
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Department of Pharmacy- KBHSS Trust’s Institute of Pharmacy, Malegaon, Nashik, Maharashtra- 423203.
Effervescent granules or tablets are unique dosage form having drug and Effervescent base which is composed of sodium hydrogen carbonate, citric acid and tartaric acid, this combination when added to water react to liberate co2, resulting in effervescence. There are many uses for these granules in daily life. Even while oral dose forms have certain drawbacks when compared to other methods, such as the possibility of medication flow absorption, which can be mitigated by administering the medicine in liquid form, they are still the most often used type of pharmaceutical administration. Thus, the use of all-over dose may be permitted.
One or more active pharmaceutical substances, with or without other components, are present in granules, a special kind of dosage form made of dried aggregates of powdered solid particles. [1]. The word "effervescent" comes from a Latin word that refers to the release of gas from a water or aqueous solution. [2]. Effervescent pills are becoming increasingly popular in a range of sectors include supplement and pharmaceutical use, thanks to the simplicity during which they will be ingested. Effervescence is the term used to describe the removal of carbon dioxide gas from a fluid as a result of a chemical reaction. This action starts when the preparation comes into contact with water, which serves as a catalytic agent. Effervescent tablets need to dissolve in water before being taken. The tablet breaks down on schedule by releasing carbon dioxide into the water. Carbon dioxide is produced by the effervescent process, which enhances the active ingredient's absorption by facilitating its passage through the paracellular pathway. This kind of patient can benefit from such dose forms because the effervescent formulation prevents direct interaction with the gastrointestinal tract. Liquid dosage forms decrease the onset of impact since they absorb formulations faster than tablet formulations. [1-2] Effervescent pills are becoming more and more popular in a variety of industries, including vitamins and pharmaceuticals, due to their ease of usage. Effervescent tablets are designed to shatter when they come into contact with liquids like water or juice, which usually causes the tablet to dissolve into a solution. [3] The emission of CO2 gas in reaction to acids and bicarbonates in the presence of H2O is known as effervescence. This reaction also commonly uses fumaric, adipic, tartaric, malic, and citric acids. The bicarbonates employed in the effervescent process are potassium, sodium, and potassium bicarbonate. The most frequently utilised drug reaction in pharmaceuticals is the acid-base interaction between sodium bicarbonate and citric acid. 3NaHCO3 + H3C6H5O7 = 3Na3C6H5O7 + 3H2O + 3CO2 in water. Even in tiny levels, water is present during this process and acts as a catalytic agent, accelerating it. All products that are effervescent or sensitive to moisture should be stored in a dry environment since water catalyses the process.

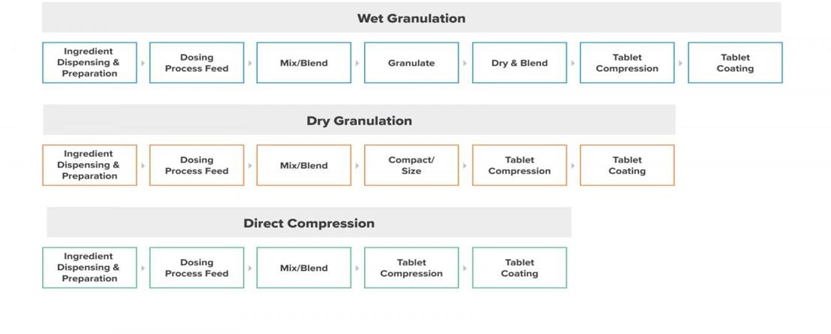
Effervescent Tablet Methodology A] Wet Granulation:
Wet granulation is the most widely used method of agglomeration in the pharmaceutical industry. It involves wet massing of the powder mixture with a granulating liquid and wet filler, followed by drying. [5-6] Despite some disadvantages, wet granulation remains the preferred method for effervescent granulation. It produces homogeneous granules suitable for compression and ensures uniformity in tablet weight and active ingredient content. Wet granulation can be divided into two types depending on the number of process steps involved. [7]
Key steps in the wet granulation process: -
Mixing of the drug(s) and excipients.
ADVANTAGES:
Limitations: -
The main drawback is its high cost due to labor, time, equipment, energy, and space requirements.
B] Dry Granulation:
Dry granulation involves compressing the powder mixture without using heat or solvents. This method is considered the least attractive among granulation techniques.
Two main methods are used:
Compressing the powder mixture between two rollers to create a sheet, which is then milled into granules. [8-15]
Roller compaction:-
A softer method that creates compressed sheets by squeezing powder between rollers.- Sheets break into flakes, which are further milled into granules.- This technique avoids heat and liquid use and is vital in increasing powder bulk density and particle size for better flow. [16-17]
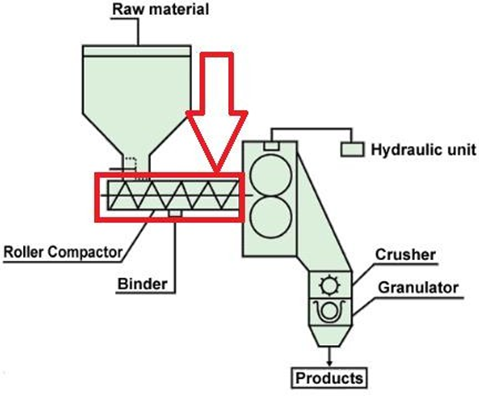
Applications: -
Used in producing directly compressible excipients.
Direct Compression:
Direct compression is a viable alternative for making effervescent tablets with acetylsalicylic acid. It addresses issues with operational efficiency and stability. However, it requires compressible, free-flowing, non-segregating materials, making it suitable only in optimal manufacturing environments.
Advancements in Granulation:
A variation of wet granulation using steam as a binding agent instead of water. Benefits include:- Uniform distribution.- Shorter processing time and increased tablet production.- Environmentally friendly (no toxic solvent residues).- Higher dissolution rates due to larger granule surface area. - Sterile steam avoids contamination. [18-19]
In this method, an elastic binder that melts at elevated temperatures is used. On melting, the binder acts as a binding liquid. Granules form upon cooling without requiring a drying step. [20]
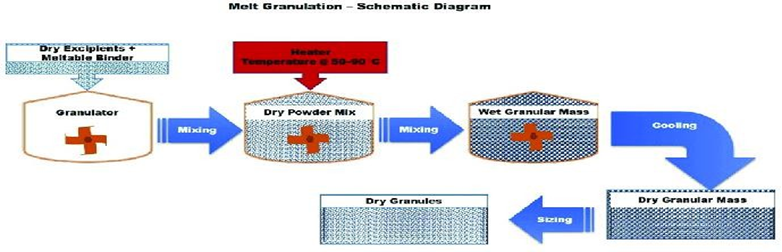
4. Shear Granulation:
Used for decades in pharmaceutical granulation. Involves high-shear mixing in a closed tank with a chopper and agitator blade.
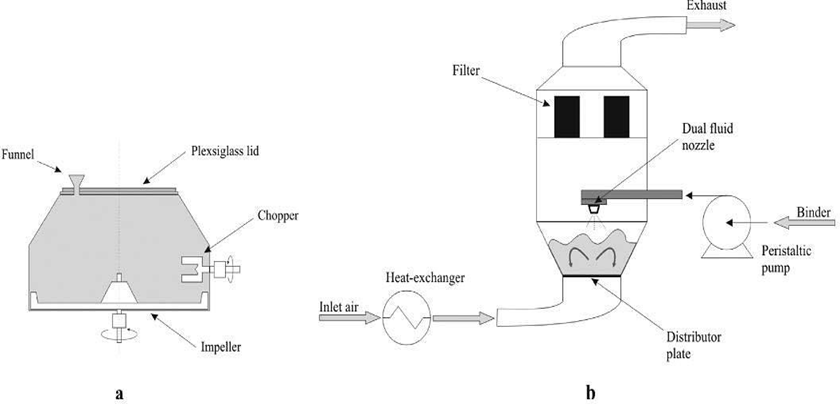
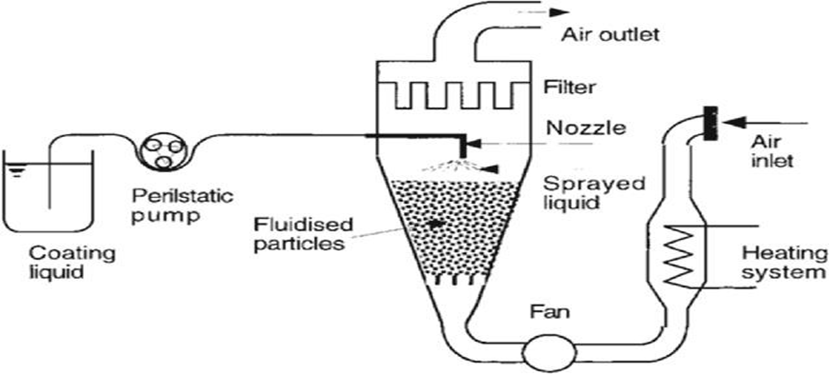
3. Fluid Bed Granulation:
Effervescent granules are produced by suspending a dry mixture of acid and carbonate in hot air.
Excipients used in EDDS
Acid materials
The three main sources of acidity are necessary for effervescence. Food acids such as ascorbic, tartaric, and citric acids are frequently utilized. These are favored due to their easy handling, affordable price, excellent flavor, and lack of odor.[24]
Citric acid
It is the chosen acidic component in effervescent products due to its outstanding solubility as well as incredibly good taste. It is a white, crystalline powder that is colorless. There are several different sizes of particles available, such as fine, medium, coarse, and powder (for the anhydrous form). Citric acid is soluble in both ethanol and water. It can be utilized as either a monohydrate or an anhydrate, depending on the machinery chosen and the specifics of the process. Compared to the monohydrate, the anhydrous form of citric acid is less prone to absorbing moisture. But if kept under high relative humidity (over 70%), the anhydrous form might aggregate. Although the anhydrous form is widely used in the formulation of effervescent tablets, citric acid monohydrate is frequently used in the preparation of effervescent granules. The monohydrate form is a suitable option for use as a binder in hot melt granulation processes since it melts at 100°C and releases its water of hydration at 75°C[25]
Tartaric acid
It is highly soluble in water and is more very hygroscopic in nature. Similar to citric acid, tartaric acid produces considerable effervescence when it reacts with sodium bicarbonate in effervescence reactions. Nevertheless, because it is diprotic (containing two hydrogen atoms that are acidic), Since citric acid is a triprotic acid, tartaric acid must be used in greater amounts to attain the proper stoichiometric proportions. When it comes to compression, tartaric acid and citric acid show similar characteristics.[26]
Ascorbic acid
It appears as white crystals when it is crystalline and pale yellow when it is finely powdered. It can be useful during production because it is easy to handle and is not hygroscopic like certain other acids. It exhibits great water solubility; around one gram dissolves in three milliliters of pure ethanol and water). On the other hand, it gradually darkens when exposed to light. The rate at which carbon dioxide is emitted in an effervescent reaction involving sodium bicarbonate is comparable to that of other acids, such as citric and tartaric acids.[27]
Acid anhydrides
Because food acid anhydrides can act as hydroxylases in water to produce the appropriate acid, they can be a practical source of acidity in effervescent compositions. This hydroxylation process continuously produces acid in the solution, ensuring a powerful and long-lasting effervescence effect. It is essential to stay away from water at all times when using anhydrides in a formulation. Before being used, the anhydrides would hydrolyze and change into the appropriate acids if water were present[28].
Binders
Generally, incorporating a binder is often essential to achieve the desired hardness in effervescent tablets for safe handling. However, the use of binders in effervescent formulations needs enhancement due to their tendency to hinder tablet disintegration and absorb moisture. Achieving an optimal balance between granule strength and disintegration time is vital when deciding on the amount of binder in a formulation. Water can serve as an efficient binder during wet granulation when all components are granulated together. A small amount of water, evenly spread across the powder bed, partially dissolves the ingredients and aids in agglomeration. Alternatively, solvents such as ethanol and isopropanol can be employed as granulating liquids to dissolve dry binders. The selection of a binder in wet granulation is influenced by the production method and the amount of granulating liquid utilized. For example, when combining alkaline and acidic ingredients with water, incorporating a binder into the mixture may not be necessary because the limited amount of water does not sufficiently activate it. The binder most frequently employed for effervescent tablets is polyvinylpyrrolidone (PVP) due to its effective binding properties, even in low doses (generally around 2%). PVP K25 and K30 are preferred choices because of their excellent solubility in water and their capacity to uphold the desired dissolution rate of the active compound, which aligns with the objectives of effervescent tablets. They can be employed in aqueous, alcoholic, and hydroalcoholic solutions, and they may also be applied in dry granulation methods.[29]. Binders typically used in dry granulation, such as lactose, mannitol, and dextrose, are usually not appropriate for effervescent formulations because they would necessitate larger quantities than permitted.[29]
Lubricants
The process of tableting is essential for the formulation of effervescent products, making the choice of a suitable lubricant critically important due to the distinct chemical and physical properties of these lubricants. Because many lubricants are poorly soluble in water, they can impede the rapid disintegration necessary for effervescent tablets. It is vital for effervescent tablets to generate a clear, transparent solution without leaving any insoluble film or residue on the surface of the water. When choosing a lubricant, it is important to carefully assess its water solubility and how well it aligns with the therapeutic effects of the active ingredient. Various lubricants have been evaluated to identify the optimal conditions for effervescent tablets, including the option of externally lubricating the granules directly within the tablet press dies. Frequently utilized lubricants in the production of effervescent products include sodium benzoate, sodium acetate, L-leucine, and carbowax 4000. Research has also investigated combinations of lubricants, such as calcium and potassium sorbates, along with micronized polyethylene glycol (PEG) paired with calcium ascorbate or trisodium citrate. One effective blend consists of spray dried L- leucine and PEG 6000. While lubricants with lower solubility continue to be used in the formulation of effervescent tablets, it is essential to find a proper balance between the efficiency of compression and water solubility for optimal lubrication. Magnesium stearate available commercially is commonly paired with sodium lauryl sulfate, a surfactant that enhances dispersion.[30]
Additives
Small amounts of additives are incorporated into the formulas to improve the flavor and visual appeal of effervescent products. Commonly used water- soluble flavors, such as lemon, orange, and fruit essences, are utilized to fulfill sensory expectations. Usually, these flavors make up approximately 0.5 to 3.0% of the final formulation. Sweeteners like sorbitol, sucrose, aspartame, stevia, and sodium saccharin are frequently blended with flavors to achieve the desired level of sweetness. Coloring agents, which can be either synthetic FD & C dyes or natural coloring agents, may be included in quantities ranging from 0.1 to 3.5% of the product. Additionally, surfactants or antifoaming agents might be used to enhance the functionality of the effervescent preparation during application.[31]
Surfactants
Surfactants such as sodium lauryl sulfate and magnesium lauryl sulfate also function as lubricants. In herbal effervescent tablets, surfactants are utilized to improve the solubility and dissolution of the active ingredients, enhance the disintegration of the tablet, and support the overall effervescence process. They contribute to producing a more consistent and stable product by lowering surface tension and facilitating better interaction between the tablet components and water.[32]
Types of Surfactants:
A variety of surfactants can be utilized in effervescent tablets, such as:
Sugar esters: These natural surfactants are obtained from sugars and fatty acids.
Polysorbates: These nonionic surfactants are frequently used in food and pharmaceutical applications.
Phospholipids: These molecules exhibit both hydrophilic and hydrophobic properties and can serve as emulsifiers and solubilizers.
Sodium lauryl sulfate: This is a widely-used anionic surfactant found in numerous products.
Other surfactants: Depending on the particular formulation, additional surfactants may be used to achieve the intended results.[32]
Anti-adherents
Ants that adhere to Discs such as poly tetra fluorethylene or polyurethane are used to stop the granules from adhering.
Disintegrates or dissolution aids
Aids for breakdown or disintegration Disintegrates are chosen based on the requirement that, after a few minutes, a transparent solution be produced by mixing the tablet with a glass of cold water.[33]
Antifoaming agents
Agents that prevent foaming to lessen the froth that forms and, as a result, the medication's propensity to remain on the glass wall above the water level. It uses polydimethylsiloxane as an antifoaming agent[34]
Sweeteners
Various natural sweeteners, including sucrose and saccharin, were utilized. Sweeteners are frequently used in effervescent herbal tablets to enhance their flavor and palatability. Common sweeteners include sugar alcohols like sorbitol and maltitol, which can also serve as binders and fillers, and natural alternatives like stevia, which is well-liked for its sweetness and low calorie content. Furthermore, some formulas use tea extracts or fruit juices to add flavor and improve taste even further.[35]
Natural Sweeteners:
Stevia: Known for its low calorie content and high sweetness intensity, stevia is a natural sweetener made from the stevia plant.
Siraitia grosvenorii: Also referred to as monk fruit, this natural sweetener enhances the flavor of fizzy tablets.
Flavors
In order to cover up the taste of sweets, flavors are utilized as additives. Flavors like orange, blueberry, and mixed berry are frequently used in herbal effervescent pills to improve palatability and cover up the taste of the herbal constituents. Lemon, lime, and strawberry are other well-liked flavors that are frequently paired with tea scents or fruit extracts. Making the tablets more palatable and simpler to take is the aim, particularly when working with components that could taste harsh or powerful. [35]
Flavor Masking:
To produce a fizzing effect and cover up the taste of some herbal extracts, effervescent pills frequently combine acidic and alkaline ingredients (such as citric acid and sodium bicarbonate).
Colors
To provide the pleasing appearance, a color that dissolves in water is also applied. Both natural and synthetic colors are frequently used in herbal medications' effervescent tablets to improve aesthetic appeal, facilitate product identification, and possibly affect consumer perception. Beetroot, turmeric, and berry-based natural colorants are frequently employed. Additionally, synthetic colors are used, which have undergone extensive safety testing. [35]
Tablets of Bael Fruit:
The fruit extract in bael fruit powder can give tablets a brownish or yellowish hue.
Evaluation Parameters of Effervescent Tablets
Effervescent tablets are innovative dosage forms designed to disintegrate and dissolve upon contact with water, releasing carbon dioxide gas. This reaction enhances the absorption and palatability of drugs, making effervescent tablets ideal for patients who struggle with swallowing conventional solid dosage forms. This review focuses on the detailed evaluation parameters of effervescent tablets as derived from multiple scientific sources.
Effervescent Reaction Formula
Citric acid + Sodium bicarbonate → Sodium citrate + Water + Carbon dioxide C?H?O? (aq) + 3NaHCO? (aq) → Na?C?H?O? (aq) + 4H?O + 3CO? (g) ↑
Evaluation of precompression:
It Was measured by fixed funnel method The fixed funnel method employ a funnel that was secured with its tip at a given height h", above graph paper that was placed on a flat horizontal Granules were carefully poured through the funnel until the apex of the conical pile just touches the tip of the funnel. Thus, with being the radius of the base of the conical pile[36]
tan-h/r
An accurately weighed sample of granulation was carefully added to the measuring cylinder with the aid of funnel. The level was observed without compacting and and noted apparent volume (V0),14,15 The bulk density was calculated by the formula as given below: [36]
Bulk density= M/VO
After bulk density measurement the cylinder was placed on the tapped density tester (ETD 1060, Electrolah) and was mechanically tapped. The cylinder was tapped for 500 times initially and the tapped volume (V1) was measured. to the nearest graduated units. The tapping was repeated for additional 750 times and the tap volume (V2) nearest tapped graduated sumits was noted. 15 The tappe density was calculated by the formala as given below:[37]
Tapped density= M/VB
The percentage compressibility of powder is direct measure of the potential of powder arch or bridge strength is calculated according to the equation given below.[38]
Tapped density = Tapped density-bulk density ×100
Hameer's ratio is an important character tu determine the flow property of powder and granules. This can be calculation by the following formula.[39]
Hausner's ratio= Tapped density
Bulk density Evaluation of post compression
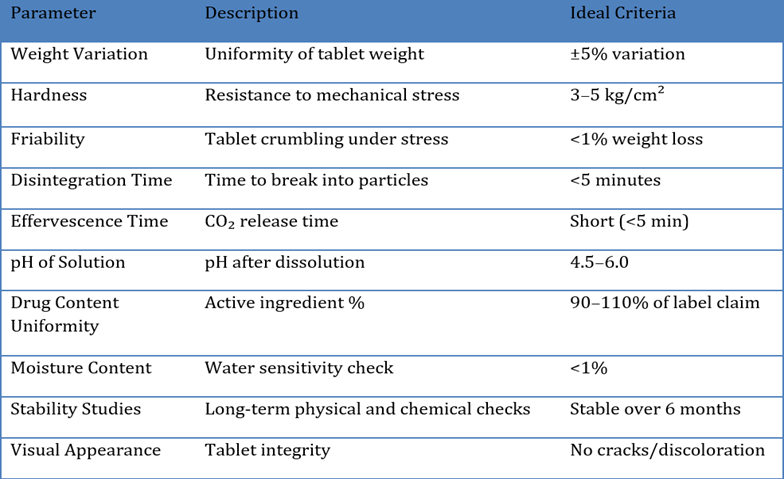
Weight variation ensures the uniformity of tablet mass. According to IP/BP standards, twenty tablets are weighed individually and their average weight is determined. Each tablet’s weight should fall within ±5% of the average weight for tablets weighing more than 250 mg.[41]
Tablet hardness is crucial for withstanding mechanical shocks during handling. A Monsanto or Pfizer hardness tester is typically used. Ideal effervescent tablets should not be too hard, as excessive hardness may delay disintegration and effervescence.[42]
Friability:
Friability measures a tablet's resistance to crumbling. Tablets are subjected to rotation in a friabilator and the weight loss percentage is calculated. For effervescent tablets, the friability should generally be less than 1%, indicating adequate mechanical integrity.[43]
Initial weight =Initial weight-final weight ×100
Disintegration time is the period required for a tablet to break down into smaller particles. Effervescent tablets must disintegrate quickly (typically within 5 minutes) when placed in water. This rapid action ensures immediate therapeutic effect.[44]
This parameter measures the time taken for complete release of carbon dioxide after the tablet comes in contact with water. Shorter effervescence time ensures better patient compliance and faster drug delivery.[44]
After dissolution, the pH of the resulting solution is evaluated. It should be within acceptable range to avoid gastrointestinal irritation and ensure drug stability. Most effervescent tablets are slightly acidic due to their acid-base composition.[44]
The active drug content must fall within 90-110% of the label claim. Spectrophotometry or HPLC is commonly used to evaluate content uniformity.[44]
Effervescent tablets are highly sensitive to moisture, which can initiate premature effervescence. Karl Fischer titration or loss on drying methods are employed to ensure moisture content remains within limits (typically<1%).[44]
Stability tests involve storing tablets under ICH-recommended conditions and evaluating physical and chemical stability over time. Parameters such as appearance, hardness, pH, and assay are monitored periodically.[44]
Tablets are inspected for cracks, color changes, and uniformity in shape and size. Good visual appearance indicates proper formulation and packaging.[44].
CONCLUSION
Constipation remains a common condition that significantly affects quality of life, requiring a comprehensive understanding of its causes, symptoms, and treatments. Herbal effervescent tablets offer a promising solution, providing benefits like faster action, better taste, and improved gastrointestinal tolerance. These tablets, made from natural ingredients such as stevia, Bael fruit pulp, psyllium husk, and Indian senna, aim to deliver effective treatment with fewer side effects. Formulated and tested according to international standards, these tablets show consistent quality, good effervescence, and improved patient compliance compared to traditional forms. However, challenges like stability, cost, and taste masking still need to be addressed. Continued research and collaboration are essential to refine these tablets and improve their accessibility and effectiveness, marking significant progress in the treatment of constipation through herbal medicine.
REFERENCES





Priyanka B. Shinde*, Roshani H. Nikam, Karuna S. Sonawane, Jagruti I. Sonawane, Shraddha P. Vaishnav, Effervescent Tablet, Int. J. of Pharm. Sci., 2025, Vol 3, Issue 9, 710-723 https://doi.org/10.5281/zenodo.17066492
 10.5281/zenodo.17066492
10.5281/zenodo.17066492