-1, which act as nucleators for crystal formation (72).
Endocytosis of CaOx Crystals:
The initial phase of kidney stone formation involves endocytosis, where renal tubular cells engulf COM crystals that first attach to microvilli. Polyanions like glycosaminoglycans, glycoproteins, and citrate may inhibit this by coating crystals. Tamm-Horsfall glycoproteins (THP) have a dual role, being either endocytosed or transported to the interstitium. Damaged cells may promote nidus formation on the renal papillary surface (72).
Cell injury and Apoptosis:
High levels of oxalate or calcium oxalate (CaOx) can harm kidney epithelial cells, raising the risk of kidney stones. Beyond triggering stone formation, cell death (apoptosis) in renal tubular cells can lead to necrosis, which may halt calcium crystal growth. Lab studies on MDCK cells exposed to oxalate confirm this, but not all cells respond the same way. Some cells may activate genes that protect against apoptosis and stone formation. More research is needed to pinpoint new molecular targets for preventing kidney stones (72).
Immune response to urinary crystals:
In kidney stone disease, immune alterations include macrophage accumulation, inflammation, and anti-inflammatory responses, contributing to renal calcium oxalate (CaOx) crystal formation. CD44, osteopontin (OPN), and fibronectin (FN) interactions in renal tubular cells promote macrophage recruitment and calcium oxalate monohydrate (COM) crystal formation. Macrophages secrete IL-8, MIP-1, and MCP-1, driving immune cell migration and renal inflammation. M1 macrophages support crystal formation, while M2 macrophages aid inVcrystal clearance and inflammation reduction (35,73,74)
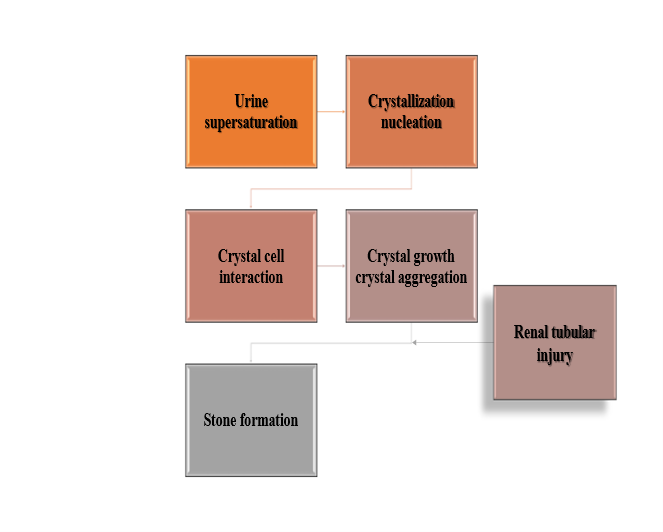
Figure 3. Mechanism of kidney stone formation.
Managing Kidney Stones: Prevention and Treatment Options:
Kidney stones are managed with NSAIDs for pain, opioids for severe cases, and alpha-blockers like tamsulosin to aid passage of small lower ureter stones, reducing discomfort (75,76). Thiazide diuretics cut urinary calcium to prevent calcium stones, while potassium citrate boosts urine alkalinity to stop calcium and uric acid stones (75,77). Allopurinol lowers uric acid, and acetohydroxamic acid helps with struvite stones (75,78). Cystine-binding drugs like tiopronin aid cystinuria but may have side effects (79). For stubborn or large stones, shock wave lithotripsy (ESWL) breaks up smaller stones, ureteroscopy (URS) uses lasers for precise treatment, and percutaneous nephrolithotomy (PCNL) handles big or complex stones (80–82). Open surgery is rare now (Nourian and Bahrami, 2022). Herbal options like Cystone, Uriflow, and Trinapanchamool can dissolve or prevent stones (23). Combining therapies—expulsion aids, pain relievers like NSAIDs, calcium channel blockers, steroids for swelling, and diuretics—tackles the complex nature of stones for better results (23).
Discussion and Future Directions:
Urolithiasis, a multifaceted urinary tract disorder, involves diverse stone types triggered by multiple causes. Our analysis identifies key risk factors such as gender, age, genetics, hormones, diabetes, obesity, pregnancy, and lifestyle. However, the precise mechanisms driving stone formation remain elusive despite thorough investigation (83), making it a significant gap in the field. The physicochemical process of kidney stone formation involves supersaturation, nucleation, crystal growth, aggregation, and retention within the renal tubules(84). Randall’s plaque, which originates in the renal papilla, serves as a nidus for stone formation, particularly in calcium oxalate stones(85). Recent evidence also suggests a crucial role of sex hormones in urolithiasis, with testosterone being linked to increased stone formation, while estrogen appears to have a protective effect(86). Additionally, emerging studies indicate that the gut and urinary microbiome influence stone formation, suggesting a potential area for therapeutic intervention(87). Current treatment strategies for urolithiasis remain inadequate. While allopathic treatments focus on pain management, hydration therapy, and surgical intervention, a definitive pharmacological therapy is still lacking(75,81). Ayurvedic formulations such as Cystone have been explored for their potential anti-lithogenic properties, but some studies suggest inconsistent efficacy, necessitating further research. While Cystone is widely used for managing urolithiasis, certain limitations must be acknowledged. One key concern is its uncertain impact on urinary supersaturation, as studies have shown no significant changes in urinary composition, a critical factor in stone formation. Additionally, although it is marketed for stone expulsion, clinical findings indicate an increase in stone burden rather than a reduction (88), Its ability to aid stone passage is uncertain. Cystone shows limited effectiveness in severe, treatment-resistant cases, indicating it may not suit complex conditions. Additionally, the absence of extensive, well-designed clinical studies hinders clear conclusions about its long-term benefits for preventing or dissolving stones (89). Cystone acts more slowly than Tamsulosin, which rapidly eases stone passage by relaxing urinary tract muscles. These observations highlight the necessity for thorough research to confirm Cystone’s effectiveness as a primary or supportive treatment for urolithiasis (90). Future urolithiasis studies seek to enhance knowledge of stone formation by investigating genetic and epigenetic factors driving molecular mechanisms(91), Uncovering these factors may lead to new treatment targets for improved outcomes. Scientists are also creating drugs to prevent kidney stones by blocking crystal formation, growth, and aggregation or altering urine composition (92). Personalized medicine is gaining traction, leveraging genomics, metabolomics, and proteomics to pinpoint individual risk factors and customize urolithiasis prevention and treatment. Advanced non-invasive imaging is also being developed to improve detection of small stones and analyze their composition accurately, with reduced radiation exposure. (93). Additionally, the discovery of urinary biomarkers may facilitate early diagnosis and predict recurrence risk, enabling proactive management (94). In surgical advancements, innovations in minimally invasive techniques, including robotic-assisted procedures, are being investigated to improve precision, safety, and outcomes in complex cases (95). These collective efforts aim to refine diagnostic, therapeutic, and preventive strategies, ultimately advancing patient-centered care in urolithiasis management.
CONCLUSION
Urolithiasis, driven by metabolic, physiological, and lifestyle factors, centers on calcium oxalate stone formation. Men face greater risk than women, with diet and hydration heavily influencing prevalence. New insights into promoters, inhibitors, immune responses, gut microbiome, and sex hormones deepen our understanding of stone development. Advances in personalized medicine, innovative drugs, and cutting-edge diagnostic and surgical methods promise better outcomes. A collaborative approach combining medical, nutritional, and surgical expertise is vital to enhance treatments, prevent recurrence, and lessen the impact of kidney stones on patients and healthcare.
REFERENCES




Naveen Maledavar, Priyanka Patil*, Kshitij Lele, Sneha Kakade, Urolithiasis: Advances in Pathophysiology, Microbiome Role, Immune Pathways, and Evolving Diagnostic and Therapeutic Strategies for Kidney Stone Formation and Management, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 5, 3396-3411. https://doi.org/10.5281/zenodo.15473206
 10.5281/zenodo.15473206
10.5281/zenodo.15473206