
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
1,3,4 Shri J G C H S College Of Pharmacy, Ghataprabha 591306
2 Shikshan Prasaraka Mandal's College of Pharmacy, Harugeri
The herbal system of medicine has gained worldwide importance due to its safety and efficacy considerations. Herbal medicines have been used for treating various diseases since ancient times. The present article deals with the importance and method of standardization of herbal medicines in order to ensure their purity, safety and efficacy. A detailed introduction regarding various aspects of the herbal system of medicine and methods of achieving quality of herbal medicine has been presented The various quality control methods recommended by World Health Organization have been described. Effort has also been made to focus on the physical, chemical and biological evaluation to achieve a high standard of quality for herbal medicine.
The Herbal system of medicine is widespread in India, Pakistan and Bangladesh and the Hakeems practicing this system are qualified from the recognized institutions of Eastern Medicine. It is also gaining importance in other developing countries as well as in Europe and North America. This system of medicine can play an important role in our national health care program. In view of the absence or relatively low side effects of herbal formulations, people are more inclined to use these formulations to get relief from the ailments. During the past decades, public interest in natural therapies, namely herbal medicine, has increased dramatically not only in developing countries but also in industrialized countries. Until a few years ago, only small companies had interest in the marketing of herbal medicines but in the current scenario many large multinational companies are interested in commercializing herbal drugs. 1 It is very important to ensure the quality of herbal medicines and to carry out quality control tests on the plant material used in herbal formulations. The efficacy, safety, purity, and quality of herbal formulations must be ensured before their use to achieve optimum benefits in the treatment of diseases. A very important element in the standardization of herbal formulations is the assessment of the quality of plant materials used in their manufacturing. If the material meets certain standards, the formulation would be of the desired quality and thus beneficial to the patient. The manufacturers of herbal medicine may have developed suitable standards to judge the quality of their products but in many cases such standards are not available. Quality control tests and specifications for the assessment of identity, purity and potency of several plant materials are available in the herbal and allopathic drug pharmacopoeias (e.g. British Herbal Pharmacopoeia, Indian Herbal Pharmacopoeia, Chinese Herbal Pharmacopoeia). It is therefore, necessary to confirm their compliance with the prescribed standards. The quality of the raw material is reflected in the quality of the herbal formulation incorporating them as essential ingredients.
Herbal drugs have been used since ancient times as medicines for the treatment of a range of diseases and the natural products have been considered as most successful source of medicines. Each plant is capable of synthesizing an indefinite number of highly complex and remarkable chemical substances that are considered as important drugs while several other drugs are simple synthetic modifications of the natural products.2-4
According to World Health Organization5 herbal medicines include herbs, herbal materials and herbal preparations as finished products. Herbs include crude plant material, such as leaves, flowers, fruit, seeds, stems, wood, bark, roots, rhizomes or other plant parts, which may be entire, fragmented or powdered material. Herbal materials include, in addition to herbs, fresh juices, gums, fixed oils, essential oils, resins and dry powders of herbs. In some countries, these materials may be processed by various local procedures, such as steaming, roasting or stir-baking with honey, alcoholic beverages or other materials. Herbal preparations may include powders, extracts, tinctures and fatty oils of herbal materials. They are produced by extraction, fractionation, purification, concentration, or other physical or biological processes. Finished herbal products consist of herbal preparations made from one or more herbs. If more than one herb is used, the term "polyherbal product" can also be applied. Finished single and polyherbal products may contain excipients in addition to the active ingredients. However, finished herbal products to which chemically defined active substances have been added, including synthetic compounds and/or isolated constituents from herbal materials, are not considered to be herbal medicine.6•7 In order to maintain reproducible efficacy and safety of phytopharmaceuticals used in rational drug therapy they should be standardized and their pharmaceutical quality must be approved.8•9 Similarly, WHO also stresses the importance of the qualitative and quantitative methods for characterizing herbal samples, quantification of the biomarkers and/ or chemical markers and the fingerprint profiles. If the principle active components are known, it becomes easy to quantify the compounds and the herbal preparations should be standardized on the basis these compounds. However, if the active ingredients are not known a specific marker substance should be selected for analytical purpose.10•11 In most cases these markers have never been tested to evaluate whether they really account for the therapeutic action reported for the herbal drugs but still they would normally be useful in the chemical assessment of the preparations.
Plants contain several constituents and some of them are present at very low concentration. In spite of the modem analytical methods available, only few phytochemical investigations have succeeded in isolating and characterizing all constituents present in a plant extract. Apart from this, plant constituents vary considerably depending on several factors (e.g. seasonal variations) that impair the quality of phytopharmaceutical agents.12 The quality control and standardization of herbal medicines involves several steps. However, the source and quality of the raw materials play a pivotal role in guaranteeing the quality and stability of herbal preparations.13 Other factors such as the use of fresh plants, temperature, light exposure, water availability, nutrients, period and time of collection, method of collection, drying, packing, storage and transportation of raw material, age and part of the plant collected, etc., can greatly influence the quality and consequently the therapeutic value of herbal medicines.14 Some plant constituents are heat labile and the plants containing them need to be dried at low temperature. Also, some active principles are destroyed by enzymatic processes that continue for long periods of time after plant collection. This explains why frequently the composition of herbal based drugs is quite variable. Apart from these variable factors, others such as the method of extraction and contamination with microorganisms, heavy metals, pesticides, etc., can also interfere with the quality, safety and efficacy of herbal drugs.15•16 About 80% of world population is dependent on herbal drugs and to enter into the global market it is vital to maintain their quality. The quality of herbal drugs is the sum of all factors which contribute directly or indirectly to the safety, effectiveness and acceptability of the product. Standardization of drugs means confirmation of its identity and assessment of its quality and purity.17 Initially the crude drugs have been identified by comparison only with the standard description available. At present due to the advancement in the chemical knowledge of crude drugs various methods like thin-layer chromatography (TLC), gas chromatography (GC), high-performance liquid chromatography (HPLC), ultraviolet/visible spectrometry (UVNis ), infrared-spectrometry (IR), mass spectrometry (MS) and nuclear magnetic resonance (NMR) spectrometry etc., alone or in combination, can be successfully used to conduct the tests required for standardization and to control the quality of both the raw material and the finished herbal drugs.18•19
STANDARDIZATION OF HERBAL MEDICINES
With the commercialization of herbal medicines, assurance of safety, quality and efficacy of medicinal plant material and herbal products has become an important issue. The herbal raw material is prone to a lot of variation due to several factors, the important ones being the identity of the plants and seasonal variation, the ecotypic, genotypic and chemotypic variations, drying and storage conditions and the presence of xenobiotic.20 Standardization is defined by American Herbal Product Association as "The body of information and control necessary to produce material of reasonable consistency". This can be achieved through minimizing the inherent variation of natural product composition through quality assurance practices applied to agricultural and manufacturing processes. Methods of standardization should comprise all aspects that contribute to the quality of the herbal drugs such as the correct identity of the sample, compliance with organoleptic test, determination of ash values and extractive values, phytochemical evaluation, volatility, tests for the presence of xenobiotics, microbial load, toxicity, and biological activity.21•22 Of these, the phytochemical profile is of special significance since it has a direct bearing on the activity of the herbal drugs. The IR fingerprint profiles serve as a reference to the phytochemical profile of the drug in ensuring the quality, while quantification of the marker compounds would serve as an additional parameter in assessing the quality of the sample. Phytochemical standardization encompasses all possible information generated with regard to the chemical constituents present in a herbal drug. The phytochemical evaluation for standardization purpose25 includes the following:
Methods of Standardization
Phytotherapeutic agents are normally marketed as standardized preparations in the form of liquid, solid (powdered extract), or viscous preparations. They are prepared by maceration, percolation or distillation (volatile oils). Usually ethanol, water, or mixtures of ethanol and water are used for the production of fluid extracts. The solid or powdered extracts are prepared by evaporation of the solvents used in the process of extraction of the raw material while some phytotherapeutic agents are greatly concentrated in order to improve their therapeutic efficacy.26 Therefore great care is required in the standardization of herbal medicines27 and various methods of standardization have been described according to Good Manufacturing Practices (GMPs).28•29 In addition, the study of various parameters such as pharmacodynamics, pharmacokinetics, dosage, stability, shelf-life, toxicity evaluation, chemical profiling of the herbal formulations is considered essential.30•31 The evaluation of heavy metals contamination, according to Good Agricultural Practices (GAPs) in herbal drug standardization is equally important.32 The stages to be considered during the standardization of raw herbal drugs and herbal formulation under the norms followed during standardization are given in Table 1.33•34 The guidelines set by WHO for the standardization of herbal drugs include: botanical characters, sensory evaluation, foreign organic matter, microscopic, histological, histochemical assessment, quantitative measurements, physical and chemical identity, fingerprints chromatography, ash values, extractive values, moisture content, volatile oil and alkaloids tests, quantitative estimation protocols, assessment of biological activity, the values of bitterness value, astringency, hemolytic index, swelling index, foaming index, toxicity, pesticides residues, heavy metals, microbial contamination as viable count total, pathogens such as E. coli, Salmonella species, Pseudomonas aeruginosa, Staphylococcus aureus, Enterobacter species, microbial contamination and radioactive contamination are followed.35•36 General testing parameter for standardization of herbal medicines are given in Table 2.
Table 1. Stages of standardization of raw herbal drugs and herbal formulations.
|
Standardization of raw herbal drugs |
Norms to be followed during standardization |
Standardization of herbal formulations |
|
Literature data of raw plant drugs |
GSL (Good survey of literature) |
Follow define GMP |
|
Correct taxonomic identification and authentication |
GAP (Good agricultural practice) |
Toxicity evaluation |
|
Study on the medicinal part root, stem, bark etc. |
GCP (Good clinical practice |
Chemical profiling |
|
Collection details: location, stage and development time, storage etc. |
GHP (Good harvesting /handling practice) |
Pharmacodynamics |
|
Organoleptic evaluation of raw drug |
GLP (Good laboratory practice) |
Pharmacokinetic |
|
Microscopic and molecular examination |
GMP(Good manufacturing practice) |
Dosage |
|
Chemical composition |
GMT (Good marketing technique) |
Stability |
|
Shelf life of raw drugs |
|
Therapeutic merits |
|
Biological activity of whole Plant |
|
Presentation and packing |
Table 2. General testing parameters for characterization and standardization of herbal medicines.
|
Title |
Testing Parameters |
|
General data |
Geographical, harvesting time, harvesting process. |
|
Description |
|
|
Identity |
Macroscopic, microscopic, chemical, TLC finger printing. |
|
Purity |
Foreign matter, ash/sulfated ash, content of extracted matter, water content. |
|
Assay |
Constituents with known therapeutic activity(bio marker) constituent with unknown therapeutic activity (marker substances), titrimetric, photometric, HPLC/GC/TLC. |
|
Contaminants |
Pesticides, heavy metals, aflatoxins microbiological purity. |
The authenticity, quality and purity of herbal drugs are established by reference to the standards given in pharmacopoeia.37•38 The important standards mentioned in pharmacopoeia are shown in Fig. I.
QUALITY CONTROL AND STANDARDIZATION OF HERBAL DRUGS
According to WHO and European Agency for the Evaluation of Medicinal Products (EMEA)39 standardization and quality control of herbals is the process involved in the physicochemical evaluation of crude drug covering aspects, such as selection and handling of crude material, safety, efficacy and stability assessment of finished product, documentation of safety and risk based on experience, provision of product information to consumer and product promotion. Attention is normally paid to quality indices such as:
1. Macro and Microscopic Examination: For identification of right variety and search of adulterants.
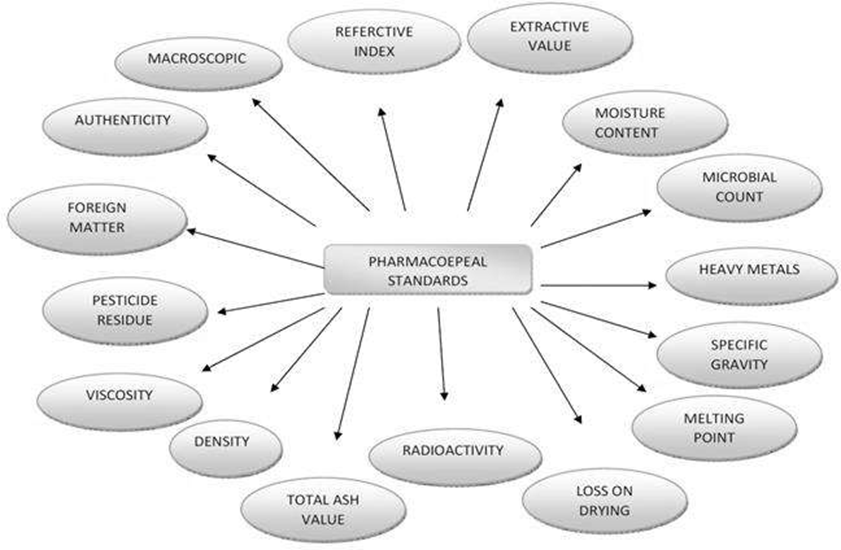
Fig. 1. Standardization parameter for herbal formulations.
2. Foreign Organic Matter: This involves the removal of matter other than plant source to get the drug in pure form.
3. Ash Value: This is the criteria to judge the identity and purity of crude drug by performing total ash, sulfated ash, water soluble ash and acid insoluble ash, etc.
4. Moisture Content: The determination of moisture content help to reduce the errors in the estimation of the actual weight of drug material. Low moisture content suggests better stability against degradation of the product.
5. Extractive Values: These are indicative weights of the extractable chemical constituents of crude drug under different solvent environment.
6. Crude Fiber: This helps to determine the woody material component, and it is a criterion for judging the purity.
7. Chromatographic Examination: Include identification of crude drug based on the use of major chemical constituents as markers.
8. Qualitative Chemical Evaluation: This covers identification and characterization of crude drug with respect to phytochemical constituent. It employs different analytical technique to detect and isolate the active constituents. Phytochemical screening techniques involve botanical identification, extraction with suitable solvents, purification, and characterization of the active constituents of pharmaceutical importance.
9. Quantitative Chemical Evaluation: To estimate the amount of the major classes of constituents.
10. Toxicological Studies: This helps to determine the pesticide residues, potentially toxic elements, safety studies in animals like LD50 and microbial assay to establish the absence or presence of potentially harmful microorganisms.
11. Pharmacological Evaluation: The herbal pharmacological activity of certain herbal drugs could be used to standardize the drug.
12. Microbial Evaluation: It involves the determination of microbial counts for compliance in the prescribed limits.
The standardization processes mentioned above involve a wide array of scientific investigations, which include physical, chemical and biological evaluation of the material employing various analytical methods and tools. The specific aim of such investigations in assuring herbal quality are as varied as the processes employed.40,41
CONCLUSION
Plants are used nowadays throughout the world as home remedies for minor ailments, in over-the-counter drug products, and as formulated products for clinical treatment. They now represent a considerable proportion of the global drug market. Therefore, it is considered essential to develop and apply internationally recognized guidelines for the assessment of the quality of herbal products. Although they have gained high popularity among the general public, Hakeem do not possess a proper understanding of their quality and safety. The indiscriminate use of herbs may be dangerous and there is a great need for standardization of herbal medicines, by established methods to safeguard their efficacy and clinical usefulness for various diseases and ailments.
REFERENCES




Pallavi Medar, Vivekananda Medar, Bhagyalakshmi Gondhali, Dafiya Soudagar, An Overview of Herbal Medicine Standardization, Int. J. of Pharm. Sci., 2026, Vol 4, Issue 2, 4001-4009. https://doi.org/10.5281/zenodo.18763012155
 10.5281/zenodo.18763012155
10.5281/zenodo.18763012155