Abstract
Garlic (Allium sativum L.), i.e., a bulbous plant from the Liliaceae family, is native to central Asia but at present grows in many countries around the world. No-one knows exactly where the name garlic comes from, but it is assumed that the Anglo-Saxon ?gar‘ refers to the spear-like foliage and the ?lic‘ is refer ring to leek. Botanically, Allium sativum is a member of the Liliaceae family, along with onions, chives and shallots. [1]
Garlic (Allium sativum L.) is a pungent bulbous crop native to central Asia it is used in both food and as a traditional medicine to treat a variety of ailments. Particularly in Asia, the Mediterranean region, and North and South America, garlic production is still booming. The biggest producer in the world is China. Most of Europe‘s production is in Spain, with Castile-La Mancha having the largest hectares dedicated to the crop. There are around 700 species in this family, which are widely scattered over the world and prized for their flavour, ease of growing, and extended storage period. Garlic consumption has more than tripled in the United States since 1980 and now stands at about 2.5 pounds per capita. Around 24,000 to 26,000 acres of garlic are planted annually in the United States with a total production of around 400 million pounds. U.S. production is concentrated in California, with smaller acreages in Oregon, Nevada, New York, and Washington. About 30 percent of U.S. garlic production is sold in the fresh market; the rest is dehydrated. The average price of garlic has been around $60–70 per cwt., but wholesale fresh- market prices have typically been around three times higher. The United States is the world's largest importer of garlic, primarily from China, Spain, Mexico, and Argentina. [2].
Keywords
Garlic (Allium sativum L.) Escherichia coli, Shigella, Pseudomonas aeruginosa, Staphylococcus aureus, S. faecalis, Klebs, S. pyogenes, Salmonella enterica. HIV, herpes simplex virus types 1 and 2, cytomegalovirus, rotavirus, and viral pneumonia.
Introduction
Garlic consumption has more than tripled in the United States since 1980 and now stands at about 2.5 pounds per capita. Around 24,000 to 26,000 acres of garlic are planted annually in the United States with a total production of around 400 million pounds. U.S. production is concentrated in California, with smaller acreages in Oregon, Nevada, New York, and Washington. About 30 percent of U.S. garlic production is sold in the fresh market; the rest is dehydrated. The average price of garlic has been around $60–70 per cwt., but wholesale fresh- market prices have typically been around three times higher. The United States is the world's largest importer of garlic, primarily from China, Spain, Mexico, and Argentina.
Taxonomical Classification:
- Kingdom: Plantae
- Clade: Angiosperms (flowering plants)
- Clade: Monocots (plants with a single seed leaf)
- Order: Asparagales
- Family: Amaryllidaceae
- Subfamily: Allioideae
- Genus: Allium
- Species: Allium sativum

History:
Morphology:
The leaves are green in colour and grow to be 12-15 cm tall (some species can grow up to 60 cm tall). Garlic leaves, also known as garlic scapes, have a different morphology compared to the bulb. The leaves are narrow, flat, and lance-shaped, growing in a tuft from the base of the plant. Garlic flowers are part of the garlic scape, which is the flower stalk that emerges from the garlic plant. The flower cluster is called an umbel. The garlic umbel consists of small, star-shaped flowers arranged in a spherical or semi-spherical shape at the top of the scape. The individual flowers typically have six petal-like tepals, which are the floral structures that resemble both petals and sepals.

Garlic flowers can vary in colour, ranging from white to shades of pink or purple. The flowers may have a delicate fragrance, although it's not as strong as the characteristic garlic scent associated with the bulbs. Garlic fruit has a pungent odour and is pale or slightly yellowish. Depending on the cultivar, the tall leaves may develop from small, rigid stems above the bulb or a softer pseudo stem consisting of overlapping leaf sheaths. [3]

The bulbs are covered with membrane skin and contain up to 20 edible bulblets known as cloves. The bulb is divided into individual sections called cloves. Each clove is enclosed in a thin, papery skin and is capable of growing into a new garlic plant when planted. The clove clusters create the bulb, which is a subterranean portion. It has long leaves with blades grouped in an alternate arrangement. The entire bulb is covered in multiple layers of thin, papery skin. These layers help protect the cloves and keep them intact. The base of the bulb is known as the root plate. This is the part from which roots emerge and anchor the garlic plant into the soil. The basal plate is the bottom part of each clove that is attached to the root plate. It's the point of attachment for the cloves within the bulb. Sometimes, a hard stem or remnant of the garlic scape (flower stalk) may be visible at the top of the bulb. This part is usually removed before culinary use. The stem, or pseudo-stem, is very short and forms a tray at the base where adventitious roots emerge. It is constituted of a string of leaves joined together by their sheaths. It has an adventitious root system, which is thick and sparsely branching, with an epidermis, a multicellular cortex, and an endoderm surrounding the central stele. It can be white, brown, light brown, violet, light violet, or dark violet, with rounded, elliptical, or circular shapes, as well as transverse broad and transverse narrow elliptical shapes. [4]
Phytoconstituents:
Garlic contains at least 33 Sulphur compounds, minerals germanium, many enzymes, potassium, copper, selenium, iron, calcium, magnesium, and zinc, as well as vitamins A, B1, and C, fibres, and water. Lysine, aspartic acid, histidine, arginine, threonine, swine, glycine, glutamine, alanine, cystine, proline, valine, leucine, methionine, phenylalanine, isoleucine, tryptophan are among the 17 amino acids included in it. Garlic contains more Sulphur compounds than other Allium species, which are responsible for garlic‘s pungent flavour as well as its therapeutic properties. Figure 2 represents the major constituents of garlic. When garlic is minced or crushed, the enzyme allinase is triggered, and allicin is produced from allin (present in intact garlic). Other important compounds discovered in garlic homogenate include ally methyl thiosulfate and 1-propenyl ally thiosulfate. Garlic contains minerals including germanium and selenium, as well as enzymes and vitamins like peroxidase, alliinase, and tyrosinase. [5]
Structure of Phytoconstituents:

Pharmacological Uses:
|
Sr.
|
Bioactive Compound
|
Medicinal Activity
|
|
1.
|
Allicin
|
Antibacterial
|
|
2.
|
Diallyl Disulphide & Diallyl Trisulphide
|
Antifungal
|
|
3.
|
Allicin & Diallyl Trisulphide
|
Antiviral, Anti-inflammatory
|
|
4.
|
Allicin & Diallyl Disulphide & Diallyl Trisulphide
|
Antioxidant
|
|
5.
|
Allicin & Diallyl Disulphide & Diallyl Trisulphide, Allin
|
Anticancer
|
It has been proven that natural substances like garlic and compounds derived from garlic can activate enzymes involved in detoxification and antioxidant activity, hence reducing intracellular ROS [36]. The endogenous enzymatic defence mechanism of many natural compounds can neutralize hazardous reactive oxygen species. Numerous plant compounds, such as flavonoids, phenolic acid, and phenolic diterpenes, have powerful antioxidant properties. Perhaps the most well-known use of garlic is as a supplementary treatment for lowering blood pressure. Garlic has been used to treat and prevent hypertension. It has been claimed that inhaling the vapour from prepared garlic has successfully treated several people with pulmonary tuberculosis. Patients were given face masks with pouches containing ethanol, eucalyptus oil, and garlic juice that had been ground up to assist cover the odour. They inhaled the fumes for two one-hour intervals each day, and pulmonary TB was successfully treated in most cases. Garlic had a more potent anti-diabetic effect than glibenclamide. [6] Garlic acts as an anti-inflammatory agent by altering cytokinesis and inhibiting NF-kB behaviour in surrounding tissue and it may be used to treat inflammation-related neurodegenerative illnesses like Alzheimer‘s disease. Garlic extract has recently been discovered to prevent the growth and germination of Rhodotorula mucilaginosa and Meyerozyma guilliermondii. Allium sativum, or garlic, has a reputation for treating several bacterial and fungal illnesses. Garlic‘s antimicrobial properties are attributed to allicin activity, which is effective against a wide range of microorganisms, including antibiotic-resistant, Gram-negative, and Gram-positive bacteria like Escherichia coli, Shigella, Pseudomonas aeruginosa, Staphylococcus aureus, S. faecalis, Klebs, S. pyogenes, Salmonella enterica. Garlic has been shown to have in vitro activity against HIV, herpes simplex virus types 1 and 2, cytomegalovirus, rotavirus, and viral pneumonia. [7]
Traditional Uses:
Garlic is widely used due to its seasoning and flavour qualities as well as health-enhancing properties. Moreover, it is used in folk medicine as a remedy for the treatment of bacterial and viral diseases. Garlic has been a kitchen superhero for centuries! Apart from adding flavour to dishes, it has a bunch of traditional uses. It's believed to have medicinal properties and has been used to treat various ailments like colds, infections, and even as a natural antibiotic. Leaves have been one of the important sources of medicine since dawn of human civilization. These are the gifts of nature to mankind for treating different types of diseases. Almost from prehistoric period herbal medicine are used for alleviation of suffering caused by different diseases in human. Further they are well documented in India and other countries: even today they are in great use in these countries. There are several beliefs or claims regarding the therapeutic utility of herbs and herbal formulation i.e;
- Herbal medicines exhibit fewer side effects and are safe.
- Herbs and herbal formulations are cheaper and easily available.
- For certain disease like hepatitis, herbs and herbal drugs are the only remedies.
- Certain chemical constituents from the herbs are serving as prototype molecules for the discovery of more effective drugs than existing ones.
Medicinal leaves are treated as a subject of serious study and undergoing intense research all over the World to identify the lead molecules. The immense surge of scientific interest in natural products as a potential source of drugs has contributed to the development of phytopharmacology research. In many developing countries, phyto- pharmaceuticals form the main base of National Health Care Programmed e.g. the use of herbal extracts in the treatment of various diseases in different countries and they are also known to possess a rich traditional medicine. In South Africa it is estimated that 80% of the black population consults traditional practitioners, perhaps this is not one and only reason for the developing countries to invest in leaves based pharmaceuticals but also has been a global resurgence of interest in leaves based drugs. [8]
Herbal therapy provides rational means for the treatment of many diseases which are considered to be obstinate and incurable in other system of medicine. It lays a great deal of emphasis upon the maintenance of positive health of an individual and thus aims at both the prevention and cure of diseases. Hyperacidity / Ulcers which require the treatment for chronic period of time and the usage of such drugs may cause various side effects, sometimes with permanent damage of vital organs. The ulcer is one of chronic disease which is normally treated with H2-receptor blockers (Cimetidine, Ranitidine) and proton pump inhibitors (Omeprazole, Pentaprazole). These drugs are known to effect microsomal enzyme system of the liver resulting in the pharmacokinetic type of drug- drug interaction with other drugs. But in herbal / traditional medicines, there are several preparations prescribed to treat hyperacidity and ulcers. [9]
Inflammation is a normal protective response to tissue injury caused by physical trauma, noxious chemical or microbiological agents. It is body‘s response for tissue repair and is triggered by the release of chemical mediators from injured tissue and migrating cells. The mechanism of anti-inflammatory drugs is considered to be inhibition of prostaglandins synthesis at the site of injury. The anti-inflammatory potency of different compounds roughly corresponds with their ability to inhibit cyclooxygenase enzyme (COX). [10]
The traditional medicine is largely gaining popularity over allopathic medicine because of the following reasons.
- Rising cost of medical care.
- Natural products are free from side effects.
- No reoccurrence after the treatment.
- Easy availability of the drugs from natural sources especially in Developing countries.
- Cure of disease by the changes in life style and social pathology.
- Renewable sources.
In the present study, we have chosen a leaves Allium sativum for which different pharmacological activities are reported with different parts of the leaves. From the literature Ayurvedic texts we found that leaves of Allium sativum have been traditionally indicated for the treatments of anti-ulcer and anti-inflammatory condition. However anti- ulcer and anti- inflammatory activities of this leaves are not reported. But for its medicinal properties reported in the Ayurvedic texts prompted us to select leaves extracts for the study of anti- ulcer and anti-inflammatory activities in different experimental animal models. [11]
MATERIAL AND METHODS:
Table No: 4.1 List of materials and equipments used during experiment
|
S. No.
|
Name of the materials and equipment
|
|
1
|
Anaesthetic ether
|
|
2
|
Ibuprofen
|
|
3
|
Carrageenan
|
|
4
|
Ethanol
|
|
5
|
Formalin
|
|
6
|
Ranitidine
|
|
7
|
Absolute alcohol
|
|
8
|
Distilled water
|
|
9
|
Microscope
|
|
10
|
Evans blue
|
|
11
|
Lead Mercury
|
|
12
|
Histamine
|
|
13
|
Restraint cages
|
|
14
|
Chloroform
|
|
15
|
Refrigerator
|
|
16
|
Sodium Hydroxide
|
Preparation of Different Extracts:
- Preparation of alcoholic extract:
Leaves of Allium sativum were collected in the month of August from the native place Ambale, Dist. Pune and dried in shade at room temperature then subjected to size reduction to a fine powder with the help of mixer grinder. [12] The leaves powder was packed in a soxhlet apparatus and extracted with 95% alcohol for 18 hr. Appearance of colourless solvent in the siphon tube was taken as the termination of extraction. The extract was then transferred into the previously weighed empty beaker and evaporated to a thick paste on the water bath, maintained at 50oC to get alcoholic (AELAS) extract. The extract was finally air dried thoroughly to remove all traces of the solvent and the percentage yield was calculated. [13]
- Preparation of aqueous extract:
About 100 g of leaves powder was taken in a round bottom flask (2000 ml) and macerated with 500 ml of distilled water with 10 ml of chloroform (preservative) for 24 h with shaking for every hour in a closed vessel. Then the marc was removed by filtering the extract (AQELAS) and then it was concentrated on a water bath maintained at 50 ?.
These two extracts were stored in airtight containers in a refrigerator below 10?. The two extracts were examined for their colour and consistency. Their percentage yield was calculated with reference to air-dried powder sample used for the extraction.
Table No: 4.2 Nature and Percentage yield of the extracts
|
S. No.
|
Name of the Extract
|
Nature
|
Colour
|
%Yield (w/w) g
|
|
1.
|
Alcohol
(AELAS)
|
Sticky
|
Greenish Brown
|
12.50
|
|
2.
|
Aqueous
(AQELAS)
|
Solid
|
Dark brown
|
12.00
|
Both the AELAS and AQELAS were subjected to the following investigations:
1. Preliminary phytochemical screening.
2. Pharmacological activities
a. Toxicity studies (LD50).
b. Anti-inflammatory activity.
Preliminary phytochemical investigations:
The preliminary phytochemical investigations were carried out with AELAS and AQELAS for qualitative identification of phytochemical constituents present with each extract and test were carried out by following standard methods74, 75, and 76. All the chemicals and reagents used were of analytical grade. [14]
Table No: 4.3 Details of qualitative phytochemical tests
|
Test
|
AELAS
|
AQELAS
|
|
I. Testsfor sterols
|
|
|
|
1. Testsolution+ConcH2SO4
|
+
|
+
|
|
2. Salkowski‘stest
|
+
|
+
|
|
3. Test solution+ sulphur
|
+
|
+
|
|
4. Libermann Burchard‘s test
|
+
|
+
|
|
II. Testsfor Glycosides
|
|
|
|
1. Baljet‘s test
|
+
|
+
|
|
2. Keller– killiani test
|
+
|
+
|
|
3. Brominewatertest.
|
+
|
+
|
|
4. Legal‘s test
|
+
|
+
|
|
III.TestforSaponins
|
|
|
|
1. Foamtest
|
_
|
_
|
|
IV.Tests for Carbohydrates
|
|
|
|
1. Molisch‘s test
|
+
|
+
|
|
2. Barfoed‘s test
|
+
|
+
|
|
3. Benedict‘s test
|
+
|
+
|
|
4. Fehlingtest
|
+
|
+
|
|
V. Testsfor Alkaloids
|
|
|
|
1.Mayer‘stest.
|
+
|
+
|
|
2.Wagner‘stest.
|
+
|
+
|
|
3.Hager‘s test
|
+
|
+
|
|
4.Dragendorff‘stest
|
+
|
+
|
|
VI. Test for Ascorbic acid
|
|
|
|
1.Dichlorophenolindophenoltest
|
_
|
_
|
|
VII. Tests for Flavonoids
|
|
|
|
1. Ferric chloride test
|
+
|
+
|
|
2. Shinoda test
|
_
|
_
|
|
3. Zn-Hcl reduction test
|
+
|
+
|
|
4. Sodium hydroxide test
|
+
|
+
|
|
5. Lead acetate test.
|
+
|
+
|
|
VIII. Tests for Tannins
|
|
|
|
1. Ferric chloride test.
|
+
|
+
|
|
2. Gelatin test
|
_
|
_
|
|
IX. Tests for Proteins
|
|
|
|
1. Million‘s test
|
_
|
_
|
|
2. Biuret‘s test
|
_
|
_
|
|
3. Ninhydrin test
|
_
|
_
|
|
X. Test for fixed oils and fats
|
|
|
|
1. Spot test
|
_
|
_
|
|
XI. Tests for triterpines
|
|
|
|
1. Salkowski‘s test
|
+
|
+
|
|
2. Libermann- Burchard test
|
+
|
+
|
(+) Indicates positive result and (–) Indicates negative result.
Pharmacological activities:
Experimental animals:
Albino rats (Wistar strain) of either sex weighing between 120-200 g and Albino mice 18-22 g were procured from National Centre for Laboratory Animal sciences, C/0 Shri. Venkateswara Enterprises, Bengaluru for experimental purpose and the animals were acclimatized for 7 days under standard husbandry condition as:
Room temperature - 26 ± 20 C
Relative humidity - 45-55%
Light/ dark cycle - 12:12 h
The procedures were followed as per rules and regulations.
Determination of acute toxicity (LD50)77 Method:
The acute toxicity of AELAS and AQELAS was determined in albino rats weighing between 18- 22 g those maintained under standard husbandry conditions. The animals were fasted 3 h prior to the experiment and ?up and down? (OECD Guideline No. 425) method of CPCSEA was adopted for toxicity studies. Animals were administered with single dose of extracts and observed for its mortality during 48 h study period (short term) toxicity. Based on the short-term toxicity profile of the extracts the doses of the next animals were determined as per as OECD Guidelines No: 425. All the animals were observed for long term toxicity (7 days) and then 1/5th, 1/10th and 1/20th of the maximum dose tested for LD50 of the individual extract was taken as effective dose ED50 and were used throughout the experimental studies. [16]
Determination of anti-inflammatory activity:
- Carrageenan induced paw edema:
Group A: Toxicant control (0.1 ml of 1% w/v Carrageenan, hind paw)
Group B: Standard (Ibuprofen 40 mg/ kg, p.o)
Group C: AELAS (100 mg/kg p.o)
Group D: AELAS (200 mg/kg p.o)
Group E: AELAS (400 mg/kg p.o)
Group F: AQELAS (100 mg/kg p.o)
Group G: AQELAS (200 mg/kg p.o)
Group H: AQELAS (400 mg/kg p.o)
Experimental Procedure:
Male albino rats (125-150 g) of 8 groups were housed as groups of six, fasted overnight prior to and during the experiment but have free access to water. Group A was served as normal toxicant control treated with toxicant carrageenan, group B with Ibuprofen (40 mg/kg p.o.) served as standard, groups C, D and E administered with AELAS and groups F, G and H with AQELAS (low, medium and high doses p.o) respectively. The rats in Groups B, C, D, E, F, G and H were administered with 0.1 ml of 1% w/v of carrageenan into of right hind paw of rats 1 h after the administration of Ibuprofen/extracts. Immediately thereafter the oedema volumes of the injected paws were measured plethysmographically at prefixed time intervals. [27-30]
- Histamine induced paw edema:
Group A: Toxicant control (0.1 ml of 1% w/v histamine, hind paw)
Group B: Standard (Ibuprofen 40 mg/ kg)
Group C: AELAS (100 mg/kg p.o)
Group D: AELAS (200 mg/kg p.o)
Group E: AELAS (400 mg/kg p.o)
Group F: AQELAS (100 mg/kg p.o)
Group G: AQELAS (200 mg/kg p.o)
Group H: AQELAS (400 mg/kg p.o)
Experimental Procedure:
Male albino rats (125-150 g) of 8 groups were housed as groups of 6, fasted overnight prior to and during the experiment but have free access to water. Group A was served as toxicant control treated with inflammogen histamine, group B with Ibuprofen (40 mg/kg p.o.) that served as standard. Groups C, D, E and F, G, H will administered with AELAS and AQELAS (low, medium and high dose p.o) respectively. The rats of groups B, C, D, E, F, G and H were administered with 1% w/v of histamine into sub plantar region of right hind paw of rats 1 h after administration of Ibuprofen/extracts. Immediately thereafter the oedema volumes of the injected paws were measured plethysmographically at prefixed time intervals. [31]
- Formalin Induced Paw Oedema:
Group A: Toxicant control (Formalin 1%, hind paw)
Group B: Standard (Ibuprofen 40 mg/ kg)
Group C: AELAS (100 mg/kg p.o)
Group D: AELAS (200 mg/kg p.o)
Group E: AELAS (400 mg/kg p.o)
Group F: AQELAS (100 mg/kg p.o)
Group G: AQELAS (200 mg/kg p.o)
Group H: AQELAS (400 mg/kg p.o)
Experimental Procedure:
Male albino rats (125-150 g) of 8 groups were housed as groups of 6, fasted overnight prior to and during the experiment but have free access to water. Group A was served as toxicant control treated with toxicant Formalin, group B with Ibuprofen (40 mg/kg p.o.) that served as standard. Groups C, D, E and F, G, H were administered with AELAS and AQELAS (low, medium and high dose p.o) respectively. The rats of groups B, C, D, E, F, G and H were administered with 1% of Formalin into sub plantar region of right hind paw of rats 1 h after administration of Ibuprofen/extracts. Immediately thereafter the oedema volumes of the injected paws were measured plethysmographically at prefixed time intervals. [32] For comparison purpose, the volume of oedema was measured at prefixed time intervals. The difference between paw volumes of the treated animals was measured and the mean oedema volume was calculated. Percentage reduction in oedema volume was calculated by using the formula,
Vo - Vt
Percentage reduction = x 100
Vo
Where,
Vo = Volume of the paw of control at time?t‘.
Vt = Volume of the paw of drug treated at time?t‘.
Statistical analysis:
All results will be expressed as mean ± SEM from 6 animals. Statistical difference in mean will be analysed using one-way ANOVA (analysis of variance) followed by Post hoc test (Dunnett?s?t‘ test). P< 0>
RESULTS AND DISCUSSION:
Preliminary Phytochemical studies Preliminary phytochemical screening:
AELAS and AQELAS were subjected for phytochemical screening and found to contain
tannins, carbohydrate, sterols, flavonoids, glycosides, alkaloid and triterpenes both in alcoholic and aqueous extracts. [34]
Pharmacological activities
Acute oral toxicity study:
The mice treated with AELAS and AQELAS at a dose of 2000 mg/kg, p.o. exhibited normal behaviour, without any signs of passivity, stereotypy and vocalization. Their motor activity and secretory signs were also normal and no sign of depression. AELAS and AQELAS even up to the dose level of 2000 mg/kg body weight did not produce any behavioural symptoms or mortality. So 1/5th, 1/10th and 1/20th doses of LD50 (maximum dose tested for each extract) was selected as low, medium and high doses and were used in the present study to explore anti-ulcer and anti-inflammatory activities.
Anti-inflammatory activity:
- Anti-inflammatory activity of AELAS and AQELAS in Carrageenan induced paw oedema model in rats:
The AELAS and AQELAS with three selected doses i.e. 100, 200 and 400 mg/kg have exhibited a significant reduction in paw oedema volume in carrageenan induced paw oedema in rats at different time intervals. Results are tabulated in Table No. 5.7. Ibuprofen (40 mg/kg) was used as standard reference and it has significantly reduced paw oedema volume by 43.35% at 1st h, 61.93% at 2nd h, 79.79% at 3rd h and 85.83% at 4th h, thus standard drug has exhibited time dependent reduction in oedema volume. During 1st h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 5.35%, 14.78%, 24.50% and 10.21%, 19.64%, 29.07% respectively, which was found to be a time dependent effect. During 2nd h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 8.12%, 23.59%, 31.21% and 11.94%, 27.28%, 38.83% respectively a time dependent effect.
During 3rd h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 14.16%, 28.03%, 38.38% and 17.64%, 31.55%, 45.22% respectively a time dependent effect was noted. During 4th h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 22.72%, 33.83%, 59.75% and 30.05%, 41.16%, 67.16% respectively a time dependent effect was noted and result are graphically represented in Fig No.5.7. ANOVA indicates a significant difference among the AELAS and AQELAS treated groups. Dunnett‘s?t‘ test confirms a significant anti-inflammatory activity with both the extracts, but more with AQELAS than AELAS.
Table No: 5.6 Anti-inflammatory effects of AELAS and AQELAS in Carrageenan induced paw oedema model in rats
|
Time after
|
Paw oedema volume (ml)
|
|
Treat ment
|
Anim als
|
Toxican t
|
Standard
|
AELAS 100
|
AELAS 200
|
AELAS 400
|
AQELAS 100
|
AQELAS 200
|
AQELAS 400
|
|
1 h
|
H
|
0.85
|
0.7
|
0.85
|
0.75
|
0.75
|
0.8
|
0.75
|
0.8
|
|
B
|
0.9
|
0.75
|
0.9
|
0.8
|
0.8
|
0.8
|
0.8
|
0.75
|
|
T
|
0.8
|
0.6
|
0.75
|
0.8
|
0.75
|
0.85
|
0.7
|
0.7
|
|
HB
|
0.85
|
0.8
|
0.85
|
0.85
|
0.8
|
0.8
|
0.8
|
0.8
|
|
BT
|
0.85
|
0.7
|
0.8
|
0.8
|
0.8
|
0.85
|
0.85
|
0.75
|
|
HT
|
0.85
|
0.65
|
0.85
|
0.8
|
0.7
|
0.8
|
0.8
|
0.7
|
|
mean±SEM
|
0.850±0
.012
|
0.700±0.0
28***
|
0.833±0.
021ns
|
0.800±0.0
12ns
|
0.766±0.0
16*
|
0.816±0.0
10ns
|
0.783±0.0
21ns
|
0.750±0.0
18**
|
|
2 h
|
H
|
1
|
0.65
|
0.85
|
0.75
|
0.75
|
0.85
|
0.8
|
0.7
|
|
B
|
0.9
|
0.75
|
0.9
|
0.9
|
0.8
|
0.9
|
0.8
|
0.75
|
|
T
|
0.95
|
0.6
|
0.85
|
0.85
|
0.85
|
0.9
|
0.75
|
0.8
|
|
HB
|
0.9
|
0.75
|
0.95
|
0.9
|
0.8
|
0.85
|
0.9
|
0.8
|
|
BT
|
0.95
|
0.65
|
0.95
|
0.8
|
0.75
|
0.95
|
0.8
|
0.75
|
|
HT
|
0.9
|
0.6
|
0.9
|
0.8
|
0.85
|
0.85
|
0.85
|
0.8
|
|
Time after
|
Paw oedema volume (ml)
|
|
mean±SEM
|
0.933±0
.016
|
0.667±0.0
27***
|
0.900±0.
018ns
|
0.833±0.0
24**
|
0.800±0.0
18***
|
0.883±0.0
16ns
|
0.817±0.0
21**
|
0.767±0.0
16***
|
|
3 h
|
H
|
1
|
0.6
|
0.9
|
0.8
|
0.8
|
0.9
|
0.9
|
0.7
|
|
B
|
0.95
|
0.65
|
0.95
|
0.9
|
0.8
|
0.9
|
0.85
|
0.75
|
|
T
|
1
|
0.5
|
0.9
|
0.85
|
0.85
|
0.9
|
0.8
|
0.8
|
|
HB
|
1
|
0.65
|
0.95
|
0.9
|
0.75
|
0.85
|
0.8
|
0.8
|
|
BT
|
0.95
|
0.6
|
0.9
|
0.85
|
0.8
|
0.95
|
0.85
|
0.85
|
|
HT
|
1
|
0.6
|
0.9
|
0.8
|
0.8
|
0.9
|
0.8
|
0.7
|
|
mean±SEM
|
0.983±0
.010
|
0.600±0.0
22***
|
0.917±0.
010*
|
0.850±0.0
18***
|
0.800±0.0
12***
|
0.900±0.0
12**
|
0.833±0.0
16***
|
0.767±0.0
24***
|
|
4 h
|
H
|
1
|
0.6
|
0.8
|
0.8
|
0.7
|
0.8
|
0.8
|
0.65
|
|
B
|
0.95
|
0.5
|
0.9
|
0.85
|
0.65
|
0.85
|
0.8
|
0.6
|
|
T
|
0.9
|
0.65
|
0.85
|
0.8
|
0.8
|
0.8
|
0.75
|
0.7
|
|
HB
|
1
|
0.6
|
0.9
|
0.75
|
0.6
|
0.75
|
0.8
|
0.65
|
|
BT
|
0.9
|
0.55
|
0.85
|
0.85
|
0.65
|
0.9
|
0.75
|
0.7
|
|
HT
|
0.95
|
0.5
|
0.8
|
0.75
|
0.7
|
0.8
|
0.7
|
0.6
|
|
mean±SEM
|
0.950±0
.018
|
0.566±0.0
24***
|
0.850±0.
018**
|
0.800±0.0
18***
|
0.683±0.0
27***
|
0.817±0.0
21***
|
0.767±0.0
16***
|
0.650±0.0
18***
|
n = 6, Significant at P < 0 xss=removed>
AELAS- Alcoholic Extract of Leaf of Allium Sativum, AQELAS- Aqueous Extract of Leaf of Allium Sativum.
Table No: 5.7 Anti-inflammatory effects of AELAS and AQELAS in Carrageenan induced paw oedema model in rats at different time intervals
|
S. N
|
Group s
|
Treatment
|
1 h
|
%
ROV
|
2 h
|
% ROV
|
3 h
|
% RO V
|
4 h
|
% ROV
|
|
A
|
Toxica nt
|
Carrageenan (1% w/v)
|
0.850 ±0.012
|
--
|
0.933
±0.016
|
--
|
0.983
±0.010
|
--
|
0.950 ±0.0
18
|
--
|
|
B
|
Standar d
|
Ibuprofen40 mg/kg
|
0.700±0.028***
|
43.35
|
0.667±0.027***
|
61.93
|
0.600
±0.022***
|
79.7
9
|
0.566±0.04***
|
85.
|
|
C
|
AELA S
|
100 mg/kg
|
0.833
±0.021ns
|
5.35
|
0.900±0.018ns
|
8.12
|
0.917
±0.010*
|
14.16
|
0.850±0.018**
|
22.
|
|
D
|
AELA S
|
200 mg/kg
|
0.800±0.012ns
|
14.78
|
0.833±0.024**
|
23.59
|
0.850±0.018***
|
28.03
|
0.800±0.01
8***
|
33.
|
|
E
|
AELA S
|
400 mg/kg
|
0.766±0.016*
|
24.5
|
0.800±0.018***
|
31.21
|
0.800±0.012***
|
38.3
8
|
0.683±0.027***
|
59.
|
|
F
|
AQELA S
|
100 mg/kg
|
0.816
±0.010ns
|
10.21
|
0.883±0.016ns
|
11.94
|
0.900±0.012**
|
17.6
4
|
0.817±0.021***
|
30
|
|
G
|
AQELA S
|
200 mg/kg
|
0.783±0.021ns
|
19.64
|
0.817±0.021**
|
27.28
|
0.833±0.016***
|
31.5
5
|
0.767±0.01
6***
|
41.
|
|
H
|
AQELA S
|
400 mg/kg
|
0.750±0.018**
|
29.07
|
0.767±0.016***
|
38.83
|
0.767±0.024***
|
45.2
2
|
0.650m±0.018***
|
67.
|
n = 6, Significant at P < 0 xss=removed>Allium Sativum, AQELAS- Aqueous Extract of Leaf of Allium Sativum
- Anti-inflammatory activity of AELAS and AQELAS in Histamine induced paw oedema model in rats:
The AELAS and AQELAS with three selected doses i.e. 100, 200 and 400 mg/kg have exhibited a significant reduction in paw oedema volume in histamine induced paw oedema in rats at different time intervals. Results are tabulated in Table No. 5.9. Ibuprofen (40 mg/kg) was used as standard reference and it has significantly reduced paw oedema volume by 56.89% at 1st h, 70.94% at 2nd h, 87.30% at 3rd h and 93.43% at 4th h, thus exhibited a time dependent reduction in oedema volume. During 1st h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 4.67%, 17.73%, 39.66% and 9.11%, 26.60%, 48.28% respectively, which was found to be a time dependent effect. During 2nd h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 11.61%, 26.50%, 44.94% and 18.94%, 30.05%, 52.27% respectively noted a time dependent effect. During 3rd h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 17.10%, 37.16%, 57.16% and 27.10%, 43.90%, 63.90% respectively a time dependent effect was noted.
During 4th h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 25.55%, 46.96%, 68.38% and 36.26%, 54.03%, 75.44% respectively a time dependent effect was noted and result are graphically represented in Fig No.5.8.
ANOVA indicates a significant difference among the AELAS and AQELAS treated groups. Dunnett‘s?t‘ test confirms a significant anti-inflammatory activity with both the extracts, but more with AQELAS than AELAS.
Table No: 5.8 Anti-inflammatory effects of AELAS and AQELAS in Histamine induced paw oedema model in rats
|
Time after
|
Paw oedema volume (ml)
|
|
Treat ment
|
Anim als
|
Toxican t
|
Standard
|
AELAS 100
mg/kg
|
AELAS 200
mg/kg
|
AELAS 400
mg/kg
|
AQELAS 100
mg/kg
|
AQELAS 200
mg/kg
|
AQELAS 400
mg/kg
|
|
1 h
|
H
|
0.85
|
0.65
|
0.9
|
0.75
|
0.7
|
0.8
|
0.75
|
0.7
|
|
B
|
0.9
|
0.6
|
0.85
|
0.75
|
0.75
|
0.9
|
0.8
|
0.65
|
|
T
|
0.9
|
0.7
|
0.8
|
0.85
|
0.8
|
0.9
|
0.85
|
0.7
|
|
HB
|
0.9
|
0.7
|
0.9
|
0.9
|
0.7
|
0.85
|
0.8
|
0.7
|
|
BT
|
0.9
|
0.7
|
0.85
|
0.8
|
0.75
|
0.8
|
0.75
|
0.75
|
|
HT
|
0.85
|
0.65
|
0.9
|
0.85
|
0.7
|
0.85
|
0.75
|
0.7
|
|
mean±SEM
|
0.883±0
.010
|
0.667±0.0
16***
|
0.867±0.0
16ns
|
0.817±0.0
24*
|
0.733±0.0
16***
|
0.850±0.0
18ns
|
0.783±0.0
16**
|
0.700±0.0
12***
|
|
2 h
|
H
|
0.95
|
0.7
|
0.95
|
0.8
|
0.7
|
0.85
|
0.85
|
0.75
|
|
B
|
1
|
0.65
|
0.8
|
0.8
|
0.75
|
0.9
|
0.8
|
0.7
|
|
T
|
0.9
|
0.6
|
0.8
|
0.85
|
0.8
|
0.9
|
0.85
|
0.75
|
|
HB
|
1
|
0.65
|
1
|
0.85
|
0.7
|
0.8
|
0.8
|
0.7
|
|
BT
|
0.95
|
0.6
|
0.95
|
0.85
|
0.75
|
0.85
|
0.8
|
0.65
|
|
HT
|
0.9
|
0.6
|
0.9
|
0.85
|
0.8
|
0.9
|
0.8
|
0.75
|
|
mean±SEM
|
0.950±0
.018
|
0.633±0.0
16***
|
0.900±0.0
34ns
|
0.833±0.0
10***
|
0.750±0.0
18***
|
0.867±0.0
16*
|
0.817±0.0
10***
|
0.717±0.0
16***
|
|
3 h
|
H
|
0.95
|
0.6
|
0.95
|
0.8
|
0.7
|
0.8
|
0.8
|
0.65
|
|
B
|
1
|
0.65
|
0.9
|
0.8
|
0.7
|
0.85
|
0.8
|
0.65
|
|
T
|
1
|
0.5
|
0.9
|
0.85
|
0.75
|
0.9
|
0.75
|
0.7
|
|
HB
|
1.05
|
0.5
|
0.9
|
0.8
|
0.7
|
0.85
|
0.8
|
0.7
|
|
BT
|
1
|
0.55
|
0.95
|
0.85
|
0.75
|
0.9
|
0.75
|
0.65
|
|
HT
|
1
|
0.6
|
0.9
|
0.8
|
0.7
|
0.9
|
0.8
|
0.75
|
|
Time after
|
Paw oedema volume (ml)
|
|
mean±SEM
|
1.000±0
.012
|
0.566±0.0
24***
|
0.917±0.0
10**
|
0.817±0.0
10***
|
0.717±0.0
10***
|
0.867±0.0
16***
|
0.783±0.0
10***
|
0.683±0.0
16***
|
|
4 h
|
H
|
0.95
|
0.5
|
0.9
|
0.7
|
0.65
|
0.75
|
0.7
|
0.6
|
|
B
|
0.9
|
0.6
|
0.85
|
0.75
|
0.65
|
0.8
|
0.7
|
0.6
|
|
T
|
1
|
0.5
|
0.8
|
0.8
|
0.7
|
0.8
|
0.65
|
0.65
|
|
HB
|
1
|
0.5
|
0.8
|
0.75
|
0.6
|
0.8
|
0.75
|
0.6
|
|
BT
|
1
|
0.5
|
0.9
|
0.8
|
0.65
|
0.8
|
0.7
|
0.6
|
|
HT
|
0.95
|
0.6
|
0.85
|
0.7
|
0.65
|
0.85
|
0.8
|
0.65
|
|
mean±SEM
|
0.967±0
.016
|
0.533±0.0
21***
|
0.850±0.0
18***
|
0.750±0.0
18***
|
0.650±0.0
12***
|
0.800±0.0
12***
|
0.717±0.0
21***
|
0.617±0.0
10***
|
n = 6, Significant at P < 0 xss=removed>
AELAS- Alcoholic Extract of Leaf of Allium Sativum, AQELAS- Aqueous Extract of Leaf of
Allium Sativum.
Table No: 5.9 Anti-inflammatory effects of AELAS and AQELAS in Histamine induced paw oedema model in rats at different time intervals
|
S.N
|
Group s
|
Treatm ent
|
1 h
|
% ROV
|
2 h
|
% ROV
|
3 h
|
% RO
V
|
4 h
|
% RO
V
|
|
A
|
Toxica nt
|
Histamine(1%w/)
|
0.883
±0.010
|
--
|
0.950±0.018
|
--
|
1.000±0.012
|
--
|
0.967±0.016
|
--
|
|
B
|
Standa rd
|
Ibupr ofen 40
mg/kg
|
0.667±0.016
***
|
56.
89
|
0.633±0.016
***
|
70.
94
|
0.566±0.024
*
|
87.
3
|
0.533±0.021
***
|
93.
43
|
|
C
|
AELA S
|
100mg/kg
|
0.867±0.016n
s
|
4.6
7
|
0.900±0.034
ns
|
11.
61
|
0.917±0.010
**
|
17.
1
|
0.850±0.018
***
|
25.
55
|
|
D
|
AELA S
|
200
mg/kg
|
0.817
±0.024*
|
17.
73
|
0.833±0.010
***
|
26.
5
|
0.817±0.010
***
|
37.
16
|
0.750±0.018
***
|
46.
96
|
|
E
|
AELA S
|
400
mg/kg
|
0.733±0.016
***
|
39.
66
|
0.750±0.018
**
|
44.
94
|
0.717±0.010
***
|
57.
16
|
0.650±0.012
***
|
68.
38
|
|
F
|
AQEL AS
|
100
mg/kg
|
0.850±0.018
ns
|
9.1
1
|
0.867±0.016*
|
18.
94
|
0.867±0.016
***
|
27.
1
|
0.800
±0.012
***
|
36.
26
|
|
G
|
AQEL AS
|
200
mg/kg
|
0.783±0.016*
*
|
26.
6
|
0.817±0.010
***
|
30.
05
|
0.783±0.010
***
|
43.
9
|
0.717±0.021
***
|
54.
03
|
|
H
|
AQEL AS
|
400
mg/kg
|
0.700±0.012
***
|
48.
28
|
0.717±0.016
***
|
52.
27
|
0.683±0.016
***
|
63.9
|
0.617±0.010
***
|
75.
44
|
n = 6, Significant at P < 0 xss=removed>
- Anti-inflammatory activity of AELAS and AQELAS in Formalin induced paw oedema model in rats:
The AELAS and AQELAS with three selected doses i.e. 100, 200 and 400 mg/kg have exhibited a significant reduction in paw oedema volume in Formalin induced paw oedema in rats at different time intervals. Results are tabulated in Table No. 5.11. Ibuprofen (40 mg/kg) was used as standard reference and it has significantly reduced paw oedema volume by 54.75% at 1st h, 64.73% at 2nd h, 84.40% at 3rd h and 90.14% at 4th h, which was found to be a time dependent effect. During 1st h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 4.75%, 17.25%, 38.00% and 8.75%, 21.25%, 42.25% respectively noted as time dependent effect. During 2nd h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 7.78%, 21.91%, 43.32% and 11.20%, 29.19%, 50.60% respectively noted as time dependent effect. During 3rd h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 19.45%, 32.80%, 55.42% and 19.84%, 39.18%, 61.81% respectively recorded as time dependent effect.
During 4th h of study AELAS and AQELAS with low, medium and high doses have significantly reduced oedema volume by 24.51%, 45.22%, 65.92% and 34.86%, 52.25%, 72.96% respectively which was recorded as time dependent effect and result are graphically represented in Fig No.5.9.
ANOVA indicates a significant difference among the AELAS and AQELAS treated groups. Dunnett‘s?t‘ test confirms a significant anti-inflammatory activity with both the extracts, but more with AQELAS than AELAS.
Table No: 5.10 Anti-inflammatory effects of AELAS and AQELAS in Formalin induced paw edema model in rats
|
Time after
|
Paw oedema volume (ml)
|
|
Treatm ent
|
Anim als
|
Toxicant
|
Standard
|
AELAS 100
mg/kg
|
AELAS 200
mg/kg
|
AELAS 400
mg/kg
|
AQELAS
100 mg/kg
|
AQELAS
200 mg/kg
|
AQELAS
400 mg/kg
|
|
1 h
|
H
|
0.9
|
0.7
|
0.9
|
0.8
|
0.75
|
0.9
|
0.85
|
0.75
|
|
B
|
0.9
|
0.7
|
0.9
|
0.85
|
0.8
|
0.85
|
0.85
|
0.7
|
|
T
|
0.95
|
0.65
|
0.75
|
0.8
|
0.7
|
0.9
|
0.75
|
0.7
|
|
HB
|
0.9
|
0.75
|
0.9
|
0.9
|
0.75
|
0.85
|
0.8
|
0.75
|
|
BT
|
0.85
|
0.7
|
0.95
|
0.8
|
0.8
|
0.8
|
0.8
|
0.8
|
|
Time after
|
Paw oedema volume (ml)
|
|
|
HT
|
0.9
|
0.6
|
0.9
|
0.85
|
0.7
|
0.9
|
0.85
|
0.7
|
|
mean±SEM
|
0.900±0.
012
|
0.683±0.02
1***
|
0.883±0.0
27ns
|
0.833±0.01
6ns
|
0.750±0.01
8***
|
0.867±0.01
6ns
|
0.817±0.01
6*
|
0.733±0.01
6***
|
|
2 h
|
H
|
1
|
0.7
|
1
|
0.8
|
0.8
|
1
|
0.8
|
0.7
|
|
B
|
0.95
|
0.65
|
0.9
|
0.9
|
0.75
|
0.85
|
0.85
|
0.75
|
|
T
|
1
|
0.7
|
0.95
|
0.85
|
0.8
|
0.9
|
0.8
|
0.7
|
|
HB
|
0.95
|
0.7
|
0.9
|
0.9
|
0.7
|
0.85
|
0.85
|
0.8
|
|
BT
|
0.9
|
0.65
|
0.95
|
0.9
|
0.75
|
0.9
|
0.8
|
0.7
|
|
HT
|
1
|
0.6
|
0.9
|
0.85
|
0.8
|
1
|
0.9
|
0.75
|
|
mean±SEM
|
0.967±0.
016
|
0.667±0.01
6***
|
0.933±0.0
16ns
|
0.867±0.01
6**
|
0.767±0.01
6***
|
0.917±0.02
7ns
|
0.833±0.01
6***
|
±0.733±0.0
16***
|
|
3 h
|
H
|
1.05
|
0.6
|
0.95
|
0.9
|
0.8
|
1
|
0.75
|
0.65
|
|
B
|
1
|
0.55
|
1
|
0.85
|
0.75
|
0.95
|
0.8
|
0.7
|
|
T
|
1
|
0.6
|
0.9
|
0.8
|
0.7
|
0.9
|
0.85
|
0.7
|
|
HB
|
1
|
0.6
|
0.95
|
0.8
|
0.75
|
0.8
|
0.85
|
0.75
|
|
BT
|
1
|
0.6
|
0.9
|
0.85
|
0.7
|
0.95
|
0.85
|
0.7
|
|
HT
|
1.05
|
0.55
|
1
|
0.9
|
0.7
|
0.9
|
0.8
|
0.7
|
|
mean±SEM
|
1.017±0.
010
|
0.583±0.01
0***
|
0.950±0.0
18ns
|
0.850±0.01
8***
|
0.733±0.01
6***
|
0.917±0.02
7**
|
0.817±0.01
6***
|
0.700±0.01
2***
|
|
4 h
|
H
|
1
|
0.6
|
0.85
|
0.8
|
0.7
|
0.9
|
0.7
|
0.6
|
|
B
|
0.95
|
0.5
|
0.9
|
0.75
|
0.7
|
0.8
|
0.7
|
0.65
|
|
T
|
1
|
0.5
|
0.8
|
0.7
|
0.65
|
0.8
|
0.8
|
0.6
|
|
HB
|
0.95
|
0.6
|
0.9
|
0.75
|
0.7
|
0.75
|
0.7
|
0.7
|
|
BT
|
1
|
0.6
|
0.85
|
0.8
|
0.6
|
0.85
|
0.75
|
0.6
|
|
Time after
|
Paw oedema volume (ml)
|
|
|
HT
|
1
|
0.5
|
0.9
|
0.8
|
0.65
|
0.8
|
0.75
|
0.65
|
|
mean±SEM
|
0.983±0.
010
|
0.550±0.02
2***
|
0.867±0.0
16**
|
0.767±0.01
6***
|
0.667±0.01
6***
|
0.817±0.02
1***
|
0.733±0.01
6***
|
0.633±0.01
6***
|
| |
|
|
|
|
|
|
|
|
|
n = 6, Significant at P < 0 xss=removed>
AELAS- Alcoholic Extract of Leaf of Allium Sativum, AQELAS- Aqueous Extract of Leaf of Allium Sativum.
Table No: 5.11 Anti-inflammatory effects of AELAS and AQELAS in Formalin induced paw oedema model in rats at different time intervals
|
S. N
|
Groups
|
Treatment
|
1 h
|
% ROV
|
2 h
|
% ROV
|
3 h
|
% ROV
|
4 h
|
% ROV
|
|
A
|
Toxicant
|
Formalin (1% w/v)
|
0.900±0.012
|
--
|
0.967±0.016
|
--
|
01.017±0.010
|
--
|
0.983±0.010
|
--
|
|
B
|
Standard
|
Ibuprofen 40mg/kg
|
0.683±0.021***
|
54.75
|
0.667±0.016***
|
64.73
|
0.583±0.010***
|
84.4
|
0.550±0.022***
|
90.1
4
|
|
C
|
AELA S
|
100 mg/kg
|
0.883±0.027ns
|
4.75
|
0.933±0.016ns
|
7.78
|
0.950±0.018ns
|
13.4
5
|
0.867±0.016**
|
24.5
1
|
|
D
|
AELA S
|
200 mg/kg
|
0.833±0.016ns
|
17.25
|
0.867±0.016**
|
21.91
|
0.850±0.018***
|
32.8
|
0.767±0.016***
|
45.2
2
|
|
E
|
AELA S
|
400 mg/kg
|
0.750±0.018***
|
38
|
0.767±0.016***
|
43.32
|
0.733±0.016***
|
55.4
2
|
0.667±0.016***
|
65.9
2
|
|
F
|
AQELA S
|
100 mg/kg
|
0.867±0.016ns
|
8.75
|
0.917±0.027ns
|
11.2
|
0.917±0.027**
|
19.8
4
|
0.817±0.021***
|
34.8
6
|
|
G
|
AQELA S
|
200 mg/kg
|
0.817±0.016*
|
21.25
|
0.833±0.016***
|
29.19
|
0.817±0.016***
|
39.1
8
|
0.733±0.016***
|
52.2
5
|
|
H
|
AQELA S
|
400 mg/kg
|
0.733±0.016***
|
42.25
|
0.733±0.016***
|
50.6
|
0.700±0.012***
|
61.8
1
|
0.633±0.016***
|
72.9
6
|
n = 6, Significant at P < 0 xss=removed>
AELAS- Alcoholic Extract of Leaf of Allium Sativum, AQELAS- Aqueous Extract of Leaf of Allium Sativum.

Fig No.5.7
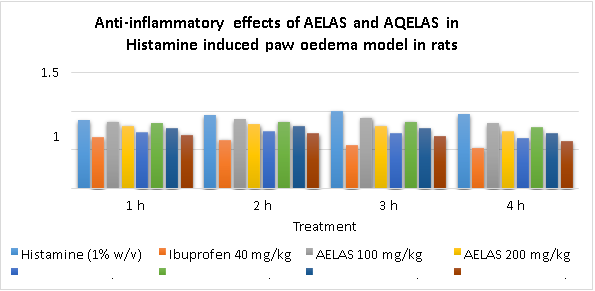
Fig No.5.8

Fig No.5.9
DISCUSSION:
- Carrageenan induced paw oedema model is used for screening of NSAIDs and inflammation produced by its biphasic in nature with the release of serotonin, bradykinin and histamine at I Phase followed by release of prostaglandins in II Phase
- Histamine being an important mediator of inflammation and also a potent vasodilator that causes increase in vascular permeability. In both phases due to release of these mediators causes pain and fever and both the extracts significantly reduced paw oedema in II Phase of the inflammation indicating there effect on prostaglandins.
- Formalin induced paw oedema model consist of two phases too with nociception in neurogenic and inflammatory phases. Drugs that primarily act on central nervous system are capable to inhibit both phases equally and by inhibiting the late phase, peripherally acting drugs produce there anti-inflammatory activity. The neurogenic and inflammatory phases are due to release of substance P, serotonin, histamine, prostaglandins and leukotrienes respectively. Both the extracts significantly reduced both phases of inflammation in formalin induced paw oedema model in rats.
- Tannins, sterols, flavonoids, alkaloids and triterpenes are reported for their anti- inflammatory activity. Both AELAS and AQELAS contained all the above mentioned Phytoconstituents and hence these might have contributed for the anti- inflammatory activity.
SUMMARY:
In the present investigation, the focus is on evaluating the leaf extract of Allium sativum for its potential anti-inflammatory activity. The study begins with an introduction detailing the significance of exploring the leaf extract's therapeutic potential, emphasizing why this research is crucial. The first chapter provides an overview of the leaf extract and highlights the necessity of this investigation to address gaps in current knowledge. It outlines the study's objectives and the necessity for the research. It includes the extraction of leaf material using various solvents, analysis of phytochemical constituents, and a toxicity study of the extracts. This chapter also covers the collection and analysis of data, setting the foundation for the subsequent evaluation. In this, a review of literature is presented, summarizing past research related to the extraction and evaluation of the anti-inflammatory properties of Allium sativum. This review draws from various scientific journals to contextualize the current study within the broader field. It also describes the materials and methods employed in the study. It details the procedures used for assessing the anti-inflammatory activities of the Allium sativum leaf extract, ensuring a comprehensive understanding of the experimental approach. The results and discussion are covered, where the biological activity of the leaf extract is evaluated and discussed in the context of its potential therapeutic effects. This chapter presents the findings and interprets their significance. It provides a conclusion based on the results, suggesting that while the leaf extract shows promise, further research is needed to isolate and identify the active principles responsible for its effects. Finally, a summary of the study, and the bibliography, including a compilation of reviewed journals and other references relevant to the research.
CONCLUSION:
Both AELAS and AQELAS contained tannins, carbohydrates, sterols, flavonoids, glycosides, alkaloids, and triterpenes, according to a preliminary phytochemical analysis of the extracts. Even at the dosage level of 2000 mg/kg body weight, neither of the extracts was associated with any fatality in acute oral toxicity trials. Both extracts demonstrated anti-inflammatory properties in rats used as experimental subjects with various inflammation models. Anti-inflammatory effects were obtained by the extracts at low, medium, and high doses in inflammatory models (P < 0>
REFERENCES
- Li F, Li Q, Wu S, Tan Z. Salting-out extraction of allicin from garlic (Allium sativum L.) based on ethanol/ammonium sulphate in laboratory and pilot scale. Food Chemistry. 2017 Feb 15; 217:91-7.
- Meriga B, Mopuri R, Murali Krishna T. Insecticidal, antimicrobial and antioxidant activities of bulb extracts of Allium sativum. Asian Pacific journal of tropical medicine. 2012 May 1;5(5):391-5.
- Anukworji CA, Putheti RR, Okigbo2012 RN. Isolation of fungi causing rot of cocoyam (Colocasia esculenta L.) Schott) and control with plant extracts :( Allium sativum L., Garcinia kola, heckel., Azadirachta indica, L. and Carica papaya, L.). Global Advanced Research Journal of Agricultural Science. 2012;1(1):1-5.
- Eja ME, Asikong BE, Abriba C, Arikpo GE, Anwan EE, Enyi-Idoh KH. A comparative assessment of the antimicrobial effects of garlic (Allium sativum) and antibiotics on diarrheagenic organisms. Southeast Asian journal of tropical medicine and public health. 2007 Mar 1;38(2):343.
- Belguith H, Kthiri F, Chati A, Abu Sofah A, Ben Hamida J, Ladoulsi A. Inhibitory effect of aqueous garlic extract (Allium sativum) on some isolated Salmonella serovars. Afr J Microbiol Res. 2010 Mar 4;4(5):328-8.
- Karuppiah P, Rajaram S. Antibacterial effect of Allium sativum cloves and Zingiber officinale rhizomes against multiple-drug resistant clinical pathogens. Asian Pacific journal of tropical biomedicine. 2012 Aug 1;2(8):597-601.
- Harris JC, Cottrell SL, Plummer S, Lloyd D. Antimicrobial properties of Allium sativum (garlic). Applied microbiology and biotechnology. 2001 Oct;57(3):282-6.
- Gatsing D, Aliyu R, Kuiate JR, Garba IH, Jaryum KH, Tedongmo N, Tchouanguep FM, Adoga GI. Toxicological evaluation of the aqueous extract of Allium sativum bulbs on laboratory mice and rats. Cameroon Journal of Experimental Biology. 2005;1(1):39-45.
- Choudhary S, Noor MU, Hussain MS, Mishra M, Tyagi S. Pharmacological properties and phytoconstituents of garlic (Allium sativum L.): A review. Biological Sciences. 2022 Dec 25;2(4):338-46.
- Fehri B, Ahmed MK, Aiache JM. The relaxant effect induced by Allium sativum L. bulb aqueous extract on rat isolated trachea. Pharmacognosy Magazine. 2011 Jan;7(25):14.
- Montes-Bayón M, Molet MJ, González EB, Sanz-Medel A. Evaluation of different sample extraction strategies for selenium determination in selenium-enriched plants (Allium sativum and Brassica juncea) and Se speciation by HPLC-ICP-MS. Talanta. 2006 Feb 15;68(4):1287-93.
- Bar M, Binduga UE, Szychowski KA. Methods of isolation of active substances from garlic (Allium sativum L.) and its impact on the composition and biological properties of garlic extracts. Antioxidants. 2022 Jul 9;11(7):1345.
- Chavan RD, Shinde P, Girkar K, Madage R, Chowdhary A. Assessment of anti-influenza activity and hemagglutination inhibition of Plumbago indica and Allium sativum extracts. Pharmacognosy Research. 2016 Apr;8(2):105.
- Montes-Bayón M, Molet MJ, González EB, Sanz-Medel A. Evaluation of different sample extraction strategies for selenium determination in selenium-enriched plants (Allium sativum and Brassica juncea) and Se speciation by HPLC-ICP-MS. Talanta. 2006 Feb 15;68(4):1287-93.
- Carreón-Delgado DF, Hernández-Montesinos IY, Rivera-Hernández KN, del Sugeyrol Villa-Ramírez M, Ochoa-Velasco CE, Ramírez-López C. Evaluation of Pretreatments and Extraction Conditions on the Antifungal and Antioxidant Effects of Garlic (Allium sativum) Peel Extracts. Plants. 2023 Jan;12(1):217.
- Vaseeharan B, Prasad GS, Ramasamy P, Brennan G. Antibacterial activity of Allium sativum against multidrug-resistant Vibrio harveyi isolated from black gill–diseased Fenneropenaeus indicus. Aquaculture international. 2011 Jun;19(3):531-9.
- Ziarlarimi A, Irani M, Gharahveysi S, Rahmani Z. Investigation of antibacterial effects of garlic (Allium sativum), mint (Menthe spp.) and onion (Allium cepa) herbal extracts on Escherichia coli isolated from broiler chickens. African Journal of Biotechnology. 2011;10(50):10320-2.
- Suetsuna K. Isolation and characterization of angiotensin I-converting enzyme inhibitor dipeptides derived from Allium sativum L (garlic). The Journal of Nutritional Biochemistry. 1998 Jul 1;9(7):415-9.
- Harazem R, El Rahman SA, El-Kenawy A. Evaluation of Antiviral Activity of Allium Cepa and Allium Sativum Extracts Against Newcastle Disease Virus. Alexandria Journal for Veterinary Sciences. 2019 Apr 15;61(1).
- Mathialagan R, Mansor N, Shamsuddin MR, Uemura Y, Majeed Z. Optimisation of ultrasonic-assisted extraction (UAE) of allicin from garlic (Allium sativum L.). Chemical Engineering Transactions. 2017 Mar 20; 56:1747-52.
- Bhattacharya S, Gupta D, Sen D, Bhattacharjee C. Process intensification on the enhancement of allicin yield from Allium sativum through ultrasound attenuated nonionic micellar extraction. Chemical Engineering and Processing-Process Intensification. 2021 Dec 1; 169:108610.
- Lanzotti V, Barile E, Antignani V, Bonanomi G, Scala F. Antifungal saponins from bulbs of garlic, Allium sativum L. var. Voghiera. Phytochemistry. 2012 Jun 1; 78:126-34.
- Epaminondas PS, Araújo KL, Nascimento JA, Souza AL, Rosenhaim R, Souza AG. Assessment of the antioxidant effect of ethanol extract of Allium sativum L., isolated and/or synergistically associated with synthetic antioxidants, applied to linseed oil. Journal of Thermal Analysis and Calorimetry. 2015 Apr;120(1):617-25.
- Fratianni F, Ombra MN, Cozzolino A, Riccardi R, Spigno P, Tremonte P, Coppola R, Nazzaro F. Phenolic constituents, antioxidant, antimicrobial and anti-proliferative activities of different endemic Italian varieties of garlic (Allium sativum L.). Journal of Functional Foods. 2016 Mar 1; 21:240-8.
- Hwang IG, Woo KS, Kim DJ, Hong JT, Hwang BY, Lee YR, Jeong HS. Isolation and identification of an antioxidant substance from heated garlic (Allium sativum L.). Food Science and Biotechnology. 2007;16(6):963-6.
- Fani MM, Kohanteb J, Dayaghi M. Inhibitory activity of garlic (Allium sativum) extract on multidrug-resistant Streptococcus mutans. Journal of Indian Society of Pedodontics and Preventive Dentistry. 2007 Oct 1;25(4):164.
- Cañizares P, Gracia I, Gómez LA, de Argila CM, de Rafael L, García A. Optimization of Allium sativum solvent extraction for the inhibition of in vitro growth of Helicobacter pylori. Biotechnology progress. 2002;18(6):1227-32.
- Li R, Chen WC, Wang WP, Tian WY, Zhang XG. Extraction of essential oils from garlic (Allium sativum) using ligarine as solvent and its immunity activity in gastric cancer rat. Medicinal Chemistry Research. 2010 Dec;19(9):1092-105.
- Rahim MA, Yasmin A, Imran M, Nisa MU, Khalid W, Esatbeyoglu T, Korma SA. Optimization of the Ultrasound Operating Conditions for Extraction and Quantification of Fructo oligosaccharides from Garlic (Allium sativum L.) via High-Performance Liquid Chromatography with Refractive Index Detector. Molecules. 2022 Sep 27;27(19):6388.
- Corbu VM, Gheorghe I, Marina? IC, Gean? EI, Moza MI, Csutak O, Chifiriuc MC. Demonstration of Allium sativum Extract Inhibitory Effect on Biodeteriogenic Microbial Strain Growth, Biofilm Development, and Enzymatic and Organic Acid Production. Molecules. 2021 Nov 27;26(23):7195.
- Samuel JK, Andrews B, Jebashree HS. In vitro evaluation of the antifungal activity of Allium sativum bulb extract against Trichophyton rubrum, a human skin pathogen. World Journal of Microbiology and Biotechnology. 2000 Aug;16(7):617-20.
- Ataee RA, Araqizade H, Yoosefi R, Tavana AM, Ataee MH. Effect of Allium sativum Extract on Erythromycin and Methicillin Resistant Bacteria Isolated from Hospital Operating Room. Journal of Medical Bacteriology. 2016 Dec 31;5(1-2):7-14.
- Cavallito CJ, Bailey JH. Allicin, the antibacterial principle of Allium sativum. I. Isolation, physical properties and antibacterial action. Journal of the American Chemical Society. 1944 Nov;66(11):1950-1.
- Rout A, Banerjee P, Bhadra P. The Role of Allium sativum Extract in Treating Aeromonas Infection of Labeo rohita. Biosc. Biotech. Res. Comm. 2020;13(12):98-110.
- Keshetty V, Pabba S, Gudipati R, Kandukuri JM, Allenki V. Antihyperlipidemic Activity of methanolic extract of Garlic (Allium sativum L.) in Triton X-100 induced hyperlipidemic rats. J Pharm Res. 2009 May;2(5):23-31.
- Nair SS, Gaikwad SS, Kulkarni SP, Mukne AP. Allium sativum constituents exhibit anti- tubercular activity in vitro and in RAW 264.7 mouse macrophage cells infected with Mycobacterium tuberculosis H37Rv. Pharmacognosy magazine. 2017 Jul; 13(Suppl 2):S209.
- EL-mahmood MA. Efficacy of crude extracts of garlic (Allium sativum Linn.) against nosocomial Escherichia coli, Staphylococcus aureus, Streptococcus pneumoniea and Pseudomonas aeruginosa. Journal of Medicinal Plants Research. 2009 Apr 30;3(4):179- 85.
- Foroutan-Rad M, Tappeh KH, Khademvatan S. Antileishmanial and Immunomodulatory Activity of Allium sativum (Garlic) A Review. Journal of evidence-based complementary & alternative medicine. 2017 Jan;22(1):141-55.
- Salem WM, El-Hamed DM, Sayed WF, Elamary RB. Alterations in virulence and antibiotic resistant genes of multidrug-resistant Salmonella serovars isolated from poultry: The bactericidal efficacy of Allium sativum. Microbial Pathogenesis. 2017 Jul 1; 108:91-100.
- Yan JK, Wang C, Yu YB, Wu LX, Chen TT, Wang ZW. Physicochemical characteristics and in vitro biological activities of polysaccharides derived from raw garlic (Allium sativum L.) bulbs via three-phase partitioning combined with gradient ethanol precipitation method. Food Chemistry. 2021 Mar 1; 339:128081.
- Eftekhari Z, Bahmani M, Mohsenzadeghan A, Gholami Ahangaran M, Abbasi J, Alighazi N. Evaluating the anti-leech (Limnatis nilotica) activity of methanolic extract of Allium sativum L. compared with levamisole and metronidazole. Comparative Clinical Pathology. 2012 Dec;21(6):1219-22.
- Divya BJ, Suman B, Venkataswamy M, Thyagaraju K. A study on phytochemicals, functional groups and mineral composition of Allium sativum (garlic) cloves. Int J Curr Pharm Res. 2017 May 5;9(3):42-5.
- Roy J, Shakleya DM, Callery PS, Thomas JG. Chemical constituents and antimicrobial activity of a traditional herbal medicine containing garlic and black cumin. African Journal of Traditional, Complementary and Alternative Medicines. 2006 Jan 12;3(2):8- 20.
- Bhandari SR, Yoon MK, Kwak JH. Contents of phytochemical constituents and antioxidant activity of 19 garlic (Allium sativum L.) parental lines and cultivars. Horticulture, Environment, and Biotechnology. 2014 Apr;55(2):138-47.
- Sulaiman FA, Kazeem MO, Waheed AM, Temowo SO, Azeez IO, Zubair FI, Adeyemi TA, Nyang A, Adeyemi OS. Antimicrobial and toxic potential of aqueous extracts of Allium sativum, Hibiscus sabdariffa and Zingiber officinale in Wistar rats. Journal of Taibah University for Science. 2014 Oct 1;8(4):315-22.
- Vlase L, Parvu M, Parvu EA, Toiu A. Chemical constituents of three Allium species from Romania. Molecules. 2012 Dec 21;18(1):114-27.
- Attia S, Grissa KL, Mailleux AC, Lognay G, Heuskin S, Mayoufi S, Hance T. Effective concentrations of garlic distillate (Allium sativum) for the control of Tetranychus urticae (Tetranychidae). Journal of Applied Entomology. 2012 May;136(4):302-12.
- Herrera-Calderon O, Chacaltana-Ramos LJ, Huayanca-Gutiérrez IC, Algarni MA, Alqarni M, Batiha GE. Chemical constituents, in vitro antioxidant activity and in silico study on NADPH oxidase of Allium sativum L.(garlic) essential oil. Antioxidants. 2021 Nov 20;10(11):1844.
- Sharma V. Analysis of phytoconstituents and free radical scavenging activity of hydroethanol extract of Allium sativum bulb. International Journal of Green Pharmacy (IJGP). 2019 Dec 18;13(04).
- Bin C, Al-Dhabi NA, Esmail GA, Arokiyaraj S, Arasu MV. Potential effect of Allium sativum bulb for the treatment of biofilm forming clinical pathogens recovered from periodontal and dental caries. Saudi journal of biological sciences. 2020 Jun 1;27(6):1428-34.
- Sabiu S, Madende M, Ajao AA, Aladodo RA, Nurain IO, Ahmad JB. The Genus Allium (Amaryllidaceae: Alloideae): Features, phytoconstituents, and mechanisms of antidiabetic potential of Allium cepa and Allium sativum. Bioactive food as dietary interventions for diabetes. 2019 Jan 1:137-54.
- Liaqat A, Zahoor T, Atif Randhawa M, Shahid M. Characterization and antimicrobial potential of bioactive components of sonicated extract from garlic (Allium sativum) against foodborne pathogens. Journal of Food Processing and Preservation. 2019 May;43(5):e13936.
- Szychowski KA, Rybczynska-Tkaczyk K, Gawel-Beben K, Swieca M, Karas M, Jakuczyk A, Matysiak M, Binduga UE, Gminski J. Characterization of active compounds of different garlic (Allium sativum L.) cultivars. Polish Journal of Food and Nutrition Sciences. 2018;68(1).


 Shital Darekar* 1
Shital Darekar* 1








 10.5281/zenodo.14160125
10.5281/zenodo.14160125